Abstract
Objective:
Struma ovarii (SO) is a rare ovarian tumor, primarily arising from ovarian dermoid cysts that contain thyroid parenchyma. Due to the rarity of cases, we aimed to present clinical factors, management strategies, and outcomes related to SO patients.
Methods:
This is a retrospective descriptive cohort study of SO patients from 2010 to 2023 in a tertiary referral center. Data were extracted from the local electronic records, and variables were analyzed. Pathology slides and radiological images were re-evaluated by an expert pathologist and radiologist.
Results:
The study included 12 cases of SO, with a median age of 47.92 years (SD: 13.55). The average lesion size in imaging was 12.1 cm (range 7–37.5 cm). Ten patients presented with unilateral tumors (83.3%) and two presented with bilateral tumors (16.7%). Mild ascites was present in two cases (16.7%) while moderate ascites was present in one case (8.3%), who had also presented with marked left pleural effusion. Salpingo-oophorectomy was performed in six patients (50%), while the other six patients underwent total abdominal hysterectomy and bilateral salpingo-oophorectomy. The thyroid tissue of the SO was benign in 10 cases (83.3%) and malignant in 2 cases (16.7%), including 1 case of highly differentiated follicular carcinoma and 1 case of papillary thyroid carcinoma. Univariate and multivariate logistic regression analyses showed no statistically significant associations for predictors of malignancy. Univariate and multivariate logistic regression analyses showed no statistically significant associations for predictors of malignancy.
Conclusion:
SO is a very rare ovarian tumor with no characteristic clinical or imaging findings. Follow-up is necessary to demonstrate the prognosis of both benign and malignant SO.
Introduction
Struma ovarii (SO) is a rare monodermal variant of ovarian teratoma, primarily arising from ovarian dermoid cysts that contain thyroid parenchyma. 1 It constitutes approximately 0.3%–1% of all ovarian neoplasms and about 2%–5% of mature ovarian teratomas. 2 While 5%–15% of dermoid tumors contain thyroid tissue, only 2% meet the criteria for SO. 3 For a tumor to be classified as an SO tumor, the thyroid fraction must make up more than 50% of the total tissue, form a macroscopically visible part of the dermoid tumor, or harbor malignant or functional thyroid tissue. 4
The median age upon diagnosis was reported to be 43 years old, with the range being generally between 12 and 77 years old. 5 SO can present with a wide range of clinical manifestations, making its preoperative diagnosis quite difficult. Many cases are asymptomatic, with 41.2% being detected incidentally during routine ultrasound (US) examinations, which underscores the challenge of identifying SO early. 6 Clinical symptoms, when present, are nonspecific and may include lower abdominal pain, pelvic mass, or, less frequently, ascites. 7 Despite the predominance of thyroid tissue in these tumors, only 5%–8% are associated with clinical hyperthyroidism. 8
SO is often benign; Malignant SO (MSO) is extremely rare, occurring in less than 5% of cases and rarely metastasizing. 9 Pelvic US is the first-line imaging modality for identifying ovarian abnormalities, although SO can show variable nonspecific sonographic features. 10 Imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) may suggest the presence of a complex ovarian mass, but often fail to provide definitive differentiation between benign and malignant cases.11,12 MSO is characterized by differentiated thyroid carcinoma within mature cystic teratomas of the ovaries. Its diagnosis is typically confirmed postoperatively through histopathological examination of surgical specimens. 5 Radiological features typically include a complex, multilobulated mass with cystic and solid components, with solid portions showing contrast uptake corresponding to thyroid tissue rich in blood vessels and fibrous tissue. 12
The rarity of MSO has led to ongoing debates regarding its optimal diagnosis and management strategies. 13 Surgical resection is the primary treatment; it generally involves localized approaches, such as unilateral salpingo-oophorectomy, as with benign SO or hysterectomy with bilateral Salpingo-oophorectomy.14,15 Despite the benign nature of most cases, when malignant transformation occurs, treatment strategies are more aligned with protocols for thyroid cancer rather than ovarian cancer, reflecting the unique pathophysiology of SO. 12 Management interventions include ovarian surgery, thyroidectomy, radioactive iodine (131-I) ablation, and thyroid hormone suppressive therapy, depending on the presence of metastatic disease, recurrence risk, and molecular genetic findings. 16
This study aimed to identify clinical factors related to SO patients, including clinical picture, diagnostic criteria, histopathological variants, recurrence rates, and the association with thyroid disease.
Methods
Study design and setting
This is a retrospective descriptive cohort study of all the patients who underwent surgical management for SO from January 2010 to December 2023 in a tertiary referral center. Ethical approval was obtained from the IRB under the number R.25.03.3116.
Patient selection and inclusion criteria
Inclusion criteria
Histopathologically confirmed cases of SO.
Exclusion criteria
Incomplete records. Lack of histopathological confirmation.
Data collection and sources
The prospectively maintained electronic database of our center was retrospectively investigated, and the variables were statistically analyzed. The pathology slides were reevaluated by an expert pathologist, and the imaging details were rechecked by an expert radiologist.
Demographic and clinical variables
Age, clinical presentation, tumor size, tumor side, imaging findings, surgery details, tumor markers, associated thyroid disease, thyroidectomy, and follow-up.
Imaging
Sonographic examination
All patients underwent routine pelvic US examinations, including Doppler imaging with conventional transabdominal and transvaginal techniques.
CT technique
CT examination was performed on a 128 multidetector CT scanner. Scanning extended from the diaphragm to the symphysis pubis utilizing the following acquisition parameters: 220–400 mAs, 120 kVp, window width 400, matrix 512 × 512, and section thickness of 5 mm. Nonionic contrast medium (Omnipaque 350, GE Health care) was provided at a 1.5 mL/kg dose.
MRI technique
MRI was performed using a 1.5-T MR scanner. Imaging started with T2-weighted images (T2-WI) in the axial and sagittal planes with and without fat suppression, followed by T1WI with and without fat suppression. The postcontrast images (3D gradient echo sequence with fat saturation, THRIVE, Philips) were gained after IV injection of Gadoterate meglumine contrast medium at a dose of 0.1 mmol/kg by an automatic injector. MRI images were processed on an extended MR Workspace 2.6.3.5, Philips Medical Systems.
Image interpretation
Image analysis was done by a consultant radiologist dedicated to interpreting gynecological imaging. The following features were analyzed for each lesion: (1) the size (the maximum diameter) (2) the side (left, right, or bilateral); (3) the presence of cystic changes; (4) MRI signal intensity on T2-WI compared with the neighboring pelvic muscles (hypointense, intermediate, hyperintense, mixed-signal); (5) presence of high attenuation areas or calcification on noncontrast CT images; (6) presence of enhancing solid component on CT or MRI; (7) degree of ascites (mild, moderate, or marked); (8) presence of pelvic lymph nodes; (9) other radiological signs (pleural effusion).
Statistical analysis
Data were analyzed using the IBM SPSS software package version 27. Qualitative data were described using numbers and percentages. Quantitative data were described using median (minimum and maximum) for nonparametric data and mean, and standard deviation for parametric data after testing normality. The significance of the obtained results was judged at the 0.05 level.
Results
The study included 12 cases of SO, with a median age of 47.92 years (SD 13.55). Clinically, four patients (33.3%) presented with pain, three patients (25%) with a mass, and two cases (16.7%) presented with bleeding, while the remaining three patients were asymptomatic. None of the patients presented with symptoms of hypo or hyperthyroidism.
Radiologically, all the patients had multilocular cystic lesions with internal septae by ultrasonography. Four cases showed internal mural nodules and soft tissue. On Doppler examination, seven cases revealed mild septal vascularity, while four cases revealed high vascularity of the soft tissue components. Three patients (25%) underwent a CT examination, while the other nine patients (75%) underwent pelvic MRI examination; three of them performed noncontrast MRI examinations. The average lesion size was 12.1 cm (range 7–37.5 cm). Five tumors (41.7%) were right-sided, and another five were left-sided, while two cases (16.7%) showed bilateral tumors. All the tumors exhibited cystic changes. MRI T2 signal intensity was mixed in eight cases with focal areas of low T2 signal (88.9%), and hyperintense only in one case (11.1%) (Fig. 1). Postcontrast enhancing solid component was detected on CT in one case, while on MRI it was present in four cases (44.4%). Mild ascites was present in two cases (16.7%) while moderate ascites was present only in one case (8.3%), who had also presented with marked left pleural effusion. Pelvic lymphadenopathy was not demonstrated in any of the cases. Tumor markers results showed all normal median ranges: a CA-125 median of 29 (range: 4.59–372), CA 19–9 median of 24.5 (range: 15.75–205), CEA median of 1.7 (range: 0.99–4), and LDH median of 155 (range: 151–164). The clinical, radiological, and laboratory variables are summarized in Table 1.

MRI of struma ovarii A 43-year-old woman with left ovarian struma ovarii and right ovarian simple cyst.
Clinical, Laboratory, and Imaging Characteristics

Continuous data were expressed as mean ± SD for parametric data or median and range for nonparametric data.
Categorical data were expressed as percentages.
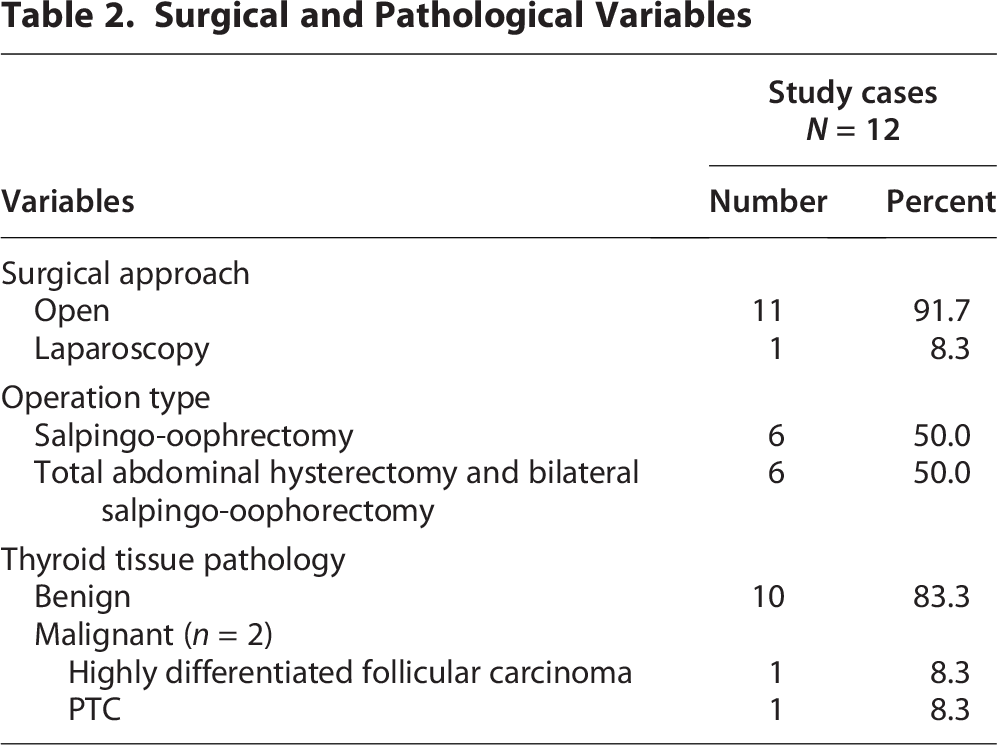
Surgical intervention was performed through an open approach in 11 patients (91.7%) while laparoscopic resection was performed only in 1 patient (8.3%). Salpingo-oophorectomy only was performed in six patients (50%), while the other six patients underwent total abdominal hysterectomy and bilateral salpingo-oophorectomy.
Grossly, 10 cases presented with unilateral ovarian mass and 2 cases with bilateral ovarian masses, ranging in size from 7 to 18 cm (median, 11.1 cm). The cut section was solid and cystic with gelatinous brown content.
Microscopically, benign tumors (Fig. 2) were formed predominantly of mature thyroid tissue that was formed of variable-sized thyroid follicles with frequent cystic changes and variable intervening fibrotic stroma. These were lined by cytologically bland cuboidal follicular cells, exhibiting rounded nuclei and eosinophilic cytoplasm, and containing eosinophilic colloid. The thyroid tissue was benign in 10 cases (83.3%) and malignant in 2 cases (16.7%). As for the two patients with bilateral tumors, one patient had bilateral SO, and the other had left SO and a mature teratoma on the right side. Regarding the two patients with pathologically malignant tumors, one case showed a highly differentiated follicular carcinoma, and the other one showed the picture of papillary thyroid carcinoma (PTC). These two patients underwent thyroidectomy, which was free from tumor tissue. IHC was performed in four patients, including the two malignant cases showing positivity for thyroglobulin and TTF1. In the patient with PTC, IHC also showed positivity for CK7 and CK19. Two of the benign cases needed IHC for confirmation of the final diagnosis. One was negative for chromogranin, while the other was positive for CK7, CD56, and negative for CK19, synaptophysin, chromogranin, galectin 3, inhibin, calretinin, CK20, and WT1. None of the patients showed recurrence during follow-up. The surgical and pathological data are summarized in Table 2.

Microscopic examination of struma ovarii:
Surgical and Pathological Variables
Discussion
SO can present with a wide range of clinical manifestations, so its preoperative diagnosis is quite difficult. 6 In our study, four patients (33.3%) presented with pain, three patients (25%) with a mass, and two cases (16.7%) presented with bleeding, while the remaining three patients were asymptomatic. These findings are consistent with other studies that reported similar presentations, including pain, pelvic swelling, vaginal bleeding, ascites, and pleural effusion 6 and confirm the nonspecific nature of the clinical presentation.
In terms of radiological findings, 10 patients had unilateral swellings (five on each side), and two patients only had bilateral tumors, suggesting that bilateral involvement in SO is uncommon. All cases showed cystic changes, which were in concordance with previous studies that suggested that the presence of a multicystic swelling with a solid element, a multilobulated surface, and viscid gelatinous content is characteristic of SO. 17 These features overlap with features of malignant ovarian masses, like mixed solid and cystic components, mixed T2 signal intensity, and an enhancing solid component.10,18–20
In our cohort, CT images revealed an enhancing solid component in one case, areas of high attenuation in another one, and no cases showed calcification foci. MRI T2 signal intensity was mixed in eight cases, with areas of low T2 signal intensity (88.89%), and hyperintense only in one case (11.1%). Similarly, previous studies revealed variable MR signal intensity with focal areas of very low T2 signal intensity and high attenuation on CT images, which can be explained by the presence of thyroid tissue that contains colloid material with iodine content. 10 Furthermore, four cases exhibited an MRI-enhancing solid component (44.4%), and two cases (22.2%) showed no enhancing soft tissue component. The presence of ascites is not a common feature of SO, but there are case reports documenting its presence even in marked form.21–24 In our cohort, nine patients had no ascites, two (16.67%) had mild ascites, and one patient (8.33%) had moderate ascites. The same patient was presented with a marked pleural effusion similar to previously published literature. 25
Laparoscopic removal of large ovarian cysts, including SO, is feasible, although most of the patients are managed via open surgery,26–28 which is consistent with our cohort. This can be explained by the absence of distinguishing clinical and imaging features, making the diagnosis mainly based on postoperative histopathological evaluation. Although Ezon et al. reported no adverse effect on disease-free or overall survival from laparoscopic surgery. 26 There are concerns about reduced staging accuracy, increased risks of port site metastasis, and cancer cell spillage when removing malignant masses laparoscopically. 29 Surgical methods vary between salpingo-oophorectomy and TAH with BSO, potentially influenced by fertility desires and tumor size. 30
Postoperatively, only two cases were MSO, including a highly differentiated follicular carcinoma 31 and PTC. Both cases revealed enhanced thick internal septae and nodularity without pathological pelvic lymph nodes. In our study, we found no significant association between the prediction of SO malignancy and commonly elevated tumor markers in other tumors, indicating these are not specific markers for SO, except for the histological diagnosis of MSO.
This study reinforces the importance of considering SO in the differential diagnosis of large, mixed-signal ovarian masses, especially in middle-aged women with normal tumor markers. The characteristic imaging features, particularly on MRI, can guide preoperative suspicion. Nonetheless, definitive diagnosis requires histopathological confirmation. The surgical approach should be personalized, taking into account fertility preservation and intraoperative findings.
To conclude, SO is a very rare ovarian tumor with few cases reported in the literature. It has no characteristic clinical or imaging findings. The diagnosis -even in malignant SO- is mainly based on postoperative histopathological evaluation. An extended follow-up is necessary to demonstrate the prognosis of both benign and malignant SO. Future research should include molecular and genetic analyses to identify novel biomarkers or genetic mutations that could predict malignancy and inform treatment decisions.
Ethics Approval and Consent to Participate
All procedures performed in the study involving human participants followed the ethical standards of the institutional research committee and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All the patients signed informed consent for the surgical maneuvers whenever indicated. This is a retrospective study. Consent for participation in the study itself is not applicable.
Consent for Publication
N/A.
Authors’ Contributions
All authors have read and approved the article. A.H.A., M.S., A.H., E.R., A.Ashraf., and A.Ayman.: Data collection and editing. A.H.A: Original draft writing. O.H.: Conceptualization and writing and revision. G.A.S.: Preparation and editing of the radiology part. A.E.E.: Preparation and editing of the pathology part. M.M.S.: Revision and editing. All authors read and approved the final version of the article.
Footnotes
Author Disclosure Statement
All authors declare they have no conflict of interest.
Funding Information
No funding was received.
