Abstract
Background:
Short-acting reversible contraceptives (SACs) are birth control methods with proven additional benefits beyond contraception. Even so, specific evidence on which SAC formulation is the most effective for a particular noncontraceptive benefit is still lacking. This study aimed to investigate the superiority of specific SAC formulations over others for particular extracontraception benefits.
Materials and Methods:
A literature search of systematic reviews and/or meta-analyses providing data on the noncontraceptive clinical benefits of SAC versus other SAC formulations or SAC versus placebo or no treatment, including the period from 2014 to 2024, was conducted. Information was extracted as individual items and classified into predefined objective health benefits.
Results:
A total of 70 articles were included in the review, with 52 items describing additional benefits of SAC versus other SAC formulations and 86 items of SAC versus placebo or no treatment. Most of the items reported information on polycystic ovary syndrome, followed by androgenism and endometriosis. For consistency, the reported clinical benefits were reclassified into five broad categories: gynecological disorders, cancer protection, menstrual bleeding disorders, hyperandrogenic conditions, and miscellanea.
Conclusions:
The use of SAC is accompanied by a range of health benefits beyond contraception that should be taken into account when prescribing. Distinguishing the particular benefits of different SAC formulations may help support informed, women-centered prescription in clinical practice.
Keywords
Introduction
Short-acting reversible contraceptives (SACs) are birth control methods used by 46.1% of contraceptive users worldwide. 1 SACs include oral contraceptive (OC) pill, progestin-only pill (POP), vaginal ring, patches, and monthly injectable contraceptives. 2 Although SACs are mostly used to prevent unintended pregnancies, additional benefits have been extensively reported, including prevention of certain types of cancer and treatment of common gynecological and nongynecological medical conditions.3,4 A range of hormonal contraception formulations are beneficial to treat other gynecological conditions such as heavy menstrual bleeding (HMB), acne, hirsutism, endometriosis, and polycystic ovary syndrome (PCOS), among others.5–7
Since the US Food and Drug administration (FDA) approved the first combined OC (COC) in 1960, numerous hormonal SAC formulations with varying components, doses, and delivery methods have been developed. SAC can be classified into progestin-only and combined estrogen–progestin formulations. 8 While all authorized SACs share fundamental similarities, each formulation contains different combinations of estrogen and progestin, doses, regimens, route of administration, and galenics, thus influencing their therapeutic outcomes beyond contraception. 8 The noncontraceptive health benefits of SAC have been extensively studied, with a recent review by Bennink et al. providing a thorough update on these benefits. 4 However, specific evidence on which SAC formulation is the most effective for each noncontraceptive benefit is still lacking. Understanding the evidence behind the clinical benefits of these formulations might support clinicians in giving informed advice that aligns with women’s health needs and preferences.
Patient-centered care (PCC) models are gaining prominence as a way to address this gap. This approach integrates patients’ preferences, values, and beliefs into health decision-making. When women choose a contraceptive method, women and clinicians should ideally engage in a thorough discussion about the benefits (both gynecological and nongynecological), risks, and personal preferences (route of administration, beliefs, etc.). 9 While the clinician may prioritize aspects such as efficacy, safety, or adherence, the woman may consider other attributes of a method (associated side effects, such as weight gain and fluid retention, or noncontraceptive add-on benefits, for instance). Therefore, the PCC model ensures that women’s unique circumstances are globally addressed.9,10 This integrated approach would allow for more personalized treatment, potentially enhancing adherence, satisfaction, and clinical efficacy. This narrative review is framed in the Agnodice project, named after the legendary figure said to be the first female gynecologist in ancient Athens, and aims to explore the current evidence of the noncontraceptive benefits of different SAC formulations, supporting informed, women-centered prescription in clinical practice.
Materials and Methods
Study design
The study was conducted in multiple successive phases under the supervision of the Scientific Committee (SC), which was composed of three gynecology expert specialists in endocrine gynecology with recognition in the medical community (the summary of the process can be found in Fig. 1).

Summary of the study design and main results.
Search strategy
The SC, based on their clinical expertise, SAC Summary of Product Characteristics’ technical data sheets, and literature search, identified the most frequently reported clinical benefits of SAC beyond contraception. The SC transformed the previously 31 identified additional clinical benefits of SAC into Medical Subject Headings (MeSH) terms and their synonyms. Then, a systematic search of all published studies from 2014 to 2024 was performed in PubMed. The selected 10-year period was chosen to capture the most recent advances in the field of hormonal contraceptives and their additional benefits, ensuring that the findings are aligned with the latest scientific developments. The search query was composed using two concepts: (i) Contraceptive search term: contracep* and (ii) MeSH OR other common synonyms in the scientific literature of benefit validated by the SC. In the Supplementary Appendix S1, the executed query can be found.
In parallel, a recently published narrative review on the clinical benefits of SAC by Bennink et al. 4 served as a starting point for conducting a bottom-to-top search for information regarding benefits not addressed by the original search strategy.
Study selection
Two reviewers (Sara García and Nerea Toro), alongside the SC, assessed the studies’ eligibility by reading the titles and the abstracts of the studies according to predefined inclusion and exclusion criteria. Publications providing quantitative data on the noncontraceptive clinical benefits of SAC when used as contraceptives, that is, comparative data between SAC versus SAC, SAC and placebo, and SAC versus no-SAC were included. Only systematic reviews and/or meta-analyses in English with available abstracts were included. Studies were excluded if they (i) were not about contraception, (ii) compared SAC with other treatments, (iii) evaluated SAC in combination with other treatments, (iv) analyzed non-SAC, or (v) lacked statistical significance testing (p-values). Finally, the two reviewers thoroughly read the preselected studies to accurately discern whether they were relevant for the review. In case of disagreement, a third reviewer additionally assessed the controversial study, and an agreed decision was reached between the three reviewers.
Data extraction
Data from the selected articles were extracted using a standardized data extraction sheet specifically created for this project (see Supplementary Appendix S2 for further details). It included key data from each article, and information was extracted as individual items. Any comparison between two or more interventions within a specific population examining particular clinical benefits and outcomes was considered “item.” According to this definition, each included article may yield several items. To note, in the present study, the term “women” refers to individuals for whom ovulation is biologically expected.
Data analysis
Based on the data obtained, the originally proposed clinical benefits were reclassified for coherence into five broad categories: gynecological disorders, cancer protection, menstrual bleeding disorders, hyperandrogenic conditions, and miscellanea (Supplementary Table S1). “Gynecological disorders” included all abnormalities related to the menstrual cycle and the reproductive system; “Cancer protection” included all benefits related to cancer; “Menstrual bleeding disorders” only included alterations in bleeding patterns; “Hyperandrogenic conditions” included pathologies related to sensitivity to androgens; and “Miscellanea” included all entities with a nonspecific relationship to cyclic changes (Supplementary Table S1).
Results
Description of included studies
The search query in PubMed retrieved a total of 258 studies. After an initial screening of title and abstract, 148 studies were excluded based on the following reasons: they did not focus on contraception, SAC, or the clinical benefits of SAC, and they did not meet the methodological criteria (systematic review or meta-analysis); abstract/text was not available; or they fell outside the defined time frame. The remaining 110 articles were fully assessed for eligibility through a complete reading of the text. Among these, 35 studies were excluded for not meeting the inclusion/exclusion criteria (Fig. 2). The list of all the excluded publications and reasons for exclusion can be found in Supplementary Appendix S3. In addition, 11 articles addressing clinical benefits not covered in the original search were extracted by bottom-to-top search from Bennink et al. 4 (Fig. 1). These additional 11 articles were six controlled studies, one retrospective study, one double-blind study, and three population-level questionnaires. Finally, 86 studies were included in the review. The list of all included studies can be found in Supplementary Appendix S4.
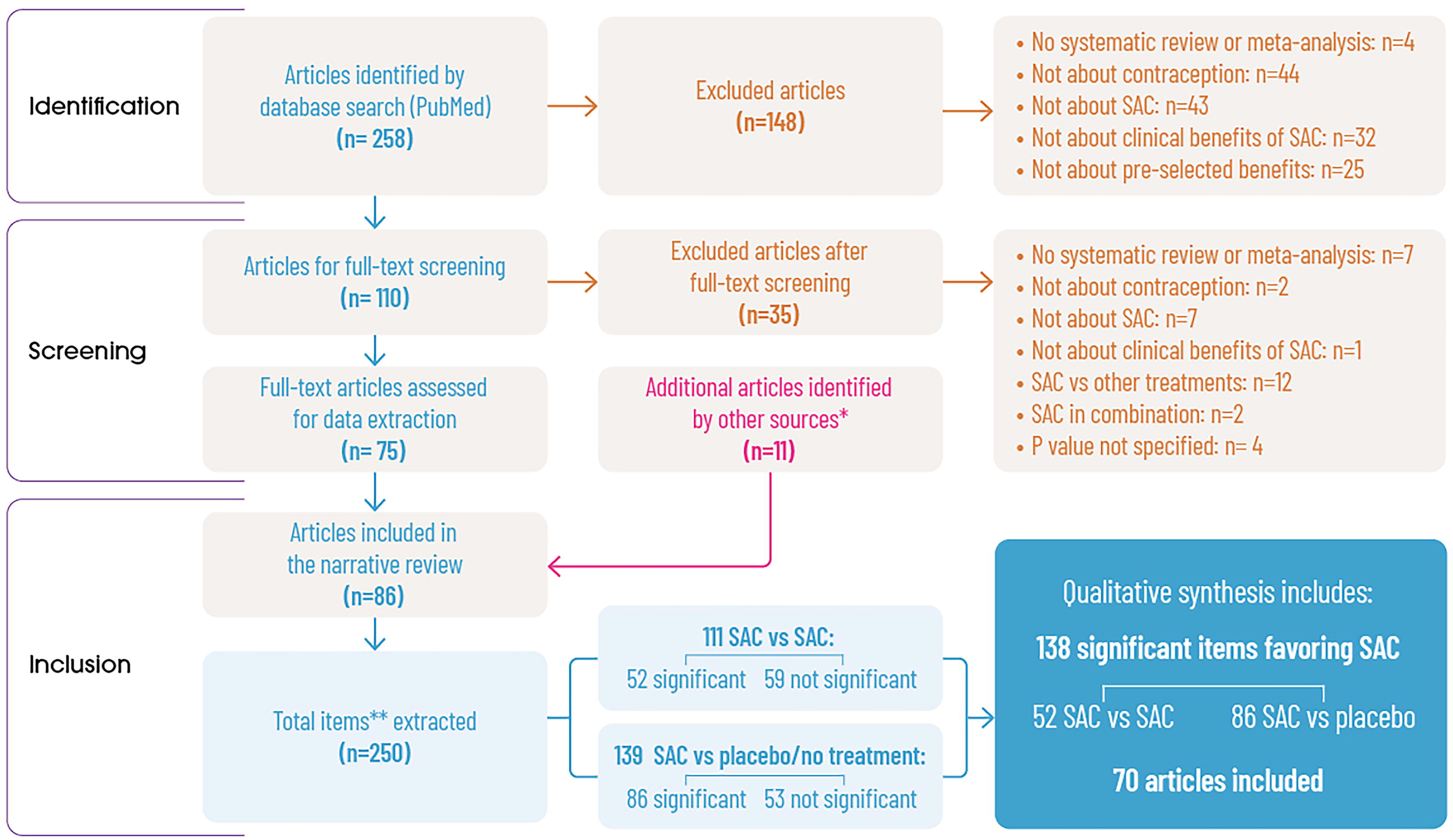
PRISMA flowchart. *A recently published review on the clinical benefits of SAC (Bennink et al., 2024) served as a starting point for conducting a bottom-to-top search for information on benefits not addressed by the original search strategy. **“Item” refers to data comparing two or more interventions within a specific population, examining particular clinical benefits and outcomes. “Not significant” refers to those comparisons that did not find differences in the effects of the compared interventions (p value <0.05). SAC, short-acting contraceptive; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Given the heterogeneity of available evidence regarding SAC benefits, with differences in study designs, outcome measures, and populations, data were presented as a narrative review. This approach allows for a comprehensive synthesis of existing literature while identifying knowledge gaps that require further investigation.
In the data extraction, the 86 articles reported 250 items of information, that is, data comparing two or more interventions for a particular clinical benefit in a specific population. However, only 138 items (from 70 articles) reported statistically significant differences between the compared interventions, and they were finally considered in the present review (Fig. 2).
SAC benefits beyond contraception compared with placebo or no treatment
A total of 139 comparisons regarding the additional benefits of SAC versus placebo or versus no-treatment on a range of conditions were extracted from the literature. Of these 139 items, 86 showed significant differences between SAC and placebo/no intervention (Fig. 2). The intervention with SAC demonstrated superiority for various formulations in 86 cases (Table 1). Most of the evidence refers to the benefits of SAC versus placebo/no treatment in gynecological disorders (35 items), followed by hyperandrogenic conditions (18 items) and a miscellanea of conditions (16 items) (Table 1). When no evidence on SAC benefits beyond contraception versus placebo or no treatment for a particular clinical benefit was obtained in the search, the row for that specific clinical benefit was not added in the results table (e.g., ovarian cysts). Further details can be found in Supplementary Table S2.
Number of Items Favoring SAC Versus Placebo/No Treatment Regarding Clinical Benefits Beyond Contraception

In this review, the term “androgenism” refers to hirsutism and acne in women without a diagnosis of polycystic ovary syndrome (PCOS), in accordance with the populations assessed in the included studies. SAC, short-acting contraceptive.
SAC formulation choice per benefit
Among the 250 comparisons extracted, 111 compared two or more SAC formulations. Of those, 52 reported a significant difference between two or more SAC formulations for the benefit of a specific condition (Fig. 3). Most of them reported information on PCOS (20 items), followed by androgenism (12 items) and endometriosis (8 items) (Table 2). When no evidence on SAC benefits beyond contraception for a particular clinical benefit was obtained in the search, the row for that specific clinical benefit was not added in the results table (e.g., pelvic inflammatory disease). Further details can be found in Supplementary Table S2.

Summary of preferred SAC formulations for each specific clinical benefit. CHCs, combined hormonal contraceptives; CMA, chlormadinone acetate; CPA, cyproterone acetate; DNG, dienogest; DRSP, drospirenone; DSG, desogestrel; EE, ethinylestradiol; ENG, etonogestrel; GSD, gestodene; LNG, levonorgestrel; NET, norethisterone; NGMN, norelgestromin; OCs, oral contraceptives.
Summary of SAC Comparisons per Benefit
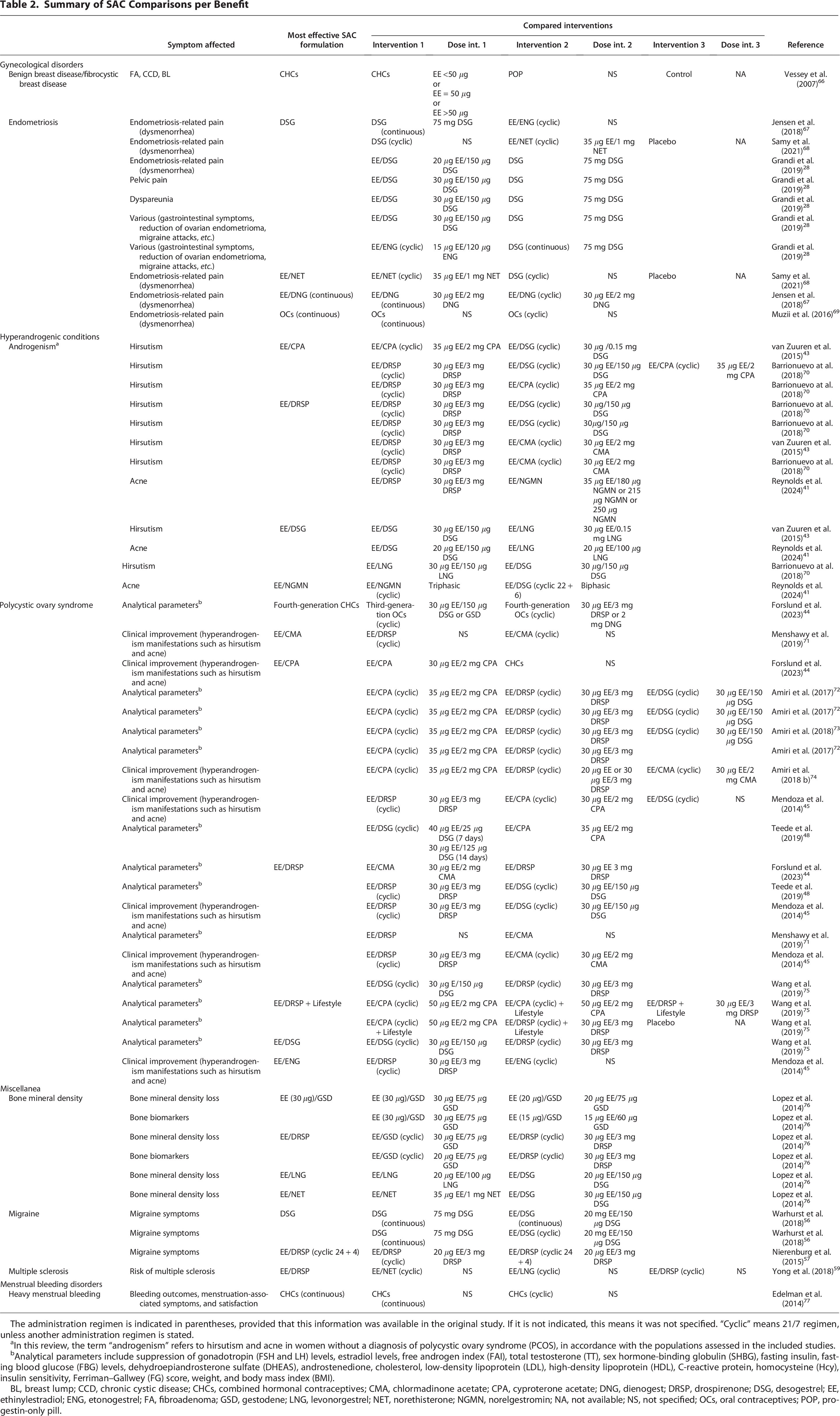
The administration regimen is indicated in parentheses, provided that this information was available in the original study. If it is not indicated, this means it was not specified. “Cyclic” means 21/7 regimen, unless another administration regimen is stated.
In this review, the term “androgenism” refers to hirsutism and acne in women without a diagnosis of polycystic ovary syndrome (PCOS), in accordance with the populations assessed in the included studies.
Analytical parameters include suppression of gonadotropin (FSH and LH) levels, estradiol levels, free androgen index (FAI), total testosterone (TT), sex hormone-binding globulin (SHBG), fasting insulin, fasting blood glucose (FBG) levels, dehydroepiandrosterone sulfate (DHEAS), androstenedione, cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), C-reactive protein, homocysteine (Hcy), insulin sensitivity, Ferriman–Gallwey (FG) score, weight, and body mass index (BMI).
BL, breast lump; CCD, chronic cystic disease; CHCs, combined hormonal contraceptives; CMA, chlormadinone acetate; CPA, cyproterone acetate; DNG, dienogest; DRSP, drospirenone; DSG, desogestrel; EE, ethinylestradiol; ENG, etonogestrel; FA, fibroadenoma; GSD, gestodene; LNG, levonorgestrel; NET, norethisterone; NGMN, norelgestromin; NA, not available; NS, not specified; OCs, oral contraceptives; POP, progestin-only pill.
A summary of the reported SAC clinical benefits beyond contraception, according to previously defined categories is explained hereafter.
Gynecological disorders
Various SAC formulations showed efficacy in managing symptoms related to endometriosis, such as intermenstrual pain, dysmenorrhea, and gastrointestinal symptoms. For endometriosis-related pain, desogestrel (DSG) alone demonstrated significant efficacy compared with ethinylestradiol (EE)/etonogestrel (ENG) 67 ; it also showed superiority over EE/norethisterone (NET) and placebo, 68 and it was more effective than EE/DSG in multiple instances. 28 Besides this, EE/dienogest (DNG) on a continuous regimen was superior to cyclic EE/DNG for addressing endometriosis-related pain. 67 For managing dysmenorrhea symptoms in endometriosis, EE/NET was more effective than DSG and placebo, 68 and OCs on a continuous regimen were superior to cyclic OCs. 69 When considering rate of satisfaction regarding changes in gastrointestinal symptoms in endometriosis, DSG was reported to be more effective than EE/ENG. 28 Briefly, these findings highlight DSG and continuous regimens as particularly effective for symptom management, with EE/DSG offering additional benefits in certain cases. The results of SAC formulation benefits on endometriosis-related symptoms are summarized in Table 2.
Regarding benign breast disease (including fibroadenoma, chronic cystic disease, and breast lump), combined SAC including progestin plus estrogen (containing <50 μg, 50 μg vg, or >50 μg ethinylestradiol) demonstrated a significantly greater benefit versus POP and controls 66 (Table 2).
Hyperandrogenic conditions
Different SAC formulations showed efficacy in managing a range of androgenism-related symptoms (Table 2). For hirsutism management, EE/cyproterone acetate (CPA) (cyclic) was more effective than EE/DSG (cyclic), 43 EE/drospirenone (DRSP), and EE/DSG. 70 The combination EE/DRSP was also reported to be more effective than EE/chlormadinone acetate (CMA)43,70 and EE/DSG, 70 and the combination EE/DSG showed superiority over EE/levonorgestrel (LNG). 43 In improving acne severity, the combination of EE/DSG was superior to EE/LNG41,43; and EE/DRSP was more effective than EE/norelgestromin (NGMN), 41 whereas EE/NGMN outperformed EE/DSG in a different study 41 (Table 2).
Various SAC formulations demonstrated efficacy in managing symptoms related to PCOS: EE/DRSP was the most versatile and effective option across multiple analytical parameters, including endocrine regulation, hyperandrogenism, and lipid profiles,44,45,48,71,75 while EE/CPA and EE/DSG also offered strong support for medical improvement, including specific conditions such as hyperandrogenism and hirsutism.44,45,74 EE/CPA was superior to EE/DSG in addressing hirsutism. 43 Further details can be found in Table 2.
Miscellanea
Different SAC formulations showed efficacy in managing a range of entities with a nonspecific relationship to cyclic changes. In this category, outcomes reporting significant differences regarding bone mineral density (BMD) loss, migraine, and multiple sclerosis (MS) were included (Table 2).
BMD loss
Various SAC formulations showed efficacy regarding BMD and biochemical markers of bone turnover in women (Table 2). For BMD, EE (30 μg)/gestodene (GSD) showed superiority over the same combination with lower EE doses (20 μg) 76 ; the combination of EE/DRSP (cyclic) showed superiority over the combination of EE/GSD (cyclic), 76 and the combinations of EE/LNG and EE/NET showed superiority over EE/DSG. 76 For bone biomarkers, EE (30 μg)/GSD showed significant results over lower EE doses (20 μg and 10 μg) 76 ; and the combination of EE/DRSP (cyclic) showed superiority over the combination of EE/GSD (cyclic) 76 (Table 2).
Migraine
Various SAC formulations demonstrated efficacy in managing migraine symptoms (Table 2). DSG on a continuous regimen alone demonstrated significant efficacy compared with EE/DSG, both on continuous and cyclic regimens. 56 Furthermore, the combination EE/DRSP (cyclic 24 + 4) showed superiority over EE/DRSP (cyclic 21 + 7) 57 (Table 2).
Multiple sclerosis
Finally, the combination of EE/DRSP (cyclic) showed superiority over the combination of EE/NET (cyclic) and EE/LNG (cyclic) for risk of presenting with MS 59 (Table 2).
Menstrual bleeding disorders
Combined hormonal contraceptives (CHC) on a continuous regimen showed significant efficacy in managing HMB outcomes, menstruation-associated symptoms, and satisfaction, compared with cyclically administered CHC 77 (Table 2).
Discussion
The present review gathered evidence of clinical benefits beyond contraception of different hormonal SAC formulations. In particular, it has outlined the evidence regarding the add-on benefits of specific SAC formulations over other SAC for particular conditions. Overall, 52 comparisons reporting significant differences between two or more SAC formulations were synthesized. The most frequently reported additional benefits of SAC were focused on hyperandrogenic conditions, a miscellanea of conditions (migraine, MS, and BMD), and gynecological disorders or menstrual bleeding disorders.
Endometriosis is an estrogen-dependent condition with symptoms associated with hormonal changes along the menstrual cycle, accompanied by pelvic pain, or dysmenorrhea and dyspareunia. 78 Current guidelines recommend a range of hormonal treatments to reduce endometriosis-related pain, and their effects are similar for all treatments. 79 In the present study, however, DSG alone was the SAC formulation most frequently reported as beneficial in managing endometriosis-related symptoms. Given the high disease burden and the impact on quality of life, 78 the links between specific COC formulations and the benefits on endometriosis-related symptoms should be further explored. In the present study, continuous administration of EE/DNG was superior to cyclic EE/DNG for addressing endometriosis-related pain, thus reinforcing the superiority of continuous regimens. 67
SACs are also useful for managing menstrual bleeding disorders, given that they help reduce endometrial thickness and maintain a thin and stable endometrium, which results in a decrease in blood loss. In the present study, CHC on a continuous regimen showed significant efficacy in managing HMB outcomes, menstruation-associated symptoms, and satisfaction, compared with cyclically administered CHC. In fact, bleeding patterns in women on continuous CHC regimens have been reported to be better than in women on cyclic CHC regimens. Continuous use may result in amenorrhea and further improve outcomes compared with cyclic use. 67 Contraceptive-induced menstrual bleeding changes have been reported as top reasons for contraceptive dissatisfaction and discontinuation. 80 In the present study, we focused on data comparing two or more SACs to highlight the specific benefits of each formulation. The combination estradiol valerate (E2V/DNG) was not included in our main results due to the lack of comparative studies with other SAC formulations. However, as shown in Supplementary Table S2, a substantial body of evidence supports E2V/DNG as a safe and effective treatment for women with HMB without organic pathology. 65 Moreover, two randomized double-blind phase III clinical trials showed a significant mean reduction in menstrual blood loss volume in women receiving E2V/DNG compared with placebo. In addition, the percentage of women on E2V/DNG prematurely discontinuing treatment because of adverse events was similar to women on placebo, thus confirming the efficacy and safety of E2V/DNG combination for HMB.81,82 The combination E2V/DNG is authorized for HMB treatment in women without an underlying pathology.
For hyperandrogenic conditions, the combinations of EE/DRSP and EE/CPA were the most frequently reported as beneficial over other SACs when administered for contraceptive reasons. Although all SACs improve hyperandrogenic conditions, the progestins CPA and DRSP have an additional antiandrogenic effect beyond their antiestrogenic activity, which may partially explain this superiority. 8 Nevertheless, no direct trials comparing the additional activities of progestins have been conducted so far, and whether the additional benefit varies by progestin type or dose should be further explored.
According to hirsutism treatment guidelines, CHC should be offered as first-line therapy 83 ; however, women with mild hirsutism and no evidence of an endocrine disorder may not necessarily receive treatment. Taking into account the impact of hirsutism on woman’s psychological health, social performance, and quality of life, 84 women with mild hirsutism or signs of androgenization seeking contraception might benefit from receiving these combinations. The combination EE/DNG is effective in controlling hyperandrogenism and hirsutism and ameliorates the abnormal metabolic profile of women with hirsutism. 85
More research is needed to evaluate the benefits of administering a particular SAC formulation to women with hyperandrogenic conditions.
COCs have been demonstrated to increase BMD and to reduce the risk of fracture in postmenopausal women. 86 In this study, the combinations of EE/DRSP and of EE (30 μg)/GSD were the most frequently reported as beneficial over other SACs in BMD, a surrogate marker. Larger studies should be designed to disentangle the precise benefits in long-term fracture prevention.
DSG alone was also reported to be beneficial over other SACs for migraine treatment. Epidemiological, pathophysiological, and clinical evidence associates estrogen with migraine. 87 Accordingly, the World Health Organization (WHO) states that women with migraine with aura or women >35 years old suffering migraine without aura are not eligible for estrogen use. 88 Therefore, women with mild migraine, or those not receiving concomitant preventive medication for headache and seeking contraception, might also benefit from receiving specific SAC formulations.
MS is most prevalent in women, and physiological/hormonal states such as puberty, pregnancy, or menopause have an impact on the disease course. 89 MS is a chronic inflammatory disease, and hormones have a role in regulating the immune system; thus, SAC may provide additional benefits for MS. 89 In the present study, the combination of EE/DRSP was identified as superior to the combinations EE/NET and EE/LNG for the risk of presenting with MS. Although the mechanism of action of DRSP in MS is not well known, its role may be partially explained by its antimineralocorticoid activity. 8
The findings of the present study provide evidence-based data to support health care practitioners (HCPs) during contraceptive counseling. When choosing a hormonal contraceptive, clinicians should consider a shared decision-making approach, being aware of individual preferences, side effects, individual efficacy, costs, and availability. During contraceptive counseling, the clinician should also look for those methods that may bring additional benefits to the woman. 9 Personalized prescription is especially relevant in women with gynecological pathology, because they can benefit most from this tailored counseling. To decide on the management of specific treatment alternatives, clinicians should consider the studies where admission conditions coincide with actual patient characteristics.
Beyond comparative effectiveness, safety considerations are essential when recommending short-acting contraceptives to individual patients. Clinical decision-making should integrate both efficacy and tolerability, as adverse effect profiles may significantly influence patient preference and adherence. In this context, international guidance such as the WHO Medical Eligibility Criteria for Contraceptive Use 88 provides a standardized framework to balance effectiveness with safety and to support individualized contraceptive counseling.
Having clear and trustworthy information regarding contraception is very important for weighing the advantages and disadvantages of each option and for planning in advance regarding potential risks or discomforts associated with medication. In fact, according to a European survey on contraception, almost two-thirds (64%) of women currently using an SAC method were interested in receiving more information, although HCPs often underestimate women’s desires for information regarding SAC methods. 19
HCPs have the responsibility to disseminate the scientific information without bias. By expanding knowledge regarding efficacy beyond contraception, we can address both contraceptive and noncontraceptive needs. Prescribing the treatment that best fits women’s needs may increase their satisfaction with the method and their willingness to use it, thus producing a virtuous circle of increased adherence, satisfaction, and clinical efficacy.
Strengths
The main strengths of the present study arise from including data exclusively from systematic reviews and meta-analyses. Moreover, data extraction was systematically performed by two expert reviewers, with the support of the SC, and following a comprehensive, thoughtful procedure with predefined inclusion criteria, exclusion criteria, classification categories, and standardized data extraction forms. Both the information sources (only systematic reviews and meta-analyses) and the carefully followed procedure ensure the robustness of the included data. The findings of this study provide evidence on SAC benefits beyond contraception that may eventually have an impact on clinical practice guidelines to improve patient care and outcomes.
Limitations
The authors acknowledge several limitations with the present study, mainly that it is not a systematic review, although an explicit methodology was followed with comprehensive and reproducible results. However, data included were heterogeneous in terms of study settings, interventions, and measurement variables. The search was performed exclusively in PubMed due to its comprehensive coverage of medical and clinical studies, particularly in the field of gynecology and hormonal contraception. However, using additional databases might have provided a more exhaustive collection of relevant studies. The search covered the 10 previous years of publications (2014–2024); this period was chosen to capture the most recent advances in the field of hormonal contraceptives and their additional benefits, ensuring that the findings are aligned with the latest scientific developments. A language bias cannot be excluded, since only English-written publications were considered.
A key limitation of the present review is that adverse effects were not extracted or analyzed, and therefore, no detailed discussion or results on safety profiles are provided, being a critical factor in clinical decision-making alongside comparative effectiveness. Future syntheses should integrate both efficacy and safety data to better guide individualized patient care.
A further limitation of this review is the absence of comparative studies for certain short-acting contraceptives in noncontraceptive indications, which restricts the ability to draw balanced conclusions across formulations. This gap in the evidence underscores the need for future studies to directly assess the comparative efficacy and safety of these regimens.
Conclusions
The present study provides new insights into the association between benefits beyond contraception and specific hormonal SAC formulations, offering valuable evidence to guide clinicians in making more informed decisions when prescribing contraceptives. By shedding light on the noncontraceptive health benefits associated with various SAC options, this research supports a more personalized approach to contraceptive care, ensuring that women’s individual health needs and preferences are better addressed.
The findings from this study pave the way for further research into the specific benefits of SAC formulations for specific conditions, which shall be explored in upcoming publications. These studies will help to refine treatment strategies by providing deeper insights into how different SAC options may address specific medical needs.
In addition, while the present study identifies the most clinically effective SAC for noncontraceptive benefits, further research is needed to understand women’s preferences and their perceptions of these benefits. Understanding these preferences is critical, as it will allow clinicians to further personalize treatment strategies and ensure that the contraceptive method chosen aligns not only with clinical needs but also with women’s values and lifestyle.
Clinicians should incorporate the findings from this study into their contraceptive counseling, emphasizing the noncontraceptive benefits of specific SAC formulations in addressing women’s broader health needs. By personalizing contraceptive care, health care providers can improve patient satisfaction and health outcomes.
Authors’ Contributions
All authors have made substantial contributions to the conception and design of the work, have drafted the initial version of the article, critically revised the article for its intellectual content, approved the final version of the article, and agreed to be accountable for all aspects of the work.
Footnotes
Acknowledgments
The authors thank Gloria González, Montse Pérez, Nerea Toro, and Sara García from Adelphi Targis SL for their technical and editorial support. Medical writing support was provided by Alba Gómez, PhD. These contributions supported the preparation of the article but did not influence the study design or interpretation of results.
Author Disclosure Statement
J.C.A. declare no conflicts of interest related to this work.
J.C.Q.C. has received consulting fees from Pfizer, honoraria for lectures and presentations from Gedeon Richter, MSD, Bayer and Theramex; and has participated in advisory boards for Exeltis, Bayer and Viñas.
J.P.C. has received honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Theramex, Organon, Cinfa, Bayer, Medtronic and Exeltis; support for attending meetings and/or travel from Theramex and Exeltis; and participated on a Data Safety Monitoring Board or Advisory Board from Theramex and Cinfa.
Funding Information
The Agnodice project was sponsored by Cinfa Salud de la Mujer, providing the necessary financial support for its development. The sponsor had no role in the study design, data collection, analysis, interpretation, or article drafting.
