Abstract
Background:
The menopause transition and postmenopause are accompanied by a wide range of cognitive, emotional, and physical symptoms that can significantly affect women’s quality of life and brain health. Despite the growing population of menopausal women, menopausal hormone therapy (MHT) remains under-researched in relation to cognitive and emotional well-being. This pilot study evaluated the effects of personalized, continuous MHT on brain health using the validated, multidimensional BrainHealth™ Index (BHI).
Methods:
Forty-two postmenopausal women completed the BHI at baseline (T1), 6 months (T2), and 12 months (T3). After T1, participants began personalized, titrated MHT with close follow-up at 5 weeks. Thirty-six women from a separate database, matched on age and education, served as controls. Mixed-effects modeling examined main effects of time, group, and the time × group interaction on four measures: the BHI composite and its components—clarity, connectedness, and emotional balance.
Results:
At T1 (before MHT), the MHT group exhibited significantly lower scores across all BHI indices, indicating greater impairment in cognitive, emotional, and social domains relative to controls. A significant group × time interaction revealed that after starting MHT, the MHT group showed greater improvement from T1 to T2 across all indices compared with the control group. By T2, MHT participants reached scores comparable to controls, and these gains were maintained at T3, with no further group differences in score change.
Conclusions:
Personalized MHT in this pilot sample of symptomatic postmenopausal women led to improvements in cognitive clarity, emotional well-being, and social connectedness. These preliminary findings suggest that MHT may serve as a whole-person intervention integrating neuroendocrine, cognitive, and emotional domains. Confirmation in larger, randomized controlled trials is needed to evaluate generalizability, adoption of personalized MHT, and the sensitivity of the BHI as a sensitive clinical assessment tool for this population.
Keywords
Introduction
In the United States, approximately 1.3 million women become menopausal each year. 1 The menopause transition, or perimenopause, is defined as a time period in a woman’s life where ovarian production of estrogen begins to fluctuate dramatically, can last from 2 to 8 years, and has been reported to be one of the most challenging times for women as it relates to health-related quality of life (HRQOL). Symptoms of the menopause transition and postmenopause vary greatly from woman to woman and can include hot flashes, urogenital complaints, generalized fatigue, irritability, depression, mood swings, anxiety, memory impairment, difficulty focusing, decreased word recall, and insomnia. Menopause is often associated with cognitive complaints, especially memory disturbances, 2 mood changes, and decreased overall cognitive function. Studies have shown objective declines in verbal memory, attention, and working memory during the menopausal transition. 3 Based on workdays missed due to menopause symptoms, researchers estimate an annual loss of $1.8 billion in the United States. 4 All of the aforementioned symptoms have been reported to greatly decrease quality of life and desire to fully engage in life as before the menopause transition. Menopause is an area of women’s health that has been underfunded and understudied, leaving women to navigate significant brain and health changes with few resources and solutions.
Sex hormones, including estradiol, progesterone, and testosterone, play important roles in cognitive function, social, and emotional well-being.5,6 For example, estradiol enhances verbal memory, attention, and processing speed.5–7 Research indicates that higher levels of estradiol are associated with improved verbal memory performance, particularly in women during the follicular phase of the menstrual cycle when estradiol levels are elevated. 8 Progesterone modulates mood and emotional processing. 8 Higher progesterone levels may lead to increased anxiety or mood swings, which can indirectly affect cognitive functions such as decision making and problem solving. 8 Testosterone has been linked to mood regulation and adequate levels of testosterone can contribute to a positive mood and emotional stability. 9 Conversely, low testosterone levels may be associated with increased feelings of anxiety and depression, impacting overall emotional health. 10 Additionally, women with higher testosterone levels have shown better cognitive performance, particularly in tasks requiring spatial awareness and verbal memory. 11 While the intricate interplay and underlying mechanisms of these hormones in relation to cognition and brain health are beyond the scope of this article, it is clear that hormonal balance is a critical component of women’s brain health and well-being.
The link between cognitive and emotional symptoms and hormonal decline in women is well established in the literature. Numerous studies have demonstrated that sex hormone therapy can alleviate many of these symptoms, leading to meaningful improvements in HRQOL and overall well-being. Menopausal hormone therapy (MHT), also referred to as hormone replacement therapy, has shown direct benefits on the most frequently reported clinical complaints during the menopause transition, including mood disturbances, sleep disruption, and cognitive difficulties. 12
Although there is growing recognition that menopause may negatively affect many domains of brain health, there are few studies that holistically evaluate the impact of MHT across the multidimensional aspects of brain health. The traditional medically limited scope of menopause symptoms fails to capture the complexity of women’s experiences during the menopausal transition, when cognitive complaints such as memory difficulties, reduced concentration, and mental fatigue often co-occur with mood fluctuations and disrupted sleep.3,13,14 Moreover, few studies have taken a longitudinal approach to examine how MHT may influence brain function over time across different domains.
The current pilot study seeks to address this gap by leveraging a novel combination of assessments collectively called the BrainHealth Index™ (BHI). The BHI is a multidimensional approach to measuring brain health. Unlike traditional assessments focused on identifying deficits, the BHI is designed to assess brain potential and track meaningful change (gains or losses) across time. It integrates measures across three key factors—clarity, emotional balance, and connectedness and recognizes the complex interplay across these dimensions in shaping overall brain performance. The BHI is particularly well suited to study brain health in the context of menopause and MHT in a relatively healthy population because it is sensitive to the detection of improvements and declines that may be missed by narrowly focused measures based on norms. The BHI enables precision brain health by charting each person’s unique trajectory of cognitive, emotional, and social functioning, so that it may be optimized over time. Moreover, the BHI was developed with an emphasis on capturing neuroplasticity-driven change and is scalable for remote, real-world use, making it an ideal tool for longitudinal interventions. 15
This study is among the first to use the BHI to examine brain health outcomes in postmenopausal women undergoing continuous combined MHT, offering a unique lens on how hormonal interventions may influence not just symptoms but multidimensional markers of brain performance. The primary objective of this study was to evaluate the impact of continuous combined MHT—estradiol and testosterone delivered via subcutaneous implants, and nightly oral micronized progesterone—on holistic brain health as well as on cognitive, emotional, and social aspects of brain health in postmenopausal women, using the BHI. We hypothesized that the women receiving MHT would show significant improvements, particularly between baseline (T1) and the 6-month mark (T2), and maintain improvements at the 12-month time point (T3).
Materials and Methods
This study was approved by the University of Texas at Dallas Institutional Review Board (IRB Protocol #21-409). All participants were informed about the study protocol before obtaining written informed consent.
Study population
A total of 78 women were recruited to participate in the study: 42 in the MHT group and 36 in the control (CNT) group at T1/baseline. See Table 1 for participant demographic information. In the MHT group, 23 participants completed assessments at T2/6 months. Among them, 21 completed assessments at T3/12 months. In the CNT group, 36 participants completed assessments at T2/6 months, and 15 of them completed assessments at T3/12 months.
Demographic Information (N = 78)

CNT, control; MHT, menopausal hormone therapy; SD, standard deviation.
Recruitment was performed online through the EVEXIAS Medical Center website. Postmenopausal women who were experiencing any of the following symptoms related to hormone imbalance were invited to take a screening survey (symptoms: fatigue; lethargy and lack of motivation; inability to focus and remember things; mood swings; anxiety and depression; insomnia and night sweats; stubborn weight gain, belly fat, and food cravings; frequent illnesses, hair loss, saggy, dry, aging skin, aches, joint pain, and longer recovery time from injuries; loss of libido; difficulty achieving arousal and orgasm; vaginal dryness). Those who passed the screening survey, endorsing all or some of the symptoms mentioned, were then invited to schedule a phone call to determine eligibility.
Inclusion criteria:
Postmenopausal female (determined by cessation of menstrual cycles ≥12 months with elevation of serum follicle-stimulating hormone [FSH] levels >25). Fluent English speaker. Clinically determined to require treatment with all three sex hormones (estradiol, progesterone, and testosterone). Endorsed moderate or greater psychological complaints (e.g., mood disturbance, irritability, anxiety, or mental exhaustion) on the Menopause Rating Scale (MRS) or the HRQOL, which may be associated with subjective brain health concerns during midlife. The MRS is an open-access, validated tool initially developed to assess symptoms of menopause (independent from those that are disease related), to evaluate the severity of symptoms over time, and to measure changes in reported symptoms after treatment with hormone therapies. The scale is a 5-point rating scale which allows the patient to describe the perceived severity of complaints of each item (none 0, mild 1, moderate 2, severe 3, and extremely severe 4) by checking the appropriate box. Three dimensions (subscales) of symptomatic complaints are identified: psychological, somatic, and urogenital.
Exclusion criteria:
Premenopausal (last menstrual cycle <12 months from assessment date). Not fluent in English. Unable or unwilling to receive continuous MHT with the three described sex hormones. Concurrent medications that may impact/influence cognitive function:
Selective Serotonin Reuptake Inhibitor Anxiolytics Benzodiazepines Antipsychotics Medications for sleep/insomnia, including natural remedies such as melatonin, 5-Hydroxytryptophan, or Gamma-aminobutyric acid Nonbenzo hypnotics Attention Deficit Disorder/Attention Deficit Hyperactivity Disorder medications or other stimulants
We created a control group to assess the efficacy of hormone replacement on changes in brain health indices. To accomplish this, we considered a large list of candidate controls from The BrainHealth Project who were required to be female and have no more than a single training module completed to prevent confounding with the cognitive training delivered in that study. From the candidate list, we applied propensity score matching based on age, education, and race. Using the MatchIt library in the R statistical computing language (http://cran.r-project.org), we created controls by matching propensity scores using a logit distance metric. The resulting sample of controls was well matched to the MHT sample (see Table 1).
Menopausal hormone therapy
The MHT administration was conducted by nurse practitioners at a local medical center specializing in continuous combined MHT. This study utilized subcutaneous hormone pellet implants for estradiol and testosterone replacement and oral micronized progesterone. Participants who were qualified and chose to enroll in the study underwent baseline clinical labs, hormone level assessments, and completion of the MRS assessment. The lab values used for MHT dosing calculation included a total and free testosterone level, FSH, and estradiol level. Lab panels to ascertain baseline liver and kidney function, as well as complete blood counts, were also evaluated to assess general health. A proprietary dosing calculator that assesses all datasets from the hormone lab markers, weight, and MRS scores for symptom presentation was used to calculate the dose of testosterone and estradiol for each individual patient. The standard postmenopausal dose of micronized progesterone of 200 mg orally taken at night was also administered to this treatment group. Median starting doses were 12.5 mg for estradiol pellets and 137.5 mg for testosterone pellets, with adjustments guided by both laboratory values and symptom resolution (see Supplementary Appendix). Participants remained on MHT for 12 months.
Assessment
Participant brain health was assessed using the validated BHI, utilizing online assessments, at three time points (baseline, 6 months, and 12 months) during the study. The BHI is composed of four separate scores: one composite BHI score plus three empirically derived factor scores:
The BHI can be completed in 60–70 minutes. Participants can complete it all in one sitting, or they can break it up over time (up to 2 weeks). As previously described, the BHI is a comprehensive, multifaceted metric of brain health and performance. It blends novel and well-established clinical measures that assess complex cognition, emotional well-being, social connection, purpose, and lifestyle factors. The cognitive tasks assess higher-order abilities—reasoning, mental flexibility, strategic thinking, and memory—using alternate, randomized stimuli at each time point. Surveys cover emotional well-being, quality of life, purpose, happiness, resilience, social support, physical fitness, and sleep, employing validated instruments whenever possible (see Table 2 for full measure list16–29 ).
Measures Included in the BrainHealth Index
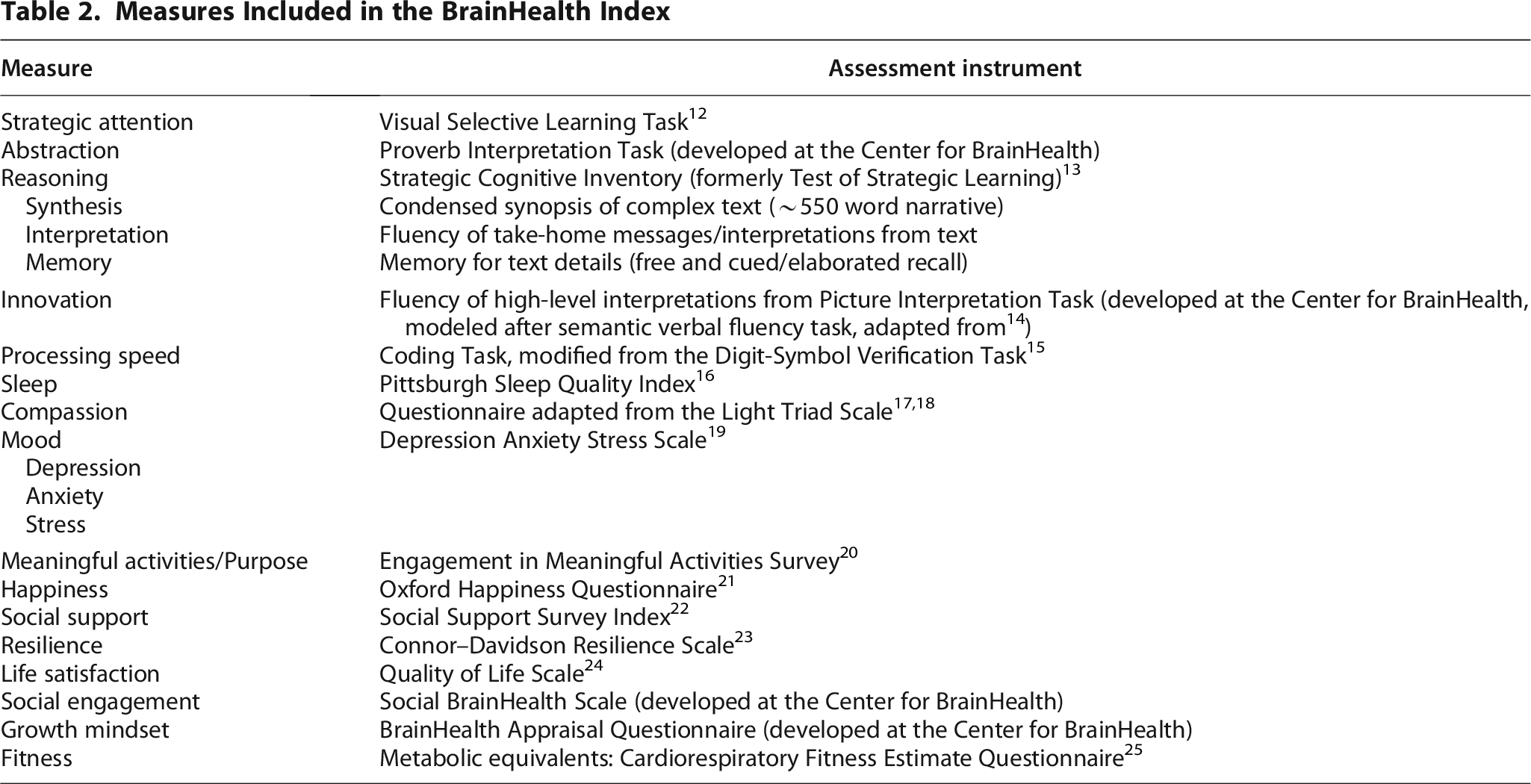
Researchers can examine isolated components of the BHI or view all components together to understand their collective influence on overall function.
Change scores on individual measures (Fig. 2) were analyzed using factor analysis, with machine learning methods used to capture how the domains interrelate rather than applying fixed weights. 30 The resulting factors, displayed in Figure 1, have been validated against brain biomarkers (predictive hemodynamic response functions) collected from a subsample who underwent functional neuroimaging alongside their BHI assessments. 31

BrainHealth Index™ Factors. Factors were derived using machine learning–based factor analysis to capture interrelated domains of function. The BrainHealth Index enables assessment of both global brain health and specific functional domains.

BrainHealth Index™ Change Scores Over Time. CNT, control; MHT, menopausal hormone therapy.
Procedure
Participants first completed a baseline BHI (T1). They had the option to complete the Index at home or could use a computer at the clinic. The BHI is accessible on computer, phone, or tablet. Next, after a comprehensive lab evaluation and consultation with the health care provider, they received their initial, personalized dose of estradiol and testosterone subcutaneous hormone insertion based on lab and symptom presentation and a standard prescription for oral micronized progesterone (see Supplementary Appendix for dosing). At 5 weeks post initial insertion, a follow-up visit was conducted to assess response to therapy. At this visit, a hormone level lab draw and MRS assessment were again performed and results compared with baseline. Adjustments were made to the participants’ dose based on post MHT lab and symptom assessment review. A follow-up visit for the next subcutaneous MHT insertion was conducted approximately 4–5 months post initial insertion. After 6 months of MHT, they were asked to complete the BHI for a second time (T2). Participants continued receiving continuous combined MHT at 4–5 month intervals and received oral micronized progesterone refills as needed to maintain continuous MHT therapy. At 12 months, they completed the BHI for the third time (T3). The control group followed the same schedule for the BrainHealth assessments at the same T1, T2, and T3.
Statistical analysis
The study was designed with the BHI, clarity, connectedness, and emotional balance as dependent variables, with group as a between-subject variable (two levels: MHT vs. CNT) and time as a within-subject variable (three levels: T1/baseline vs. T2/6 months vs. T3/12 months). Mixed-effects modeling was conducted including the effects of time, effects of group, and the interaction effect of time and group. Since measures within the same participant across time points are positively correlated, we included the error term to account for a between-subject variance and a within-subject variance. The interaction effect of time and group on the dependent variables is of our primary interest. Significance level was set at 0.05. Post hoc analysis with multiple comparisons was conducted using Bonferroni correction.
Results
The MHT group showed significantly lower scores than the CNT group at T1/baseline in all the indices, suggesting that, compared with the CNT group, symptoms in the MHT group negatively affected the cognitive, emotional, and social aspects of behavioral health (see Table 3). Additionally, there was a significant interaction between group and time on all the indices (see Table 4 and Fig. 2). Post hoc analysis showed that, compared with the CNT group, the MHT group demonstrated a significantly greater score improvement in all the measures from T1/baseline to T2/6 months. No significant differences in score changes were observed between the groups from T2/6 months to T3/12 months (see Table 5). These results suggested that the MHT intervention facilitated significant progress in the MHT group, enabling them to reach performance levels comparable to those in the CNT group between T1/baseline and T2/6 months. Subsequently, both groups exhibited similar patterns of score changes from T2/6 months to T3/12 months.
Group Differences in BrainHealth Indices at T1/Baseline

p < 0.05, **p < 0.01.
Main Effects of Time, Main Effects of Group, and the Interaction Effects of Time and Group on the BrainHealth Indices

p < 0.05, **p < 0.01, ***p < 0.001.
Score Change Differences Between Groups Across Time Intervals

p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
This pilot study sought to understand the impact of MHT on postmenopausal women’s brain health, as measured by a validated, holistic composite, the BHI. At baseline, the MHT group exhibited significantly lower scores across all four indices (holistic brain health, clarity, connectedness, and emotional balance) compared with the CNT group data from a large project using the BHI. The MHT group showed significantly greater improvements from T1 to T2 compared with the CNT group. There were no significant group differences in score changes between T2 and T3.
The lower baseline brain health scores observed in the participants reinforce the negative impact that menopausal symptoms can have on cognitive function, emotional regulation, and social engagement. These initial differences underscore the need for targeted assessments to guide interventions during this challenging period of a woman’s life. The results of this pilot study suggest that continuous, tailored MHT may offer an effective intervention to improve overall brain health, given that the majority of participants demonstrated marked improvements across multiple domains given the large effect size. The improvements from baseline (T1) to 6 months (T2) in the MHT group suggest that the intervention helped them recover to the level of age- and education-matched controls. Importantly, the ability for the participants to maintain the T1–T2 gains while continuing MHT therapy was demonstrated.
Data are lacking regarding the full impact of sex hormones on women’s brain health. While several studies have examined the role of estrogen in mood and cognition, few, if any, have explored the combined influence of estradiol, progesterone, and testosterone on cognitive and emotional functioning. In particular, data on testosterone in women is especially scarce, despite the fact that the ovaries produce this hormone and androgen receptors are widely distributed in the brain, especially in key emotional regions like the amygdala. Existing research on androgens in women has largely focused on sexual desire and reproductive health, historically minimizing their potential role in brain health. Additionally, subcutaneous pellet therapy as a delivery method for MHT in women remains under-researched, with most available studies dating back more than four decades. This study contributes new insights by examining how testosterone combined with estradiol—administered via subcutaneous pellets and in conjunction with oral progesterone—may influence cognitive function and brain health in women, addressing a critical gap in the current scientific understanding. Finally, the standard of care for MHT is a personalized approach based on clinical factors and patient preference for the hormone type, dose, administration route, and use duration.32–35 Given the heterogeneity of MHT intervention, the authors want to acknowledge that the findings from this pilot study may not generalize to other postmenopausal women who are receiving a different MHT protocol.
In addition to the implications of cognitive and behavioral benefits from MHT, a second strength of this pilot study lies in the outcomes provided by use of the BHI. The Index is a validated and multidimensional tool that offers a more nuanced approach to assessing brain health compared with traditional cognitive assessments. In short, the BHI provides a sensitive and comprehensive tool for capturing multidimensional changes over time at an individual level, thus highlighting its utility in evaluating personalized effectiveness of interventions that may affect multiple domains of brain health differently over time and in response to treatment protocols. The BHI was specifically designed to measure not only potential cognitive losses, as seen with many cognitive assessments, but also performs equally well in measuring the upward gains in cognitive, social, and emotional well-being performance. This unique design contrasts with widely used measures such as the Montreal Cognitive Assessment and the Mini-Mental State Examination, which were primarily developed as screening tools to detect cognitive deficits and typically only register significant changes once impairment is already clinically apparent.
The BHI is a validated, sensitive tool for detecting incremental improvements and subtle losses that may result from interventions like MHT. Moreover, the BHI is intentionally constructed to capture dynamic change over time, not just a static snapshot. This is particularly important given the brain’s continuous adaptation to environmental, hormonal, and lifestyle factors. By using the BHI, this study provides a more informative and actionable understanding of how MHT influences menopausal women’s brain health in everyday life. This approach not only highlights the value of viewing brain health through a broader, more proactive and sensitive lens but also sets a precedent for future research and clinical applications focused on optimizing cognitive and emotional function during menopause and beyond.
The results from this promising pilot study must be interpreted cautiously due to several limitations. As with any pilot, the relatively small sample size necessitates cautious interpretation of the findings, as the results may not be generalizable to a broader, more diverse population. Additionally, this pilot trial was nonrandomized. However, the observation that the participants demonstrated significant improvements from baseline to 6 months suggests a potential treatment-related benefit that is not likely due solely to practice effects. Notably, prior research indicates that practice effects on the BHI in randomized control groups are minimal, supporting the interpretation that these gains extend beyond improvements due to repeated testing alone. 36 Additionally, since both estradiol and testosterone were administered concurrently, the study design did not allow us to isolate the individual effects of each hormone. Given that both have been independently associated with cognitive and emotional benefits, future randomized, placebo-controlled studies with separate treatment arms of the individual components of this MHT would offer greater clarity and statistical power.
As with many studies, these preliminary data are hypothesis generating. Future studies could build on this work by comparing subcutaneous pellet therapy with other MHT modalities, such as oral or transdermal administration. Subcutaneous pellets offer steady-state hormone release, whereas oral and transdermal routes may produce fluctuating levels and are subject to first-pass metabolism, potentially affecting both efficacy and safety. These differences remain underexplored in literature and are essential to understanding the full impact of MHT on women’s brain health. A comparative approach may help determine whether the route of hormone delivery influences both clinical outcomes and adherence to treatment protocols.
Future studies may also differentiate between hormone combinations (e.g., estradiol plus progesterone vs. the addition of testosterone) to help isolate the contribution of androgens, which remain insufficiently studied in women despite their known presence and receptor distribution in the brain. To ensure meaningful comparisons, standardized use of bioidentical formulations (such as bioequivalent estradiol, micronized progesterone, and testosterone) is recommended, given that synthetic alternatives, particularly synthetic progestins, have been shown to exert different effects on mood, cognition, and cardiovascular outcomes.
Finally, incorporating objective biomarkers such as C-reactive protein, hemoglobin A1c, liver function tests, and lipid profiles, at baseline and follow-up, would strengthen the physiological characterization of MHT’s impact. Such markers may capture additional benefits of hormone therapy on inflammation, metabolic function, and cardiovascular health. Collectively, this study provides important early insights and lays the groundwork for more comprehensive, mechanistic, and longitudinal investigations into the role of sex hormone therapy in women’s brain health, extending beyond reproductive and sexual function.
Conclusions
This pilot study evaluated the impact of continuous, individualized MHT in postmenopausal women as measured by the BHI, a validated, multidimensional tool for brain health. Participants showed significant improvements in cognitive, emotional, and social well-being in the first 6 months, compared with a CNT group, and maintained those gains at 12 months. These findings underscore the importance of addressing hormonal changes as a potential key factor in supporting cognitive, social, and emotional well-being during the menopause transition. While results are promising, they are preliminary. Larger, randomized controlled studies are required to confirm these findings, further evaluate causality, and determine the utility of the BHI as an assessment tool for widespread clinical use in midlife women.
Authors’ Contributions
J.F.: Conceptualization, project administration, writing—original draft, and writing—review and editing. T.D.: Conceptualization, investigation, and writing—original draft. Z.C.: Data curation and formal analysis. E.V. and J.G.W.: Writing—review and editing. J.S.S.: Data curation, formal analysis, and methodology. S.B.C.: Conceptualization, funding acquisition, supervision, and writing—original draft and editing. All authors contributed to the revision of the article, read, and approved the final version.
Footnotes
Acknowledgments
We gratefully acknowledge Ms. Cindy Thomas and Mr. Bert Headden for their generous funding support for The Cindy Thomas Women’s BrainHealth Study, which made this research possible.
Author Disclosure Statement
T.D. is the founder, researcher, lead faculty, and Chief Science Officer at EVEXIAS Health Solutions. J.F., Z.C., E.V., J.G.W., J.S.S., and S.B.C. have nothing to declare.
Funding Information
This research was supported by funding from philanthropic donors Ms. Cindy Thomas and Mr. Bert Headden.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
