Abstract
This paper presents the swelling mechanisms of mortar bars subjected to alkali-silica reactions when confined with carbon fiber reinforced polymer (CFRP) sheets. Alkali-silica reactions are a critical concern in concrete structures because they can degrade durability, leading to cracking, disintegration, and malfunctioning of elements. A standard test protocol (ASTM C1293) is employed to examine the expansions of the mortar which contains a variable amount of reactive aggregates (i.e., replacement ratios of regular sand with rhyolite powder ranging from 0% to 100% in 25% increments, intended to control the degree of alkali-silica reactions) and is immersed in water and sodium hydroxide solutions. The properties and behavior of the CFRP and conditioned bars are assessed using supplementary tests such as particle gradation, surface microscopy, tension, and bending. The distribution of the regular sand and rhyolite particles is comparable to each other at an average difference of 5.1%. The discrepancy in moisture-related adhesion between the cement paste and rhyolite leads to the local spalling of the mortar. Upon activation of the chemical reactions, amorphous silica gels form and cover the mortar surface. The volumetric expansions of the plain mortar and CFRP-confined cases are investigated (0.45% and 0.06% at 28 days, respectively), followed by a comparative assessment against the 0.2% limit of ASTM C1260. The replacement ratio affects the flexural capacity and energy dissipation of the plain and CFRP-confined mortar bars (e.g., 8.1% and 30.3% decreases in the capacity with and without CFRP, respectively). Chemo-mechanical modeling characterizes the reactivity of the test specimens and substantiates the evolution of the silica gels and the efficacy of CFRP in mitigating detrimental alkali-silica reactions.
Keywords
Introduction
The durability of concrete is essential in built-environments. Various exposure conditions impinge upon the functionality of structural systems and result in premature failure. The mechanical and chemical attributes of concrete engage in the process of deterioration, which dominates the ultimate and serviceability limit states of the system. On many occasions, durability issues arise from outside a concrete element such as the use of deicing agents in the winter months; otherwise, the cause often emerges within the concrete itself. The constituent materials of concrete interact with one another under a certain circumstance and provoke damage. A typical mechanism is as follows: aggregates react against Portland cement and incur durability problems, depending upon the component (i.e., siliceous minerals (SiO2) for alkali-silica reactions and dolomite (CaMg(CO3)2) for alkali-carbonate reactions 1 ). Compared with alkali-carbonate reactions that are noticed in certain regions, alkali-silica reactions are prevalent around the world. 2 These deleterious reactions are also reliant upon both internal and external factors (i.e., mixture design and service conditions 1 ). Once a concrete structure is impaired by alkali-silica reactions, the probability of synergistic degradation increases when encountering other environmental surroundings. 3
Alkali-silica reactions are a slow process taking over 5 to 15 years 4 ; accordingly, accelerated testing prevails in the construction community to appraise the risk of specific concrete mixtures within a reasonable time frame and at affordable expense.1,5 For evaluating the possibility of alkali-silica reactions in concrete, standard organizations have developed test protocols1,5,6 with the aim of examining acceptance criteria, rather than determining exact deterioration amounts. 7 Several methods have been used to alleviate the repercussions of alkali-silica reactions, namely, selecting certain aggregate types, adding supplementary cementitious materials, incorporating nano particles, and blending chemicals.8–11 All these are, however, for new construction projects and limited approaches are available for existing structures. Because the complete prevention of alkali-silica reactions is not likely, 1 unexpected reactivity may occur during a concrete member’s service life. Therefore, an effective technique should be proposed to improve the sustainability of constructed civil infrastructure.
Carbon fiber reinforced polymer (CFRP) composites, outperforming other FRP types, have been employed to rehabilitate deteriorated concrete structures for decades. 12 The composite sheets are bonded to a concrete substrate using an adhesive. The majority of literature on the subject of durability has focused on the performance of CFRP-bonded concrete members under aggressive environments (e.g., freeze-thaw, 13 elevated temperatures, 14 and wet-dry 15 ). Experimental findings substantiate that CFRP sheets are a suitable material for strengthening and have a great potential for repairing environmentally degraded structures, including alkali-silica reactions. A feasible scenario is that deteriorated concrete members, expected to undergo substantial expansion and spalling due to alkali-silica reactions, can be confined with the CFRP repair system to preserve structural integrity, thereby extending the longevity of the members. However, given scarce test data, current knowledge is insufficient to implement CFRP-based rehabilitation for concrete suffering from alkali-silica reactions. Moreover, published specifications related to CFRP do not contain provisions concerning structures damaged by alkali-silica reactions.16,17 As such, it is unclear whether the state-of-the-art repair method is applicable to this particular circumstance accompanying swelling-induced stresses that may disintegrate the concrete. 18 Before developing design guidelines, a fundamental understanding is imperative on the mechanism of alkali-silica reactions with the application of CFRP.
In the present experimental research, the behavior of mortar bars subjected to variable degrees of alkali-silica reactions is investigated per ASTM C1260. 5 Notwithstanding the ideal nature with mortar bars, the ASTM approach is frequently used on account of its practicality and convenience. 19 The primary objective of the research is to examine the effectiveness of CFRP-confinement with an emphasis on precluding the volumetric expansion of the mortar. Owing to the absence of standard test methods, the ASTM C1260 5 protocol is adopted to evaluate its applicability to CFRP-confined mortar under alkali-silica reactions. In addition, ancillary testing is conducted for constituent materials and flexural loading for the conditioned bars with and without CFRP. Chemo-mechanical modeling is performed to study the material characteristics of the plain and CFRP-confined mortar bars. The quantification of acceptable risk is elaborated when alkali-silica reactions and ensuing expansions are associated with CFRP-confinement at the material level.
Experimental program
A test program is described to explore alkali-silica reactions in mortar with and without CFRP. In addition, supplementary tests are detailed with the intention of comprehending the consequences of these reactions, which deteriorate the mortar and weaken the mechanical resistance of the cementitious and retrofit systems, including the relationship between the reactions and the conformational properties of constituent particles.
Materials
Two types of fine aggregates were used (Figure 1(a)): regular sand with natural loose grains of spherical shapes and rhyolite powder comprising quartz, plagioclase, and sanidine. The rhyolite powder was reactive to alkalinity (compressive strength = 140 MPa as per the supplier). The binder of mortar was ordinary Portland cement, classified as Type I in ASTM C150.
20
To satisfy the requirements of ASTM C1260,
5
the aggregates and cement were graded by sieves (Figure 1(b)). CFRP sheets consisted of unidirectional carbon fabrics and a two-part solvent-free epoxy matrix. A hardener and a resin were mixed at a ratio of 1 to 3 by volume in order to produce the matrix (nominal tensile strength = 52 MPa and elastic modulus = 2.6 GPa) where the fabrics were impregnated and cured for 7 days at room temperature, as instructed by the producer’s application guides: the adhesive would be sufficient to provide the adequate bond of CFRP against existing concrete substrates in the field. While the manufacturer-reported glass transition temperature of the epoxy is 71°C, previous testing shows that its residual strength remained unchanged after heating up to 125°C because of the phase transition related to a conformational recovery (the rubbery state of the epoxy adhesive returned to the glassy state when the thermal distress was removed).
21
The composite CFRP sheet has the following design properties based on an equivalent fiber thickness of 0.165 mm: tensile strength = 3800 MPa, elastic modulus = 227 GPa, and rupture strain = 1.67%. Materials and test specimens: (a) fine aggregates; (b) sieves; (c) plain mortar bars (25 mm by 25 mm by 285 mm, each); (c) CFRP-confined mortar bars (25 mm by 25 mm by 285 mm, each).
Specimens
Mixture design by mass ratio.
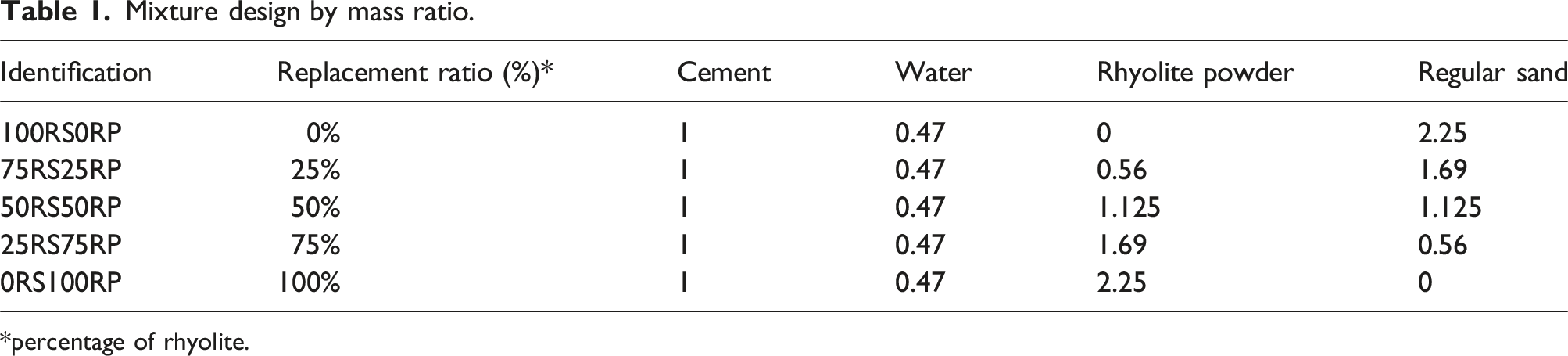
*percentage of rhyolite.
Accelerated conditioning
The test protocol specified in ASTM C1260
5
was employed for conditioning the aforementioned mortar bars and coupons (Figure 2). A sodium hydroxide solution was mixed with NaOH and distilled water (40 g and 900 mL for 1.0 L of the solution, respectively) and poured in containers where the specimens (120 bars, 12 replications for each replacement category) had been placed. It should be noted that hydrogen oxide (H2O) is a prerequisite to the occurrence of alkali-silica reactions.
23
The volume of the solution was 4.5 times the volume of the bars. According to the procedure of ASTM C1260,
5
the containers were covered and placed in an electric oven heated at 80°C. For comparison, the remaining 120 bars were submerged in water at room temperature. All specimens were then conditioned for up to 28 days, which was conservatively longer than the required period of 16 days in ASTM C1260.
5
The bars were taken out of the containers and tested immediately, then returned to the conditioning environment. Conditioning schemes: (a) immersion of plain mortar bars in sodium hydroxide solution at 80°C; (b) immersion of plain mortar bars in water at room temperature; (c) immersion of CFRP-confined mortar bars in sodium hydroxide solution at 80°C; (d) immersion of CFRP-confined mortar bars in water at room temperature.
Test methods
Particle gradation
The particle distributions of the regular sand and rhyolite powder were determined by sieve analysis. These granular materials (500 g, each) were poured through the sieves stacked in order and shaken for 10 min, then the mass of each sieve was weighted. As a supplementary approach, digital image processing was carried out. The sand and rhyolite grains were dispersed on a piece of black paper under two illumination lamps (Figure 3(a)), which eliminated shadows, and digital pictures were taken by a 24-million-pixel camera. The image files were converted to a binary format and processed using Java-based software called ImageJ.
24
Test methods: (a) particle imaging; (b) digital microscopy; (c) monotonic tension; (d) expansion of plain mortar bar (ASTM 1260); (e) expansion of CFRP-confined mortar bar (ASTM 1260); (f) three-point bending.
Surface microscopy
A high-resolution digital microscope equipped with a polarizer was utilized for qualitatively assessing the surface of the conditioned epoxy adhesive and mortar (Figure 3(b)). The CMOS (complementary metal-oxide semiconductor)-sensor-based microscope generated 1280-by-1024-pixel images at 30 frames per second. A computer with graphics software received scanned data and analyzed.
Tension
The epoxy and CFRP coupons were located in a universal testing machine and monotonically tensioned at a rate of 2.5 mm/min until failure (Figure 3(c)). A non-contacting laser extensometer measured the strain of the coupons (an initial gage length was defined by the two reflection tapes shown in the inset of Figure 3(c)). The detection range of the extensometer was 8 to 127 mm at a resolution of 0.001 mm. Five coupons were replicated for each test.
Expansion
The length change of the plain and CFRP-confined mortar bars was recorded periodically using a digital gage (Figure 3(d) and (e)). The first reading was taken after 24 h of casting based on ASTM C1293, 22 then continued until 28 days to log the persistent swelling of the specimens. Representative expansions were calculated per test category by averaging the readings of four mortar bars. ASTM C1260 states that the alkali-silica reactions of the bars are insignificant if the expansions are less than 0.1% after 16 days of casting, while the reactions are deleterious if more than 0.2%.
Three-point bending
Upon completion of the conditioning task in the sodium hydroxide solution, the bars were monotonically loaded under three-point bending with a span length of 118 mm (Figure 3(f), the 285-mm mortar bar was cut in half for convenience). The flexural test was conducted at a rate of 0.1 mm/min until the specimens failed. A load cell was mounted to log the applied load and a linear potentiometer was positioned at midspan for measuring the displacement.
Test results
Below are discussions on the physical and mechanical properties of mortar and CFRP exposed to alkali-silica reactions. Focus is given to the volumetric expansions, deterioration mechanisms, and flexural responses of the conditioned specimens.
Ancillary test
Particle distribution
Figure 4(a) exhibits a binary image for the rhyolite powder. It is seen that the particles were randomly spread over the scanned domain, which was analogous to the case of the regular sand. The observation frequencies of the sand and rhyolite are graphed in Figure 4(b) and (c), respectively. The sieve and image analyses provided similar distributions, including average absolute margins of 13.0% and 11.5% for the sand and rhyolite, respectively. While the maximum frequencies occurred at a particle size of 1.18 mm for both aggregates, the image analysis revealed that the average size of the sand was 5.1% smaller than that of the rhyolite. Such a negligible difference implies that the size of these fine aggregates would not impact on the composition of the mortar bars that were composed of variable replacement ratios. Particle gradation: (a) binary image of rhyolite powder; (b) regular sand; (c) rhyolite powder.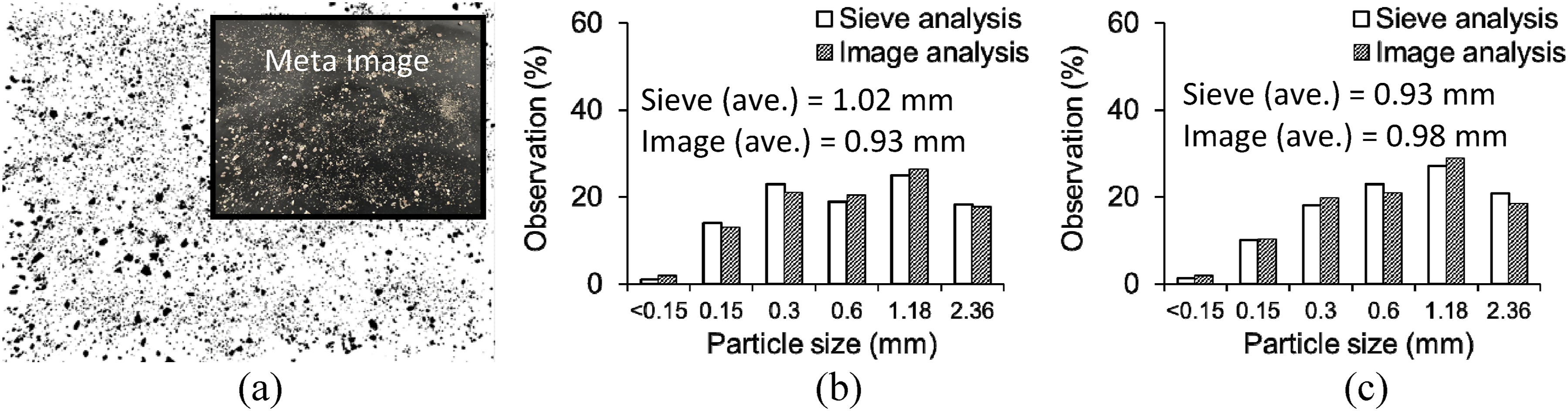
Surface degradation of mortar and epoxy
Pictured in Figure 5 is the surface morphology of the intact and conditioned mortar bars (100 times magnification), as well as of the epoxy coupons (650 times magnification). Without exposure to the sodium hydroxide solution (0 days in Figure 5(a)), alkali-silica reactions were not visible even in the 100%-replaced specimen (0RS100RP). Local spalling was, however, noticed in the 50%- and 100%-replaced cases and the extent enlarged with an increase in the replacement ratio. This observation can be explained by the fact that the adhesion of the rhyolite to the cement paste was not as good as that of the regular sand, which is important for maintaining the integrity of the mortar under the influence of alkali-silica reactions to avoid untimely disintegration of the interfacial transition zone between the paste and aggregates. When the conditioning period progressed to 14 days (Figure 5(b)), the consequences of the aggregate replacement became apparent. The amorphous byproducts from the alkali-silica reactions were deposited on the surface of the mortar; specifically, the hydroxyl ions interacted with the reactive aggregates,
25
leading to the volumetric expansion of the bars that involves the growth of internal stresses, destructive cracking, and amplified permeability. When the exposure time reached 28 days (Figure 5(c)), the byproducts fully enveloped the mortar surface. The cement paste was believed to have degraded through a silica dissolution process.
26
Dissimilar to the nearly pristine surface of the epoxy at 0 and 14 days (insets of Figure 5(a) and (b), the morphological condition of the epoxy was remarkably altered at 28 days (Figure 5(c), inset). The chemical interaction between the epoxy and sodium hydroxide discolored the exterior layers, degenerated the molecular chains of the polymeric adhesive, and softened the compositional bond.27,28 Microscopic images of mortar immersed in NaOH (100 times magnification; percentage = replacement ratio): (a) 0 days; (b) 14 days; (c) 28 days.
Properties of epoxy and CFRP
The exposure-dependent ultimate stress of the epoxy is provided in Figure 6(a). The average control capacity at 0 days was 44.7 MPa, which was lower than the nominal strength of 52 MPa. Nonetheless, the test value is still acceptable because the manufacturer’s data sheet states that the guaranteed tensile capacity of the epoxy is 30 MPa. As the exposure time in the sodium hydroxide solution increased, the ultimate stress declined at 14 days and leveled off at 28 days alongside the linear stress–strain relationship (Figure 6(a), inset). The average capacity of the conditioned epoxy coupons normalized by that of the intact coupons is given in Figure 6(b). The drops were 36.6% and 34.9% at 14 and 28 days, respectively. This fact illustrates that supplementary curing occurred in the epoxy coupons that had been saturated by the solution after 14 days; consequently, their cohesion strength increased.
29
Figure 6(c) shows the variation of CFRP capacities with the exposure period. The average 0-day capacity was 17.6% higher than the manufacturer-guaranteed strength of 3800 MPa, which is reasonable since the latter is determined by the mean capacity minus three standard deviations for design purposes. The failure of the CFRP coupon initiated in the epoxy matrix, which inhibited a proper stress distribution between adjacent fibers (Figure 6(c), inset). The normalized capacity drops of 14.8% and 26.8% at 14 and 28 days, respectively, as shown in Figure 6(d), indicate that the carbon fibers continuously resisted the applied tension after the local failure of the epoxy matrix (Figure 6(b)) and that the sodium hydroxide was a detrimental chemical in terms of degrading the CFRP capacity. Ancillary test under sodium hydroxide solution: (a) ultimate stress of epoxy; (b) average relative capacity of epoxy; (c) ultimate stress of CFRP; (d) average relative capacity of CFRP.
ASR-induced expansion
Plain mortar
Figure 7(a) and (b) compare the expansions of the plain mortar bars immersed in water and the sodium hydroxide solution, respectively. The expansions are ascribed to the reactions between the rhyolite powder and the alkali hydroxides of the mortar (i.e., Na2O and K2O in the cement).1,23 In other words, the porous silica gel, accompanied by osmosis and the differential kinetics of the ions, was responsible for the swelling of the mortar.18,30 When the specimens were immersed in water (Figure 7(a)), the expansion range at 28 days varied from 0.07% to 0.12%, which was lower than the 0.2% limit of ASTM C1260.
5
In accordance with ASTM C1778,
1
the reactivity class of R0 (Non-reactive: expansion less than 0.1% at 14 days) can be assigned. Literature reports that immersion in water eliminates soluble alkalis and tends to hinder alkali-silica reactions.
7
On the other hand, when subjected to the sodium hydroxide solution (Figure 7(b)), alkali-silica reactions were activated and the magnitude of the expansion became as high as 0.31% and 0.45% at 14 and 28 days, respectively. These values exceeded the ASTM C1260 limit of 0.2% and led to the R2 Class of ASTM C1778 (Highly reactive: expansion between 0.30% and 0.45% at 14 days). From a strength point of view, such expansions brought about the degraded bond between the cement paste and aggregates
31
; hence, the load-carrying capacity of the bars was reduced (to be elaborated in Sec. 3.3). Expansion of plain mortar bar: (a) average expansion in water; (b) average expansion in sodium hydroxide; (c) coefficient of variation (COV) in water; (d) coefficient of variation (COV) in sodium hydroxide.
Although the level of uncertainty associated with the expansions was a function of the conditioning period and replacement ratio, the activation of the alkali-silica reactions was a primary factor (Figure 7(c) and (d)). For instance, the coefficient of variation (COV) in the water-immersed mortar bars ascended from 0.037 to 0.076 (Figure 7(c)); conversely, the COV of the sodium hydroxide–conditioned bars spanned from 0.042 to 0.131 (Figure 7(d)), which was particularly noticeable for the replacement ratios of 75% and 100% (25RS75RP and 0RS100RP, respectively) after 14 days of exposure due to the increased degree of the random reactions.
CFRP-confined mortar
The expansions of the mortar bars confined with CFRP are plotted in Figure 8(a) and (b). The maximum expansion of the water-immersed bars was 0.06% at 28 days (Figure 8(a)) and the corresponding expansion of the sodium hydroxide case (Figure 8(b)) was 0.09%. These values were significantly lower than the 0.2% limit of ASTM C1260.
5
Furthermore, the CFRP-confined bars can be considered non-reactive (i.e., expansion less than 0.1% after 16 days of casting
5
), which corroborates the efficacy of CFRP-confinement in controlling the adverse effects of the alkali-silica reactions. A hypothesis is thus proposed that the volumetric changes, constrained by CFRP, align with a reduced swelling pressure around the rhyolite aggregates, which would need microscopic investigations in future research. Expansion of CFRP-confined mortar bar: (a) average expansion in water; (b) average expansion in sodium hydroxide; (c) coefficient of variation (COV) in water; (d) coefficient of variation (COV) in sodium hydroxide.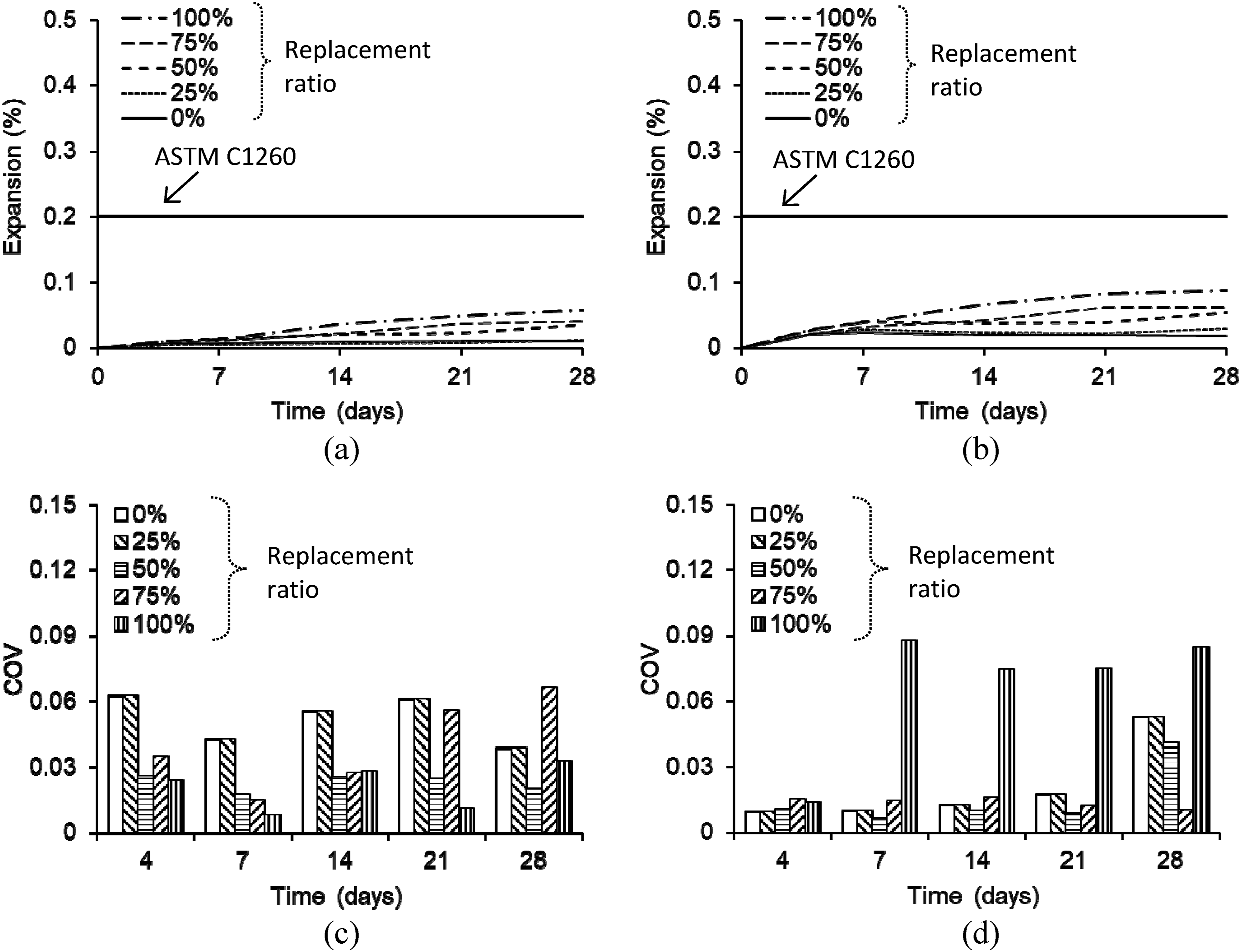
Unlike the gradually elevating coefficients of variation in the plain mortar bars (Figure 7(c) and (d)), an irregular trend was noted in the CFRP-confined bars (Figure 8(c) and (d)). This is attributable to the wet-layup process that was implemented for bonding the CFRP sheets to the bars (i.e., an epoxy layer was pasted and the precut carbon fabrics were impregnated); accordingly, the amount of the epoxy was not strictly controlled and differed in the specimens. Considering that CFRP-confinement is used to increase the load-carrying capacity of a structural member, such a minor discrepancy is not a critical factor in the rehabilitation of concrete members that have suffered from alkali-silica reactions.
Flexural capacity
The average flexural capacities of the plain mortar bars are given in Figure 9(a). The capacities of the intact bars (0 days) increased when the replacement ratio rose, which alludes that the strength of the rhyolite was greater than that of the regular sand. Although the compressive strength of the sand was not tested, its generic compressive strength is 131 MPa,
23
which is 6.4% lower than the strength of the rhyolite (140 MPa). Contrary to the case of the bars with 100% sand (100RS0RP), the capacities of the conditioned bars mixed with the rhyolite noticeably decreased and were susceptible to the exposure time; as a result, the benefit of the above-discussed initial strength disappeared. The CFRP-confined bars revealed a similar trend, as shown in Figure 9(b), whereas their variation range was narrower. Figure 9(c) and (d) further assess the gain and loss of the capacity, contingent upon the replacement ratio and exposure time. A clear divergence was observed in the ascending and descending capacities of the plain bars (Figure 9(c)), while a marginal difference was visible in the CFRP-confined bars (Figure 9(d)). These observations emphasize the significance of the CFRP-retrofit system in the context of improving the load-carrying capacity of the bars exposed to alkali-silica reactions, suggesting that the system can effectively improve the structural performance and durability of concrete members affected by these reactions. Flexural capacity of specimens immersed in sodium hydroxide solution: (a) plain mortar; (b) CFRP-confined mortar; (c) ultimate load comparison of plain mortar; (d) ultimate load comparison of CFRP-confined mortar.
Flexural response
Regardless of the replacement ratio, the load-displacement behavior of the plain bars was linear up to abrupt failure (Figure 10(a)). By contrast, the progressive bond failure between the mortar substrate and the bonded CFRP sheet was the source of the locally fluctuating responses (Figure 10(b)). Since the performance of the CFRP-retrofit system relies on the adequacy of the bond, any bond failure leads to a degradation in the system’s functionality. From a long-term durability perspective, the randomly deteriorated system can cause irregular behavior and reduce the effectiveness of the retrofit. The slippage between the supports and the smooth epoxy surface of the confined bars caused a low stiffness during the early load stage. Since the fiber direction of the confinement was perpendicular to the loading span, the failure of the confined bars was across the epoxy matrix in which the fibers were absent (Figure 10(b), inset). The flexural energy of the test specimens was calculated by numerically integrating the area under the load-displacement curves and is summarized in Figure 10(c) and (d). The energy range of the plain bars with respect to the replacement ratio was between 0.12 and 0.15 kNmm at 0 days (25.0% variation); however, their range at 28 days was between 0.06 and 0.11 kNmm (83.3% variation), as shown in Figure 10(c), which signifies the unfavorable ramifications of the alkali-silica reactions from a durability standpoint. As far as the CFRP-confined bars are concerned (Figure 10(d)), the energy variation with the replacement ratio was substantially reduced to be 19.6% and 27.6% at 0 and 28 days, respectively. Flexural response of specimen immersed in sodium hydroxide solution: (a) load-displacement of plain mortar; (b) load-displacement of CFRP-confined mortar; (c) energy comparison of plain mortar; (d) energy comparison of CFRP-confined mortar.
Chemo-mechanical investigation
Analytical modeling is conducted to elucidate the expansion mechanisms of the mortar bars with and without CFRP-confinement subjected to alkali-silica reactions. For the characterization of swelling factors, the theory of chemo-mechanics is applied, as described in this section.
Modeling
The extent of alkali-silica reactions (ξ(t)) is defined by32,33
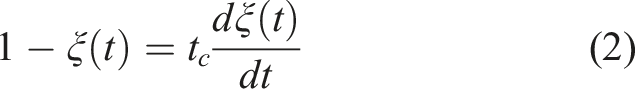
Rearranging equation (2) yields
The local imbalance (A
m
(ξ)) that induces the formation of swelling gels is expressed as
33


Implementation
Susceptibility to alkali-silica reactions
Figure 11(a) and (b) display the determined characteristic time (τ
c
) of the plain bars immersed in water and sodium hydroxide, respectively. Given that the characteristic time in equation (1) is construed to be a projected time corresponding to the maximum extent of alkali-silica reactions, an increase in τ
c
is equated with the decreased response slope of ξ(t) (i.e., more physical time is necessary to reach the full expansion strain of ε(t
∞
)). Irrespective of the conditioning method, the characteristic time rapidly declined within the replacement ratios varying from 0% to 50% in comparison with the time for ratios between 50% and 100%, which are susceptible to the alkali-silica reactions. Likewise, the characteristic time was reduced as the replacement ratio went up. The time-dependent characteristic time of the plain and CFRP-confined bars is given in Figure 11(c) and (d), respectively. The magnitude of the characteristic time considerably increased with the presence of CFRP, reaffirming the controlled expansion of the mortar bars. Determination of characteristic time: (a) plain bars immersed in water with replacement ratio; (b) plain bars immersed in sodium hydroxide with replacement ratio; (c) plain bars immersed in sodium hydroxide with exposure time; (d) CFRP-confined bars immersed in sodium hydroxide with exposure time.
Formation of silica gel
The normalized local imbalance (G(ξ)) of the plain bars immersed in water is shown in Figure 12(a). The conspicuous reduction of the imbalance from 4 to 14 days illustrates that the formation of the silica gel was active with the rhyolite content; afterward, the formation became slow between 14 and 28 days. When the plain bars were immersed in the sodium hydroxide solution (Figure 12(a), inset), the imbalance dropped owing to the prompt chemical reaction, which explains the fully-covered mortar surface pictured in Figure 5. Regarding the CFRP-confined bars, the variation range of the imbalance was within 12.9% and 19.6% for the water (Figure 12(b)) and sodium hydroxide (Figure 12(b), inset) solutions, respectively. It is, thus, stated that the CFRP sheets inhibited the formation of the silica gel. There were insignificant differences between the plain and CFRP-confined bars immersed in water (Figure 12(c) and (d), respectively); contrarily, when subjected to the sodium hydroxide solution, the controlled gel formation was apparent due to the use of CFRP (insets of Figure 12(c) and (d)). Local imbalance for formation of silica gel: (a) plain bars immersed in water with replacement ratio; (b) CFRP-confined bars immersed in water with replacement ratio; (c) plain bars immersed in water with exposure time; (d) CFRP-confined bars immersed in water with exposure time.
Extent of alkali-silica reactions
Figure 13(a) graphs the influence of the conditioning schemes in the extent of alkali-silica reactions (the ratio of the extent under sodium hydroxide to that under water is given). The exposure to sodium hydroxide was notable in the plain bars without rhyolite (0% replacement) and gradually raised the normalized extent up to 3.65, which differed from the case of the CFRP-confined bars that exhibited an almost constant value of 1.73 in all replacement ratios (not shown for brevity). The extent of the plain and CFRP-confined bars was dominated by the replacement ratio (Figure 13(b)), while their development rates were analogous when the physical time was normalized by the characteristic time (t/τ
c
), justifying the importance of the characteristic time (Figure 11) in analyzing the alkali-silica reactions. According to the plain bars subjected to sodium hydroxide (Figure 13(c)), the intrinsic reaction time (t
c
, unavailable at 0 days due to the inactivation of the alkali-silica reactions) increased with the physical time when the rhyolite was not mixed (0% replacement). In contrast, as the replacement ratio rose, the dependency of the reaction time on the physical time abated because the extent of ASR had already been sufficiently activated for the high replacement ratio cases. As shown in the inset of Figure 13(c), the relationship between the reaction and physical times was enhanced by the CFRP-confinement. Figure 13(d) clarifies the sensitivity of the reaction time. At a replacement ratio of 0%, the gap in the reaction time was large between the 4- and 28-day exposure periods, whereas the discrepancy diminished at a ratio of 100%. The tendency of the reaction time with respect to the replacement ratio in the water-immersed bars was akin to its sodium hydroxide counterpart, except for the magnitudes (Figure 13(d), inset). Extent of alkali-silica reaction (ASR): (a) comparison of plain bars between sodium hydroxide and water immersion conditions; (b) ASR extent of plain bars under sodium hydroxide with normalized time; (c) intrinsic reaction time with exposure time; (d) intrinsic reaction time with replacement ratio.
Summary and conclusions
This paper has discussed the mechanisms and consequences of alkali-silica reactions in mortar bars with and without CFRP-confinement. To simulate various levels of the reactions, regular sand was replaced by rhyolite powder from 0% to 100%. The ASTM C1293 test protocol was adopted for evaluating the expansions of the specimens at variable replacement ratios in water and sodium hydroxide solutions. Based on supplementary tests (i.e., particle gradation, surface microscopy, tension, and bending), the behavior of the conditioned mortar and the CFRP system was comprehended. Chemo-mechanical modeling accounted for the fundamental characteristics of the plain and CFRP-confined bars subjected to alkali-silica reactions. The technical findings are expected to broaden the applicability of CFRP composites in strengthening concrete structures damaged by alkali-silica reactions and to support the relevance of the composite-based repair for real-world applications. Although the outcomes of the study are meaningful, further research could address its limitations such as size-dependent swelling control, CFRP-confinement of concrete elements suffering from alkali-silica reactions, and full-scale reinforced concrete columns with CFRP. The following are concluded: • The particle distributions of the regular sand and rhyolite powder were alike, while the former was 5.1% smaller in terms of size, on average. Owing to the exposure to sodium hydroxide, the strength of the epoxy and CFRP decreased. The effects of additional curing in the epoxy after 14 days of immersion improved its cohesion strength. • Even if the water-immersed specimens did not manifest alkali-silica reactions, local spalling of the mortar was observed with the increased rhyolite replacement ratio possibly due to the adhesion discrepancy between the cement paste and rhyolite. The amorphous chemical byproducts (i.e., porous silica gels created from the reactions) covered the mortar surface and caused volumetric expansion. • The expansions of the plain specimens immersed in water were marginal (0.12% at 28 days); on the contrary, those of the specimens conditioned in the sodium hydroxide solution were significant up to 0.45% at 28 days, which exceeded the limit of ASTM C1260. The activation of the alkali-silica reactions governed the degree of uncertainty in swelling. After confinement with CFRP, the expansion of the conditioned mortar substantially decreased to 0.09% at 28 days, indicating mitigated swelling caused by alkali-silica reactions. The amount of the rhyolite dominated the flexural capacity and energy dissipation of the plain and CFRP-confined mortar bars. • As the alkali-silica reaction progressed and the replacement ratio rose, the characteristic time of the conditioned mortar decreased; however, with the presence of CFRP, the time was elevated to reduce the degree of expansion. The predicted local imbalance explained the formation mechanism of the silica gels. The relationship between the intrinsic reaction time and the physical time of the conditioned mortar was a function of the rhyolite replacement that controlled the chemical reactivity.
Footnotes
Acknowledgments
The authors would like to acknowledge support from the Fundamental Research Funds for the China Central Universities (No. 2572023CT17-05) and the US Department of Transportation through the Mountain-Plains Consortium. Proprietary information such as product names and manufacturers was not included to avoid commercialism. The technical content presented in this paper is based on the opinion of the authors and does not necessarily represent those of others.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the China Central Universities (No. 2572023CT17-05) and the US Department of Transportation through the Mountain-Plains Consortium.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
