Abstract
Using coated implant materials has been demonstrated to enhance bone regeneration and expedite healing around implant sites significantly. Generally, employing a polymeric matrix reinforced with ceramic materials has been considered a promising composite material for the coating of implants. The present study aimed to evaluate the effect of mixing varying concentrations of nano-barium titanate (nanoBaTiO3) (9, 18, and 36 wt%) to polycaprolactone (PCL) (18 wt%) on the properties of coatings applied to commercially pure titanium (CpTi) and Ti13Nb13Zr alloys implant materials. The electrospinning technique was utilised to fabricate the coatings, and the samples were characterised using atomic force microscopy (AFM) to investigate the composite coating’s surface roughness and topography; the incorporation of a high amount of BaTiO3 resulted in increased roughness of the coating layer on CpTi and Ti13Nb13Zr alloys (69.78 nm and 96.88 nm, respectively). Field emission scanning electron microscopy (FE-SEM) was used to investigate the surface morphology; the fibre diameters of BT/PCL composite were 80 to 534 nm for different mixture concentrations. Fourier transform infrared spectroscopy (FTIR) verified the chemical bonds in the composite coating. Results indicated that increasing the proportion of nano-barium titanate in the coating composition reduced water contact angles and enhanced the adhesion strength of the composite coating to the substrate. These findings provide valuable information for developing new coating materials to promote the growth of new bone and accelerate healing around implants.
Introduction
The loss of natural teeth due to oral diseases such as caries, periodontal disease, and trauma can affect the chewing system, speech, and appearance.1,2 Dental implants are a popular solution as they can replace missing teeth and restore both function and aesthetics, improving the patient's overall quality of life.3,4 The term ‘osseointegration’ was coined by the Swedish orthopaedic surgeon, Brnemark, who studied the bone growth and regeneration behaviour of titanium (Ti) implants. The process creates a strong and reliable interaction between the implant surface and the surrounding bone. The long-term success of dental implants depends on this osseointegration.5,6 The process that creates a reliable and useful interaction between the implant surface and bone is known as osseointegration. The long-term prognosis of dental implants depends on their osseointegration. 7 Long-term studies and clinical experience have shown that titanium and its alloys, particularly CpTi alloy, adapt well to the bone. As a result, titanium has become the material of choice for implants. Even though they are widely used, these so-called first-generation alloys are imperfect and might have negative effects after implantation. The major issue is the high Young’s modulus, which is over 100 GPa, compared to a normal bone’s modulus, which ranges between 15 and 30 GPa. Additionally, it has been noted that the alloying elements (Al, Fe, and V) can, to some extent, trigger toxic and allergic reactions.8–10 An innovative class of biomedical alloys, such as the Ti13Nb13Zr (TNZ) alloy, is used in medical implants. The TNZ alloy is made up of beta-alloys and has the advantage of high corrosion resistance due to its persistent passive layer, good adherence to the metallic substrate, and low rate of tissue disintegration in surrounding physiological fluids.11,12
On the other hand, in both saline and acidic conditions, the Ti13Nb13Zr alloy’s corrosion resistance was often somewhat more than or equivalent to that of CpTi. The value of Young’s modulus, one of the most crucial characteristics from the viewpoint of biomedical engineering, was also determined to be 84.1 ± 1.8 GPa for Ti13Nb13Zr alloy, which is somewhat lower than CpTi. 13
The material with good mechanical properties that is non-toxic and biocompatible is essential for biomedical applications. 14
Barium titanate (BaTiO3) is a ceramic material with piezoelectric and ferroelectric characteristics and is part of the perovskite family. The structure of its elementary cell changes with temperature. At temperatures above the Curie temperature (120°C), the cell is cubic and symmetric. However, when the temperature drops below the Curie temperature, the cell becomes slightly distorted and tetragonal. Between temperatures of 5°C and −90°C, the stable structure is orthorhombic and below −90°C, and the stable structure becomes rhombohedral.15–17 The bioceramics’ surface-to-volume ratio produces a strong bond with the bone. These materials’ high and open porosity design promotes tissue formation, allowing nutrients and osteoblasts to enter the matrix. Due to these materials’ great biocompatibility, they progressively degrade at a rate consistent with tissue growth. After a while, the tissue fills in and replaces the damaged area of the body with ceramic or composite. 18
In recent years, polycaprolactone (PCL), a biocompatible and biodegradable polyester approved by the U.S. Food and Drug Administration (FDA), has gained popularity as a biomaterial in biological tissue engineering due to its ease of accessibility, cost-effectiveness, and versatility for modification.19–21 However, due to the encapsulation by fibrous tissue, which prevents PCL from adhering to the bone, PCL implantation into bone tissue alone may not be effective. It appears that PCL has to improve its characteristics to be used as a biomaterial in biomedical applications. Therefore, different materials (such as ceramic materials) are considered to progress PCL osteoconductivity and bioactivity.22,23 Biodegradable polymers and bioactive ceramics combine to form a novel composite material with potential uses in tissue engineering, such as short-term bone implants, dental fillers, and biocompatible scaffolds. Polymers’ advantages, such as flexibility and deformability under stress, are preserved while the composite material’s strength and bioactivity are improved. 24 Particles or fillers are frequently utilised in polymeric materials to improve their mechanical properties under various loading and service situations.25–27
Composite biomaterials made of a polymeric matrix, such as polycaprolactone (PCL) or polylactic acid (PLA), reinforced with bioactive ceramic particles are well-suited for tissue engineering and bone regeneration. These composites combine the advantages of polymers, such as biodegradability, processability, and mechanical properties, with the bioactivity of the ceramic phase to more closely mimic the properties of natural bone tissue.28–31
Recently, the fabrication technology of electrospinning has gained popularity due to its ability to provide configurable building blocks for intelligent devices across various industries.32,33 Over the past two decades, electrospinning has been extensively utilised in the field of nanoscience and nanotechnology. 34 Electrospun nanofibers have demonstrated significant potential for use in various industries, including the environment, energy, pharmaceuticals, and functional materials. Consequently, there has been an increase in interest in the manufacture of nanofibers recently.35,36 Electrospinning is a powerful, simple, and efficient method for producing an ultrafine fibre from a wide range of materials, including polymer, ceramic, and composite. Two main electrospinning setups are available: vertical and horizontal setups.37–40 Polycaprolactone (PCL) has gained considerable interest among the diverse range of polymers that can be utilised for electrospinning because of its availability, non-toxicity, and stability. More significantly, the PCL electrospun fibres offer superior mechanical qualities. 41
The electrospun barium titanate/polycaprolactone composite’s characteristics as a coating material for commercially pure titanium and Ti13Zr13Nb implants that will affect the healing quality around implants have not been studied. The effect of different concentrations of barium titanate (ceramic material) to the polycaprolactone (polymer) on the coating material’s wettability and adhesion strength to the metallic substrate has not been clarified. The novelty of this work is to evaluate if increasing or decreasing the amount of barium titanate material mixed with polycaprolactone affects the performance of composite as a coating material; therefore, this study aims to investigate and characterise various concentrations of nano-barium titanate and polycaprolactone composite coating (with different concentrations 1:1 (18%–18% wt.), 1:2 (9%–18% wt.), and 2:1 (36%–18% wt.) of barium titanate: polycaprolactone on commercially pure titanium (CpTi) and Ti13Nb13Zr alloy dental implants.
Materials and method
Preparation solution of PCL and BT/PCL
Polycaprolactone (PCL) solutions of varying concentrations (10%, 14%, and 18% w/v) were prepared by dissolving PCL particles (average molecular weight of 80,000 g/mol, obtained from Sigma Aldrich, USA) in glacial acetic acid (extra pure, molecular weight of 60.05 g/mol, obtained from Thomas Baker (Chemicals) Pvt. Ltd, India). The solutions were mixed at room temperature using a magnetic stirrer overnight to ensure complete polymer dissolution. The solutions were subsequently sonicated for 30 min to further aid in the dissolution process. 42
Nano-barium titanate (N-BT) powder with a particle size of 50–70 nm and a purity of 99.9% (provided by Skyspring Nanomateraials, Inc.) was utilised to synthesise the composite coating. A series of solutions were prepared by mixing N-BT powder with polycaprolactone (PCL) in different weight ratios (1:1, 1:2, and 2:1) at room temperature using a magnetic stirrer for 4 h.
Electrospinning process
In this study, the coating is done by using the electrospinning method. The mixture was placed in a syringe of 20 mL, then the fixed parameter of the machine, including the 13 Kv voltage source after trying of (6, 8, 10, and 13 Kv), 10 cm distance of nozzle of needle (diameter 0.7 mm) from the collector with 1 mL flow rate for an hour, at first electrospinning done on aluminium foil to adjust the parameters before preparing the Ti and Ti13Nb13Zr disks and coating it.
The electrospinning process was done for 20 min after trying electrospinning at (20, 40, and 60 min) to adjust the thickness of the coating layer.
Preparation and cleaning of CpTi and Ti13Nb13Zr alloy disks
Commercially pure titanium (grade II) and Ti13Nb13Zr alloy were employed as the substrate materials in this study. The material was cut into smaller circular disks, with a thickness of 1 mm and diameter of 18 mm for Ti and 20 mm for the alloy (obtained from Baoji Jinsheng Metal Material Co, Ltd, China). The surfaces of the disks were abraded using silicon carbide paper, then polishing using a rotary technique at 250 revolutions per minute for 2 min per each abrasive step to obtain a smooth surface. The disks were then cleaned with ethanol for 15 min to remove any traces of contamination and debris. 43
Acid etching
Acid etching of the samples was performed by immersing them for 5 min in a 10% (v/v) hydrofluoric acid (HF) solution under magnetic stirring conditions. The samples were positioned such that the surface was perpendicular to the flow of the solution. After rinsing the samples with distilled water, the samples were passivated for 3 min in a 30% (v/v) nitric acid solution. Subsequently, they underwent ultrasonication for 10 min in a 50% ethanol and distilled water mixture. The etched samples were dried at 37°C for 1 h. 44
Characterisation of coating composite
Atomic force microscopy (AFM) (NanoAFM 2022, Nanosurf, Switzerland) was used to investigate the composite coating’s surface morphology, roughness, topography, and particle size distribution. The AFM measurements were performed in tapping mode with a silicon nitride cantilever with a nominal spring constant of 40 N/m. The samples were scanned in a XYZ range of [15 × 15] µm with a resolution of [1 × 1 × 0.5] nm. Data analysis and image processing were performed using the Nanosurf software, and the data were corrected for the tip shape and the substrate-induced deformation before the analysis. The results are presented as 3D height images.
Surface morphology of the CpTi and Ti alloy disks coated with (BT/PCL) beside the control disks and surface treated disks were examined by FE-SEM (inspect f50 FE-SEM; Netherland).
The chemical bonds present in the composite coating were identified using Fourier transform infrared spectroscopy (FTIR) (8400S, Shimadzu, Japan). The spectra were recorded in the mid-infrared region (400–400 cm−1) with a resolution of 4 cm−1. The spectra obtained were analysed using the OMNIC software, and the resulting absorption bands were assigned to specific chemical functional groups using a combination of literature data and the library search function of the software. The FTIR spectra were thought of as a molecular fingerprint of the sample, allowing for the identification of the chemical functional groups present in the composite coating.
The samples were subjected to phase analysis using the coating process with different concentrations of BT/PCL. Cu-K radiation (=1.5406 Ao) was used in an automated X-ray diffractometer for phase analysis. The procedure was carried out at 30 mA and 40 kV. At room temperature, diffraction patterns were captured at 10 s per angular step (1 angular step = .02o). Peak indexing was done in accordance with the Joint Committee on Powder Diffraction Standards (JCPDS) of the International Center for Diffraction Data.
Wettability test
The water contact angle of the CpTi and Ti alloy disks coated with varying concentrations of PCL/BT composite were measured and compared to uncoated control disks to evaluate the improvement in hydrophilicity of the coated samples. The measurements were performed by applying a droplet of deionised water (DW) to the surface of the samples using a small syringe and then measuring the contact angle after 30 s with a contact angle measurement tool (Si-plasma CAM 110, Creating Nano Technologies, Taiwan) connected to a digital camera. The images were analysed using the software provided by the manufacturer to determine the contact angles.
Adhesion test
All the coated specimens were subjected to adhesion testing (pull-out test) to investigate the adhesion bond between the composite and CpTi and Ti alloy disks.
Statistical analysis
The data obtained from the characterisation and mechanical tests were analysed using IBM SPSS software (ver. 23, SPSS Inc., IL, USA). Analysis of variance (ANOVA) test and least significant difference (LSD) test were used to compare the significance difference between the means of the different samples. The level of significance was set at p-values less than .05. All experimental data are presented as mean ± standard deviation (SD).
Results and discussion
The electrospinning processing variables.

The average fibre diameters related to the concentration of PCL.

Fibre diameters of BT/PCL for different concentrations of the mixture.


SEM analysis for (A) 10% PCL; (B) 14% PCL; and (C) 18% PCL.
Figure 2 demonstrates how increasing the electrospinning time increases the coating layer thickness; at 20 min, the coating thickness was approximately 5–8 µm; at 40 min, the coating thickness was approximately 77 µm. After 60 min of coating, the coating thickness is approximately 199 µm. A coating thickness over 50 µm increases the risk of debonding from the implant and might result in a partial breakdown.
51
Sopcak et al.
52
demonstrated that thinner coatings with an average thickness of less than 50 µm indicated greater adhesion strength than implants with a thickness of 200 µm. However, although extremely thin coatings cannot meet protective requirements, coating adherence improves as thickness decreases. On the other hand, it is known that thick ceramic coatings may break or crack following the deposition process.
53
Stress accumulation occurs with increased coating thickness, which encourages coating cracking or delamination. A thin covering can prematurely resorb during bone regeneration, whereas a thicker coating may cause the outer layer to separate from the implant.
54
(A) Coating thickness after 20 min of electrospinning, (B) coating thickness after 40 min of electrospinning, and (C coating thickness after 60 min of electrospinning.
The surface of CpTi and Ti alloy disks was altered by immersion in hydrofluoric acid (HF) solution as shown in Figure 3. The polished Ti disks lost their initial brightness and became opaque grey. This change in surface colour is likely a result of changes in the surface topography at the micro and nano scales, as previously observed by Lamolle et al.
55
(A) Etched Ti disk and (B) etched Ti13Nb13Zr disk.
For both CpTi and Ti alloy specimens (control and coated), surface roughness analysis was evaluated by atomic force microscopy (Figure 4). To detect the number of morphological changes on the surface, which is one of the important aspects of implant success with coating. Values of surface roughness for control, etched, and coated specimens are listed in Table 4. AFM analysis for (A) control CpTi, (B) etched CpTi, (C) Ti alloy, (D) etched Ti alloy, (E) 2:1 (BT/PCL) coated CpTi, (F) 1:1 (BT/PCL) coated CpTi, (G) 1:2 (BT/PCL) coated CpTi, (H) 2:1 (BT/PCL) coated Ti alloy, (I) 1:1 (BT/PCL) coated Ti alloy, and (J) 2:1 (BT/PCL) coated Ti alloy. Measured roughness according to AFM analysis.

Acid etching with HF acid was performed on CpTi and Ti alloy samples and increased surface roughness six times for CpTi and three times for Ti13Nb13Zr alloy. The etching process led to an increase in the surface roughness due to the loss of titanium mass and disruption of the crystal structure of the titanium. These topographic alterations have been linked to an enhancement in the biological response of the cells on the surface.44,56
Surface morphology was recognised to significantly impact cell behaviour, with pore size, pore connectivity, and surface properties having the most influence on interactions between cells and surfaces of implants, including cell adhesion, migration, and proliferation. It seemed from SEM micrographs of surfaces with and without etching (Figure 5) that the etched surface had a feature or a structure of almost nano-rods. Acid etching of Ti alloy resulted in step or ridge shaped grain boundaries.44,57 Chemically treating of Ti alloy induced the samples’ surfaces to develop a nano topography sponge-like and completely cracked shape.
58
On the other hand, Jonáová et al. explained that the etching of Ti alloy produces a micro-roughened surface.
59
Yi et al. reported a surface texture with a three-dimensional sponge-like porosity on a chemically treated CpTi surface.
60
High-magnification images of the composite-based samples (Figure 6) revealed no aggregation of the ceramic particles, indicating that the two materials were properly mixed before electrospinning and that there was no or lower difference between the surface energy of PCL and ceramic material. The main reason for the agglomeration of ceramic particles in the PCL matrix may be caused to a higher difference between the surface energy of PCL and ceramic material, and this reduced interfacial interaction between ceramic material and PCL phase; therefore, ceramic nanoparticles strongly attracted each other in the PCL matrix.
61
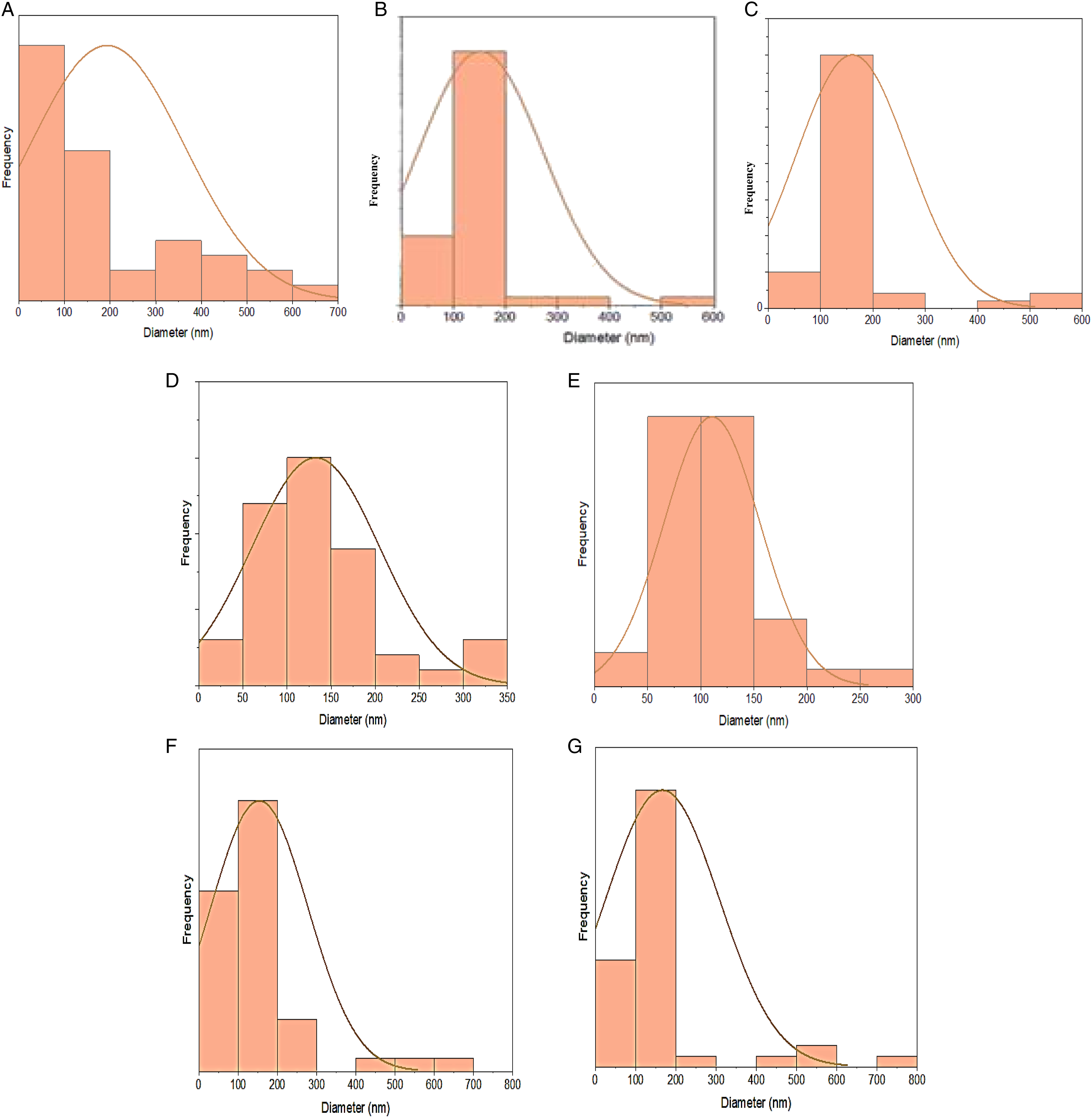
All coating groups had uniform strand diameters, with an average strand diameter of roughly 170–358 nm (Figure 7). FE-SEM analysis for (A) polished CpTi, (B) etched CpTi, (C) polished Ti13Nb13Zr, and (D) etched Ti13Nb13Zr. FE-SEM analysis for: (A) 2:1 (BT/PCL) coated CpTi, (B) 1:1 (BT/PCL) coated CpTi, (C) 1:2 (BT/PCL) coated CpTi, (D) 2:1 (BT/PCL) coated Ti alloy, (E) 1:1 (BT/PCL) coated Ti alloy, and (F) 2:1 (BT/PCL) coated Ti alloy. Fibre diameter distribution histogram determined from SEM for (A) 18% PCL; (B) 1:2 BT:PCL coated CpTi; (C) 1:1 BT:PCL coated CpTi; (D) 2:1 BT:PCL coated CpTi; (E) 1:2 BT:PCL coated Ti13Nb13Zr; (F) 1:1 BT:PCL coated Ti13Nb13Zr; and (G) 2:1 BT:PCL coated Ti13Nb13Zr.

The mixed PCL-based powders were subjected to FTIR analysis to establish the inclusion of BaTiO3 inorganic phases. As shown in Figure 8, the PCL/BaTiO3 spectra displayed the characteristic bands of PCL (Figure 8, green line); in particular, the C-H stretching at 2943 and 2865 cm−1, the C = O carbonyl group at 1722 cm−1, the CH2 deformation band between 1161 and 1471 cm−1, the C-O and C-C stretching of the crystalline phase at 1293 cm−1, and the Ti-O stretching vibration peak at 432 cm−1 and Ti-OH bond at 530 cm−1. Additionally, the spectra also displayed the particular shape of the BaTiO3 spectrum (Figure 8, purple line), which confirmed the presence of this phase in the PCL/BaTiO3 composite material; these results are in agreement with previous studies by Jin et al. (2009), Singh et al. (2017), and Porta et al. (2020).62–64 FTIR analysis for barium titanate (BT), polycaprolactone (PCL), and the mixture of BT/PCL with different concentrations.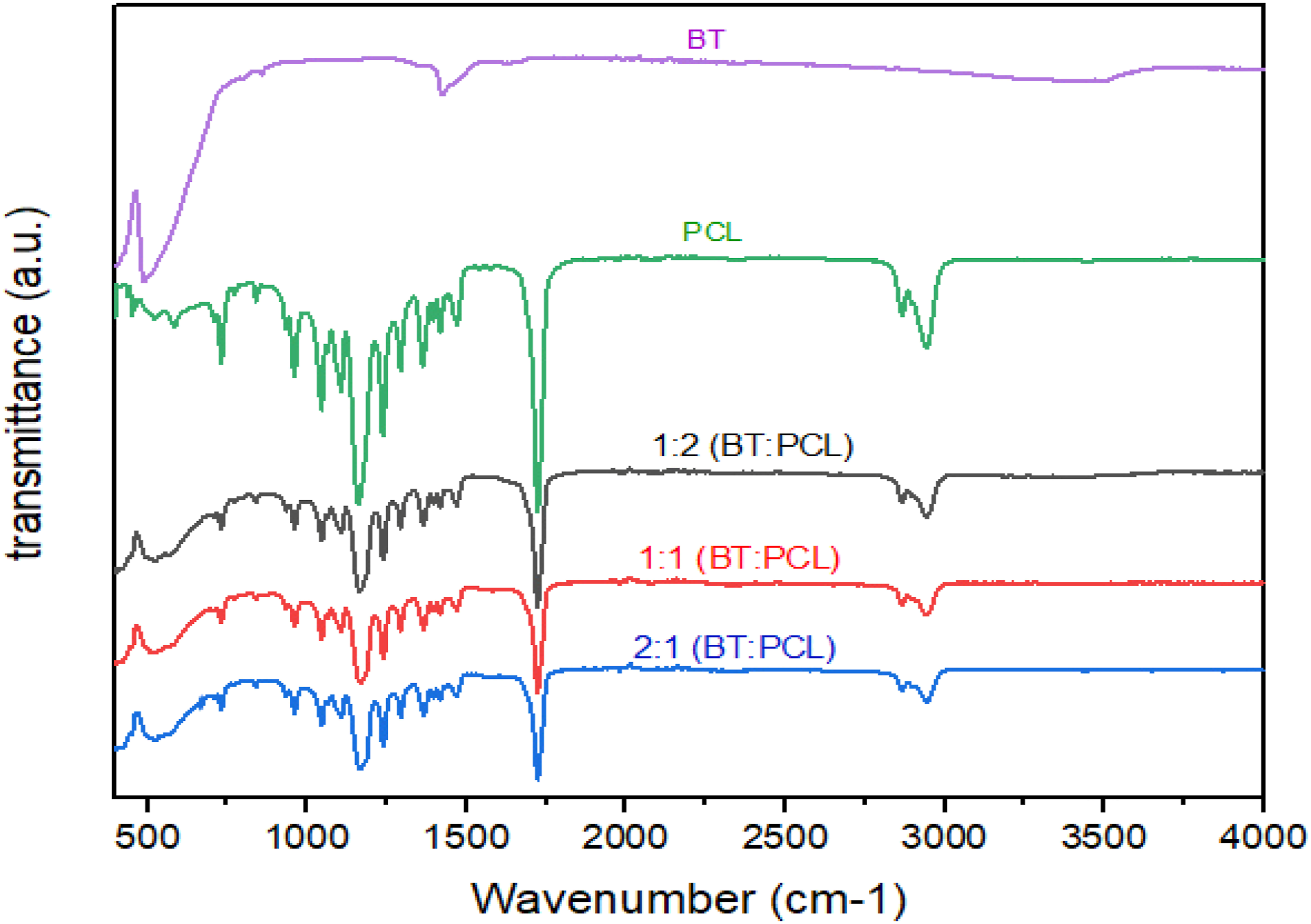
X-ray diffractometer (XRD) is a potent technique frequently used to analyse materials and explore crystalline properties, including the crystalline phases and the associated planes; in order to understand the impact of the ceramic’s integration in a polymer matrix, the XRD pattern of the BT/PCL composites with varying ceramic concentrations of 9, 18, and 36% were investigated, as shown in Figure 9. For composite-coated CpTi and Ti13Nb13Zr alloy, the Bragg reflection peak (201) at 2 = 50.9° was the most prominent feature. In the composite (JCPDS # 01-089-1428), the peaks at 2 = 31.7, 38.8, 45.4, 50.9, 62.1, 64.7, and 74.5 are assigned to the crystal phase of BT. PCL is responsible for the peaks at 2 = 21.3 and 23.6 (JCPDS # 0-1431). After the coating process, there may have been X-ray penetration through the coated layer, so there are Ti peaks in the XRD pattern.
65
The α phase of CpTi (JCPDS# 44.1294) and Ti13Nb13Zr (JCPDS # 253841 for α phase) and (JCPDS # 645545 for β phase)) alloy is represented by the diffraction peaks (100) and (101) of coated samples at two theta values (38.4 and 40.1) for CpTi coated and (38.3 and 40.08) for coated Ti13Nb13Zr alloy. The XRD diffractogram showed that the peaks of PCL decreased and widened as the BT concentration increased, whereas the peaks of BT increased, especially at 2ϴ = 50.9 for samples coated with (2:1 BT:PCL). This study’s results agree with those of a study by Moura et al.
66
and Ismail et al.
67
that contrasted different concentrations of hydroxyapatite combined with polymer material. As the concentration of ceramic increased, the XRD diffractogram revealed that the peaks of polymer reduced while the peaks of ceramic increased. Other study comparing the effects of different concentrations of TiO2 combined with PCL, the relative intensities of TiO2 increase as TiO2 content rises. Additionally, the insertion of TiO2 nanoparticles into the PCL fibrous structure caused the peak height to decease and peak widths to broaden, indicating a decrease in the crystallinity of the PCL structure.
68
XRD pattern with both the ceramic and polymer peaks suggesting the formations of the composites.
69
(A) XRD analysis of CpTi coated with different concentrations of BT/PCL, and (B) XRD analysis of CpTi coated with different concentrations of BT/PCL different concentrations.
Water contact angle images were taken for all study groups, as shown in Figure 10; the control CpTi specimens showed a water contact angle of (69.4 ± 1.8°), while the contact angle for Ti alloy was (56.8 ± 1.6°). The CpTi and Ti alloy coated with a 2:1 (BT:PCL) ratio showed the lowest water contact angle (45.7° ± 7.6 and 41.6 ± 5.4, respectively). The means of water contact angles for all groups are compared in Figure 11. The obtained values of water contact angles of other concentrations on CpTi and Ti alloy are summarised in Table 5. The equality of means was tested by the analysis of variance (ANOVA) test, as shown in Table 7. This table presents highly significant differences in wettability means between all studied groups. The least significant difference (LSD) test was used to differentiate between groups, as shown in Table 8, which manifests a highly significant difference between coated surfaces when compared with the uncoated group. In this study, a water contact angle test was performed on commercially pure titanium (CpTi) and Ti13Nb13Zr alloy samples coated with a composite material made of 18% polycaprolactone (PCL) and variable concentrations of nano-barium titanate (9, 18, and 36% w/w). The surfaces of the PCL/BT composite particles were between hydrophobic (PCL) and super hydrophilic (BaTiO3),
70
and the surface hydrophilicity steadily increased with BaTiO3 concentration which agreed with the results of Liu et al. in 2019,
71
and contradicts previous studies that reported water contact angles above 100 when different concentrations of nano-hydroxyapatite were mixed with PCL using different techniques and solvents.
72
However, this finding agrees with studies on zirconia implants that reported decreased water contact angle with increased ceramic material in the polymer coating.
73
The correlation between roughness and wettability is that increasing the surface’s roughness will increase wettability (decrease the contact angle) due to the surface’s chemistry.
74
The property of hydrophilicity has drawn more attention as a factor influencing the osseointegration of dental implants. A key element in controlling osteogenesis is the implant surface energy. Depending on the surface energy, the surfaces could be classified as hydrophilic or hydrophobic. Many plasma proteins that initiate the initial osteogenic interactions adsorb to hydrophilic surfaces. Early wound healing and the events that occur during osseointegration, which facilitates bone integration, benefit greatly from hydrophilicity.
75
Balanced protein adsorption, initial cell contact, motility, proliferation, and differentiation are all benefits of intermediate hydrophilicity (40°–70°).
76
Water contact angle for (A) control CpTi; (B) untreated Ti alloy; (C) 1:2 (BT/PCL) coated CpTi; (D) 1:1 (BT/PCL) coated CpTi; (E) 2:1 (BT/PCL) coated CpTi; (F) 1:2 (BT/PCL) coated Ti alloy; (G) 1:1 (BT/PCL) coated Ti alloy; and (H) 2:1 (BT/PCL) coated Ti alloy. Comparison of water contact angle for all studied groups. Statistical analysis of wettability test on CpTi and Ti13Nb13Zr.


Statistical analysis of adhesion test on CpTi and Ti13Nb13Zr.

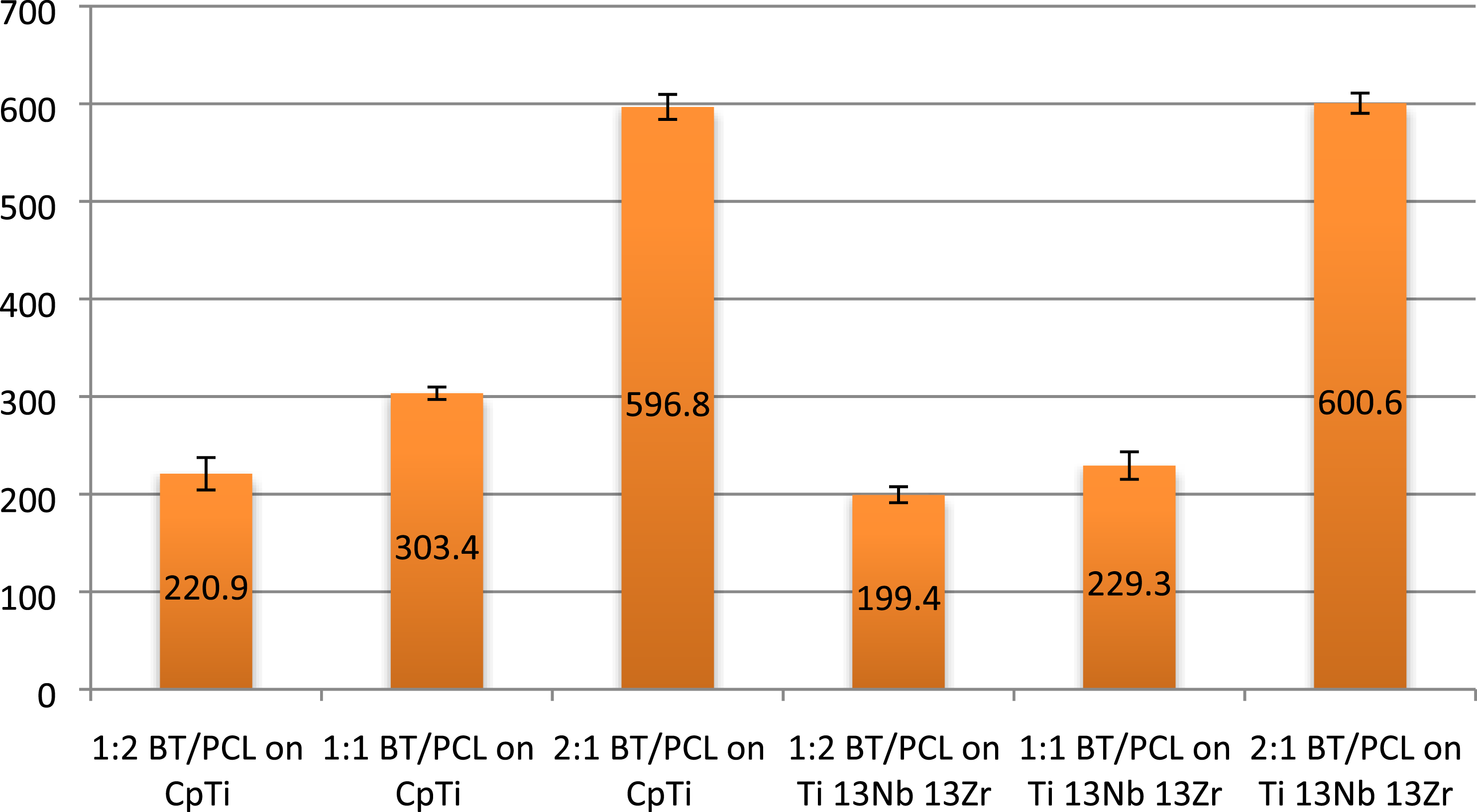
Comparison of adhesion values for all studied groups.
ANOVA test of wettability and adhesion tests on CpTi and Ti13Nb13Zr.

Multiple comparisons (LSD) among all groups.

Conclusion
In conclusion, the present study aimed to evaluate the potential effect of polycaprolactone (PCL) reinforced with different concentrations of barium titanate (BT) as a coating material on commercially pure titanium (CpTi) and Ti13Nb13Zr alloy. The study’s results indicated that the inclusion of 36% BT into 18% PCL improved the hydrophilicity performance of the coating material and increased the adhesion strength of the coating material to the substrate. These findings suggest that PCL reinforced with BT may be a promising coating material for load-bearing dental and orthopaedic implant applications. Additionally, these results are consistent with previous studies, which have shown that incorporating inorganic materials into polymeric matrices can improve the properties of the resulting composites.
Supplemental Material
Supplemental Material - Electrospun nano-barium titanate/polycaprolactone composite coatings on titanium and Ti13Nb13Zr alloy
Electrospun nano-barium titanate/polycaprolactone composite coatings on titanium and Ti13Nb13Zr alloy by Sabreen Waleed Ibrahim and Thekra Ismael Hamad in Composites and Advanced Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
