Abstract
In this paper, Ag3Sn nanoparticles were prepared by chemical method and added into SAC305 solder in different proportions to explore the effects of the Ag3Sn on the wettability, melting performance, metallographic structure, and mechanical property of the solder. The results show that a small amount of Ag3Sn nanoparticles were able to reduce the wetting time, increase the maximum wetting force, and expand the spreading area. The melting performance was also improved. Ag3Sn nanoparticles were also found to significantly refine the solder structure in the metallographical. The best comprehensive performance was achieved with the addition of 0.5% Ag3Sn nanoparticles. These Ag3Sn nanoparticles effectively improved the morphology of the IMC at the interface and effectively reduced the thickness of the IMC layer. The addition of nano Ag3Sn increased the diffusion activation energy and stabilized the interface. Moreover, the growth of the IMC layer was inhibited by 0.5% Ag3Sn, as indicated by aging results. The addition of Ag3Sn nanoparticles increases the tensile strength and Vickers hardness of the solder joint. When the amount of Ag3Sn nanoparticles was 0.5 wt%, the tensile strength reached the maximum of 17.45 MPa. The microhardness value of the solder is greatly increased, which reaches 311 HV. Finite element simulation showed that the service life of the solder was prolonged by adding the appropriate amount of Ag3Sn nanoparticles. The paper demonstrates a method of “nano” solder paste preparation. Results show that the addition of Ag3Sn nanoparticles plays an important role in the interfacial reaction and mechanical properties between the SAC solder and Cu substrate.
1. Introduction
To promote human health and the environment, electronic packaging industries have pursued the goals of lead-free solders. Sn-3.0Ag-0.5Cu (SAC305) solder demonstrates high mechanical strength, good soldering performance, and high solder joint reliability, and SAC305 solder has become the main lead-free solder 1–4
Nanomaterials materials that have at least one dimension in nanometer size (1–100 nm) or are composed of them as basic units in three-dimensional space. Due to their high potential for enhancing properties, nanoparticles can also be used to improve the performance of solder.5–6 Nanoparticles have the characteristics of small size, large specific surface area, high surface energy, and a large proportion of surface atoms.7–9
Ag3Sn IMCs usually exist in Sn xAg solder. The precipitation of nanoparticles on the surface of IMC can reduce the interface free energy and inhibit the growth of IMC. At the same time, they can prevent the disappearance of liquid-phase grain boundary channels and prevent copper atoms from flowing into the copper substrate.10–11 The overall effect of nanoparticles on the growth kinetics of IMC is still unclear. With the increase of Ag concentration, the number and size of Ag3Sn nanoparticles increased. At high silver concentration and soldering temperature, some flake and long fibrous Ag3Sn will appear on the interface.12–13
So far, there are also some achievements in the research of adding nanoparticles into solder to try to develop new solder. Chen et al. mechanically mixed TiC nanoparticles with SAC305 solder powder to prepare a SAC305 composite solder containing TiC nanoparticles. The wettability of this improved composite solder was enhanced and its melting range became larger due to the addition of the TiC nanoparticles. Moreover, the interface IMC layer was suppressed and the mechanical properties were improved. 14 Tsao et al. studied the microstructure of a composite solder prepared with TiO2 nanopowder and Sn-3.5Ag-0.5Cu, reporting that the nanoparticles affected the morphology of the IMC layer at the interface and hindered the growth of the IMC layer. 15 . Nanoparticles apparently decrease the surface energy and hinder the growth of the Cu6Sn5 IMC layer. 16 Ma et al. studied the effect of Ag3Sn nanoparticles and temperature on Cu6Sn5 IMC growth in Sn-xAg/Cu solder joints, reporting that the competition between promoting of Cu and pinning of Ag3Sn for IMC growth in specimens is analyzed. 17 X Liu et al. studied the average size of the Ag3Sn nanoparticles adsorbed on the Cu6Sn5 intermetallic compounds as a function of soldering temperature and time were studied, reporting that the adsorption of Ag3Sn particles occurs during solidification process and the number of Ag3Sn particles increases with the morphology changing of Cu6Sn5 intermetallic compounds. 18 Guo et al. studied the effect of Ag3Sn nanoparticles and temperature on Cu6Sn5 IMC growth in Sn-xAg/Cu solder joints, reporting that the blockading effect of Ag3Sn particles on Cu6Sn5 IMC growth is more pronounced in Sn-3Ag and Sn-3.5Ag alloys. 19 Nai S et al. studied varying weight fractions of multi-walled carbon nanotubes were successfully incorporated into 95.8Sn-3.5Ag-0.7Cu solder, to synthesize novel lead-free composite solders. An improvement in the mechanical properties was also recorded with the presence of increasing carbon nanotubes. 20
In this study, Ag3Sn nanoparticles were prepared by chemical method and added into SAC305 solder in different proportions to explore the effects of the Ag3Sn on the wettability, melting performance, metallographic structure, and mechanical property of the solder.
2. Experiment
Ag3Sn nanoparticles were prepared by chemical synthesis method.
21
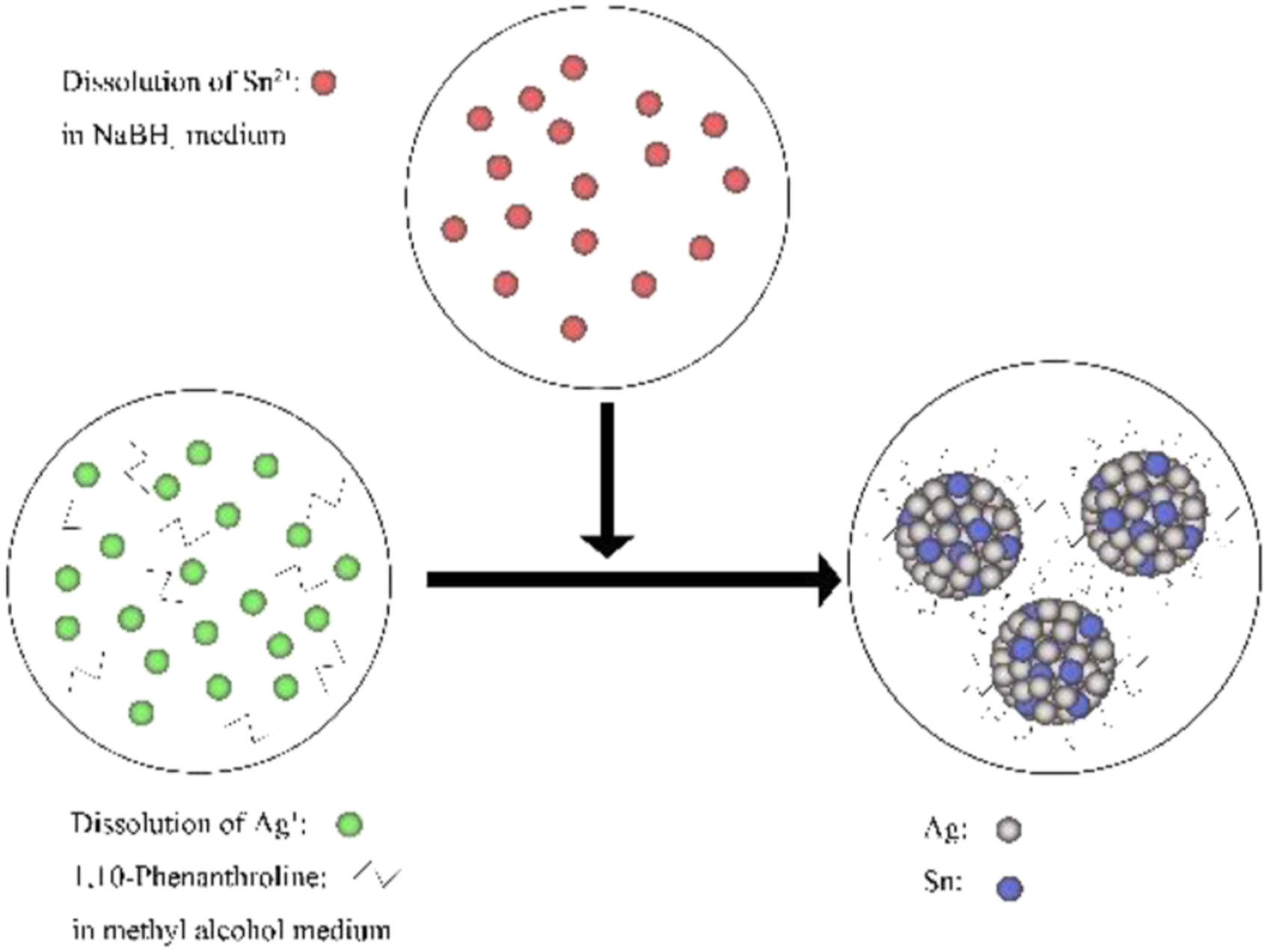
The metal cations were reduced in organic solvents to obtain intermetallic compound nanoparticles. Figure 1 shows formation mechanism of the Ag3Sn nanoparticle particles. To prepare solution A1, C16H30O4Sn, AgNO3, and 1,10-Phenanthroline monohydrate (C12H8N2·H2O) were mixed to achieve the following concentrations: n(C16H30O4Sn) = 0.01 mol/L, n(AgNO3) = 0.03 mol/L, and n(C12H8N2·H2O) = 0.025 mol/L. The solvent for solution A1 was anhydrous methanol. NaBH4 + NH3·H2O + H2O mixed solution B1 was prepared with an NH3·H2O:H2O ratio of 4:1: n(NaBH4) = 0.01 mol/L. At a reaction temperature of 40°C, solution B1 was slowly added to solution A1. This mixed solution was ultrasonicated and stirred, upon which the solution gradually shifted from yellow to colorless, black particles were generated, and a large number of bubbles were generated. After the reaction, the solution was allowed to stand for 2°h. Next, the reaction product was separated and washed by centrifugation. The obtained solid was dried in a vacuum drying oven for 8 h. The composition of the dried powder was analyzed by powder X-ray diffraction (XRD), and the morphology was observed by transmission electron microscopy (TEM). Formation mechanism of the Ag3Sn nanoparticle particles.
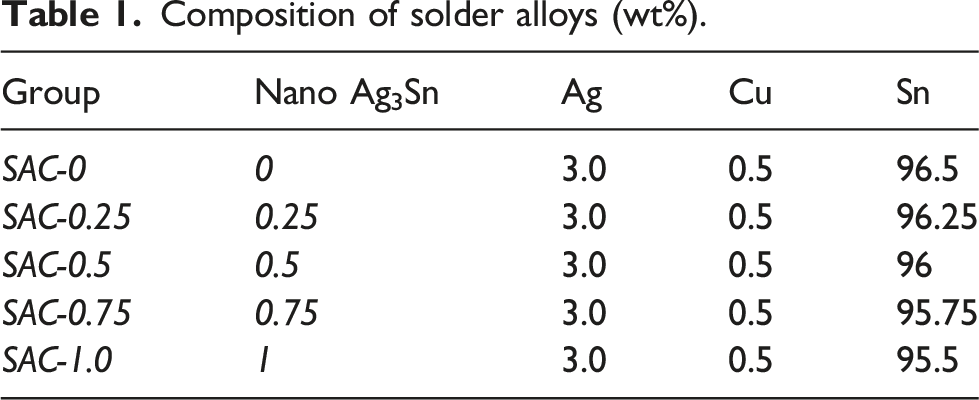
Composition of solder alloys (wt%).
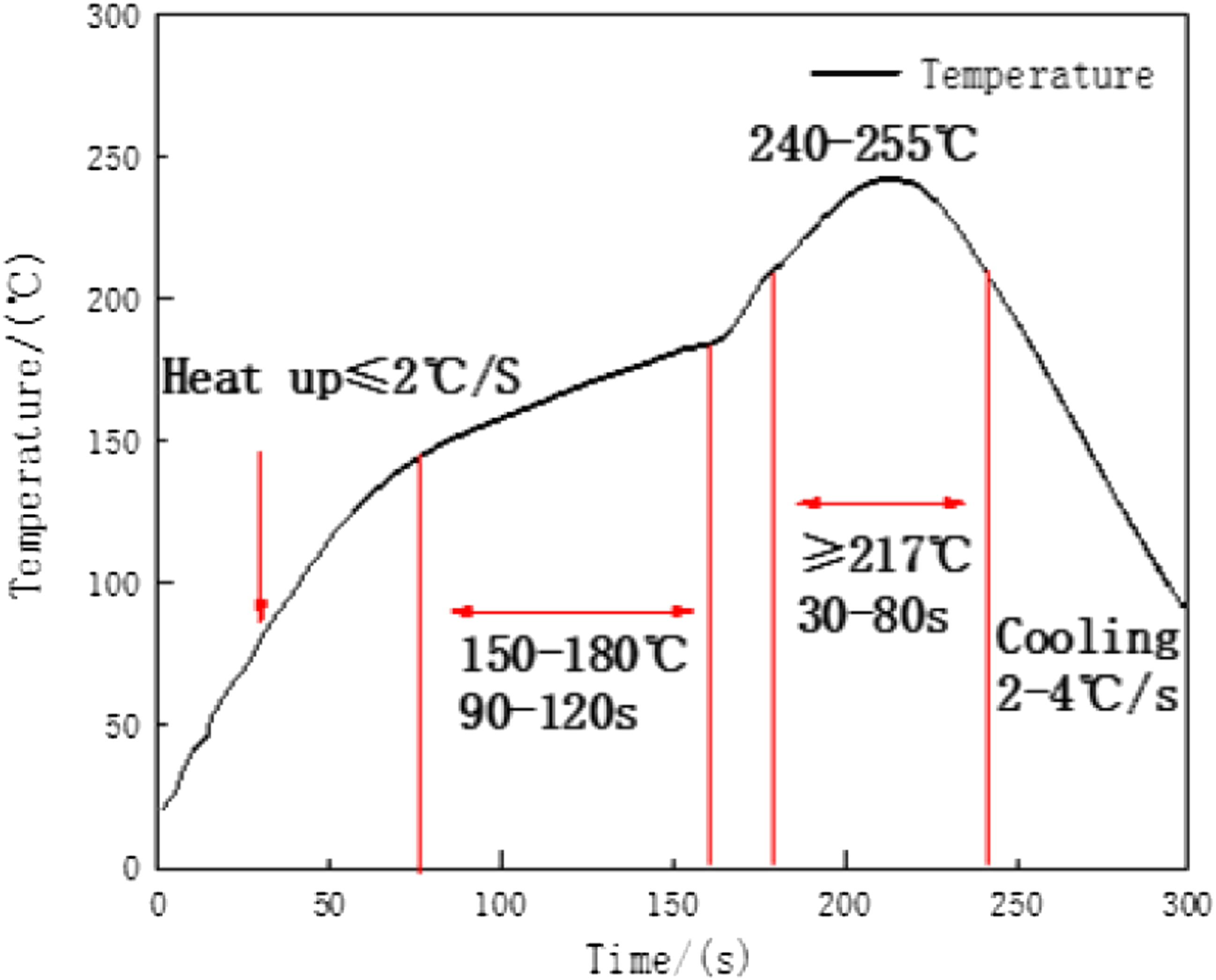
Reflow loading curve.
Wetting is a prerequisite for soldering. High-quality solder joints can only be obtained in the soldering process when there is good wettability between the solder and the substrate. The wetting and spreading ability of the prepared solder alloys was analyzed by wetting balance method. Prior to wettability test, a repolished Cu sheet (99.99% purity) was cut into square pieces (20 mm × 20 mm × 1 mm) and then ultrasonically cleaned with acetone followed by 5.0 vol% HCl acid to remove oil and oxidation. The wettability of the composite solder paste was measured by a MUST SYSTEM II weldability testing instrument (Be First Technology Co., Ltd., Beijing).
Differential scanning calorimetry (DSC) and thermogravimetric (TG) analysis were used to further investigate the composite solder paste with a DSC131ev. For each test, about 18 mg of sample was weighed and added to a crucible. The crucible was covered and loaded in the DSC instrument. Under a nitrogen atmosphere with a pressure of 0.2 MPa, the sample was heated at a temperature ramp rate of 5 K/min to 600° C, held for 10 min, and then cooled at 10 K/min to 100° C.
After cold inlaying with epoxy resin and curing agent, each solder joint was fully cured for more than 12°h. Each joint was then polished on a grinding machine with sandpaper, cleaned with absolute ethanol, dried, and corroded with a corrosive agent. Corrosive agent is 93% methanol+5% nitric acid+2% hydrochloric acid. The microstructure and IMC morphology of the solder joints were observed with an XJT-201A metallographic microscope. The soldering joints were aged for 120°h, 240°h, 360°h, and 480°h. Vickers hardness tests were done on each sample with a MH-6 digital microhardness tester with a load of 50 g (0.49 N) for 10 s. The tensile strength, elastic modulus, and other mechanical properties are tested on the REGER mechanical testing machine, and the tensile speed is set to 5 mm/min.
Finally, thermodynamic calculations and finite element simulation were used to analyze the effects of different IMC growth conditions on the quality and life of solder joints
3. Results and Discussion
The nano Ag3Sn prepared by chemical synthesis method was a black particle precipitate, and a black powder was obtained after vacuum drying. Figure 3 (a) shows the X-ray diffraction pattern of the obtained sample. Three strong diffraction peaks appeared at 2θ positions of 34.656°, 37.602°, and 39.588°, corresponding to the (201), (020), and (211) crystal planes of Ag3Sn, respectively, (PDF#71–0530). Thus, the prepared nanoparticles exhibited the Ag3Sn phase. (a) X-ray diffraction pattern of Ag3Sn nanoparticles, (b) TEM micrograph of Ag3Sn nanoparticles.
The particle size of the Ag3Sn nanoparticles was calculated by the Scherrer formula:
Grain sizes of 28.683, 29.781, and 30.617 nm were calculated using the (201), (020), and (211) peaks, respectively. The weighted average grain size was 30.127 nm.
Figure 3 (b) shows a TEM image displaying the morphology of the Ag3Sn nanoparticles. These Ag3Sn nanoparticles exhibited a relatively uniform particle size and good dispersion. The average particle size of the Ag3Sn nanoparticles was 30–40 nm.
Figure 4 shows the DSC and TG curves of the Ag3Sn nanoparticles. The DSC curve showed a strong endothermic reaction peak in the temperature range of 461.8–534.1°C, with a peak temperature of 489.9°C. Moreover, the melting point of the Ag3Sn nanoparticles was 480.1°C. The TG curve showed a weight loss of 0.45% at 40.0–101.8°C, 7.72% at 101.8–224.1°C, and 4.47% at 224.1–600.0°C. The total weight loss was 12.62%°C DSC and TG curves of Ag3Sn nanoparticles.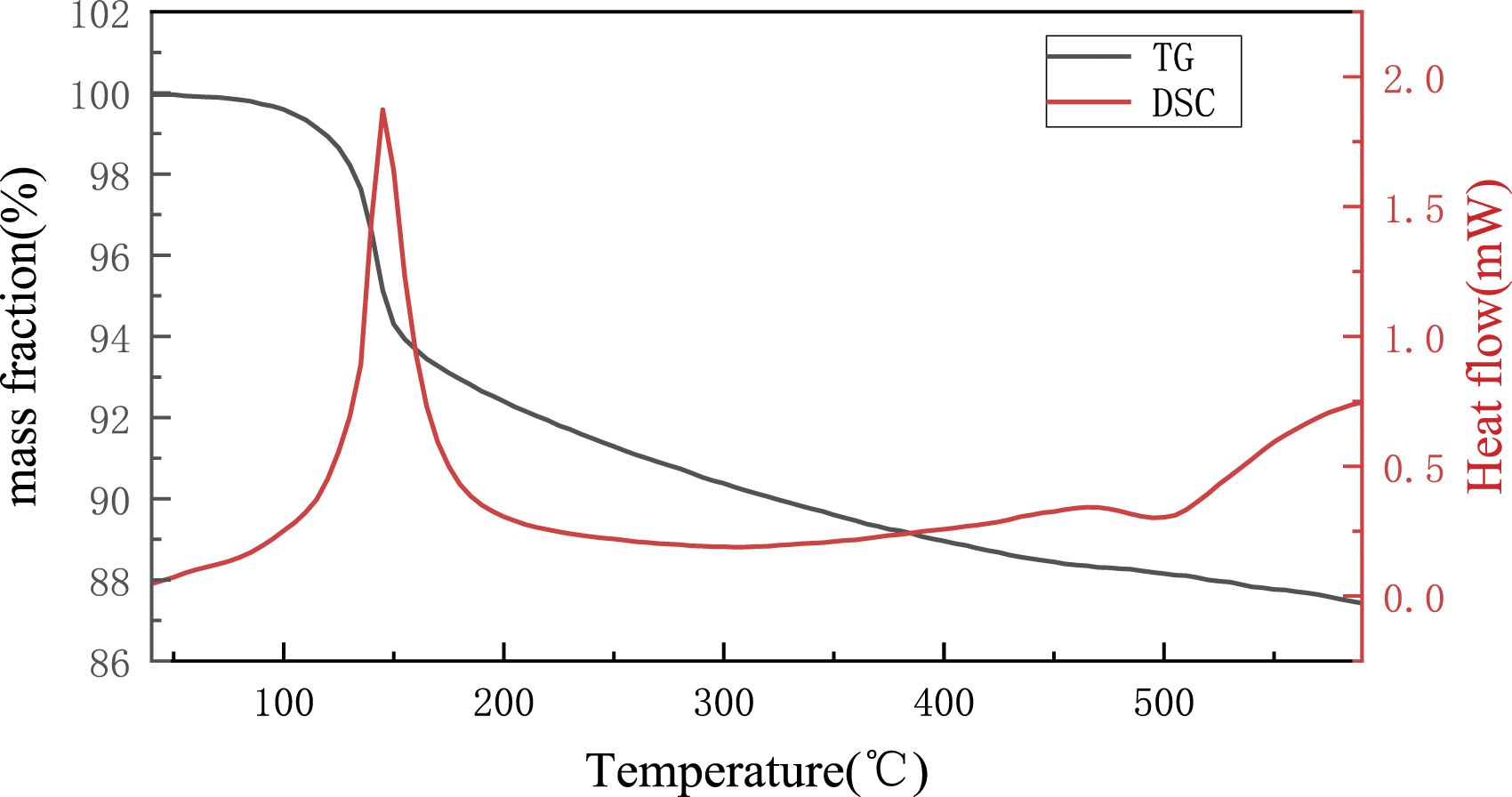
Effect of nano Ag3Sn on wettability of solder.

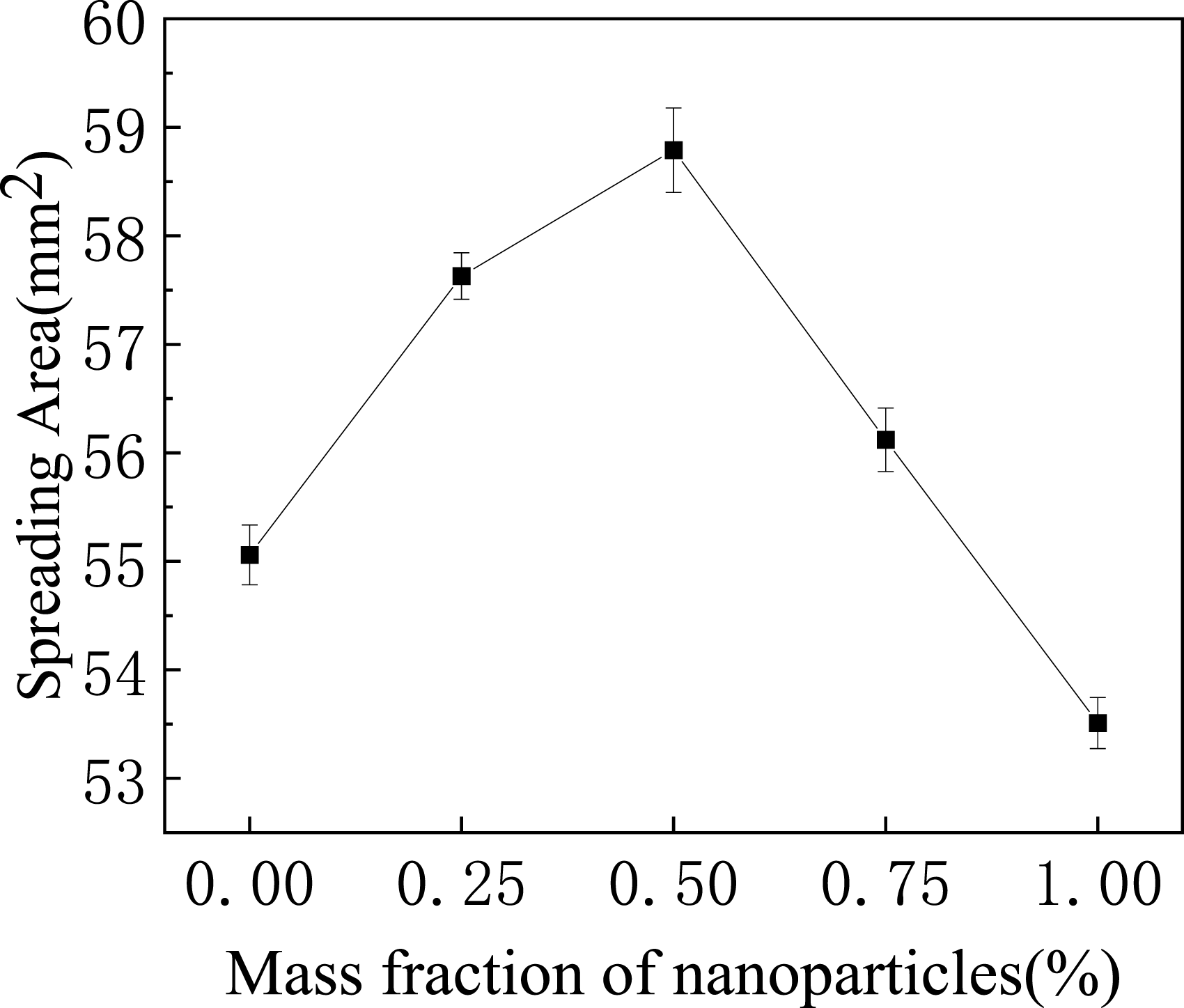
Spreading area of SAC305 solder with increasing Ag3Sn nanoparticle content.
Effect of nano Ag3Sn on melting properties of solder.

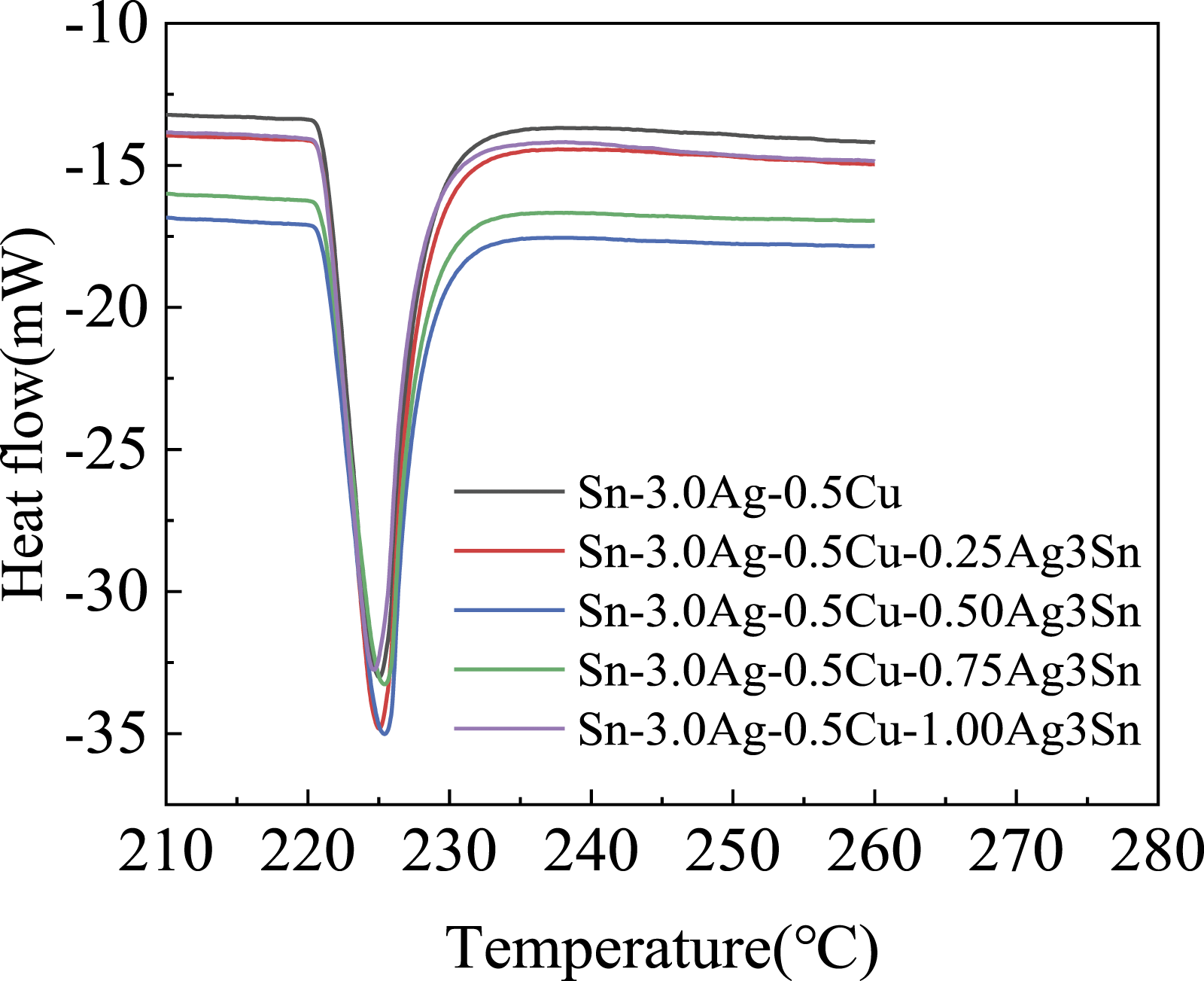
DSC curves of SAC305 prepared with different amounts of nano Ag3Sn.
Figure 7 shows the growth of the interfacial intermetallic (IMC) of Sn-3.0Ag-0.5Cu-xAg3Sn nanoparticle composite solder. The upper layer is solder, the middle layer is intermetallic, and the lower layer is the Cu substrate. Without the addition of Ag3Sn, the IMC layer showed a large size difference and different growth directions. However, with the addition of nano Ag3Sn, in 0.5% amount added the IMC layer had a flat appearance and uniform size. This shows that nano Ag3Sn enhanced the IMC morphology and refined the microstructure of the solder. The most significant solder matrix refinement was achieved with 0.5% Ag3Sn nanoparticle content. Interfacial intermetallic compound growth of SAC composite solder.
Figure 8 shows the interface line scanning analysis of SAC-0.5 solder after aging for 120°h. The IMC exhibited obvious delamination after aging, so the distribution structure and thickness of the intermetallic compounds were able to be accurately distinguished. EDX analysis showed that both Cu6Sn5 and Cu3Sn existed in the interface after aging. The diffusion activation energy of Cu atoms required for the formation of Cu6Sn5 IMC layers is very low. Therefore, the Cu6Sn5 IMC layers is often formed in the process of liquid soldering, and generally in the later solid state high-temperature aging, the atoms between the Cu6Sn5 IMC layers and the Cu substrate diffuse and react to form the Cu3Sn IMC layers. Interface line scanning analysis of SAC-0.5 composite solder after aging for 120 h.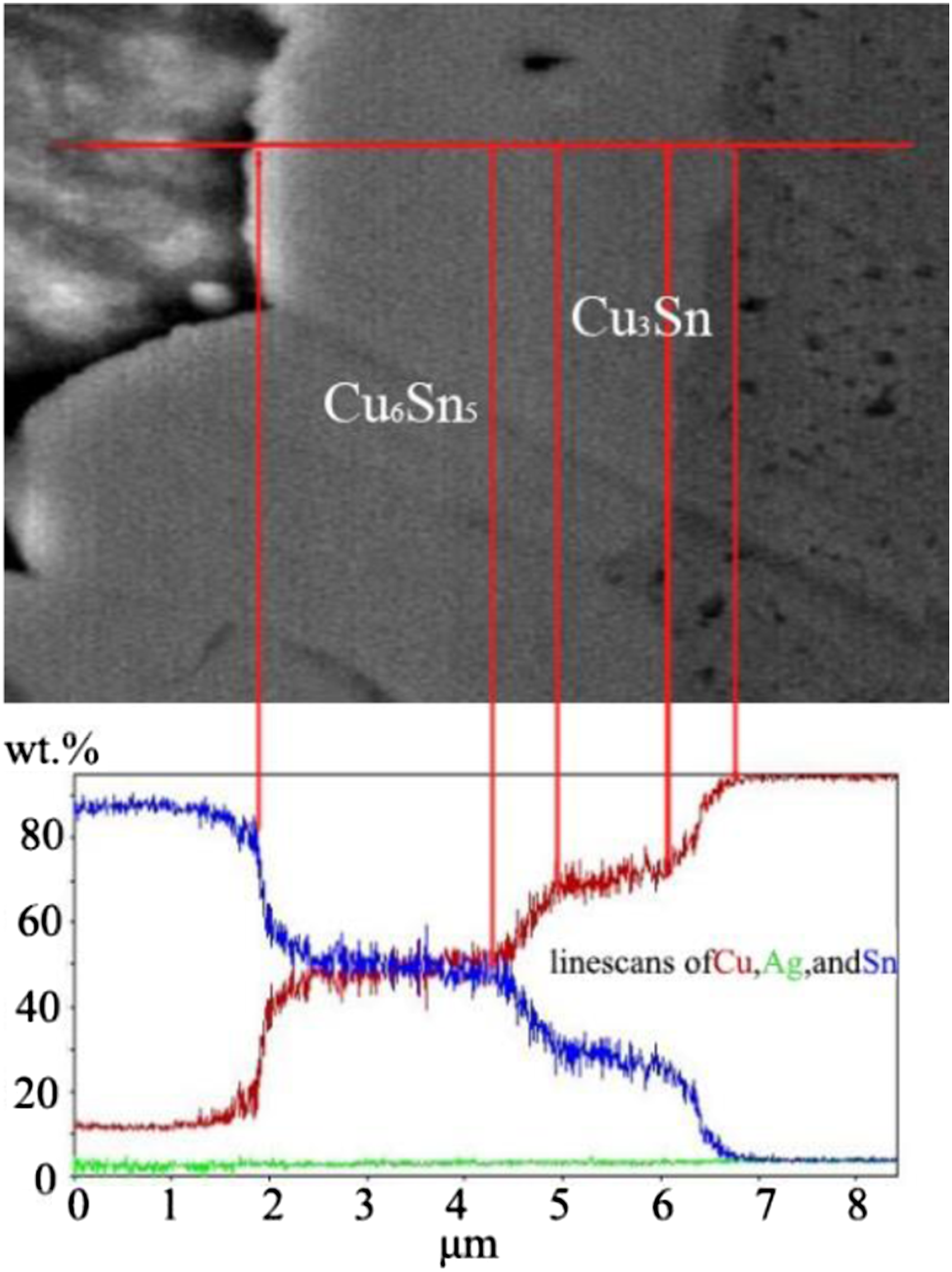
Figures 9 show the IMC morphology of the interface after aging for 120°h, 240°h, 360°h, and 480°h with different amounts of composite solder. With increasing aging time, the delamination of Cu6Sn5 and Cu3Sn became more obvious and the morphology of the IMC layer tended to be flatter. The change in IMC thickness at the interface of SAC305 for solder with different amounts of nano Ag3Sn was plotted by measuring the IMC layer thickness, as shown in Figures 10. With increasing aging time, the IMC grew and the thickness gradually increased. With increasing Ag3Sn nanoparticle concentration, the IMC thickness first decreased and then increased. This phenomenon was most significant after aging for 480°h. The addition of 0.5% Ag3Sn nanoparticles before aging reduced the thickness of the IMC by 15%, while the thickness of the IMC layer decreased by 9% after 480°h of aging. Thus, the delamination of Cu3Sn was clearly inhibited. Excessive addition of Ag3Sn nanoparticles to the SAC solder will lead to a decrease in wettability and an increase in the thickness of the interface IMC layer. The influence of the weight percentage of Ag3Sn nanoparticles may be attributed to the adsorption capacity of the solid surface. If Ag3Sn nanoparticles can be adsorbed on the surface of Cu substrate, the total interface energy of nanocomposite solder and Cu substrate system will decrease. When a small amount of Ag3Sn nanoparticles are added to the SAC solder, the nanoparticles tend to be adsorbed on the solid surface of the interface IMC or Cu substrate to minimize the surface energy. This demonstrated that Ag3Sn nanoparticles can effectively improve the stability of solder joints, providing guidance for the research and development of new solder materials. Growth of interfacial intermetallic compounds of SAC composite solder after aging. Changes in the interfacial IMC thickness of SAC305 with increasing Ag3Sn nanoparticle content (a) IMC, (b) Cu3Sn, and (c) Cu6Sn5.

The late growth of the interface between the solder alloy and Cu was controlled by diffusion, with atoms migrating under the action of the chemical potential gradient. Therefore, studying the change of the interface structure during aging from the perspective of dynamics was crucial. The relationship between the interface thickness and time can be described by the following formula 22
].
The average activation energy of intermetallic compound growth was calculated using the Dybkov model, and only diffusion was considered. Thus, the growth of the intermetallic compound layer can also be described by the following formula
The Arrhenius formula was used to study the influence of temperature and activation energy on the growth of the intermetallic compound layer.
Thermodynamic related parameters.

Greater activation energy means that a higher energy barrier needs to be overcome for the diffusion of interface elements. This means that the growth of interface intermetallics is more difficult. The addition of 0.5% Ag3Sn nanoparticles led to a small interface thickness in the initial growth process and the lowest interface growth rate in the later stage. This showed promise for increasing the service time of electronic components and meeting the requirements of high reliability for electronic component soldering.
Figure 11 shows the comparison of tensile strength and Vickers hardness of SAC solder joints with different amount of Ag3Sn nanoparticles. the ASTME415-08 standards used to conduct the mechanical properties testing. The tensile strength of the nanocomposite solder is higher than that of the solder without addition, and shows a trend of first increasing and then decreasing, as shown in Figure 11. Compared with the tensile strength (15.17 MPa) of SAC solder joints without Ag3Sn nanoparticles, the tensile strength (17.45 MPa) of composite solder joints with 0.5wt% Ag3Sn nanoparticles increased by 16.45%. However, when more than 0.5% Ag3Sn nanoparticles were added, the tensile strength decreased. In addition, the microhardness of nanocomposite solders is higher than that of alloy solders without Ag3Sn nanoparticles, and the microhardness first increases and then decreases. Among them, when the addition amount of Ag3Sn nanoparticles is 0.5%, the maximum microhardness of nanocomposite solder can reach 311 HV, which is 15.5% higher than that of SAC alloy solder (270 HV) without nanoparticles. According to the E. Orowan mechanism,
23
the non-deformable second phase particles suppress the grain boundary slip, which hinders the dislocation motion to increase the strength of the composite solder. However, when the addition amount is too large, the nanoparticles will be easier to agglomerate and transform into large particles.
24
Thus, the dispersion and strengthening effect of the second phase of nanoparticles are weakened, and the improvement degree of tensile strength and Vickers hardness is reduced. The tensile strength and Vickers hardness (HV) of SAC composite solder.
In order to better understand the fracture failure behavior, the tensile section is subject to SEM as shown in Figure 12. The fracture mode is ductile fracture, and the fracture morphology is in the form of equiaxed dimple. The fracture location of all SAC composite solders occurred in the solder after tensile test, and the equiaxed dimples in SEM were formed under tensile normal stress. The stress is evenly distributed on the whole fracture surface. The influence of different amount of nanoparticles on the solder shows that the distribution density, size, and depth of dimples are different from the appearance on the tensile fracture surface. It is not difficult to find that the addition of nanoparticles effectively reduces voids and makes dimples more dense and smaller. SEM image of fracture surface of SAC solder joint.
Materials properties of these solders used for model.


Stress nephogram of the finite element simulation of the IMC layer.
The stress and strain of the IMC layer of the solder joints were simulated under the temperature range of −40–150°C, a voltage of 38 V, and a current density of 5 × 103 A/cm2. Then, the solder joint life was estimated through the Coffin-Manson life model.
27

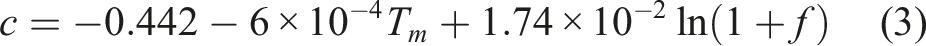
Through simulation results and theoretical formula. The service life of the composite solder with different amounts of nano Ag3Sn is shown in Figure 14. These results clearly demonstrate that the addition of a proper amount of nano Ag3Sn can effectively improve the service life of SAC305. Service life of SAC305 with increasing Ag3Sn nanoparticle content.
4. Conclusions
By studying the effect of adding different amounts of Ag3Sn nanoparticles to SAC305 lead-free solder, the following conclusions are drawn:
(1) A small amount of Ag3Sn nanoparticles can improve the wettability and melting performance of SAC305 solder. Ag3Sn nanoparticles can significantly refine the microstructure of SAC305 solder. The addition of about 0.5% can effectively reduce the thickness of the IMC layer, but higher Ag3Sn content increases the thickness of the IMC layer. After high-temperature aging, the addition of Ag3Sn nanoparticles to SAC305 leads to enhanced interface stability and a reduced IMC layer thickness, effectively improving the service life compared with Ag3Sn-free SAC30. The higher diffusion activation energy means that the interface is more stable and more reliable for long-term service.
(2) The addition of Ag3Sn nanoparticles increases the tensile strength and Vickers hardness of the solder joint. When the amount of Ag3Sn nanoparticles was 0.5 wt%, the tensile strength reached the maximum of 17.45 MPa. The microhardness value of the solder is greatly increased, which reaches 311 HV.
(3) The life of the solder joints was estimated by COMSOL finite element simulation. The results show that the service life of these solder joints can be effectively prolonged by adding 0.5% Ag3Sn nanoparticles.
Footnotes
Acknowledgements
This work was supported by the Precision Assembly Project of the Provincial Innovation Team (202105AE160028) for the Development and Application Research of Automotive Electronic High Reliability Alloys of Yunnan Tin Materials Co., Ltd., the Number of New Materials Project of the Yunnan Major Science and Technology Special Plan (202202AB08001), and the Joint Project of Basic Research and Application Basic Research Enterprises of Yunnan Tin Industry Group (holding) Co., Ltd. of the Yunnan Department of Science and Technology (202101BC070001-010).
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical statement: This material has not been published in whole or in part elsewhere. The article is not currently being considered for publication in another journal; all authors have been personally and actively involved in substantive work leading to the article, and will hold themselves jointly and individually responsible for its content.
All authors disclosed no relevant relationships.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Development and Application Research of Automotive Electronic High Reliability Alloys of Yunnan Tin Materials Co., Ltd., the Number of New Materials Project of the Yunnan Major Science and Technology Special Plan (202202AB08001), the Joint Project of Basic Research and Application Basic Research Enterprises of Yunnan Tin Industry Group (holding) Co., Ltd. of the Yunnan Department of Science and Technology (202101BC070001-010) and the Precision Assembly Project of the Provincial Innovation Team (202105AE160028).
