Abstract
Background
Human plasma-derived albumin (HA) is a critical therapeutic protein widely used in clinical applications. However, albumin aggregation remains one of the biggest challenges for plasma protein manufacturers worldwide, as it can compromise product stability, efficacy, safety, and yield. Factors that contribute to aggregation include protein concentration, ionic strength, temperature, agitation, pH fluctuations, and stabilizer concentrations during processing. This study examines how varying albumin concentrations affect aggregation under controlled pasteurisation conditions using identical parameters for complete process including agitation, temperature, and stabilizer concentration.
Methods
Albumin was purified from human plasma using a modified Cohn's fractionation method, yielding Fraction V intermediate paste. This paste underwent a series of ultrafiltration steps followed by pasteurisation at 60 ± 1 °C for 10 hours, a vital virus inactivation process. To maintain consistency, all influencing parameters such as agitation, temperature, and stabilizer concentrations were kept constant. Three experimental designs were employed: (i) analysing polymer and aggregate formation across multiple batches with albumin concentrations between 20% and 28% having similar starting material i.e. Fraction V paste at 1 kg scale, obtained from manufacturing facility from same manufacturing pool of plasma, (ii) assessing the impact of NaCl at different albumin concentrations during pasteurisation, purified from same lot of Fraction V paste at 1 kg scale, and (iii) performing a trend analysis of polymer aggregate percentage on controlled albumin concentrations (about 25%, 23%, and 21%) in a single batch started at 3 kg scale of Fraction V paste where it was divided into three parts and diluted at three different above mentioned concentrations followed by pasteurisation. Aggregation levels were quantified post-pasteurisation.
Results
The findings indicate that albumin concentrations below 23% resulted in significantly higher polymer and aggregate formation (>6.5%), whereas concentrations above 25% reduced aggregation to <. The presence of NaCl had minimal stabilizing effects, particularly at lower albumin concentrations. The trend analysis further confirmed that higher albumin concentrations mitigate heat-induced aggregation, likely due to molecular crowding effects.
Conclusion
Albumin concentration plays a crucial role in maintaining structural integrity during pasteurisation, with higher concentrations (>25%) providing enhanced stability. While NaCl contributes to stabilization, it is insufficient at lower albumin concentrations.
Keywords
Introduction
Human plasma-derived albumin (HA) is one of the most abundant proteins in human plasma having isoelectric point of 4.7–5, constituting approximately 55%–60% of the total plasma proteins. It is synthesized in the liver and plays a pivotal role in maintaining colloid osmotic pressure, transporting endogenous and exogenous compounds, and serving as an antioxidant and free radical scavenger.1,2 With its remarkable ability to bind and carry a wide variety of molecules, including hormones, fatty acids, metal ions, and drugs, HA is essential for maintaining physiological homeostasis.3,4
HA is extensively used as a therapeutic protein in clinical settings. Its primary indications include hypovolemia, hypoalbuminemia, burns, septic shock, acute liver failure, nephrotic syndrome, and as a plasma expander during surgeries.5,6 It is also utilized in extracorporeal therapies, such as therapeutic plasmapheresis and dialysis, due to its ability to restore plasma volume and bind toxins.7,8 The widespread use of HA in critical care emphasizes the importance of ensuring its safety, stability, and efficacy during manufacturing and storage.
HA is manufactured using cold ethanol fractionation, a well-established method for plasma protein separation, first developed by Cohn et al. 9 This process isolates albumin primarily from Fraction V, eliminating other plasma proteins such as fibrinogen, immunoglobulins, and coagulation factors. 10 Following fractionation, the albumin-containing fraction undergoes ultrafiltration and diafiltration to remove residual ethanol, salts, and low-molecular-weight impurities, ensuring high-purity albumin. 11 The pH is adjusted to physiological levels using sodium hydroxide, optimizing albumin stability. 12 To ensure viral safety, the purified albumin solution undergoes pasteurisation at 60 ± 1°C for 10 hours, a validated method for virus inactivation while preserving protein integrity.13,14 To maintain stability during heat treatment, sodium caprylate and N-acetyl tryptophan are added as stabilizers at the concentration of 0.08 mmol/L, preventing denaturation. 15 Sodium chloride is used as an isotonic agent, ensuring physiological compatibility.
While pasteurisation process ensures microbiological safety, it also presents a significant challenge: the tendency of albumin molecules to aggregate under high-temperature conditions.16,17 In the context of human albumin, aggregation during pasteurisation is primarily driven by:
Thermal unfolding of albumin or contaminating plasma proteins (eg, haptoglobin, transferrin). Exposure of hydrophobic regions, which tend to associate via hydrophobic or van der Waals forces.
Which leads to intermolecular interactions and the formation of soluble and insoluble aggregates. 18 These aggregates compromise the structural integrity, functionality, and therapeutic efficacy of the protein. Protein aggregation during pasteurisation not only affects product quality but also poses immunogenic risks to patients. The presence of aggregates has been linked to adverse immune responses, including the formation of anti-drug antibodies, which can neutralize the therapeutic effects of albumin and cause hypersensitivity reactions.19,20 Protein aggregation is a critical concern in the development of biotherapeutics. Aggregates can reduce product stability, increase viscosity, and affect the overall shelf life of the product. More importantly, aggregates have been shown to induce immunogenicity, which can result in neutralizing antibodies and immune-mediated side effects in patients. 21 Therefore, controlling and minimizing aggregation during the production of HA is crucial for ensuring its clinical safety and effectiveness. Recent advances in protein biophysics have highlighted the significant role of molecular crowding in stabilizing protein structures during thermal stress. Molecular crowding refers to the phenomenon where high concentrations of macromolecules occupy the cellular or formulation volume, thereby limiting the available free space for protein unfolding or aggregation. Christiansen et al. (2023) demonstrated that crowded environments slow down protein diffusion, stabilize native conformations, and inhibit aggregation by reducing effective collision rates. 22 HA is widely recognized for its protein-stabilizing properties, making it a crucial excipient in various biopharmaceutical formulations. One of the key factors influencing its stabilizing behaviour is its concentration, which plays a significant role in modulating aggregation dynamics during pasteurisation. Given this premise, the present study was initiated to investigate the relationship between albumin concentration and aggregation behaviour under heat-treatment conditions.
Recent studies reinforce the importance of maintaining high concentrations of human plasma–derived albumin to mitigate aggregation during processing. Volmer et al. (2023) demonstrated that glycation of albumin significantly alters its structural integrity, underlining how albumin's conformational sensitivity plays a role in aggregation behaviour during formulation and pasteurisation conditions. 23 In line with this, a 2025 study revealed that human serum albumin (HSA) can actively bind and dissociate amyloid aggregates in a concentration-dependent manner, confirming its inherent anti-aggregation capability at elevated levels. 24
In this study, we conducted a trend analysis based on multiple experimental trials, systematically varying albumin. concentration while maintaining a consistent pasteurisation process. For these experiments, the starting material for albumin purification was Process Intermediate Fraction V paste, obtained through cold ethanol fractionation. By evaluating aggregation patterns under different concentrations, this research aims to provide valuable insights into the impact of albumin concentration on product stability during thermal processing.
Equipments and Materials
Equipments
Plasma fractionation was conducted at the manufacturing facility using stainless steel (SS) vessels procured from DDE. A plate-frame filter press was obtained from Microfilt, while centrifugation was performed using equipment from GEA Westfalia. pH and conductivity measurements were carried out using a meter from Thermo Scientific.
Downstream processing was performed in the R&D laboratory, utilizing a peristaltic pump from Masterflex. Stainless steel vessels with a capacity of 20 L were also sourced from DDE. Tangential flow filtration (TFF) was conducted using a system and cassettes procured from Sartorius Stedim (Germany), HPLC system for polymer and aggregate estimation was procured from Agilent Technologies (USA). Additional analytical measurements were taken using a pH and conductivity meter from Mettler Toledo. A water bath from Being Lab (USA) was employed for temperature control during the process.
Materials
For Downstream processing starting material is Fraction V paste which was obtained from manufacturing facility of Plasmagen Bioscience from a single manufacturing batch so that for all the experiments same quality of paste can be used. The batch size of Fraction V paste employed in this study was determined based on operational feasibility factors, including the minimum achievable volume through Tangential Flow Filtration (TFF) and the quantity required for analytical characterisation. A formal statistical power analysis was not conducted, as the primary objective of the study was process development and product characterisation rather than hypothesis-driven statistical evaluation. This is noted as a limitation in this study.
Whereas Chemicals required for buffer preparation and stabilization were supplied by Merck (Darmstadt, Germany). Depth filters and 0.2µ filters were obtained from Sartorius Stedim (Germany). For analytical procedures, the TSKgel HPLC column was obtained from Tosoh Biosciences (USA). Total protein estimation was performed using the LIQUIXX Total Protein kit (Biuret method) from ERBA Mannheim (GmbH, Germany).
Procedure
Plasma Fractionation
The plasma fractionation process used in this study was developed at Plasmagen R&D and is a modification of the traditional Cohn fractionation method. Plasma proteins are separated based on their distinct isoelectric points, which result in differing solubilities. The solubility of proteins in this system is influenced by four primary variables: pH, salt/solvent concentration, temperature, and protein concentration. The ethanol concentration conditions range from an initial value of zero to 40%, and the pH gradually shifts from alkaline to more acidic, reaching 5.2 during the fractionation process.
Fractionation batch was conducted at the Plasmagen Biosciences manufacturing facility, where Fraction V intermediate paste was separated. This paste serves as the starting material for downstream processing of Albumin. Single fractionation batch was carried out for Albumin processing for various experimental trials to minimize variations caused by the source material.
Downstream Processing
Figure 1 presents a schematic representation of the albumin purification process. Fraction V paste, obtained during plasma fractionation, serves as the starting material to produce human albumin. The Fraction V paste was dissolved in dissolution buffer at least for 3 h, followed by depth filtration and subsequent 0.2 µm filtration. Pre-concentration was performed to reduce the volume of the albumin drug substance, followed by pH adjustment to 6.80 ± 0.1 using pH adjustment buffer. After pH adjustment, diafiltration was carried out using Diafiltration buffer to remove ethanol and other ions. The final concentration of the albumin drug substance was achieved through continued filtration until the protein content reached the desired level. Following concentration, stabilizers (sodium caprylate and N-acetyl tryptophan) were added in proportion to the protein content. The solution was then subjected to pasteurisation at 60 ± 1 °C for 10 hours. Finally, the albumin drug substance was filtered through a 0.2 µm filter before being stored.

Schematic of the HA Downstream processing.
The aforementioned process was employed for the purification of HA in all experimental trials to assess the impact of concentration on polymer and aggregate formation during the pasteurisation process. All the experimental trials were conducted by trained technicians and supervisors. In all cases, the starting material, namely Fraction V paste along with raw materials, process parameters, and process conditions remained consistent (inclusion criteria: Fraction V paste used for all experiments was sourced from a qualified manufacturing batch, ensuring uniformity in protein content and composition; exclusion criteria: Any paste exhibiting deviations in critical quality attributes such as total protein concentration and monomer +dimer s, < 2% and <80% respectively, was excluded from the study). The only variable was the concentration of albumin during pasteurisation which was adjusted to evaluate its effect on polymer aggregate formation under these conditions. Total protein and HPLC analysis were performed in triplicate to ensure statistical validation, assess reproducibility and minimize experimental errors.
This study comprised three experiments which was conducted between December 2023 and December 2024 at R&D laboratory of Plasmagen Biosciences Pvt Ltd. In the first experiment, polymer and aggregate formation in the drug substance was analysed across multiple batches, using Fraction V paste obtained from single fractionation batch, with albumin concentrations between 20% and 28% during pasteurisation. The overall purification process remained consistent in terms of process parameters, conditions with the previously described method. Following pasteurisation, the samples were further processed and analysed.
In the second experiment, polymer and aggregate formation in the drug substance was analysed in four trials at the scale of 1 kg, where Fraction V paste was kept consistent from single fractionation batch. In the first and second trial, the albumin concentration was maintained above 25% to evaluate the effect of NaCl on polymer and aggregate formation. In the first trial, NaCl was added to a final concentration of 120 mM, whereas in the second trial, pasteurisation was conducted without NaCl. However, both trails received the same quantity of stabilizers. In the third and fourth trials, the albumin concentration was maintained at approximately 21%, with NaCl conditions similar to those in the first and second trials, allowing for the assessment of NaCl's effect. The remaining purification steps were performed in accordance with the previously described method.
In the third experiment, polymer and aggregate formation in the drug substance was analysed in single batch (starting material i.e. Fraction V paste was same for all conditions) at the scale of 3 kg paste, where the final concentrated sample was divided into three parts where Albumin concentration during pasteurisation was kept about 25%, 23% and 21% respectively, followed by pasteurisation. In third experiment NaCl was also added to 120 mM concentration during pasteurisation process. The remaining purification steps were performed in accordance with the previously described method. Above experiment was performed in duplicate to assess the reproducibility.
The selection of albumin concentrations for the controlled trials was based on both practical manufacturing relevance and preliminary experimental observations. Our primary target was 20% Albumin, as it represents a widely used commercial formulation and is also the product manufactured by our facility. Additionally, the maximum achievable concentration of albumin during manufacturing is approximately 28%, while the highest commonly marketed concentration is 25%. Therefore, we selected the concentration range between 20% and 28% to reflect both the practical manufacturing window and clinical relevance. Preliminary data indicated that higher aggregation was consistently observed below 23%, whereas concentrations above 25% effectively reduced polymer and aggregate formation. This concentration gradient was intentionally designed to capture conditions ranging from optimal stability (25%) to suboptimal stability (21%), enabling a comprehensive evaluation of the concentration-dependent behaviour of aggregate formation during pasteurisation.
Results
In the first experiment, a total of 19 batches, purified from different Fraction V intermediate pastes, were analysed to assess the impact of varying Albumin concentrations during the pasteurisation process.
The findings indicate that polymer and aggregate levels in the drug substance exceeded 6.5% when the Albumin concentration was below 23% during pasteurisation. Conversely, a higher Albumin concentration during pasteurisation resulted in a reduced formation of polymers and aggregates (preliminary observed data), as illustrated in Table 1. HPLC chromatograms are represented for only few batches as in Figure 2.

HPLC chromatograms for Experiment 1. (Peak near 27 min is N-acetyl tryptophan peak which is added as a stabilizer).
Comparison of Polymer and aggregate Percentage in Batches Varying Albumin Concentration During Pasteurisation (Experiment 1).

* The Protein concentration values represent the average of triplicate measurements.
In the second experiment, the results indicate that polymer and aggregate levels in the drug substance were approximately 2% when the Albumin concentration was maintained at around 26%, regardless of the presence of NaCl. However, at a concentration of approximately 21%, polymer and aggregate levels increased to about 7%, as shown in Table 2. HPLC chromatograms are represented only for one of the triplicate samples as in Figure 3.

HPLC chromatograms for Experiment 2. (Peak near 27 min is N-acetyl tryptophan peak which is added as a stabilizer).
Comparison of Polymer and aggregate Percentage in Batches Varying Albumin Concentration During Pasteurisation When Purified from Same Fraction V Paste (Experiment 2).

* The Protein concentration values represent the average of triplicate measurements.
In the third experiment, two batches were processed, with the final concentration sample divided into three parts to assess the effect of varying albumin concentrations (about 25%, 23%, and 21%) during the pasteurisation process. The results are presented in Table 3. The data revealed an inverse relationship between albumin concentration and the percentage of polymers/aggregates. At the highest albumin concentration (26.17%), the polymer/aggregate content was the lowest (2.84%). In contrast, batches with lower albumin concentrations (21.50-23.25%) exhibited higher levels of polymers/aggregates, ranging from 7.26% to 8.41%. Notably, replicate experiments (Part No. 1 and 2) confirmed this trend, with albumin concentrations of 24.46% and 23.25% yielding 3.34% and 8.41% polymers/aggregates, respectively. HPLC chromatograms are represented only for one of the triplicate samples as in Figure 4.
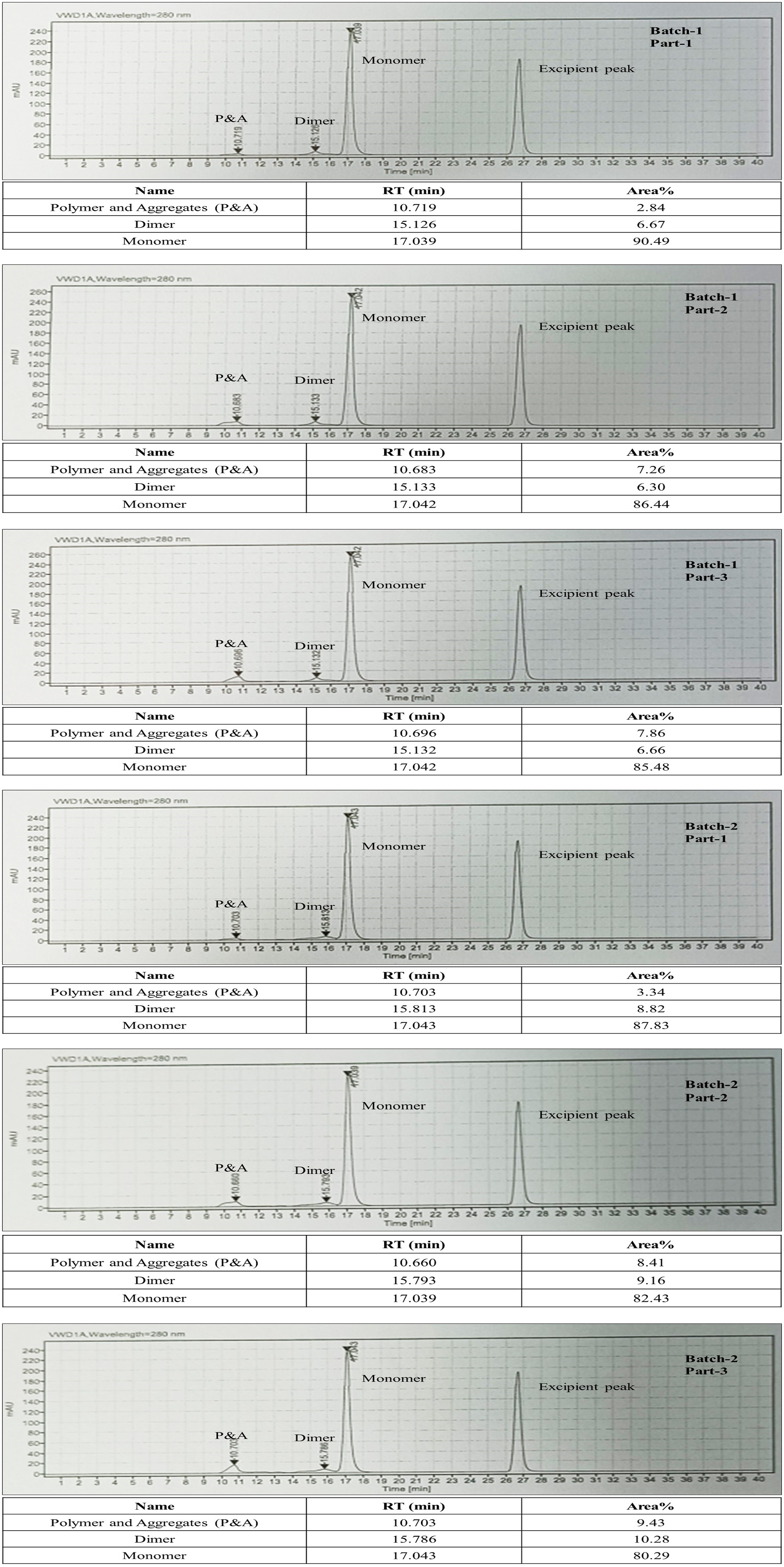
HPLC chromatograms for Experiment 3. (Peak near 27 min is N-acetyl tryptophan peak which is added as a stabilizer).
Trend Analysis of Polymer and aggregate Percentage in Batches Varying Albumin Concentration During Pasteurisation When Purified from Same Fraction V Paste (Experiment 3).

* The Protein concentration values represent the average of triplicate measurements.
Discussion
This study systematically evaluated the impact of varying albumin concentrations on polymer and aggregate formation during the pasteurisation process. The findings provide valuable insights into optimizing albumin stability, which is crucial for ensuring product safety and efficacy in clinical applications.
The results from all three experiments indicate a clear inverse correlation between albumin concentration and the formation of polymers and aggregates. Specifically, when albumin concentration was below 23% during pasteurisation, aggregate levels exceeded 6.5% depicted in Figure 5. In the case of HA, higher concentrations (>25%) replicate such a crowded molecular environment, reducing mobility of unfolded species and suppressing aggregation pathways during pasteurisation. Statistical analysis using one-way ANOVA followed by Tukey's HSD test revealed a highly significant effect of albumin concentration on polymer and aggregate formation (p < 0.001). Higher albumin concentrations (∼26%) consistently demonstrated aggregate levels below 3%, whereas concentrations below 23% showed significantly higher aggregation levels (∼7%–9%). Standard deviations are provided in Tables 1–3 and Figures 5–7 (in figures, error bars not visible as std deviation value is too less). Moreover, Finn et al. (2022) showed that albumin exhibits chaperone-like activity under stress conditions, protecting both itself and other proteins from aggregation through transient hydrophobic shielding and structural buffering. These mechanisms become particularly relevant during pasteurisation, where trace amounts of heat-labile plasma proteins—such as haptoglobin and transferrin—can denature at elevated temperatures (60 ± 1 °C) and act as nucleation sites for aggregation. However, at higher albumin concentrations, molecular crowding limits such unfolding events, keeping minor plasma proteins in a folded state or spatially separated from each other, thereby preventing aggregate propagation. This finding is consistent with previous studies, which suggest that lower protein concentrations increase susceptibility to heat-induced unfolding, leading to intermolecular interactions and aggregation.25–28 The increased aggregation at lower concentrations is attributed to reduced protein-protein interactions that stabilize the monomeric form, thereby allowing unfolded albumin molecules to aggregate.

Polymer aggregate results in same batch having varying Albumin concentrations during pasteurisation and NaCl interference (represented HPLC results only for one of the triplicate samples).

Polymer aggregate results of different batches having varying Albumin concentrations during pasteurisation (represented HPLC results only for one of the triplicate samples).

Polymer aggregate results in same batch having varying Albumin concentrations during pasteurisation (represented HPLC results only for one of the triplicate samples).
The present findings strongly correlate with the biophysical principles established by Christiansen et al (2023) and Finn et al. (2022) regarding protein stability under crowded conditions. At albumin concentrations below 23%, a noticeable increase in aggregate formation (>6.5%) was observed, which can be attributed to the absence of sufficient molecular crowding, allowing for greater molecular mobility, unfolding, and interaction of heat-labile species. Conversely, concentrations above 25% reduced aggregation to below 3%, (shown in Figure 7) indicating effective stabilization through crowding effects. In line with molecular crowding theory, densely packed albumin molecules create a steric barrier that limits the conformational space available for protein unfolding. This crowding not only preserves the folded structure of albumin itself but also shields other plasma proteins—such as haptoglobin, which is known to unfold and aggregate during pasteurisation—from intermolecular collisions and aggregation. Furthermore, unfolded proteins often expose hydrophobic residues which tend to associate via hydrophobic interactions, leading to dimer or oligomer formation. At higher albumin concentrations, diffusion is restricted, thereby reducing the probability of such non-specific interactions and aggregate growth. Additionally, Finn et al. (2022) demonstrated that albumin actively binds to destabilized proteins, functioning as a molecular chaperone to maintain solubility and prevent irreversible aggregation. These mechanisms collectively explain why high-concentration formulations of HA exhibit superior thermal stability and reduced aggregate content during pasteurisation.
25
The second experiment explored the effect of NaCl on aggregation at different albumin concentrations. Interestingly, at 26% albumin concentration, the presence or absence of NaCl did not significantly impact aggregation levels, which remained below 3% (Figure 6). However, at lower concentrations (∼21%), polymer and aggregate formation increased to approximately 7%, regardless of NaCl presence. This suggests that while NaCl can influence protein stability through electrostatic shielding, its stabilizing effect is insufficient at lower albumin concentrations. Previous research supports this finding, indicating that ionic strength alone does not prevent aggregation but can modulate protein-protein interactions.29,30
The third experiment, which involved controlled variation of albumin concentration (about 25%, 23%, and 21%), provided a clear trend analysis. The results (Figure 4) reinforce the conclusion that lower albumin concentrations promote aggregation. This observation is crucial for industrial applications, as it suggests that optimizing albumin concentration above 25% during pasteurisation can significantly enhance product stability.
Moreover, the study highlights the importance of formulation conditions in preventing aggregation-related issues in plasma-derived albumin products. By maintaining a higher albumin concentration, the exposure of hydrophobic regions is minimized, thereby reducing intermolecular interactions that drive aggregation. This aligns with previous work demonstrating that molecular crowding plays a key role in preserving protein integrity under thermal stress.
31
Beyond manufacturing stability, the study's findings hold significant clinical implications. Protein aggregates in therapeutic formulations are known to trigger undesirable immunogenic responses, which can result in the production of anti-drug antibodies (ADAs). These antibodies may neutralize the therapeutic efficacy of albumin, reduce its pharmacokinetic performance, or lead to hypersensitivity reactions, including anaphylaxis or delayed immune responses. Such immunogenic reactions are particularly critical in patients who require large-volume albumin infusions, such as those with liver failure, nephrotic syndrome, burns, or undergoing plasmapheresis.
Aggregates—especially those formed from heat-sensitive plasma proteins like haptoglobin or transferrin, which unfold during pasteurisation—can serve as immune-stimulatory particles. These protein aggregates are recognized by the immune system as foreign, leading to antigen presentation, activation of T-cells, and subsequent antibody production. This process has been widely associated with reduced therapeutic efficacy, altered pharmacodynamics, and potentially severe clinical consequences.
Maintaining albumin at higher concentrations (>25%) during pasteurisation significantly reduces aggregate formation, not only preserving the structural integrity of albumin itself but also protecting co-existing heat-sensitive proteins via molecular crowding effects. This protective mechanism prevents the exposure of hydrophobic residues that typically drive aggregation through hydrophobic interactions and non-specific intermolecular bonds. By reducing the formation of such aggregates, the risk of immunogenic responses is substantially minimized.
From an industrial perspective, this insight enables manufacturers to optimize pasteurisation processes, leading to:
Improved product safety, with lower immunogenic potential. Extended shelf life, due to reduced aggregation-related degradation. Higher product yield and consistency, minimizing batch failures.
Furthermore, regulatory agencies such as the EMA and WHO emphasize stringent control of aggregates in plasma-derived therapeutics due to their direct link to clinical adverse events and reduced treatment efficacy. Therefore, this study not only provides a scientific basis for selecting an optimal albumin concentration but also supports regulatory compliance, patient safety, and therapeutic effectiveness.
In conclusion, maintaining an optimal albumin concentration during manufacturing is a crucial parameter that directly impacts both the clinical reliability and industrial viability of human plasma-derived albumin therapeutics.
This study has certain limitations. Firstly, it did not include formal power analysis for sample size determination. Secondly, the study was limited to evaluating aggregation under pasteurisation conditions without assessing long-term stability post-formulation. Additionally, structural analyses such as FTIR or circular dichroism (CD) were not performed to confirm molecular-level stability. Future studies incorporating these aspects will be crucial to further strengthen the findings.
Future Prospects
This study highlights the impact of albumin concentration on polymer and aggregate formation during pasteurisation but lacks structural-level insights into molecular stability. Future research should employ high-resolution techniques such as x-ray crystallography, spectroscopic methods like circular dichroism (CD) and FTIR, and real-time aggregation analysis using dynamic light scattering (DLS) and small-angle x-ray scattering (SAXS) to monitor conformational changes. Investigating immunogenic risks of aggregates will be crucial for optimizing plasma-derived therapeutics. By integrating biophysical, computational, and formulation-based approaches, future studies can refine albumin processing, ensuring greater stability, efficacy, and clinical safety.
Conclusion
In conclusion, this study establishes that albumin concentration is a critical determinant of polymer and aggregate formation during pasteurisation. Higher albumin concentrations (>25%) effectively mitigate aggregation, whereas lower concentrations (<23%) result in significant polymer formation. While NaCl contributes to stabilization, it does not prevent aggregation at lower albumin concentrations. These findings provide a strong basis for optimizing albumin formulation conditions to enhance stability, ensuring both manufacturing efficiency and clinical safety.
Footnotes
Acknowledgements
We are thankful to the management of Plasmagen Biosciences Pvt Ltd for allowing the work to be conducted at the research and development laboratory.
ORCID iDs
Ethics Approval
The Fraction V paste, which served as the starting material for albumin purification in this study, was obtained from the Plasmagen Biosciences Pvt Ltd manufacturing facility. Since this material is a processed intermediate product derived from pooled plasma and not collected specifically for research purposes, separate institutional ethics approval was not required.
The human plasma used in the manufacturing of Fraction V paste was sourced from licensed and Drug Controller General of India (DCGI)-approved blood banks located across various cities in India. These blood banks operate under valid No Objection Certificates (NOC, e.g., NOC No. X11026/1/2024-BD) and comply with national regulatory standards.
Plasma collection was performed by the respective blood banks from healthy, voluntary donors, following all standard procedures for donor screening, medical fitness verification, and infectious disease testing.
For confidentiality and traceability, only unique donor IDs are shared with the manufacturer (Plasmagen), while all donor consent forms, medical records, and identifying details remain confidential and securely maintained at the respective blood banks, in accordance with ethical and regulatory requirements.
Consent to Participate
Study does not involve participation of any subjects or patients.
Consent for Publication
Study did not involve any participants or patients, so no consent was required for publication.
Author Contribution(s)
Funding
This research was funded internally by Plasmagen Biosciences Pvt Ltd, India. No external funding or support was received for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Availability of Data and Materials
The data generated or analysed during this study are the intellectual property of Plasmagen Biosciences Pvt Ltd and are not available because of confidentiality policies.
