Abstract
Background
The role of natural anticoagulant deficiency in the development of arterial thrombosis (AT) is controversial.
Objective
Our objectives were to assess the deficiency of natural anticoagulants, including protein S (PS), protein C (PC), antithrombin III (AT III) and their involvement in the occurrence of AT.
Design
Retrospective cross-sectional study.
Methods
This study was conducted in 585 patients who were examined with PS, PC, and AT III tests. The activity of PC, PS (men, women), and ATIII under 70%, 75%, 60%, and 80% was recognized as a deficiency, respectively. Peripheral blood cell and coagulation tests were performed before starting treatment. Patients with previous AT, venous thromboembolism (VTE) or anticoagulant therapy were excluded.
Results
Patients without thrombosis were 222 (38%), patients with newly diagnosed VTE were 281 (48%), and patients with newly diagnosed AT were 82 (14%). The most common AT sites were in the lungs, brain, and lower extremities (31.2%, 20.8%, and 20.8%, respectively). Compared to the nonthrombosis group, the AT group had a lower PS activity (%) (82.77 ± 24.09 vs 91.31 ± 27.27), a higher fibrinogen (g/L) (4.25 ± 1.68 vs 3.74 ± 1.51), a higher D-dimer (mg/L FEU) (6.16 vs 1.95), and a higher neutrophil count (G/L) (8.57 vs 6.50) with P < .05. Compared to the VTE group, the AT group had higher hemoglobin (g/L) (135.95 ± 23.75 vs 129.02 ± 25.22) and a higher neutrophil count (G/L) (8.57 vs 7.28) (P < .05). In the AT group, the frequencies of PC, PS, and AT III deficiency were 23.1%, 28%, and 17.1%, respectively. The AT group had a higher frequency of PS deficiency than the nonthrombosis group (28% vs 17.1%, P = .035). Patients with PS deficiency had a higher risk of AT compared to those without PS deficiency (OR = 1.888, 95% CI [1.041-3.422], P = .036).
Conclusion
PS deficiency may be considered a factor in increasing the risk of AT.
Introduction
Thrombophilia is used to refer to the hereditary/acquired condition with a predisposition to the development of thrombosis.1,2 This condition can lead to venous thromboembolism (VTE) or arterial thrombosis (AT) that causes mortality and disability.3–5 AT is usually associated with vessel wall injury (e.g. atherosclerosis) and platelet-mediated thrombi. In contrast, VTE, including deep venous thrombosis and pulmonary embolism (PE), is recognized as an association with hypercoagulability status. The most common hereditable causes of thrombophilia are mutations in FV Leiden, prothrombin genes, and deficiencies in protein C (PC), protein S (PS), and antithrombin III (AT III).1,6–10 PC, PS, and AT III are natural anticoagulants that have been shown to inhibit the blood clot formation process.1,11 PC is an inactive enzyme. In the activated form, it inactivates the factors Va and VIIIa. PS functions as a cofactor PC in the inactivation factor Va and VIIIa. Both PC and PS are vitamin K-dependent glycoproteins that were synthesized in the liver. AT III is a nondependent vitamin K protease that inhibits thrombin and factor Xa. 12 These activations limit the coagulation pathway to contribute to the balance between procoagulant activity and anticoagulant activity. Deficiencies in PC, PS, or AT III predispose to thrombophilia.1,13 The correlations of PC, PS, and AT III deficiencies with the increased risk of VTE are mostly recognized.1,14,15 On the contrary, the role of PC, PS, and AT deficiencies in the development of AT is controversial.6,16–18 We conducted this study to assess the deficiency of natural anticoagulants, including PS, PC, and AT III and their involvement in the occurrence of AT.
Materials and Methods
Designs
This was retrospective cross-sectional study.
Patients
The retrospective cross-sectional study was conducted in Bach Mai Hospital, Hanoi, Vietnam from January 2021 to December 2021. The study enrolled all consecutive patients who underwent PS, PC, and AT III tests. Patients with previous AT, VTE, or anticoagulant therapy are excluded. The Institutional Review Board of Hanoi Medical University waived the need for approval and patient consent due to the retrospective observational nature of the study. All details of the patient were deidentified.
The patients were classified into 3 groups. Group 1 included consecutive patients in whom thrombophilic factors were assessed and they were not confirmed to have VTE or AT. Group 2 included patients with newly diagnosed VTE. Group 3 included patients with newly diagnosed AT. The diagnosis of VTE or AT was confirmed by Doppler Utrasound, CT scan, and MRI. Risk factors as smoking, hypertension, dyslipidemia, diabetes, and overweight were reported.
Screening test: Peripheral blood cells including hemoglobin, WBC, neutrophils, platelet count and coagulation tests including PC, PS, AT III, PT%, APTTs, fibrinogen, and D-dimer, were performed prior to starting treatment.
The PC assay was performed with ACL TOP 750 using the HemosIL PC Kit. PC activity from 70% to 140% was recognized as normal. The PS assay was performed with ACL TOP 750 using the HemosIL Free PS Kit. PS activity from 75% to 130% (for men) and 60% to 130% (for women) were recognized as normal. The AT III assay was performed with ACL TOP 750 using the HemosIL Liquid Antithrombin Kit. AT III activity from 80% to 140% was recognized as normal.
Statistical analysis: Patients were grouped according to the presence of VTE, AT, or no thrombosis. The difference in quantitative variables (PC, PS, AT III, PT%, APTTs, fibrinogen, D-dimer, hemoglobin, WBC, neutrophils, platelet count) was analyzed by independent sample t-test with variables of normal distribution and nonparametric test (the Mann-Whitney test) with non normally distributed variables, (between 2 groups: AT and nonthrombosis, AT and VTE). Differences in qualitative variables (PC, PS, and AT III deficiency, sex, smoking, hypertension, dyslipidemia, diabetes, overweight, occurrence of thrombosis according to site) were analyzed by χ2 or Fisher's exact test (between 2 groups: AT and nonthrombosis, AT and VTE). Regression analysis was used to examine the relationship between factors (PC, PS, AT III deficiency) and the presence of AT. P < .05 was considered a statistically significant difference. The bias was controlled because there were no missing data.
The study reporting conforms to the STROBE guideline. 19
Results
Patient data: 585 patients were enrolled in our study. Their data were retrospectively analyzed. The median age was 43 years (range: 16-96 years) and the male was 55.7%. Patients without thrombosis were 222 (38%), patients with newly diagnosed VTE were 281 (48%), and patients with newly diagnosed AT were 82 (14%). Data are presented in Tables 1 and 2. No significant differences were found in factors such as smoking, hypertension, dyslipidemia, diabetes, and overweight between the AT group and the VTE group or the AT group and the nonthrombosis group, except that the proportion of men in the AT group was higher than in the VTE group (Table 1). Table 1 also shows that the most common AT sites were in the lungs, brain, and lower extremities (31.2%, 20.8%, and 20.8%, respectively). The frequency of AT in the lung and kidney was higher than VTE while it was lower in the transverse sinuses and lower extremities (P < .05).
Characteristics of Patients.

Note: P1: between AT and no thrombosis.
P2: between AT and VTE.
AT, arterial thrombosis; VTE, venous thromboembolism.
Coagulation and Peripheral Blood Cell Indices.
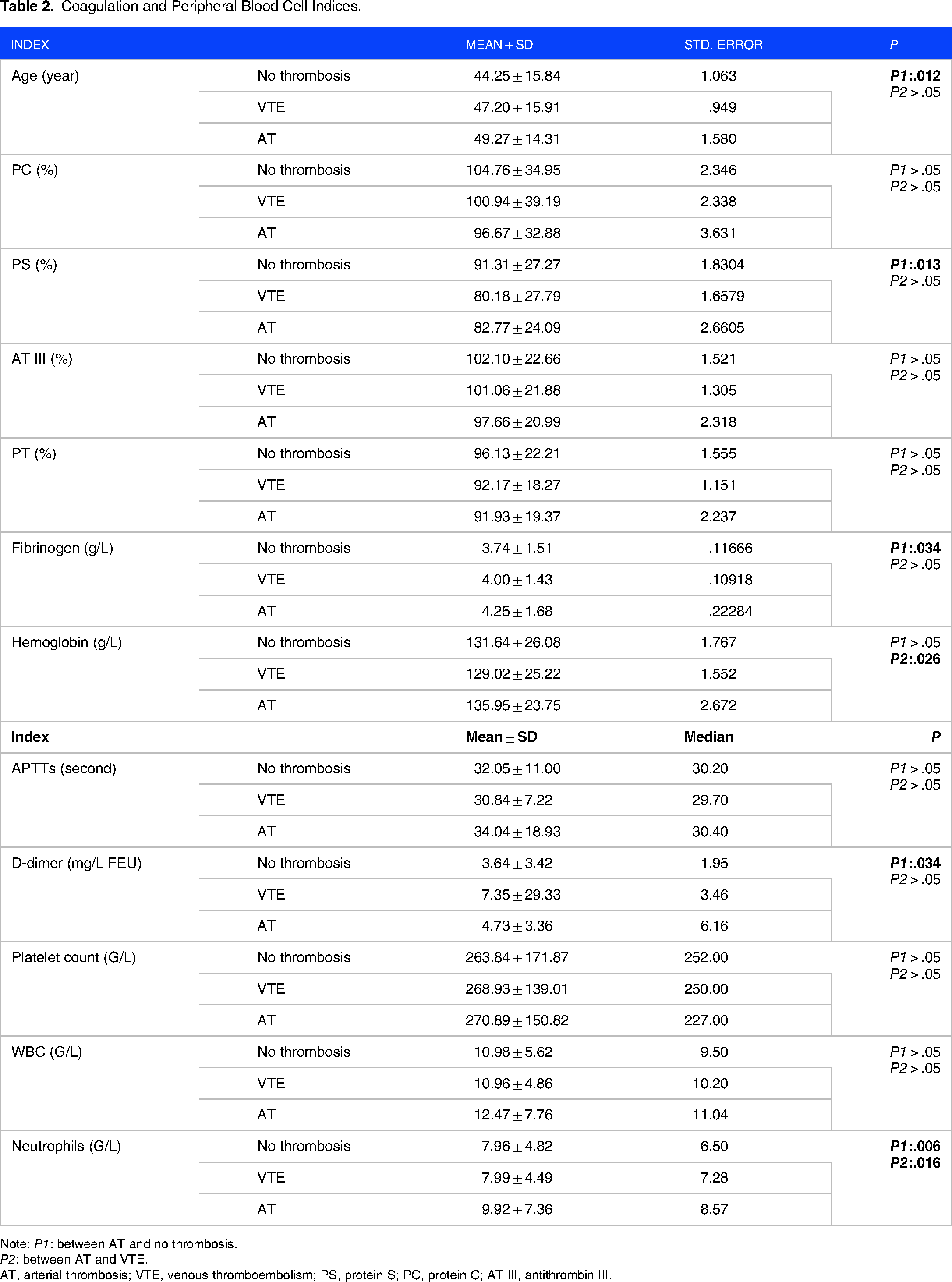
Note: P1: between AT and no thrombosis.
P2: between AT and VTE.
AT, arterial thrombosis; VTE, venous thromboembolism; PS, protein S; PC, protein C; AT III, antithrombin III.
Table 2 shows that the AT group had lower PS activity, higher fibrinogen level, higher D-dimer level, and higher neutrophil count (P < .05) in comparison with the nonthrombosis group. The age of the AT group was also higher than that of the group without thrombosis. Compared to the VTE group, the AT group had a higher hemoglobin level and a higher neutrophil count, (P < .05).
Association of the Deficiencies of PS, PC, and AT III With the Occurrence of AT
In the AT group, the frequency of PS deficiency was highest (28%) (Table 3). Table 3 shows that the AT group had a higher frequency of PS deficiency than the group without thrombosis with P = .035. But there was no significant difference between the AT and VTE groups. Regression analysis defined that patients with PS deficiency had a higher risk of AT compared to those without PS deficiency with OR = 1.888 (95% CI [1.041-3.422]), P = .036.
Frequency of Protein C (PC), Protein S (PS), and Antithrombin III (AT III) Deficiency.

Note: P1: between AT and no thrombosis.
P2: between AT and VTE.
AT, arterial thrombosis; VTE, venous thromboembolism.
Discussion
In recent decades, based on many studies, the authors have tried to review and classify risk factors for VTE and AT.1,20–22 PC, PS, and AT III are accepted as strong risk factors for VTE.1,20,21,23 However, the association between natural anticoagulants and AT was not well described as in VTE. Martinelli et al showed that there was no association between these defects and AT. 24 Ali Nadir et al supported that PC and PS deficiencies only influenced PE. 25 Linnemann et al suggested that thrombophilia except antiphospholipid antibodies did not tend to increase the risk of AT. 26 Some authors reviewed that there was no evidence supporting the hypothesis of the role of natural anticoagulants in the development of AT.1,5,20,27 On the contrary, hyperhomocysteinemia and antiphospholipid antibodies are accepted as risk factors for AT.20,27–30
However, there were rarely studies that attempted to support this hypothesis. Some studies have shown the prevalence of AT in people with PC, PS, and ATIII deficiencies, as well as other studies that had provided data on PC, PS, and AT III deficiency in patients with AT.31–33 In our study, the frequencies of PC, PS, and AT III deficiency of the AT group were 23.1%, 28%, and 17.1%, respectively. There was also no statistically significant difference between the AT and VTE groups, but the AT group had a higher frequency of PS deficiency than the nonthrombosis group with P = .035.
Chiasakul et al in meta-analysis concluded that inherited thrombophilia including PC and PS deficiencies was identified as an increasing risk of AT, but excluded AT III. 34 Tormene et al suggested that the group with inherited natural anticoagulant deficiency had a five-fold higher probability of AT compared to those without. 16 We tried to find the association between the deficiency of natural anticoagulant and the occurrence of AT. Our study also showed that there was an association between PS deficiency and AT. Regression analysis showed that patients with PS deficiency had an approximately two-fold (1.888) higher risk of AT compared to those without PS deficiency.
Our results also showed that the AT patient group had significantly higher fibrinogen level, D-dimer level, and neutrophil count than the group without thrombosis. The association between hyperfibrinogenemia and thrombosis has been studied for a long time, but the causal relationship is still unclear.35–37 Fibrinogen is also an inflammatory factor that can increase cases of thrombosis while it can also be a factor that promotes thrombosis formation. D-dimer is a fibrin degradation product, which is considered both a consequence and a marker for the diagnosis of thrombosis.38,39 Neutrophils are also an important factor in the formation of thrombi through the action of neutrophil extracellular traps.40,41 This mechanism provides new targets for the prevention and treatment of thrombosis.40,41 However, within the scope of this study, we do not have enough data to discuss further.
Limitation of the Study
Our study did not conduct long-term follow-ups on patients, so we could not monitor patients beyond the study period. We were unable to assess the relationship between natural anticoagulant deficiency and the progression of AT over time, as well as its effect on treatment outcome, and whether there may be additional VTE in the future. Hyperhomocysteinemia and antiphospholipid antibodies, which are accepted as risk factors for AT were not included in the study. In addition, some particular factors which are considered as a provocation of thrombosis were difficult to clarify. We hope that there may be further studies to answer these questions.
Conclusion
Our study demonstrates that PS deficiency may be considered a risk factor for AT. The relationship between natural anticoagulant deficiency including PS, PC, and AT III and the appearance of AT is not yet conclusive. Natural anticoagulant deficiency in AT remains a question to explore. More research is needed to evaluate the prevalence of natural anticoagulant deficiency in patients with AT on a large scale, early detect and monitor the occurrence of AT in people with natural anticoagulant deficiency, and determine its effect on the severity and treatment outcome.
Footnotes
Acknowledgments
The authors thank all technicians in the Coagulation Lab—Hematology and Blood Transfusion Center—Bach Mai Hospital, for their efforts in supporting research.
Author Contributions
Thi Tuyet Mai Nguyen: data curation, formal analysis, investigation, methodology, and writing—original draft. Minh Phuong Vu: conceptualization, data curation, formal analysis, methodology, writing—original draft, writing—review and editing. Tuan Tung Nguyen: formal analysis, writing—original draft. Hai Yen Duong: formal analysis, investigation, and writing—original draft. All authors have read and approved the final manuscript.
Data Availability of Data and Materials
Data can be obtained from the corresponding author upon request.
Ethics Approval and Consent to Participate
The Institutional Review Board of Hanoi Medical University waived the need for approval and patient consent because of the retrospective observational nature of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
