Abstract
Conventional therapeutic plasma exchange (TPE) is aimed at removing pathological agents from circulation using a medical device which separates out plasma from other components of blood. Usually, 1.0 to 1.5 plasma volumes are removed and replaced with a replacement solution. There is strong clinical evidence for the effectiveness of TPE in a wide spectrum of diseases, such as thrombotic thrombocytopenic purpura, severe acute inflammatory demyelinating polyneuropathy, and Guillain-Barré syndrome, among others. However, therapeutic approaches using lower plasma exchange volumes (LVPE) suggest some degree of effectiveness in a number of pathological conditions, although generally with low level of clinical evidence: hematology disorders (autoimmune hemolytic anemia), neuroimmunological diseases (Guillain-Barré syndrome, neuromyelitis optica, myasthenia gravis, multiple sclerosis), pulmonary disorders, liver failure, dermatology (pemphigus) and metabolic diseases (porphyria, cholesterol). Further or newer studies are needed to confirm LVPE as an alternative to TPE for such groups of pathologies. While plasma volume removed for TPE ranges 3-4.5 L per procedure, for LVPE ranges from 350 mL to 2 L (frequency and duration vary depending on the pathology treated). In the case of Alzheimer‘s disease (AD), the AMBAR trial recently investigated a combined sequence of intensive TPE with human albumin replacement followed by a maintenance phase LVPE (650-880 mL of plasma removed, similar to a plasma donation). The treatment slowed the decline or stabilized the disease symptoms. The AMBAR approach could justify investigating the use of LVPE in chronic conditions where plasma exchange strategies have proven successful.
Introduction
Transfusion technologies with apheresis procedures have opened new possibilities in the treatment and control of a complex group of diseases for which standard pharmacological approaches have not been fully successful. Plasma exchange procedures aimed at the removal of pathological agents from circulation and replacement of the plasma with fresh frozen plasma (FFP) or colloid solutions (eg, albumin) are used to treat several pathological conditions.
Depletion of harmful humours or toxic agents from circulating blood has always been considered a valuable therapy. The notion of “bloodletting”, the partial removal of a patient‘s blood for therapeutic purposes, has garnered interest throughout human history. For more than 2000 years, bloodletting was an accepted form of treatment for diverse diseases and symptoms. 1 Methods used for bloodletting included: localized procedures done by scarification using lancets and fleas, cupping or by applying leeches; or generalized procedures done by venesection of the median cubital vein or arteriotomy. In the nineteenth century bloodletting was a treatment usually managed by barbers, physicians and surgeons. 2 Further discoveries using new diagnostic techniques undermined earlier dogma and the initial enthusiasm on the use of bloodletting decreased progressively.
In the twentieth century the possibility of extracting blood and separating its cellular components and plasma, with the possibility of returning the cellular components to the body mixed in with a replacement fluid was first described and developed by Abel et al. 3 The concept of apheresis as a methodology to separate the different blood components was thus generated. Technologies and equipment evolved to ensure the feasibility and safety of plasmapheresis to allow donors to donate more frequently. 4
Conventional therapeutic plasma exchange (TPE) is a specific apheresis procedure in which large volumes of plasma (3 to 5 L) are removed from a patient and replaced by plasma from a healthy donor, or by colloid or crystalloid fluids. TPE has demonstrated to be an effective therapy in a wide number of pathological conditions. In the last two decades, a series of studies have suggested that the large volumes of plasma exchanged, and the replacement fluids used in TPE could be reduced through the introduction of non-conventional or less-conventional strategies such as small- or low-volume plasma exchange (LVPE). However, LVPE has received little attention as a topic deserving extensive bibliographical research and an in-depth discussion.
The present review describes the terminology and technologies used for various apheresis procedures. The main established indications for conventional TPE are explored to place in perspective the differences in procedures and intensities of plasma exchange as compared with LVPE in those pathologic conditions where it has been applied. Finally, this review will examine combined TPE and LVPE strategies recently described for the treatment of Alzheimer‘s disease (AD) with successful results and summarize their most relevant aspects.
Apheresis and Plasma Exchange: Terminology and Technologies
The term “apheresis” is derived from the Greek word aphaeresis, which means “to separate”, “to take away by force” or “to remove”. Apheresis were also been called pheresis or hemapheresis, though these terms have become obsolete. In essence, apheresis is the process of removing a specific portion of the blood while returning the remainder of the blood to the patient. The terminology used commonly specifies the component of blood that is being removed: a) plasmapheresis (plasma); b) plateletpheresis (platelets); c) leukocytapheresis (leukocytes), and d) erythrocytapheresis or erythropheresis (red blood cells).
Plasmapheresis refers exclusively to the removal of small volumes of plasma – not more than 15% of total blood volume – and does not imply replacement of the removed plasma. It is a procedure currently used by blood banks or plasma centers – using adequate processors – to maximize the volume of plasma donated by a single donor, without removing erythrocytes. A plasma volume equivalent to 400-600 mL can be obtained in one session from a single healthy donor using plasmapheresis (vs 200-250 mL from whole blood-derived donations). The plasma collected is used for transfusion or manufacturing other products such as albumin, immune globulins, coagulation factor concentrates, and other therapeutic proteins, as well as laboratory reagents.
TPE refers to the removal of large volumes of plasma (from 1.0 to 1.5-fold of patients’ total plasma volume -TPV-) with an appropriate volume replacement using FFP, albumin, or a combination of crystalloid/colloid solutions, depending on the pathology being treated. The volume of plasma exchanged from a patient is expected to remove a certain proportion of a circulating noxious substance responsible for the pathological condition being treated (Figure 1A). The TPV of a patient can be easily estimated using well-established nomograms and equations. 5 As summarized in Table 1, the estimated plasma volume for an adult male weighing 70 kg would be approximately 3 L. An exchange of 1.0 TPV would represent the removal of 3 L of plasma in the patient and replacement with the corresponding volume of fluids. Similarly, an exchange of 1.5 TPV would imply the process of replacing 4.5 L of plasma. In the case of a female patient weighing 60 kg, 1.0 TPV would involve replacement of approximately 2.75 L of plasma and a 1.5 TPV would imply replacement of 4 L of plasma, approximately.
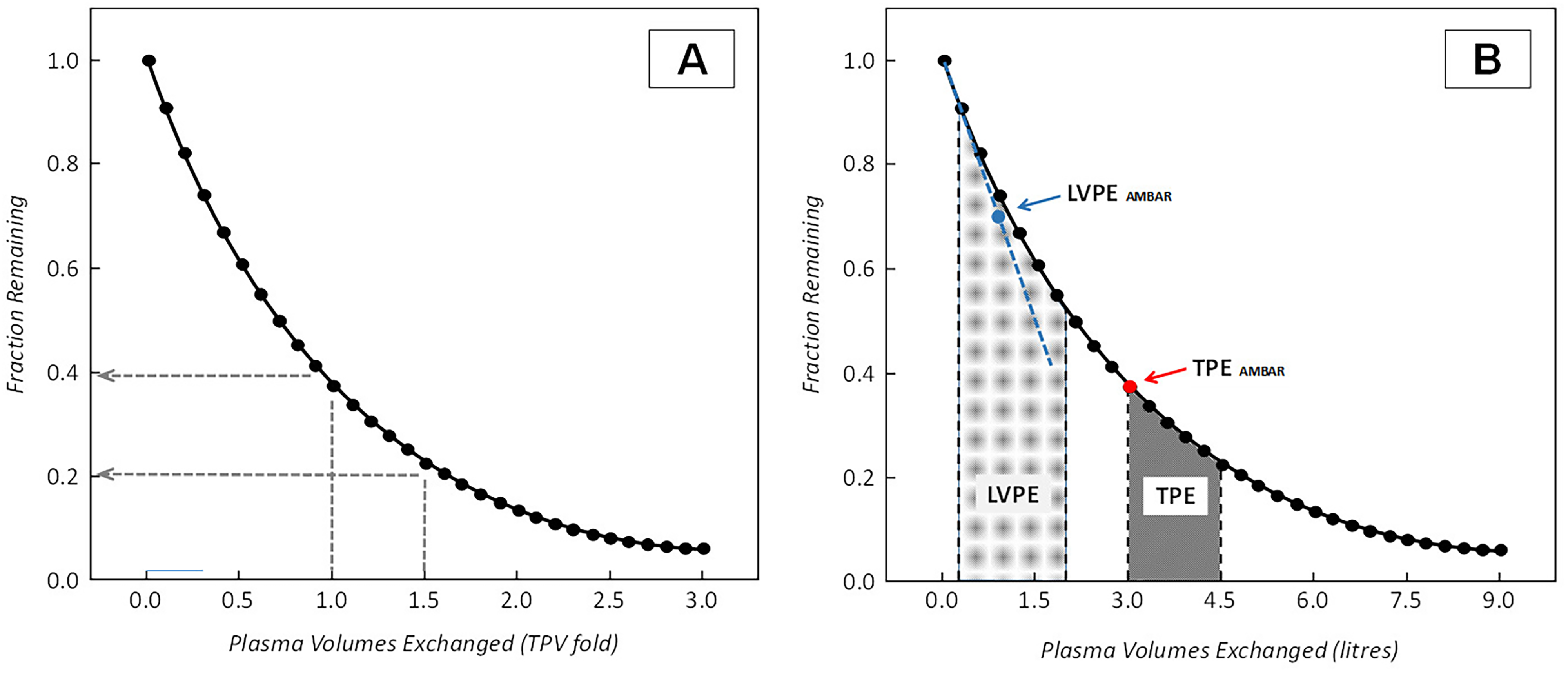
Efficiency of plasma exchange removing a circulating substance: [A] Depletion of soluble substances from plasma by plasma exchange considering their distribution in a one-compartment model. Therapeutic plasma exchange aimed at treating 1 or 1.5 total plasma volumes (TPE) should theoretically remove from 60 or 80% of the circulating substance in this model. Efficiency is lower when substances are distributed in more compartments; [B] Representative image comparing plasma volumes exchanged in TPE versus LVPE. Volumes exchanged are quantitatively represented in litres per session or procedure. Plasma volumes exchanged in LVPE are near one third of those exchanged during conventional TPE. Arrows approximately indicate the upper range of plasma volumes exchanged per procedure in the AMBAR programme: 2.5-3 L for the intensive TPE period (red dot) and 0.7-0.9 L during the LVPE maintenance period (blue dot). In the AMBAR LVPE procedures, the relationship between the plasma volume removed and the fraction remaining is lineal (blue dashed line).
Calculation of Total Plasma Volumes (TPV) in adults. TPV (L) = 0.07 × Weight (kg) × (1-Hematocrit [Expressed in Decimal Form]).
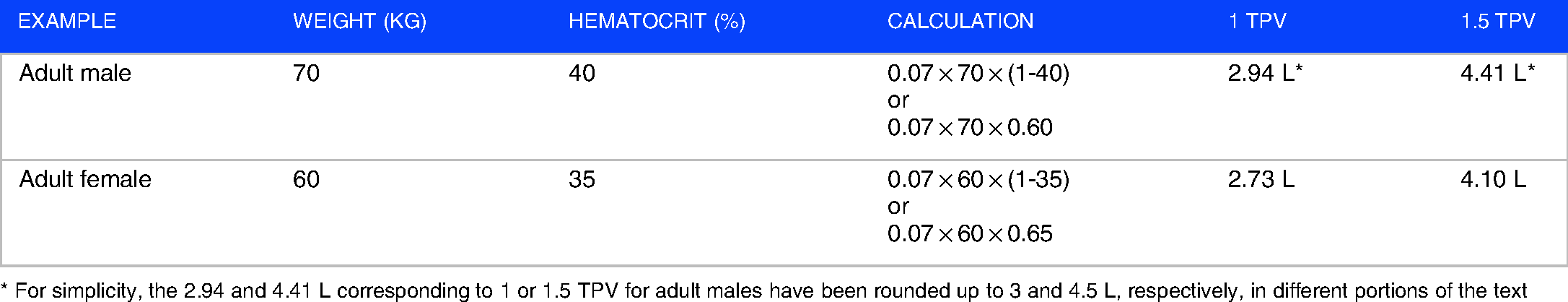
* For simplicity, the 2.94 and 4.41 L corresponding to 1 or 1.5 TPV for adult males have been rounded up to 3 and 4.5 L, respectively, in different portions of the text
There are two main procedures for plasma removal: centrifugation and filtration. In filtration, blood is pumped through a membrane with pores of 0.2 to 0.6 μm allowing plasma to pass through while retaining blood cells. 6 Older devices used parallel-plate membranes, but currently hollow fibre membrane filters are preferred.
Centrifugation is the predominant method used for TPE. 7 Early developed instruments worked under intermittent flow, with procedure performed in cycles (withdrawal, separation and reinfusion). The extracorporeal volume of blood being processed under this approach could be elevated. More modern instruments are devised to work under continuous flow. In the newer devices, whole blood is pumped into a rapidly rotating separation chamber. In these systems blood is withdrawn, processed and reinfused simultaneously making the whole procedure faster. The extracorporeal volumes are smaller, resulting in better hemodynamic stability.
In summary, TPE is nowadays considered an effective treatment for several pathological conditions. Despite the increased accessibility to plasma exchange therapies, some relevant considerations must be taken into account. TPE should be established in a suitable environment such as tertiary hospitals or specialized centres. Access to adequate equipment, need for dedicated and trained personnel and coverage of the costs of replacement fluids or additional treatments are critical elements to ensure the quality and efficacy of the TPE procedures.
Indications for Therapeutic Plasma Exchange: Recommended Volumes and Frequencies
Recommendations for the use of TPE exist for numerous pathologic conditions. The basic premise TPE is that removal of certain toxic substances from the plasma will reduce further damage and may even permit reversal of some of the pathological processes involved. The toxic substances could be autoantibodies, immune complexes, cryoglobulins, myeloma light chains, endotoxins, cholesterol/lipoproteis, or other potentially damaging components. The most recent guidelines of the American Society for Apheresis – Journal of Clinical Apheresis (ASFA-JCA) Committee for relevant diseases and medical conditions has graded and categorized 157 indications and/or therapeutic apheresis modalities. 8 Table 2 summarizes a selected group of pathologies for which TPE has been considered as a first-line therapy, either as a primary standalone treatment or in conjunction with other modes of treatment (Category I) with the highest grading quality of evidence and strength of recommendations. 9
Characteristics of indications in disorders for which therapeutic plasma exchange is considered first-line therapy, either as a primary standalone treatment or in conjunction with other modes of treatment (Category I). 8

Recommended volumes of plasma to be exchanged per session for the more relevant pathologies for which TPE is considered a first line treatment are relatively high (3 to 4.5 L). For uniformity, the most recent guidelines 8 refer to the volumes to be exchanged as a proportion of the patients TPV as shown in Table 2. Other authors prefer the calculations of the plasma volumes expressed as mL/body weight and recommend exchange volumes of 40 to 60 mL/kg for well-established TPE indications. 10 Calculations of volumes to be exchanged for using the later approach are essentially coincident with those calculated using the estimated TPV. Volumes of plasma to be exchanged calculated using 40 or 60 mL/kg for a standard 70 kg adult male are approximate 2.8 or 4.2 L, respectively; volumes quite close to the approximated 3 or 4.5 L calculated using the 1 or 1.5 TPV mentioned earlier. The following text briefly describes the most relevant pathologies, summarized in Table 2, for which TPE has become the first line treatment with the highest level of clinical evidence.
Thrombotic Thrombocytopenic purpura (TTP)
TTP and other related microangiopathic conditions are characterized by the development of a systemic thrombotic pathology affecting mostly small vessels. Today, TTP presents rarely with the classical pentad of severe thrombocytopenia, microangiopathic hemolytic anemia, altered mental status, renal failure and fever. TPE with plasma replacement has significantly improved patients’ clinical outcomes and has drastically reduced the overall mortality of immune mediated TTP from nearly uniformly fatal to <10-20%. 11 TTP is associated with a severe deficiency (<10%) of plasma ADAMTS13 enzyme activity, which is responsible for maintaining normal distribution of von Willebrand factor multimers. 12 ADAMTS13 deficiency can be congenital or, more frequently, due to autoantibodies against this enzyme. As for the beneficial effect plasma exchange in this condition, one hypothesis is that TPE removes autoantibody directed against ADAMTS13, while replacing ADAMTS13 protease activity. New treatment strategies combining TPE with immunosuppressants, such as rituximab, and caplacizumab are being introduced. 13 If TPE is not immediately available, large dose plasma infusions (25-30 mL/kg) may be given if tolerated, until TPE can be initiated. 14 It is recommended that the aim for plasma volume exchange should be at 1-1.5 TPV per session and the procedures initially repeated daily using plasma or plasma/albumin as the replacement fluid. TPE for TTP is generally performed daily until the platelet count is >150 × 109/L, and lactate dehydrogenase (LDH) is near normal for 2-3 consecutive days.
Gillain Barré Syndrome
Guillain-Barré syndrome (GBS) is an acute, usually symmetrical, and typically ascending, paralyzing disorder caused by inflammation of the peripheral nerves. Acute inflammatory demyelinating polyradiculoneuropathy (AIDP), which comprises up to 90% of GBS cases, is an acute progressive paralyzing illness affecting both motor and sensory peripheral nerves. Mortality of these conditions is estimated at 3-5%. In trials using TPE and/or intravenous immune globulin (IVIG) in GBS, AIDP patients represented the majority compared to other variants. TPE was the first therapeutic modality to impact the disease favourably. TPE is an effective treatment for GBS and should be initiated within 7 days of disease onset.15,16 The typical TPE strategy is to exchange 1-1.5 plasma volumes, 5-6 times over 10-14 days, but some patients may need additional treatments. TPE frequency reommended is daily or every other day and the replacement fluid albumin or plasma.
Acute Liver Failure
Acute liver failure (ALF) is a pathologic condition that can lead to multi-organ system failure and has a high mortality rate if not treated promptly. Liver transplant is the most effective treatment to correct the dramatic course of ALF, but the limited availability and donor resources at the moment of ALF presentation is a major problem. Artificial and bioartificial liver support systems may provide a “bridge” for patients with severe liver disease to recovery or transplantation. 17 TPE has demonstrated improved outcomes in patients with ALF by increasing liver transplant-free survival. 18 This has been attributed to attenuation of innate immune activation and amelioration of multi-organ dysfunction. TPE can remove albumin-bound toxins as well as unbound toxins, including aromatic amino acids, ammonia, endotoxin, indols, mercaptans, phenols and other factors which may be responsible for hepatic coma, hyperkinetic syndrome, and decreased systemic vascular resistance and cerebral blood flow. High-volume daily TPE is recommended until liver transplantation or self-regeneration occurs. 18 Volume exchange recommended is 1.0-1.5 TPV; with a target 8-12 L with a daily frequency, using plasma and albumin as replacement fluid.
Goodpasture's Syndrome
Anti-glomerular basement membrane (GBM) disease is defined as the presence of small-vessel vasculitis which affects the glomerular capillaries, pulmonary capillaries, or both along with anti-GBM autoantibody deposition. An early clinical trial involving a small number of patients treated with TPE demonstrated maintained kidney function and improved survival among patients. 19 The presence or absence of antibody should not be used to initiate or terminate therapy, because antibodies are not detectable in some patients with the disease and may be present in other patients without active disease. Recommended volume of plasma treated is 1-1.5 TPV with a daily or every other day schedule using albumin of plasma as replacement fluid. The minimum course of TPE should be 10-20 days. In patients with active disease, TPE should be continued until resolution of evidence of ongoing glomerular or pulmonary injury.
Catastrophic Anti-Phospholipid Syndrome (CAPS)
This is a rare but life-threatening autoimmune disease which is characterized by disseminated intravascular thrombosis resulting in multiorgan failure. A triple therapy approach of anticoagulation plus glucocorticosteroids plus TPE and/or IVIG is the recommended approach to therapy. 20 Volume exchange recommend is 1-1.5 TPV with TPE performed daily or every other day and plasma alone or in combination with albumin used as replacement fluid. Most published cases have reported daily or every other day TPE for a minimum of 3-5 days up to courses of 1-3 weeks, but some patients have been treated with longer courses.
Wilson Disease
Patients suffering from this rare inherited disease are unable to eliminate copper properly leading to its accumulation, possibly to a life-threatening level. TPE can effectively remove copper from the circulation 21 and provide a therapeutic bridge to liver transplantation in fulminant Wilson disease. The recommended plasma volume treated is 1-1.5, with a daily or every other day frequency using plasma and albumin as replacement fluids. Serum copper reduction is achieved rapidly and maintained after the first 2 treatments. However, the total number of TPE performed is variable (1-11), depending on the availability or liver transplantation or patient recovery.
In summary, the recommended plasma volumes exchanged during TPE for the previously mentioned pathologies ranges from 1 to 1.5 TPV, representing approximately 3 to 4.5 L of plasma exchange for a standard person weighing 70 kg (Figure 1B). It can be approximated that total volumes of plasma exchanged during a therapeutic course for patients with the most frequently treated conditions may range from 12 to 30 L for patients with GBS, 30 to 45 L for patients with TTP, and from 30 to 60 L for patients with Goodpasture's syndrome.
Low Volume Plasma Exchange as an Alternative to TPE in Selected Pathologies
The concept of LVPE has emerged in the last two decades as a possible alternative to TPE modality for certain groups of pathologies. LVPE, which may be also referred as small volume plasmapheresis, is based on the same principle as conventional TPE (selective removal of plasma) but uses a simpler technology with much lower costs. 22 LVPE approaches have been mainly developed in low income countries that cannot afford the expenses associated with TPE or IVIG treatments.23–27 In addition, LVPE may represent a preferred plasma exchange treatment modality in chronic conditions, in which the removal of smaller volumes of plasma would be justified for long periods.
The following text describes selected pathologies and conditions in which efficacy of LVPE has been explored, regardless of their level of evidence. This means bringing to light published studies which are old, preliminary or limited in scope. This stresses the need of further or newer studies. Information on the studies reported below is summarized in Table 3.
Characteristics of therapeutic approaches with low volume plasma exchange in various pathologies.
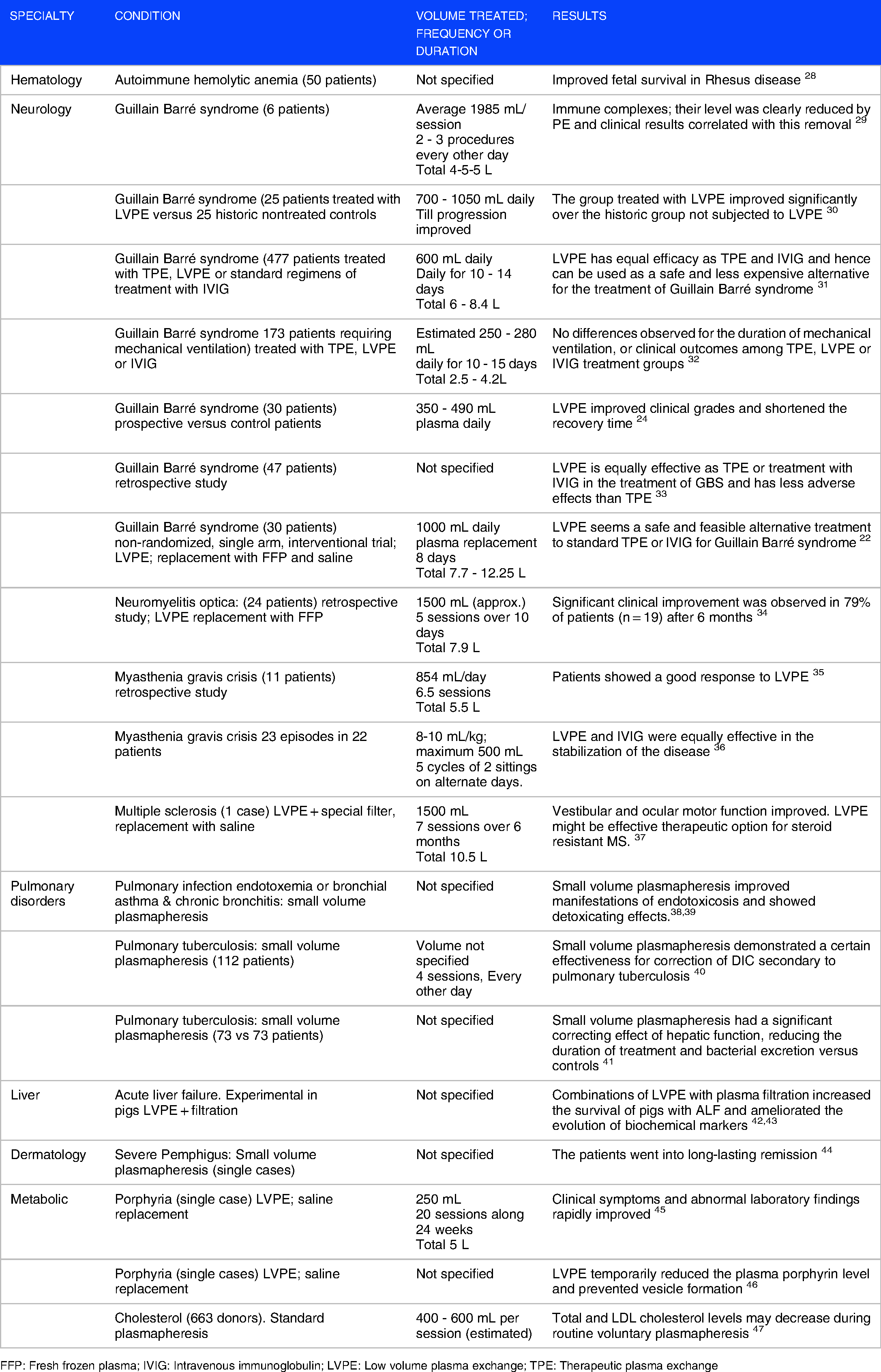
FFP: Fresh frozen plasma; IVIG: Intravenous immunoglobulin; LVPE: Low volume plasma exchange; TPE: Therapeutic plasma exchange
Hematology: Autoimmune Hemolytic anemia
Reports on the use of small volume plasmapheresis in combined treatment for patients with autoimmune hemolytic anemia unresponsive to routine therapy, have shown the utility of this approach in promoting more effective recovery of red blood cell transfusions, and reducing the need for corticosteroid treatment. Plasmapheresis could be used in the early stages of disease treatment. 48 A clinical study in 50 multiparous pregnant patients with Rhesus isoimmunization investigated the efficacy of small volume plasmapheresis treatment initiated at the end of their first trimester. This therapeutic approach proved effective in improving fetal survival in Rhesus disease. Rates of fetal survival observed were considered favourable and supported the use of small volume plasmapheresis as an alternative therapy in locations where immunoprophylaxis is not available. 28 Effects of weekly small volume plasmapheresis have also been explored by other investigators in the Rhesus D immunization. In the latter case, the good clinical results obtained with the small volume plasmapheresis could not be explained by a consistent change in the anti-D IgG subclass composition involved in rhesus-D immunization. 49 These studies were published before the current recommended treatment, rituximab, was available. This agent is still expensive and is not available or reimbursable in all countries.
Volumes of plasma exchanged, replacement fluids administered, and duration of procedures have not been clearly specified in the previous mentioned reports. However, by definition, a small volume plasmapheresis would be equivalent to a reduced plasma donation by plasmapheresis (more likely <400 mL per session).
Neurology: Guillain Barré Syndrome
Evidence-based guidelines of the American Academy of Neurology report equal strength of evidence to support the use of TPE or IVIG in the treatment of GBS. 50 The results of several controlled trials comparing TPE to supportive care alone indicate that TPE can accelerate motor recovery, decrease time on the ventilator, and speed attainment of other clinical milestones. Interestingly, LVPE procedures have been frequently applied to improve the course of neurological complications in patients affected by GBS.
In an early report, Valbonesi et al. 29 evaluated LVPE in 6 patients with acute GBS and in 1 with its chronic relapsing type. Two procedures on alternate days were carried out in 4 patients while a third procedure was performed in 3 other ones. The average volume of plasma exchanged per session was 1985 mL, ranging from 1050 to 3600 mL. After LVPE, patients showed moderate to marked improvement in motor strength, in their ventilatory function and in their sensory symptoms. Levels of immune complexes were clearly reduced after LVPE and clinical results correlated with this laboratory finding. In a later study, the impact of treatment with LVPE in GBS was studied by comparing 25 patients treated with LVPE since 1982 with 25 historic controls treated without LVPE prior to 1982. 30 LVPE was performed by removing 10-15 mL plasma/kg body weight daily till the progression of the disease was arrested or recovery started. The group treated with LVPE started recovering significantly earlier, attained better clinical grades at the end of the first and third months, and took much shorter time to recover by one clinical grade.
The efficacy of LVPE was compared with well-established treatments with TPE or IVIG in the treatment of GBS. 31 The study included 477 historic patients during a 12-year period. Of these, 353 patients were AIDP, 87 were acute motor axonal neuropathy (AMAN), and 37 were unclassified. Standard regimens of treatment for IVIG (n = 197) and TPE (n = 106) were included. In the LVPE group (n = 128), two sessions of exchanges were carried out daily for 10-14 consecutive days or till the first sign of recovery appeared. A closed, double-bag system developed for this procedure was used and 500 mL blood was centrifuged at 3000 rpm for 10 min and plasma removed on a plasma extractor. Statistical analysis did not show any significant difference among the three groups in the short- and long-term outcomes. The study concluded that the effectiveness of the LVPE was similar to the IVIG approach and hence it could be used as a safe and less expensive alternative for the treatment of GBS. In another retrospective study, Netto et al. 32 compared the effectiveness of three modes of intervention on the outcome of patients (n = 173) with GBS requiring mechanical ventilation treated with IVIG, LVPE, or conventional TPE between 1997 and 2007. In LVPE, 10% of blood volume was collected through a peripheral vein in a plastic double bag containing the standard anticoagulant, which was then centrifuged at 5,000g and the plasma was removed. An equivalent amount of physiological saline was added to the red blood cells and the mixture was infused back into the patient. The primary outcome parameters evaluated were mortality, duration of mechanical ventilation, hospital stay and Hughes scale at discharge from hospital. The mean duration of mechanical ventilation, total hospital stay, and Hughes scale at discharge did not differ among the three treatment groups. Complications were similar in the three treatment groups except for hypoalbuminemia and anemia, which were more common in patients in the LVPE group. Mortality rate was higher in the LVPE group, although statistical comparison was unresolved due to the small number of patients. In conclusion, the outcome of patients treated with these three treatments strategies were similar. Despite the described safety concerns, the beneficial effects of LVPE in this study supported the development of further randomized control trials in resource-constrained settings. 32
Therapeutic outcomes of LVPE were also evaluated in 30 GBS patients using volume exchanges around 5-7 mL plasma/kg body weight daily (350-490 mL/day for a 70 kg adult). The group of patients treated with LVPE started recovering earlier than control patients. The time taken for improvement in the clinical grades at the end of the first and third month was shorter. LVPE shortened the recovery time in patients with GBS. 24 Another study compared the efficacy of LVPE, versus IVIG or TPE in a retrospective study in 47 patients with GBS. 33 Results of this study demonstrated that LVPE was equally effective as TPE or IVIG in the treatment of GBS and had less adverse effects than large volume plasma exchange.
A feasibility study using a LVPE approach was carried out by Islam et al. 22 in patients with GBS. The study was designed as a non-randomized, single-arm, interventional trial and included 20 adult patients with GBS presented within 2 weeks of onset (NCT02780570). LVPE was performed by subjecting blood, previously drawn through a central vein catheter, to cell sedimentation in a blood bag, removal of supernatant plasma, and with red blood cells being re-transfused at the end of the procedure. This process was repeated 3 to 6 times a day, for eight consecutive days. FFP and normal saline were used as replacement fluids. The median plasma volume exchanged was 140 mL/kg (range 110-175) and removal of 8 L plasma was possible in 15 (75%) patients. It was estimated that patients in whom replacement included FFP may have received a median 1 g/kg IgG, although a substantial proportion of IgG was probably removed again in subsequent LVPE sessions. GBS disability score improved by at least one grade in 14 (70%) patients 4 weeks after LVPE started. Authors concluded that LVPE would be a safe and feasible alternative to standard treatments established for GBS. 22
Neurology: Other Conditions
LVPE approaches have been also investigated in other neurological conditions such as myasthenia gravis and multiple sclerosis (MS). Myasthenic crisis (MC) is an important and common complication in the natural history of myasthenia gravis that can be precipitated by multiple factors and requires urgent aggressive management. When treated aggressively, MC is associated with good outcome. Panda et al. 35 reported the results of a retrospective study conducted in 11 patients with MC admitted at a tertiary care centre in India. Steroid or cholinesterase inhibitor withdrawal and infections were the commonest precipitating factors for MC. Patients responded well to LVPE consisting of a mean 854 mL/ day with mean 6.5 cycles per patient. In a retrospective study of case records of 23 episodes of MC, fifteen (65%) episodes were treated with LVPE and eight (35%) episodes received IVIG. 36 LVPE consisted of five cycles performed on alternate days. Each session consisted of two sittings and for each sitting 8-10 mL/kg of plasma was exchanged, a maximum of 500 mL for each sitting. LVPE and IVIG showed to be equally effective in disease stabilization.
Some patients with MS are resistant to steroid therapy. In those patients an alternative therapy could be therapeutic apheresis. In one reported case, a woman with relapsing-remitting cerebral MS with dizziness and imbalance, resistant to steroid therapy was treated with LVPE with saline substitution. 37 During each session 1000-1500 mL of plasma were replaced. Vestibular and ocular-motor tests prior to and after treatment demonstrated improved function. The efficacy of LVPE has been also investigated in patients with other autoimmune demyelinating diseases such as the neuromyelitis optica. In a retrospective study, investigators evaluated the impact of LVPE in the evolution of disease in 24 patients. 34 Volumes of plasma exchanged averaged 112 mL/kg body weight, levels well below the 195 mL/kg/body weight usually recommended for these patients. Patients were treated with 5 sessions of plasma exchange over 10 days. FFP was used as the replacement fluid. A significant clinical improvement was observed in 79% of patients after 6 months. There were no significant differences in volume of plasma treated between patients, who had moderate and marked improvement. LVPE proved to be a safe and effective add-on therapy in patients with neuromyelitis optica, especially in steroid-resistant cases.
The overall information available supports the concept that therapeutic approaches with LVPE should be considered a very simple, non-expensive and effective procedure that improves outcomes in a wide spectrum of neuro-immunological diseases in countries with limited resources where the costs of TPE are unaffordable.51–53
Pulmonary Disorders and Endotoxemia
Data of therapeutic strategies using small volume plasmapheresis in various pulmonary conditions are only available from case reports and from local or regional studies. Combined use of small volume plasmapheresis with extracorporeal hemoxygenation prevented manifestations of endotoxicosis and improved treatment results in a case of a patient with acute infections of lungs and pleura, and considerably reinforced the detoxicating effect of plasmapheresis. 38 A comparative assessment of the clinical efficacy of standard and small volume plasmapheresis versus standard treatment was evaluated in 21 patients with chronic bronchitis aggravated by asthma. 39 Small volume plasmapheresis procedures attenuated inflammation in bronchial mucosa and were non-inferior in efficacy to the standard treatment of chronic obstructive diseases.
Karpina et al. 40 evaluated the efficiency of small volume plasmapheresis in the correction of hemostatic disorders in 112 patients with progressive destructive pulmonary tuberculosis. Patients were divided in 3 groups: a) basic, 43 subjects receiving the standard therapy supplemented by 4 small volume plasmapheresis sessions every 2 days; b) 36 patients receiving 2 plasmapheresis sessions weekly; and c) patients on standard therapy alone. The use of small volume plasmapheresis demonstrated effectiveness for the correction of disseminated intravascular coagulation secondary to pulmonary tuberculosis. In another study a total 140 patients were divided into two groups; 73 patients in whom small volume therapeutic plasmapheresis as an efferent therapy to treat drug intolerance and endotoxicosis (study group); and 67 patients not subjected to therapeutic plasmapheresis (control group). Treatment with small volume plasmapheresis reduced the duration of therapy and contributed to a 2.5-fold reduction in the level of bacterial excretion versus the control group. This can be explained by the rapid and prolonged recovery of hepatic function in 100% of plasmapheresis cases, associated with a significant correction of toxic and allergic reactions to anti-tuberculous drugs. Pulmonary infiltrate resolution was observed twice more frequently in patients receiving plasmapheresis than in the controls. 41
As previously commented for hematologic conditions, volumes of plasma removed, replacement fluids and duration of these procedures in the pulmonary disorders previously cited have not been accurately specified. Although there is no apparent evidence for LVPE in pulmonary disease, the overall impression is that plasmapheresis has been used as an additional detoxification technique in the aforementioned conditions.
Acute Liver Failure
LVPE approaches have been used in experimental and clinical artificial liver systems. Experimental studies in pigs with ALF have demonstrated that blood purification systems combining LVPE with plasma filtration absorption increased the survival of the animals, ameliorated the evolution of biochemical markers (liver enzyme, bilirubin, bile acid and blood ammonia levels), effectively removed elevated TNF-alpha, TGF-beta1, angiotensin-II and improved hepatocyte regeneration.42,43 A clinical study is being carried out to evaluate in patients if this LVPE plus plasma filtration approach could be an alternative procedure to prolong the survival time of ALF patients. 54
Dermatology
Small volume plasmapheresis proved a successful therapeutic approach in cases of severe pemphigus vulgaris and foliaceus if applied together with corticosteroids and cyclophosphamide, both at lower doses. 44 The patients went into long-lasting remission. The effect of plasmapheresis did not appear to depend on the removal of large quantity of antibodies. This therapy may be considered an alternative for patients who cannot tolerate large doses of prednisone because of severe side-effects of steroids.
Metabolic Conditions
LVPE has proven useful to improve symptoms in a reduced number of patients with porphyria. A 49-year-old man with porphyria cutanea tarda was treated with repeated LVPE. Approximately 250 mL of plasma were removed and replaced by physiological saline. Clinical disease manifestations and abnormal laboratory findings rapidly improved following a total of 20 sessions during a 24-week period. The blood hemoglobin and plasma levels of total protein were unchanged. 45 In another case, a 58-year-old male patient with diabetes developed photosensitive bullous dermatosis on his hands and face, with porphyrin levels consistent with porphyria cutanea tarda. LVPE temporarily reduced the plasma porphyrin level and prevented vesicle formation in that patient. 46
LDL cholesterol apheresis is used to treat patients with familial hypercholesterolemia, and low volume plasmapheresis for plasma donation may similarly lower cholesterol levels in some donors. In a prospective multicenter study, researchers investigated the effect of voluntary plasmapheresis on plasma cholesterol levels in 663 donors in which a blood sample was obtained for analysis before each donation. Data were analyzed using a multivariable repeated measures regression model with a general estimating equations approach with changes in cholesterol as the primary outcome measure. The model predicted a significant decrease in total and LDL cholesterol for both genders and all baseline cholesterol level (P < .01). These results suggest that, in donors with elevated baseline cholesterol levels, total and LDL cholesterol levels may decrease during routine voluntary plasmapheresis. 47
In summary, the available information confirms that LVPE is a simpler procedure that does not require expensive equipment. Separation of plasma from whole blood can be performed through simple centrifugation or sedimentation of whole blood drawn from the patients. Plasma can be easily removed and red blood cells re-transfused to the patient. In selected indications, the efficacy of LVPE could be similar to that of conventional TPE, even with plasma volumes exchanged in LVPE being typically one third of those exchanged during conventional TPE (Figure 1B). Moreover, LVPE can reduce the costs associated to replacement fluids or treatments with IVIG. However, the evidence levels for LVPE in the described conditions are rather low, and further or newer studies are necessary.
TPE and LVPE in Alzheimer’s Disease: the AMBAR Programme
In the last decade the combination of TPE and LVPE approaches has been investigated as a possible treatment for slowing down disease progression in patients with AD (Table 4). An increase in the level of amyloid-β (Aβ) has been implicated in the development of senile plaques, leading to neuronal cell death and neuro-degeneration in AD. 55 In 2005, a clinical investigation programme aimed at replacing endogenous albumin of patients with mild to moderate AD with human albumin through TPE was developed with the purpose of altering the dynamic equilibrium between albumin-bound Aβ in plasma and Aβ in cerebrospinal fluid (CSF). In a pilot proof-of-concept study (EudraCT#: 2005-001616-45), 7 patients underwent 6 PE in 3 weeks and 1 year of follow-up. 56 During each plasma exchange procedure, the full plasma volume was removed from the patient and replaced by a similar volume of 5% human albumin. Plasma Aβ determinations demonstrated a variation pattern in levels in relation with the TPEs. Cognitive status scores evaluated remained stable in comparison with the decline that would be expected in the normal course of the disease. Six patients previously enrolled in the pilot study participated in an extension study. All patients except one completed the cycle of 6 plasma exchanges in 3 weeks in an outpatient regimen. Results of neuroimaging studies showed a favourable trend consistent with that observed in the pilot study. 56
Characteristics of therapeutic approaches with therapeutic plasma exchange and low volume plasma exchange in Alzheimer´s disease.

AD: Alzheimer‘s disease; CSF: Cerebrospinal fluid; LVPE: Low volume plasma exchange; TPE: Therapeutic plasma exchange
Phase II Clinical Trial in AD
In view of the favourable results in the previous studies, a phase II clinical trial (EudraCT#: 2007-000414-36; ClinicalTrials.gov ID: NCT00742417) was designed to determine whether TPE with albumin replacement was able to modify Aβ concentrations in CSF and plasma as well as to improve cognition in patients with mild-moderate AD. 57 In a multicenter, randomized, patient- and rater-blind, controlled, parallel-group, phase II study, 42 AD patients were assigned (1:1) to TPE treatment or control (sham TPE) groups. Treated patients received a maximum of 18 TPE with 5% albumin with three different schedules: two TPE weekly for three weeks, one TPE weekly for six weeks, and one TPE every two weeks for 12 weeks, plus a six-month follow-up period. CSF Aβ1−42 levels after the last PE compared to baseline were marginally higher in PE-treated group versus controls (p = .072). Plasma Aβ1−42 levels were lower in the TPE-treated group after each treatment period (p < .05). Patients treated with TPE scored better in the Boston Naming Test and Semantic Verbal Fluency (p < .05) throughout the study. Neuropsychiatric Inventory scores were higher in controls during the PE phase (p < .05). TPE with human albumin modified CSF and plasma Aβ1-42 levels. Patients treated with TPE showed improvement in memory and language functions, which persisted after the plasma exchange procedures, were discontinued. 57 Overall, data indicated that a TPE schedule was to be selected, one TPE per week for 6 weeks should be therapeutically equivalent to two TPE per week for 3 weeks. A longitudinal neuroimaging evaluation of the patients included in the previous studies revealed that patients with AD showed decreased brain volume and impairment of brain perfusion as expected for the progression of the disease. Treatment with TPE and albumin replacement favoured the stabilization of brain perfusion in these patients. 58
Phase IIb/III AMBAR Study
On the basis of the promising results in the previous clinical investigations, a further clinical programme was designed. The AMBAR (
Comparative Volumes of Plasma Exchanged, Frequency and Duration of Treatment in AD
Table 5 provides comparative information on the plasma volumes exchanged in the AMBAR programme as well as in the different clinical conditions described in this review. It can be roughly estimated that that total volumes of plasma exchanged for indications treatable with LVPE approaches are one third of those used for indications for which TPE has become a standard. The plasma volume removed in the LVPE procedure of the AMBAR study is similar to a plasma donation (650-880 mL) and depends on patient‘s weight. Volumes of plasma exchanged per TPE or LVPE procedures in AD patients, are comparable with those used in more acutely presenting pathologies (TTP, GBS, GPS, ALF). However, in contrast to TPE and to other LVPE procedures, cell separation in AMBAR LVPE is discontinuous (ie, first the plasma is separated from the cells and albumin is infused afterwards). This implies that the volume of removed plasma is identical to the volume of albumin solution infused. Therefore, the proportion of pathologic plasma with respect to infused albumin removed in LVPE (100%) is more efficient than in TPE (60%-80%; see Figure 1B).
Comparative plasma volumes exchanged during therapeutic plasma exchange or low volume plasma exchange for the most representative pathological conditions described in this review.

LVPE: Low volume plasma exchange; TPE: Therapeutic plasma exchange: TTP: Thrombotic thrombocytopenic purpura; GBS: Guillain-Barré syndrome: AIDP: Acute inflammatory demyelinating polyradiculoneuropathy; AD: Alzheimer‘s disease
Safety of TPE and LVPE
Apheresis procedures are essentially safe. 63 Adverse events (AEs) are considered as mild, moderate (need for medication), severe (interruption due to the AE) or death (due to AE). The majority of AEs occur during the first sessions, are usually mild and affect 2.4% of the patients (problems with IV access, hypotension, and hypocalcemia). Moderate AEs occur in 3% (hypocalcemia, urticaria, hypotension and nausea). The majority of the AEs reported are related to the use of citrate-containing anticoagulants (hypocalcemia) and the replacement fluid used (plasma or albumin). 64 Severe AEs are rare but may happen in .4% of procedures (syncope/hypotension, urticaria, chills, fever, arrhythmia, nausea and vomiting). Potentially life-threatening adverse reactions can be even rarer (0.12% incidence). 65 Intensive TPE implies the exchange of large volumes of plasma and requires a reliable central access. Procedures performed through a central access are bound to more severe AEs. Infections from central accesses may be critical for the patient and complicate the continuation of the plasma exchange procedures. The frequency and severity of AEs during studies with TPE in AD patients 57 seems compatible with those previously mentioned for conventional TPE approaches.
LVPE -as performed for the majority of the conditions described in this review- appears as a less invasive procedure for the patient but is more dependent on hand-operated procedures including, multiple blood drawing, use of plastic bags, centrifugation, sedimentation and re-infusion. The number of serious AEs is apparently low. 33 Hypotension has been reported to occur in 58% of the patients treated with LVPE in an study including a reduced number of patients. 33 Islam et al. 22 reported a severe sepsis in one patient related to a central venous access. Hypoalbuminemia and anemia are more common in patients treated with LVPE. 32 However, considering the context and limitations of some studies using LVPE approaches, it is possible that the number of AEs is underreported. In AD, results from the AMBAR trial proved that both PE and LVPE were feasible and safe in such a sensitive patient population, typically aged and with concomitant pathologies. 60 Being AD a chronic condition, use of LVPE, in which a smaller volume of plasma is removed than in TPE with same efficacy, would be justified.
Mechanisms of Action of Plasma Exchange
Despite the fact that plasma exchange was first described almost 100 years ago 3 and its being widely used nowadays, the understanding of the exact mechanisms of action of TPE is still limited. 65 The main action of conventional TPE is the removal of abnormal substances such as pathologic antibodies, immune complexes, and cytokines, followed by further replacement of plasma with an appropriate fluid. It has been presumed that the removal of these substances represents the major mechanism of action of TPE. A mathematical model for removal of plasma constituents considering a theoretical distribution in one-compartment was early defined. 66 According to this theoretical model, the exchange of 1 or 1.5 TPV should remove about 60% or 80% of the intravascular constituent (Figure 1A). The efficiency of TPE in the real patient depends on the nature of the constituents to be removed (ie IgG vs IgM) and their redistribution in different compartments.
However, the previous mechanisms do not explain the persistence of the favourable response observed in some disorders. Additional evidence suggests that TPE may have an immunomodulatory effect beyond the removal of antibodies or toxic constituents. There have been reports on effects of TPE on immune function including: T-cell modulation with a shift from in the Th1/Th2 balance 67 ; suppression of IL-2 and IFN-γ production68,69; and an increase in induced suppressor cell function. 70 Thus, the beneficial effect of TPE in a wider number of indications could be the consequence of the acute removal of toxic constituents in plasma combined with more prolonged immunomodulatory mechanisms. The removal mechanisms of pathological constituents achieved with LVPE should be obviously reduced with respect to conventional TPE. It is possible that the activation of immunomodulatory mechanisms mentioned before may have a greater contribution to the favourable outcomes of LVPE approaches in the pathologic conditions mentioned in this review.
The mechanism of the beneficial action of TPE and LVPE in patients with AD is being investigated. As mentioned earlier, Aβ seems to play a critical role in the pathogenesis of AD. An increase in the levels of Aβ induces Aβ-fibril formation, and the accumulated Aβ fibrils develop into senile plaque, causing neurotoxicity and induction of tau pathology, that leads to neuronal cell death and neuro degeneration.55,71 Strategies aimed at decreasing the production of the Aβ protein, preventing its aggregation, facilitating its removal from the circulation and reducing levels of Aβ in the brain are being considered. The effectiveness of these approaches have been controversial, though very recent studies with specific antibodies are shedding some light on the Aβ pathophysiology associated with reduction of Aβ plaque.72–75 It is pertinent in this context to bear in mind that, in some pathologic conditions treated with TPE, the replacement fluid used during plasma exchange seems to be an additional contributing mechanism. 65 TTP would be an example in which ADAMTS-13 is supplied with the FFP used for replacement. 12
It has been suggested the therapeutic albumin used as replacement during TPE and LVPE in AD could partially explain the favourable action of these therapies in patients with AD. Human albumin inhibits Aβ self-association by selectively binding to Aβ aggregates and by preventing further growth of the Aβ. 76 Albumin oxidation has been found dramatically increased in CSF of patients with AD when compared to healthy controls. 77 Recent data support the role of glycation and oxidative stress in AD. 78 Albumin consistently binds peptides containing the primary sequence of human Aβ. 79 Albumin infusion could disrupt further nitro-glycative modifications of circulating albumin that can induce Aβ aggregation. 80 Removal of Aβ protein from the circulation and prevention of its aggregation by albumin could be additional mechanisms explaining de favourable action of plasma exchange in AD.
It is important to consider that, after more than one hundred years from the first published description, 3 knowledge on mechanisms and rationale for TPE have evolved for the different pathological conditions. The ideal situation is generated when there is a strong correspondence among the pathophysiology of the disease, the rationale for TPE and the clinical benefit. 81 However, there have been situations in which the rationale for TPE was unclear, but there was evidence of clinical efficacy. One example of the later is TTP, which moved from one category to another when its pathophysiology was finally recognized. There are pathologies in which controlled trials seem to support efficacy, but the rationale is not clear. This is the most challenging category, and the one that requires more investigations on their pathophysiology. 81 AD could be one of such pathologies where TPE and LVPE may function as effective therapies prior to a better understanding of its complex pathophysiology.
Caution and Limitations
Although plasma exchange (implying fluid replacement) and plasmapheresis (not necessarily requiring fluid replacement) are two different procedures, some authors use both terms interchangeably. The incorrect usage of the terms by authors may lead to incorrect categorization of the procedures.
The terms small- low- or reduced-volume plasmapheresis, which refer to very similar procedures, have been used by the authors of some original communications commented in this review. These approaches -by definition- involve the removal of a plasma volume lower than that of a standard plasmapheresis donation (400-600 mL) without fluid replacement, therefore not definitely implying plasma exchange. For consistency, ‘small volume plasmapheresis’ has been used throughout the text.
TPE procedures are performed daily or every other day, depending on the severity of the conditions. Volumes of plasma exchanged per session and frequency of the procedures are tapered down once the symptoms improve. In contrast to the precise plasma exchange schedule of the AMBAR study, the total volumes of plasma exchanged for the other different pathologies can only be approximated within an estimated range. Similarly, volumes of plasma exchanged, and frequency of procedures are not accurately provided for some of the LVPE approaches described in previous sections of this review. Whenever possible, the volumes based on the terms used by the authors within the context of the pathologies reported have been estimated.
Several communications cited in the present review refer to abstracts, proceedings or posters presented in meetings, or are reported in old articles, journal supplements, or in journals of local scope, for which the full text of the presentation or the paper was not available.
Conclusions
Conventional TPE has become a first line treatment for a wide number of pathologies and is the elective therapeutic approach for a selected number of pathological conditions. The effectiveness of TPE is particularly evident in a wide spectrum of hematologic and neurological diseases and supported by strong clinical evidence referred in guidelines from reputed International Societies.
Volumes of plasma treated for the most relevant indications for which TPE is considered a first line of treatment range from 3-4.5 L per procedure. Frequency and duration vary depending on the pathology treated, but in general extends from 1 to 3 weeks, with procedures being carried out daily or every other day. Total volumes exchanged during a full treatment with TPE may range from 12 to 49 L.
Less conventional therapeutic approaches using lower plasma exchange volumes (LVPE) seem to be clinically effective in a number of pathological conditions. The majority of the reported studies have focused on neuroimmunological diseases. The strength of the clinical evidence of these studies is lower than that supporting the main indications for TPE. Volumes of plasma treated for those pathologies for which LVPE approaches have been evaluated range from 350 mL to around 2 L per procedure. Frequency and duration of the treatments are variable depending on the pathology treated. Total volumes exchanged during a full treatment with LVPE may range from 5 to 10 L. It can be approximated that total volumes of plasma exchanged for indications treatable with LVPE approaches are one third of those used for indications for which TPE has become a standard.
A combined sequence of intensive TPE followed by a maintenance phase with LVPE has been investigated as a possible treatment for patients with AD. Volumes of plasma exchanged during TPE and LVPE were 2.5-3 L and 650-880 mL, respectively. While volumes of plasma exchanged per TPE or LVPE procedures in AD patients, are comparable with those used in more acutely presenting pathologies (TTP, GBS, GPS, ALF) duration of the treatments is more extended in AD (weeks to months) and intervals between procedures more prolonged (weekly or monthly) than when they are applied to the treatment of classic pathologies with a more acute course. The total number of procedures carried out for TPE and LVPE in AD are similar to those applied to the treatment of other pathologies for which these plasma exchange strategies have proven successful, though in the case of AD, procedures are performed less frequently though maintained for longer periods and more sustained in time. Such a chronicity issue would justify the use of LVPE strategies.
Footnotes
Abbreviations
Author contribution(s)
Acknowledgements
Jordi Bozzo PhD CMPP (Grifols) is acknowledged for editorial support in the preparation of this manuscript.
Competing Interests
GE has received honoraria/consultant fees from Bayer, BMS, Boehringer Ingelheim, CSL Behring, Grifols, and Terumo (BCT). AP is a full-time employee of Grifols, a manufacturer of plasma-derived albumin products. JC has received speaker/advisory fees from Cerus, Fresenius kabi, Grifols, MacoPharma, Pharm-Olam International, Sanofi, and TerumoBCT.
