Abstract
BACKGROUND
Vaccines have emerged as a crucial tool in combatting the COVID-19 pandemic particularly those based on SARS-CoV-2 S-protein mRNA. A crucial aspect of vaccine efficacy is the duration of antibody responses. In this study, a seroconversion panel was created to assess antibody responses to the mRNA-1273 vaccine over time (6.5 months).
METHODS
Blood samples collected from 15 healthy adult participants prior to and up to 6.5 months after vaccination with the mRNA-1273 vaccine (Moderna). Serum from these blood samples were analyzed for anti-SARS-CoV-2 antibody activity by chemiluminescent immunoassay.
RESULTS
The immunoassay results showed that one participant was positive for anti-SARS-CoV-2 antibodies prior to vaccination indicating a prior infection. All participants showed a positive antibody response after the first vaccination. Highest antibody responses were seen after the second dose (41-45 days from the first dose). Subsequent samples collected at 69-75 days, 130-135 days, and 221-229 days after the first vaccination showed positive responses but a biphasic decline in the levels of anti-SARS-CoV-2 antibodies.
CONCLUSIONS
Declining antibody levels in these participants support the use of booster vaccination to increase antibody levels 4-6 months after the initial vaccine series and continued monitoring to assess the durability of COVID-19 vaccine responses. These results are in agreement with other studies showing antibody persistence but declining the antibody levels in the months after immunization with mRNA-based vaccines. The seroconversion panels described here could be useful in the development of antibody assays and in the assessment of the duration of antibody responses to vaccine boosters compared to the initial vaccine series. This panel could also be used to assess antibody activity against emerging viral variants (eg B.1.1.529 [Omicron] and its subvariants) compared to earlier variants.
Introduction
The SARS-CoV-2 pandemic has been a major public health challenge since its emergence in December 2019. 1 Globally research has taken many approaches to vaccine development including mRNA, DNA, protein, and non-replicating virus technologies. 2
The mRNA-1273 (Spikevax, Moderna) is approved in the US for adults (≥18 years old) and available under an Emergency Use Authorization for children over six months and as a booster. In Europe, mRNA-1273 is approved for individuals over 6 years old. This vaccine is administered as two doses (100 µg) of pre-fusion stabilized spike protein mRNA given four weeks apart. 3
In this study, seroconversion panels were constructed from blood samples collected from study participants before and up to 6.5 months after vaccination and analyzed for anti-SARS-CoV-2 antibody responses. The goal of the study was to assess the anti-SARS-CoV-2 antibody response to mRNA-1273 vaccination over time.
Material and Methods
Seroconversion Panel Collection
The samples which make up these seroconversion panels were collected from volunteer healthcare workers at a Morristown, Tennessee (USA) hospital. All volunteers provided informed consent. This study was conducted under an approved IRB protocol ([SDP-003] Human Biological Specimen Collections: Diagnostic Investigational Review Board, Cummaquid, MA, USA)) . This study was conducted in compliance with all applicable regulatory guidelines.
This longitudinal panel was comprised of undiluted, unpreserved serum specimens collected from 15 participants between 25 February 2021 and 14 October 2021. Vaccine administration for this study was conducted under the original emergency use authorization (EUA) for mRNA-1273 and the dosing regimen described therein. 4 The small number of participants in this study reflect that vaccine supplies were extremely limited at the time the study was conducted. Additional (booster) vaccine doses had not been recommended or authorized at that time. 5 The participants were 15 healthy adults 30-60 years of age who had received two injections of mRNA-1273 SARS-CoV-2 vaccine (100 µg) with an objective target of 28 days apart. There were nine male and six female participants in this group. All participants were white/Caucasian.
The samples for each participant were collected from before the first dose of the mRNA-1273 vaccine to 6.5 months after the second dose. In detail, samples are collected prior to the first vaccination (between 0 to 3 days: sample 1, pre-vaccination samples), prior to the second vaccination (between 2 to 7 days: sample 2), after the second vaccination (between 13 to 15 days; sample 3), 1.5 months after the second vaccination (between 41 to 45 days; sample 4), 3.5 months after the second vaccination (between 103 to 107 days; sample 5), and 6.5 months after the second vaccination (between 194 to 199 days; sample 6).
The serum samples were divided into 1 mL aliquots and frozen at −20 °C until they were assayed. Samples were thawed at room temperature and mixed by inversion prior to testing. The serum samples were tested for anti-SARS-CoV-2 antibody activity (S1 and S2 IgG) with a chemiluminescent immunoassay according to the manufacturer's instructions (CLIA: Liaison SARS-CoV-2 IgG Assay, Diasorin, Inc, Saluggia, Italy: EUA approved). The details of the CLIA have been previously published. 6 Results of the assay are expressed as arbitrary units/mL (AU/mL) and the threshold/cutoff value was determined as described in the manufacturer's assay instructions.
Results
Figure 1 shows the dynamic changes in antibody response against SARS-CoV-2 in a longitudinal seroconversion panel from 15 participants. Sample were collected before and after vaccination and up to 6.5 months after the second vaccine dose. In this figure, time 0 corresponds to the sample obtained before the first vaccination and the time shown for subsequent samples is number of days elapsed since collection of the pre-vaccinated sample.
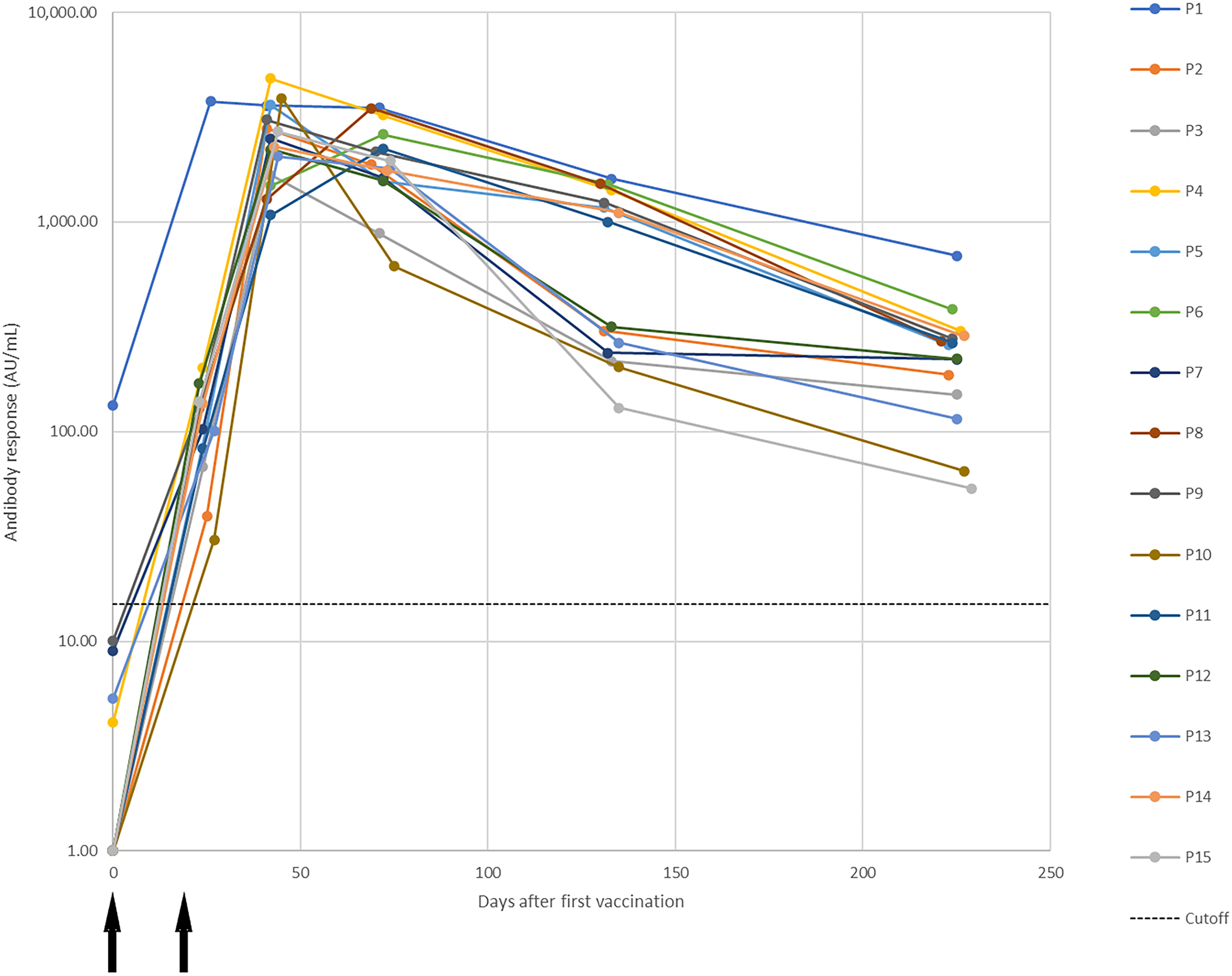
Dynamic changes in antibody response against SARS-CoV-2 in a longitudinal seroconversion panel from 15 participants (P1–P15) before and after vaccination with two doses of the mRNA-1273 SARS-CoV-2 vaccine (Moderna), indicated by arrows. Samples were collected prior to vaccination and up to 6.5 months from the second vaccine dose. IgG antibody activity results were obtained using a chemiluminescent immunoassay and are expressed as arbitrary units/mL (AU/mL). Responses ≥ 15.0 units were considered positive. Time 0 correspond with the sample obtained before the first vaccination dose and the time of the subsequent samples are the elapsed days after the pre-vaccinated sample.
Testing of pre-vaccination samples gave the following results (Figure 1: day 0): 14 negative and one positive result (Participant 1). The presence of anti-SARS-CoV-2 IgG in this participant indicates that the participant was previously infected with the virus prior to collection of this sample.
Samples collected after the first injection showed 15 positive results (all participants). (Figure 1: 23-27 days). Samples collected after the second dose (Figure 1: 41-45 days) also showed 15 positive results with the highest antibody responses. Samples collected 1.5 months post second dose (Figure 1: 69-75 days) also showed 15 positive results. The antibody responses have started to decrease at this point. Samples collected 3.5 months post second dose (Figure 1: 130-135 days) showed positive results for all participants and a decrease in antibody responses compared with the previous sample. At 6.5 months post second dose (Figure 1: 221-229 days) all participants showed positive results and the antibody response was still decreasing compared with the previous sample collection.
When the decline in antibody activity after peak response was examined (Figure 2), a biphasic decline was noted. An initial rapid decline of approximately three orders of magnitude was observed over the first 30-60 days after the peak antibody response. This was followed by a slower decline over the remaining sample collection period (up to 150 - 199 days). The pattern of the decline in antibody activity was similar for all participants.

Decrease in antibody responses after peak antibody activity in a longitudinal seroconversion panel from 15 participants (P1–P15) after vaccination with two doses of the mRNA-1273 SARS-CoV-2 vaccine (Moderna). Peak antibody response was set as Day 0 for each participant and the decline in antibody response was evaluated over the next 152-199 days (221-229 days after first injection).
Conclusions
The results of this study showed that only one participant had an antibody response before the administration of the first dose of the mRNA-1273 SARS-CoV-2 vaccine. This positive response indicates a prior infection with SARS-CoV-2. All samples showed positive antibody responses after the first dose of the vaccine and all samples collected after 6.5 months (post-second dose) were still positive. These results show a robust response to the first dose of the mRNA-1273 vaccine that is enhanced by the second dose of vaccine. The antibody response wanes over the post-vaccination period suggesting that humoral immunity declines in the months after vaccination.
These results agree with previous published observations which showed antibody persistence through 6 months after the second dose of the mRNA-1273 vaccine and a steady decay in antibody activity over time.7, 8 Early studies of antibody responses to the mRNA-1273 vaccine showed that over the short-term (∼60 days after the first injection), antibody activity remained high.9, 10 Later studies have shown that anti-S protein antibodies decline over a longer post-vaccination observation period (3-5 months).11, 12 In addition, the biphasic decline in anti-S protein antibodies in the current study, parallels the observation of a biphasic decrease in antibody activity observed after vaccination with the BNT162b2 mRNA vaccine. 13
An observational study looking at the real-world effectiveness of the mRNA-1273 vaccine (n = 352 878 per group) showed that there was a small increase in SARS-CoV-2 infections in vaccinated individuals 4-5 months after vaccination. Hospitalizations in the vaccinated group were less than 1/10 of those seen in the unvaccinated group and the number of deaths were far less in the vaccinated group (n = 1) than in the unvaccinated group (n = 25). 14
Since this study was conducted, new studies have shown a decline in vaccine effectiveness over time likely due to a combination of decreased antibody activity and the emergence of resistant variants.15–17 These factors have led to the recommendation of booster doses for continued protection.
Limitations of the current study include the small study population, which was due, in part, to the limited availability of vaccine at the time the study was conducted (25 February 2021 and 14 October 2021). In addition, the study examined antibody binding only via the CLIA and did not include neutralization studies. No attempt was made to correlate antibody responses with disease severity due to the small number of participants and the even smaller number of participants anticipated to be infected with COVID-19 after vaccination. No provision for follow-up examination, testing or surveillance was included in the study protocol.
These seroconversion panels can have utility in the development of antibody assays, the assessment of the duration of antibody responses to vaccine boosters compared to the initial vaccine series and activity against emerging viral variants (eg B.1.1.529 [Omicron] and its subvariants) compared to earlier variants.
Footnotes
Acknowledgements
Michael K. James, PhD is acknowledged for medical writing and Jordi Bozzo, PhD, CMPP for editorial assistance.
Author contributions
Availability of Data and Materials
The data underlying this manuscript is available from the corresponding author upon reasonable request. The seroconversion panel is available from Access Biologicals.
Conflicts of Interest
FB and OM are employees of Grifols. RC and MC are employees of Access Biologicals.
Funding
These studies were supported by Grifols (Barcelona, Spain) and Access Biologicals (Vista, CA, USA).
Ethics Approval and Consent to Participate
The samples which make up these seroconversion panels were collected from volunteer healthcare workers at a Morristown, Tennessee (USA) hospital that had provided informed consent. This study was conducted under an approved IRB protocol ([SDP-003] Human Biological Specimen Collections: Diagnostic Investigational Review Board, Cummaquid, MA, USA).
