Abstract
Background:
Patients undergoing Bariatric and Foregut procedures are at increased risk of complications from postoperative nausea and vomiting (PONV) and pain. In this study, we investigated enhancement of standard Enhanced Recovery After Surgery (ERAS) protocol with fosaprepitant and paragastric autonomic nerve blockade (PGANB) with bupivacaine liposome injectable suspension (BLIS).
Methods:
We analyzed 91 patients undergoing Bariatric surgery and/or Hiatal Hernia Repair (HHR) who received standard ERAS protocol alone (Group S), with fosaprepitant (Group F), or with fosaprepitant and PGANB (Group FP). We collected patient demographics, PONV Intensity Scale ratings, pain scores, and PRN pain medications at 6 and 24 hour time points.
Results:
Kruskal-Wallis testing revealed differences in PONV across intervention groups (P = .033). Post-hoc analysis with Conover-Iman (pCI), Cliff’s delta (Δ), and Cohen’s D (d) showed decreased PONV in Group F compared to Group S (pCI = 0.028, Δ = 0.280, d = 0.508). LOS for any Bariatric surgery decreased (P = .046) by approximately 19.9 hours in group F versus group S (pCI = 0.062, Δ = 0.424, d = 0.657) and by approximately 11.2 hours in group FP versus group S (pCI = 0.084, Δ = 0.389, d = 0.416). HHRs showed no significant intervention-dependent differences in LOS or PONV.
Conclusions:
Our results demonstrate positive associations between the addition of fosaprepitant to standard ERAS protocols in Bariatric patients and PONV control. Further investigation into the role of fosaprepitant is warranted in Foregut and Bariatric patients. Further studies should also focus on optimizing anesthetic agent selection and multimodal analgesia, including PGANB, in the context of enhanced ERAS protocols with fosaprepitant.
This is a visual representation of the abstract.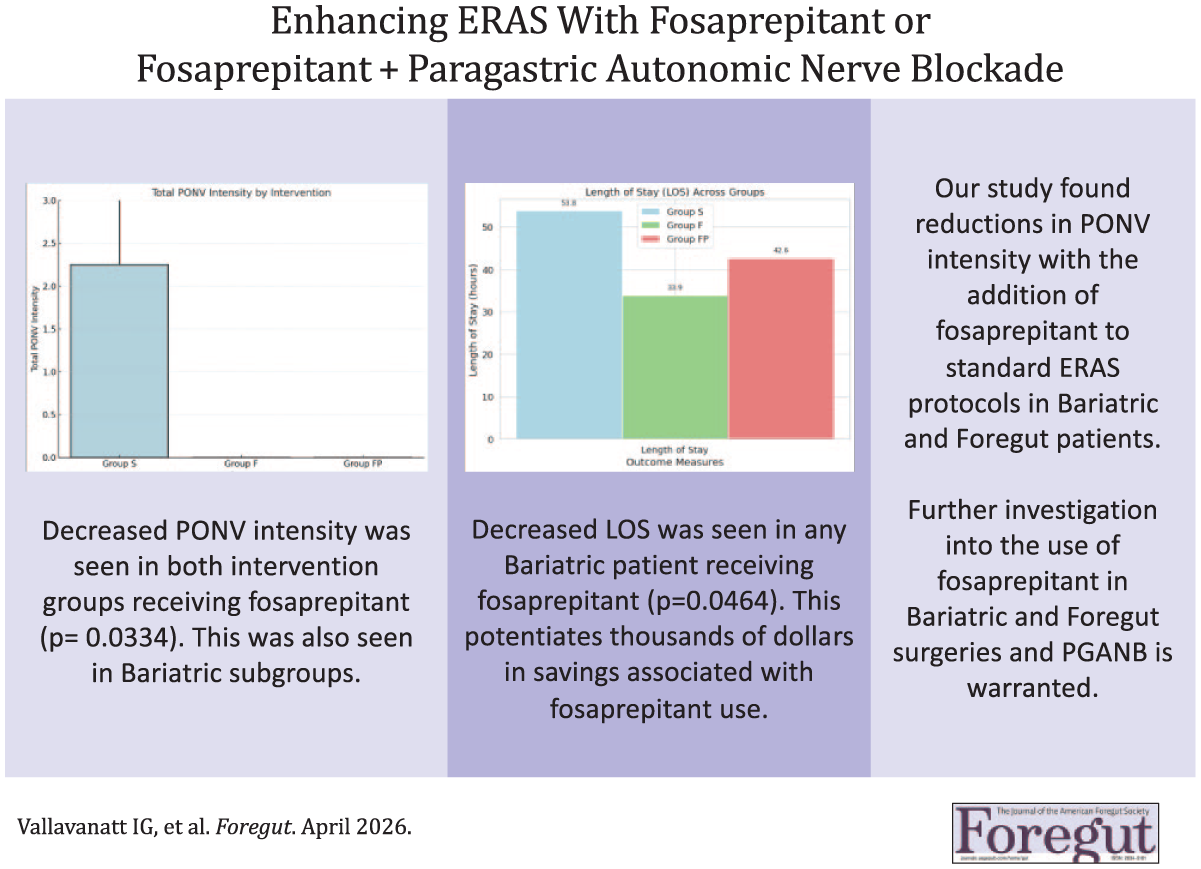
Key Learning Points
Fosaprepitant can decrease the incidence of PONV and LOS in Bariatric patients.
Fosaprepitant remains clinically promising for PONV control in Foregut patients and should be investigated further.
Additional study is warranted to determine effectiveness of PGANB for PONV control in Bariatric and Foregut patients.
Background
Patients undergoing Bariatric and Foregut procedures are at risk for increased complications related to postoperative nausea and vomiting (PONV) or pain-related sequelae. Additionally, PONV and pain increase patient discomfort and dissatisfaction, length of stay (LOS), readmission rates, and patient and hospital costs. In fact, PONV has been reported in up to 80% of patients 24 hours after abdominal surgery and is the most common reason of readmission within 30 days. 1
Postoperative pain is often controlled with opioids with well described side effects that can be particularly detrimental to Bariatric and Foregut patients.2,3 Current management of PONV utilizes pharmacologic agents postoperatively such as 5-hydroxytryptamine-3 receptor antagonists and dopamine D2 receptor blockers, such as ondansetron and prochlorperazine respectively. These agents have varying efficacy and duration, further necessitating investigation into the control of PONV and pain. 4 Fosaprepitant (Emend™) is a neurokinin-1 (NK-1) receptor antagonist that functions centrally and within the GI tract to limit nausea and reduce the incidence of PONV by blocking the binding of neuropeptide Y. Fosaprepitant has been shown to decrease the incidence of vomiting in other types of surgery; current literature on its use preventing PONV and its sequelae in Bariatric patients and Foregut patients is promising, though limited.5-7
Furthermore, autonomic nerve blockades have been investigated in the prevention of PONV associated with visceral pain. Daes et al demonstrated that patients receiving paragastric autonomic nerve blockade (PGANB) following laparoscopic sleeve gastrectomies (SG) with 0.5% bupivacaine were found to have significantly lower pain at 1 and 8 hours postoperatively as well as decreased incidence of PONV and opioid requirements. 3 Since these effects were limited to the 24 hour period, we explored the same PGANB with the longer acting bupivacaine liposome injectable suspension (BLIS) which we have used successfully for local TAP blocks in our practice.
Thus, a need exists to determine best practice for mitigating PONV and visceral pain in Bariatric and Foregut surgery patients. Primarily, our aim was to determine if the use of fosaprepitant enhanced the beneficial effects of our current ERAS protocol. Secondarily, given the previously demonstrated success with 0.5% bupivacaine PGANB, we wanted to explore the potential prolonged benefit with BLIS. The purpose of this study is to assess the effectiveness of our standard Enhanced Recovery After Surgery (ERAS) protocol in conjunction with fosaprepitant alone or fosaprepitant in combination with PGANB to reduce PONV and pain in this population.
Methods
Study Design
This is a retrospective analysis of prospectively collected data that was collected from December 27, 2022 to July 19th, 2024 at a tertiary care center. Patients were studied in 3 intervention groups and retrospectively reviewed: a standard ERAS protocol control group (S), a fosaprepitant + standard ERAS protocol group (F), and a fosaprepitant + PGANB + standard ERAS protocol group (FP). Patients were allocated into intervention groups sequentially until each group totaled 30 patients. There were no major changes in clinical care during the study period.
Study Population
91 patients undergoing Bariatric surgery including SG, Roux-en-Y Gastric Bypass (RYGB), Single Anastomosis Duodeno-Ileal Bypass with Sleeve Gastrectomy (SADI-S) with or without HHR were included (Table 1). Five patients underwent concomitant cholecystectomy. Patients that were undergoing revision of previous Bariatric or Foregut surgery, had an Edmonton Obesity Scale Severity of 4, preoperative narcotic usage or other pain management regimen, or having concomitant procedures other than those listed above were excluded.
Baseline Characteristics by Surgical Group and Intervention.
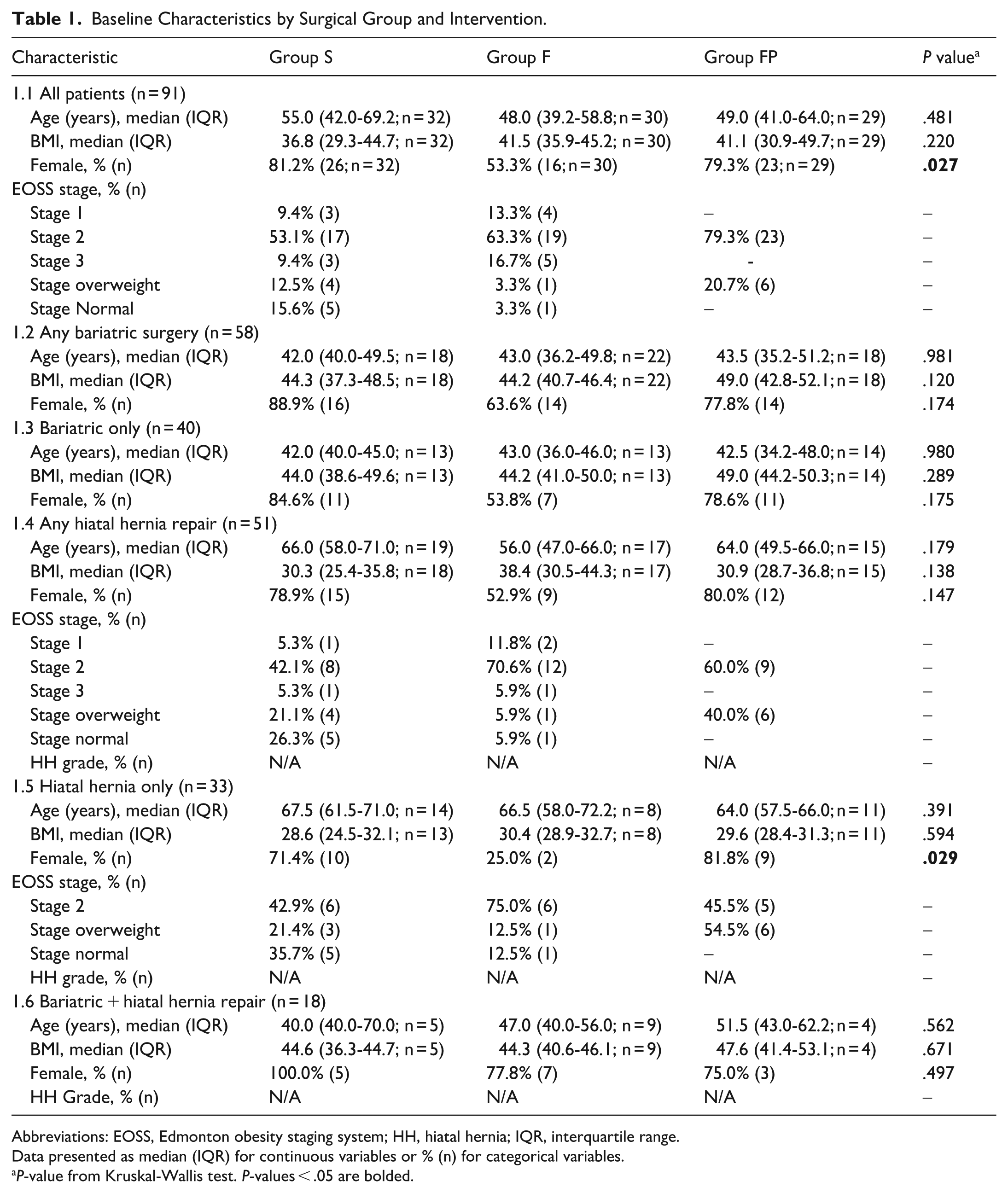
Abbreviations: EOSS, Edmonton obesity staging system; HH, hiatal hernia; IQR, interquartile range.
Data presented as median (IQR) for continuous variables or % (n) for categorical variables.
P-value from Kruskal-Wallis test. P-values < .05 are bolded.
Study Outcomes
The primary outcomes were patient reported presence, onset, severity, timing of nausea, and episodes of vomiting. These were collected via the validated PONV Intensity Scale (Cronbach’s α = .78, Figure 1). Secondary outcomes included rescue doses of analgesic, rescue doses of antiemetic medications, and 10-point pain score. Patient outcomes were collected via the validated PONV Intensity Scale (Cronbach’s α .78, Figure 1) in-person, over the phone, or via nurse communications or through the electronic medical records at 6 and 24 hours postoperatively.

Validated postoperative nausea and vomiting (PONV) intensity scale, a patient-reported tool used to quantify the severity of PONV and guide antiemetic treatment.
Statistical Analysis
Data and statistics were analyzed and visualized using Python PANDAS, Seaborn, and Microsoft Excel. Google Gemini AI assisted with drafting code for analysis and visualization. We performed our statistical analysis by first assessing for normality using the Shapiro-Wilk test. As none of the intervention groups had a normal distribution, we selected the Kruskall-Wallis test, a 1 way non parametric ANOVA for comparing 2 or more independent samples of equal or different sample sizes. Next, we conducted pairwise comparisons of each intervention for each type of surgery using the Conover-Iman test. P values from Kruskall-Wallis testing are noted as pKW and P-values from Conover-Iman testing are noted as pCI. For our post-hoc analysis, we determined effect sizes using Cohen’s D (noted as d) and Cliff’s Delta (noted as Δ). The following cutoffs were used for Cliff’s Δ: small ≥ 0.11, moderate ≥ 0.28, large ≥ 0.43. The following cutoffs were used for Cohen’s D magnitudes: small ≥ 0.2, moderate ≥ 0.5, large ≥ 0.8. Our subgroup analysis followed the same parameters.
Perioperative ERAS Protocol
Each patient received the standard ERAS protocol at minimum. Per our ERAS protocol, patients received gabapentin elixir 250 mg, heparin 5000 Units subcutaneously, acetaminophen elixir 1 g, and antibiotic prophylaxis preoperatively. Additionally, patients less than 65 years of age received a scopolamine patch (1 mg). Those patients receiving fosaprepitant received 150 mg intravenously 1 to 4 hours prior to surgery. Intraoperatively, patients received dexamethasone 10 mg and ondansetron 4 mg.
Paragastric Autonomic Nerve Blockade
BLIS (20 ml 1.3% BLIS diluted in 60 ml 0.9% normal saline) was infiltrated with 2 ml per injection with an Interjet needle in all accessible regions near the anterior vagus nerves (Figure 2) and posterior vagus nerve (Figure 3), the anterior and posterior branches of the vagus nerve along with the lesser sac, and in the preperitoneal space over the left and right crus. We blocked lesser omentum vagal branches (always anterior; posterior when accessible, as in a sleeve), peri-vagal (anterior and posterior when accessible in a hiatal dissection), and preperitoneal along the right and left crura (when dissected in a hiatal hernia repair). Our goal was to infiltrate all accessible potential areas.
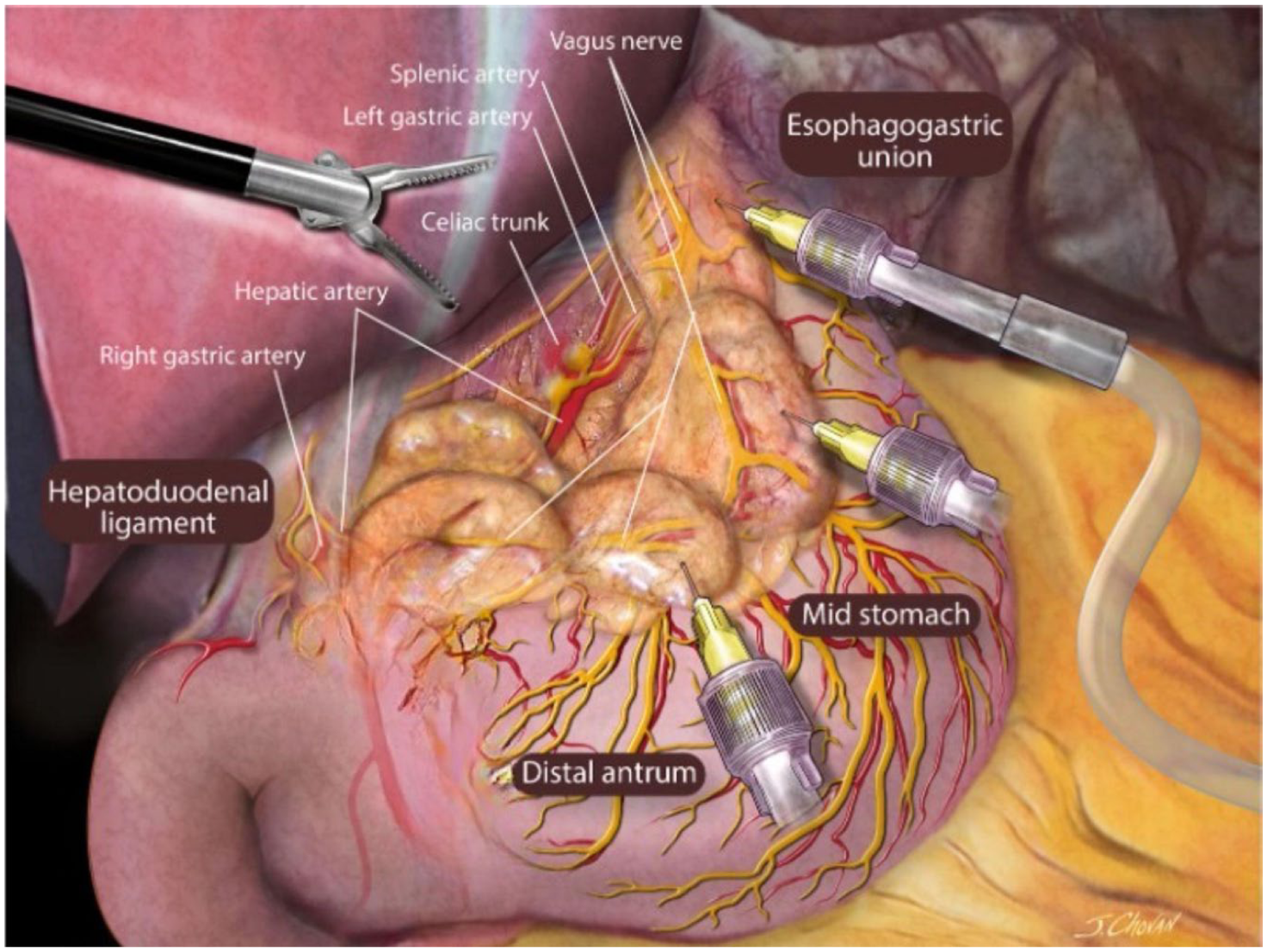
Areas of injection in proximity to the anterior vagus nerve and branches in patients receiving PGANB.
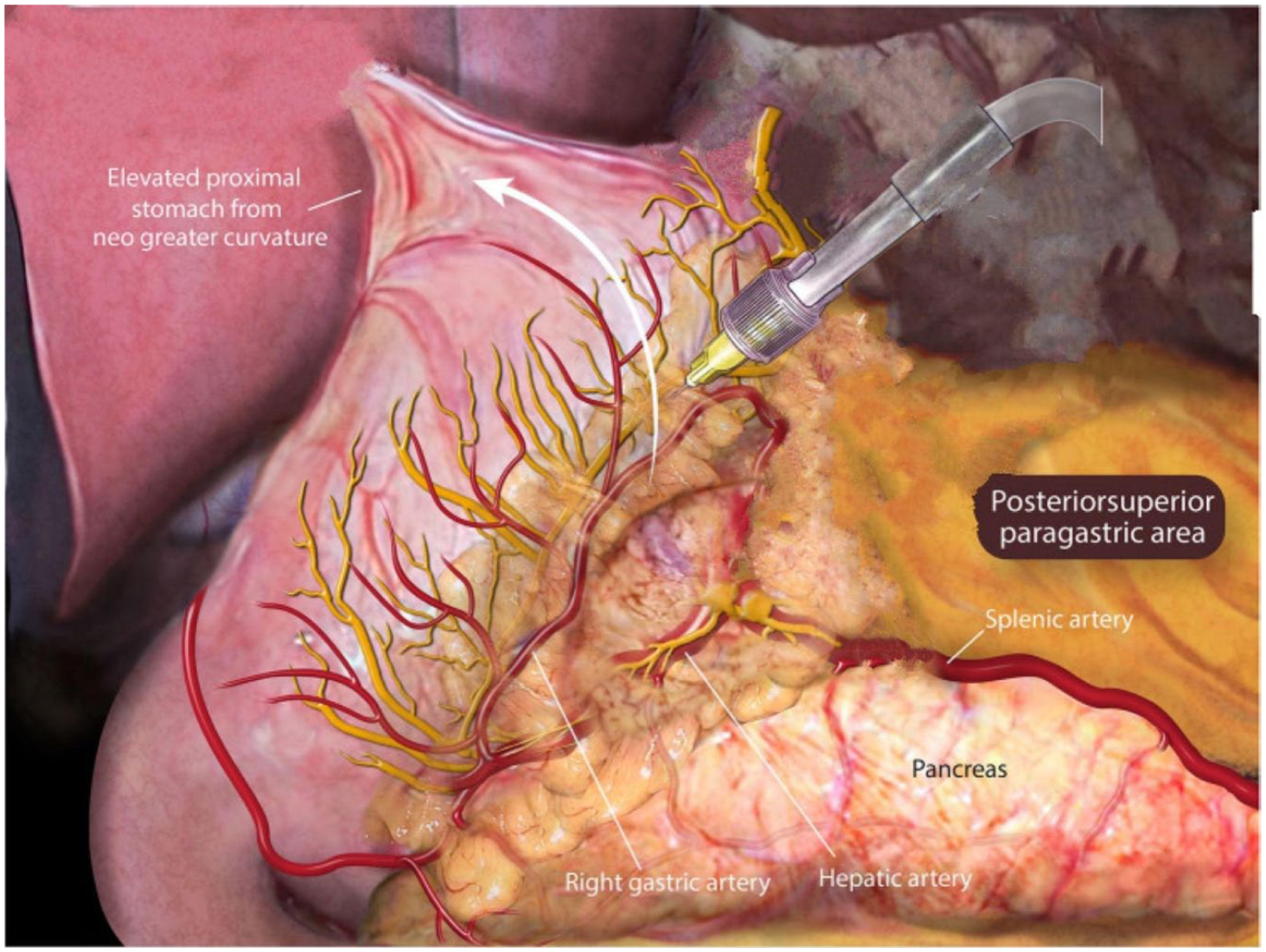
Areas of injection in proximity the posterior vagus nerve and branches in patients receiving PGANB.
Surgical Procedures
All procedures were done under general anesthesia and utilized the minimally invasive robotic surgery platform. 1.3% BLIS was injected locally at all incisions. When used for PGANB, 1.3% BLIS was injected in all described and available areas (Figures 2 and 3).
Postoperative Protocol
Postoperatively, magnesium sulfate 2 g was administered intravenously. Patients were initiated on a sugar-free clear liquid diet at least 2 hours after the completion of surgery. Ambulation also began after this. CPAP was utilized in PACU and on the floors per STOP BANG protocol. Ondansetron 4 mg was given intravenously every 4 hours in the first 24 hours after surgery and then as needed after that. Prochlorperazine 10 mg every 8 hours was given for nausea breakthrough as needed. Either hydromorphone 0.25 mg, hydrocodone/acetaminophen elixir, or codeine/acetaminophen elixir were given every 3 to 4 hours as needed for pain.
Results
We reviewed 91 patients according to the interventions they received perioperatively. Across all surgeries, significantly less PONV intensity was observed in the intervention groups compared to Group S (Table 2.1, pKW = 0.033, χ2 = 6.796). Post-hoc analysis revealed Group F had superior antiemetic efficacy compared to Group S (pCI = 0.028, Δ = 0.280 [small], d = 0.508 [moderate]). Group FP did not demonstrate significant findings in post-hoc analysis compared to Group S (pCI = 0.235, Δ = 0.170 [small], d = 0.296 [small]).
Postoperative Outcomes by Surgical Group and Intervention.
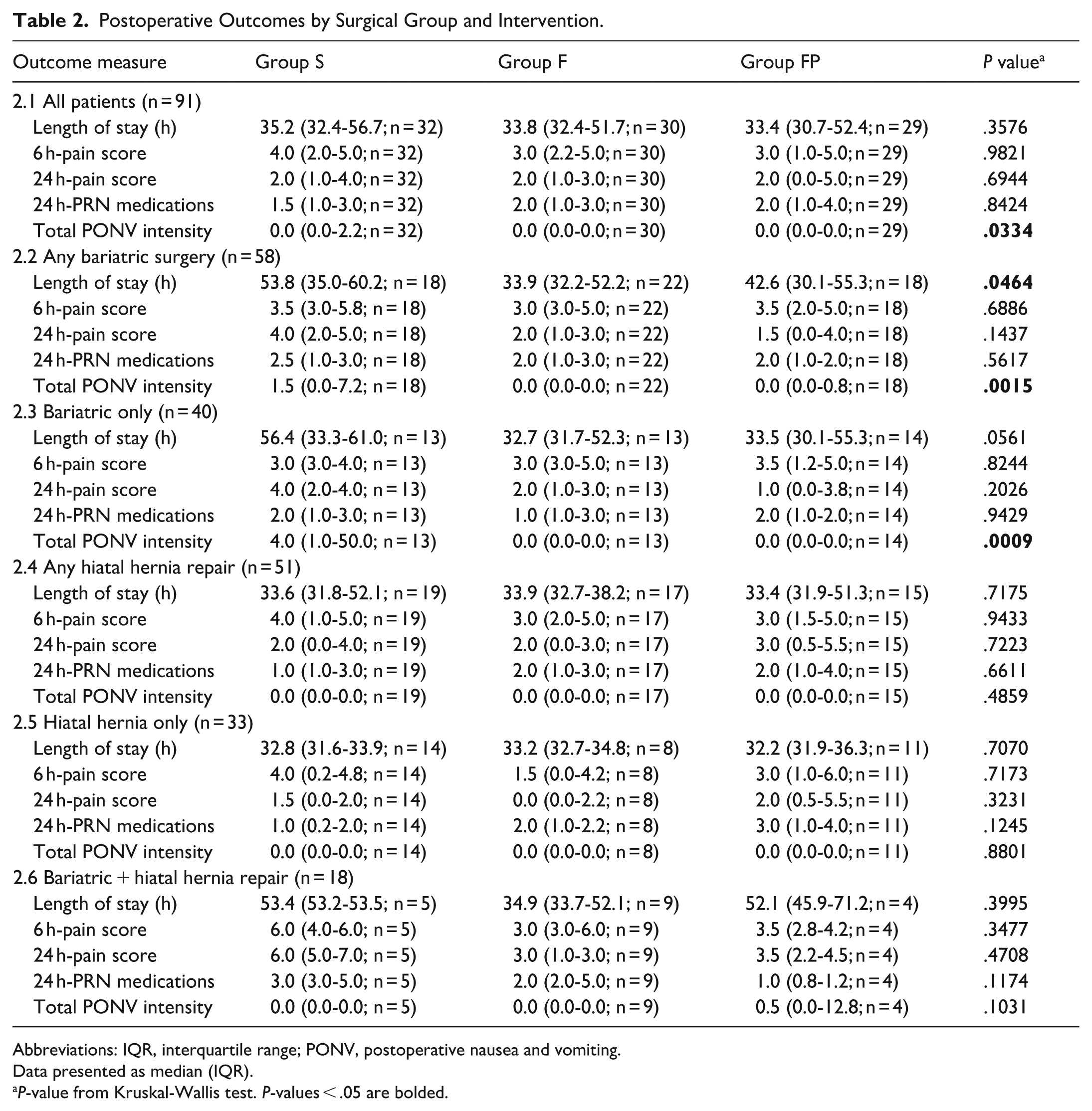
Abbreviations: IQR, interquartile range; PONV, postoperative nausea and vomiting.
Data presented as median (IQR).
P-value from Kruskal-Wallis test. P-values < .05 are bolded.
When examining intervention groups by surgery performed, we found in patients undergoing any Bariatric surgery (Table 2.2, pKW = 0.002, χ2 = 12.981), Group F exhibited strong antiemetic control compared to Group S (pCI = 0.001, Δ = 0.543 [large], d = 0.801 [large]). Group FP demonstrated moderate reductions compared to Group S (pCI = 0.036, Δ = 0.349 [moderate], d = 0.458 [small]). This was consistent in patients undergoing only Bariatric surgeries (Table 2.3, pKW = 0.001, χ2 = 13.979) in both Group F (pCI = 0.0004, Δ = 0.687 [large], d = 0.952 [large]) and Group FP (pCI = 0.002, Δ = 0.566 [large], d = 0.782 [moderate]) compared to Group S. These results were observed despite modest sample sizes.
Patients undergoing any HHR (Table 2.4, pKW = 0.458, χ2 = 1.443) and only HHR (Table 2.5, pKW = 0.880, χ2 = 0.2554) showed no significant intervention-dependent differences in PONV intensity. Effect sizes were negligible in these groups. In combined Bariatric and HHR (Table 2.6, pKW = 0.1031, χ2 = 4.5433), Group FP had higher PONV intensities compared to Group S (pCI = 0.389, Δ = 0.350 [moderate], d = 0.772 [moderate]); however, these differences did not reach statistical significance. Further, our post-hoc effect sizes showed this same subset (Table 2.6, pKW = 0.1174, χ2 = 4.2841) had higher 24-PRN pain medication requirements (pCI = 0.133, Δ = 0.800 [large], d = 1.450 [very large]).
Notably, we found reductions in LOS for patients undergoing any Bariatric surgery (Table 2.2, pKW = 0.046, χ2 = 6.141) and only Bariatric surgery (Table 2.3, pKW = 0.056, χ2 = 5.761) compared to Group S. Post-hoc Conover-Iman comparisons in any Bariatric showed the following reductions: 33.9 versus 53.8 hours for Group F versus Group S (pCI = 0.062, Δ = 0.424 [medium], d = 0.657 [moderate]) and 42.6 versus 53.8 hours for Group FP vs Group S (pCI = 0.084, Δ = 0.389 [medium], d = 0.416 [small]). We did not observe the same reductions in LOS in patients undergoing any HHR (Table 2.4, pKW = 0.718, χ2 = .664), HHR only (Table 2.5, pKW = 0.707, χ2 = 0.694), or in combined HHR and Bariatric procedures (Table 2.6, pKW = 0.400, χ2 = 1.835). Post-hoc Conover-Iman comparisons were not significant and effect sizes were small to negligible in HHR subgroups.
Analysis of the baseline characteristics using the Kruskall-Wallis test revealed that more female patients were sequentially allocated to Group S or Group FP compared to group F by the stochastic nature of sequential allocation (pKW = 0.027). Our subgroup analysis included analyses by demographics, including age, sex, and BMI. This analysis revealed limited statistically significant differences in surgical outcomes across intervention groups for the majority of patient subgroups examined. We observed notable LOS reductions in younger patients receiving fosaprepitant-based interventions (pKW = 0.005, χ2 = 10.744) with Group F (pCI = 0.007, Δ = 0.680 [large], d = 0.879 [large]) and Group FP (pCI = 0.008, Δ = 0.621 [large], d = 0.819 [large]) compared to Group S. Younger patients also demonstrated decreased PONV intensity (pKW = 0.003, χ2 = 11.354) in both Group F (pCI = 0.002, Δ = 0.621 [large], d = 0.771[moderate] and Group FP (pCI = 0.024, Δ = 0.467 [large], d = 0.596 [moderate]) compared to Group S. Further, patients with class I obesity (pKW = 0.003, χ2 = 11.557) in Group F (pCI = 0.002, Δ = 0.446 [large], d = 0.654 [moderate]) and Group FP (pCI = 0.088, Δ = 0.289 [moderate], d = 0.412 [small]) experienced less PONV intensity compared to Group S. There were no significant findings when analyzing patients further by any characteristics.
Discussion
In this study, we anticipated reduced PONV intensity in groups receiving fosaprepitant based on recent literature and demonstrated effects of NK-1 receptor antagonism. Our results are consistent with existing and emerging evidence that fosaprepitant is effective in reducing PONV intensity in patients undergoing Bariatric surgeries. 9 Further, our results may be associated with significant reductions in LOS beyond those seen with standard ERAS protocols. 8 When considering the estimated cost-benefit of fosaprepitant, the reduction in LOS by approximately 19 hours seen in Group F and 11 hours in Group FP compared to group S suggests a significant financial advantage. Estimating that 1 hour of inpatient postoperative stay equates to approximately $1886, this approximates to an average savings of approximately $2756.58 for Group F and savings of $1624.93 for Group FP for an average Bariatric surgery patient undergoing an in-patient procedure in the United States. 8 The recent introduction of generic fosaprepitant has reduced its costs to around $30 per vial. Even when accounting for drug and inpatient cost variation between institutions and local practice patterns, this could translate into additional hundreds to thousands of dollars in savings per patient.8,9
In Foregut patients, we did not observe a significant reduction in PONV intensity or LOS with fosaprepitant use. This discrepancy may be due to several factors, including small sample sizes, lower baseline LOS in this population, and greater variability in the nature of Foregut procedures compared to Bariatric procedures. Although no significant reductions were observed in Foregut patients, the magnitude of effect sizes observed in our Bariatric cohort supports further investigation into the addition of fosaprepitant to ERAS protocols, especially considering the estimated cost-benefit seen in our Bariatric patients.
Finally, we evaluated the addition of a PGANB to enhanced ERAS protocol with fosaprepitant to determine if there was additional benefit to the combination of interventions. Daes et al found that the control group had higher visual analog scale scores, increased prevalence of PONV, and required three times as many opioid doses compared to the treatment group when using 0.5% bupivacaine. 3 Since effects were limited to the 24 hour period, we pursued further investigation with BLIS, which has a 48 to 72 hour half-life, to prolong blockade duration. In doing so, we found no significant reduction in pain scores at 6 and 24 hour time points across Bariatric and Foregut groups. However, our data was limited by a very small subset of patients (n = 4), and our study varied from the retroperitoneal injection site in the Daes study, possibly contributing to a difference in outcomes. The use of BLIS in the relief of visceral pain may still have utility in Bariatric and Foregut surgical patients, with or without fosaprepitant, and should be investigated further and with larger sample sizes. Recent breakthroughs in multimodal anesthesia also compel further study, especially in conjunction with regional nerve blockade benefits for PONV mitigation. 10
Interestingly, we had a large cohort of female patients posing a potential confounder. Analysis of the 58 bariatric surgery patients in this study revealed demographic characteristics largely consistent with national benchmarks. The proportion of female patients (75.9%, 44/58) did not differ significantly from the 79.3% reported by Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) between 2015 and 2017 (pKW = 0.518, χ2 = 0.42). Similarly, the mean preoperative Body Mass Index (BMI) in our sample (45.27 ± 6.80 kg/m2) was statistically comparable to the MBSAQIP mean of 46.6 kg/m2 (One-Sample t(57) = −1.49, P = .143). These findings suggest that our study sample is representative of the broader Bariatric surgery population described in the MBSAQIP database regarding patient sex distribution and baseline BMI.
While the results of our study are interesting and provide considerations for emerging ERAS protocols and further study, the results should be interpreted in the context of its limitations. Because sequential cohort design cannot establish treatment efficacy, our findings represent associations within a protocol-evolution study. Further studies are warranted to validate our observations and address limitations inherent in the group allocation process and the limited sample size. Our post-hoc analysis suggested potential subgroup-specific benefits of fosaprepitant, particularly in younger and higher BMI patient populations; again, further study is warranted to validate these conclusions due to our limited sample size. Local practice patterns may have also influenced LOS outcomes and additional studies including multiple institutions may better estimate true cost savings. In this study, we included 3 different Bariatric procedures (SG, RYGB, and SADI-S) which have their own varying propensities for PONV. Further, multiple procedures in the same operation necessarily results in increased abdominal trauma which can increase PONV, pain, and LOS. In subsequent studies, we suggest analysis by specific surgical procedure outside of studies investigating concurrent procedures to better isolate intervention responses. Objectively assessing subjective outcomes like PONV and pain is challenging on its own and highly variable as patient perceptions are influenced by a multitude of individual factors such as age and gender. 2 Finally, it is important to note that this study was conducted at a single practice within an emerging academic center in a suburban area with limited resources and support for research.
Conclusion
Our results demonstrate positive associations between the addition of fosaprepitant to standard ERAS protocols in Bariatric patients and PONV control. Further investigation into the role of fosaprepitant is warranted in Foregut and Bariatric patients. Further studies should also focus on optimizing anesthetic agent selection and multimodal analgesia, including PGANB, in the context of enhanced ERAS protocols with fosaprepitant.
Footnotes
Acknowledgements
The authors would like to thank the Daes team for allowing the use and editing of their paragastric nerve blockade images to visualize BLIS injections for our readers. The authors would also like to thank Gregory Wilson, MD and Eiline Cai, MD for their contributions to data collection and Will Choi, MD; Tam Tran, MD; Joke’ Fasoranti, PharmD for their contributions to study design.
Ethical Considerations
Patients signed surgical consent forms per institutional policies. This was a retrospective review of prospectively collected data. This study was conducted under institutional and international ethical standards, and although IRB approval was not required, the study adhered to the local institution IRB guidelines.
Author Contributions
Study design: TAW, JMJ, GA, MY. Patient recruitment: TAW, JMJ. Data collection: TAW, JMJ, MY, IGV. Statistical analysis: CJ, IGV. Data interpretation: CJ, IGV, TAW, JMJ. Manuscript writing: IGV, CJ, TAW, JMJ, MT. Manuscript revision: IGV, TAW, CJ, JMJ, MT. Image editing: MT. Primary surgeon: TAW. Final approval of version submitted for publication: all authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Use of Artificial Intelligence
Google Gemini AI assisted with drafting code for analysis and visualization which was reviewed and disclosed per journal policy.
