Abstract
The Endoluminal Functional Lumen Imaging Probe (EndoFLIPTM) impedance planimetry system is a tool that allows for real-time, objective feedback of gastrointestinal sphincter geometry in both the gastrointestinal suite and the operating room. Eight experienced foregut surgeons and one gastroenterologist, with a total of over 3750 FLIP cases, reviewed published data, unpublished data, and personal experience to update protocols for FLIP impedance planimetry system use in the operating room during fundoplication, magnetic sphincter augmentation (MSA), laparoscopic Heller myotomy (LHM), and peroral endoscopic myotomy (POEM). Additionally, they created standardized protocols for FLIP impedance planimetry system use in the operating room during peroral pyloromyotomy (POP), cricopharyngeal myotomy (CPM), and for collecting FLIP Panometry (ie, esophageal motility evaluation) in either the gastrointestinal (GI) suite or the operating room.
Introduction
The Endoluminal Functional Lumen Imaging Probe (EndoFLIP™) impedance planimetry system is a tool that allows for real-time, objective feedback of gastrointestinal sphincter geometry in both the gastrointestinal suite and the operating room. A previously published expert consensus statement established protocols for standardized use in the operating room during several common foregut procedures. 1 At the time of the consensus group’s meeting in 2019 there remained wide heterogeneity in protocols used in the published literature, limiting the group’s ability to provide recommendations related to target values correlated with patient outcomes. Since then, use of FLIP technology in the operating room has become more widespread and standardized. In an effort to describe target values and protocols for expanded practice use, as well as to review data that have been published to date, an expert panel was reconvened.
Eight experienced foregut surgeons (Drs. Shahin Ayazi, Christy Dunst, Jon Gould, Min P. Kim, Kirsten Newhams, Aaron N. Sachs, Paul Severson, and Michael B. Ujiki), with a total of over 3750 FLIP cases, reconvened in Nashville, Tennessee on September 23rd, 2021. The group included authors of the original expert-based consensus published in 2021 and Dr. Ayazi, who has published extensively on his use of FLIP technology in foregut surgery. 1 All were selected based on expertise and experience in using FLIP technology in foregut surgery. Conflicts of interest were declared up front, with specific attention to relevant industry conflicts. The group reviewed published data, unpublished data, and personal experience to update protocols for FLIP impedance planimetry system use in the operating room during fundoplication, magnetic sphincter augmentation (MSA), laparoscopic Heller myotomy (LHM), and peroral endoscopic myotomy (POEM). Additionally, they created standardized protocols for FLIP impedance planimetry system use in the operating room during peroral pyloromyotomy (POP), cricopharyngeal myotomy (CPM), and for collecting FLIP Panometry (ie, esophageal motility evaluation) in either the gastrointestinal (GI) suite or the operating room. After convening in Nashville, Tennessee, Dr. Carlson collaborated with the group of foregut surgeon to lend his expertise with FLIP panometry. Following the in-person meeting in Nashville, Tennesse, iterations of the working document were circulated amongst all authors for comment until unanimous consensus was reached. As limited high-quality evidence is available, this methodology was pursued. Institutional review board approval and consent were not required as the conference did not require patient participation or patient health information.
The FLIP Impedance Planimetry System
The FLIP system is described in detail in Supplemental File 1. Values reported on the FLIP display include the minimum diameter in mm (Dmin), intra-bag pressure in mmHg (P), narrowest cross-sectional area in mm2 (CSA), distensibility index in mm2/mmHg (DI), and compliance in mm3/mmHg (C). 2 When deciding which catheter is appropriate for use during a procedure, the below protocols describe published target values for use of an 8 cm (EF-325) or a 16 cm (EF-322) catheter. If information about esophageal motility is required at the time of the procedure, the 16 cm (EF-322) catheter is set up.
FLIP Panometry
FLIP Panometry, the measurement and visual depiction of secondary esophageal peristalsis induced by catheter-based stepwise endoluminal volumetric distension, can be measured with a 16 cm (EF-322) catheter and the FLIP 2.0 or FLIP 300 system. Secondary esophageal peristalsis differs from primary esophageal peristalsis in that it is the response of the esophagus to endoluminal distension rather than passage of a bolus. Further description of the software required for use of FLIP panometry can be found in Supplemental File 2.3,4
First described in 2014, panometry has been well described in the gastrointestinal literature and has been shown to correlate with high-resolution manometry/Chicago Classification version 4.0 esophageal motility diagnoses when performed during upper endoscopy.5-9 The ability to detect major motility disorders using panometry has spurred efforts to establish and validate a formal secondary motility classification system, though high-resolution maneomtry remains the gold standard. 10 Although panometry classification and its clinical correlation with the Chicago Classification v4.0 system are being refined, there are general principles of assessment to determine normal motility from a spectrum of diminished or disordered contractile responses. 4 A guide to interpreting panometry findings, including normal contractile response, impaired/disordered contractile response, spastic-reactive contractile response, and absent contractile response can be found in Supplemental File 2. Any panometry findings that do not correlate with the clinical picture should spur further evaluation with HRM.
Minimal literature exists on the use of intraoperative panometry and the effects of general anesthesia on esophageal motility. The authors have noted absent or severely diminished secondary esophageal peristalsis when halogenated anesthetic gases such as sevoflurane are used, discordant with manometry and panometry findings obtained preoperatively. An impact of sevoflurance on FLIP findings has been reported. 11 However, other endoscopic sedation with propofol or midazolam with fentanyl was shown to not significantly impact the relationship of FLIP Panometry with awake HRM. 12 Panometry has been found in a single center pilot study to be highly sensitive, 95.7%, to detect disorders of esophageal primary peristalsis when performed at the start of procedure during laparoscopic anti-reflux surgery. 13
FLIP System Configuration and Placement
After initial set up (Supplemental Video 1), the catheter is purged and zeroed to atmospheric pressure while being held in a horizontal position. Zeroing should be performed immediately prior to catheter placement. The catheter is typically placed trans-orally in a manner similar to an orogastric tube. The catheter should readily pass. If significant or unexpected resistance is encountered, in order to maintain the integrity of the catheter and avoid any inadvertent esophageal injury, the authors recommend use of endoscopy to confirm catheter placement. For placement across the pylorus, it is helpful to manually guide the FLIP catheter across the sphincter with an endoscopic snare. At the upper esophageal sphincter (UES), esophago-gastric junction (EGJ) and pylorus, the sphincter should be centered on the screen, generating an hourglass.14-18
For panometry, a 16 cm (EF-322) catheter is used. After initial set up, the catheter is purged, zeroed to atmospheric pressure, and placed trans-orally, in the same manner as described above. Again, the authors recommend endoscopic guidance if any unexpected resistance to catheter passage is encountered. The catheter should be positioned to have one to three distal sensors within the stomach, with the remaining sensors positioned along the length of the esophagus (Supplemental Video 2).
Previous publications have documented significant fluctuation in FLIP output under varying conditions in the operating room. With different volumes of probe fill, patient positioning, and presence or absence of pneumoperitoneum the FLIP output varies significantly.19,20
Filters
As previously descried by Su et al, 1 there are several different filter settings available on the FLIP system. The following protocols, with the exception of panometry, utilize a high-weighted filter, with use of the low-weighted filter at the time of initial catheter placement for improved sphincter identification. For panometry, the filter is turned off.
Data Points
While distensibility index is the most highly reported metric provided by the FLIP impedance planimetry system, it remains unclear which parameter(s) correlate best with patient outcomes. Therefore, the authors continue to recommend documentation of Dmin, P, DI, and C for all procedures, at a standardized fill. Though target ranges are reported for DI only in this manuscript, we would encourage all users to continue to investigate target values of Dmin, P, DI, and C that may help predict clinical outcomes. At this time, Dmin can also be of particular use when ruling in or out esophagogastric junction outflow disorders.
The company user guide recommends a minimum intra-bag pressure of >15 mmHg, while the authors recommend a minimum intra-bag pressure of >20 mmHg when using the calculated distensibility index to ensure appropriate apposition to the walls of the esophagus.1,21,22 The authors recommend significant caution when interpreting any values where the P did not reach a minimum threshold of 20 mmHg, as more reliable, reproducible measurements occur when this threshold is met.
FLIP Protocol During Hiatal Hernia Repair and Fundoplication or Magnetic Sphincter Augmentation (MSA)
The ability of FLIP technology, in conjunction with upper endoscopy, to correctly identify the EGJ and evaluate EGJ competence makes it an ideal adjunct for use during anti-reflux surgery. By ensuring all wraps are performed on the esophagus and providing real-time evaluations of EGJ configuration following crural closure and fundoplication or MSA placement, postoperative quality of life outcomes are optimized and there is less need for revisional surgery. 1
There are minimal data on FLIP measurements of the EGJ in normal controls, with several small studies reporting on 8 cm (EF-325) catheter use with a 30 mL fill. Nathanson et al 20 reported on 50 patients without symptoms of gastroesophageal reflux disease (GERD) undergoing laparoscopy with general anesthesia and found a median Dmin of 6.3 mm, median CSA of 31 mm2, median DI of 1.4 mm2/mmHg, and median C of 42.9 mm3/mmHg. From the gastroenterology (GI) literature, Kwiatek et al 23 evaluated 20 healthy controls undergoing EGD with moderate sedation and found a median EGJ DI of 4 mm2/mmHg (1-14 mm2/mmHg). In a study of 20 healthy volunteers at time of high-resolution manometry, median EGJ DI at 30 mL fill was found to be 5.1 mm2/mmHg (0.8-21.7 mm2/mmHg). 24 It is important to note that surgical studies like the one from Nathanson et al, 20 measured EGJ DI under CO2 insufflation and, thus, this likely accounts for the lower EGJ DI seen compared to those measured by gastroenterologists without capnoperitoneum.
Additional publications on FLIP measurements of the EGJ in normal controls include seven studies reporting on 8 cm (EF-325) catheter use with a 40 mL fill. Kwiatek et al 23 reported on 20 healthy controls undergoing EGD with moderate sedation, and found a median EGJ CSA of 264 mm2 (5-95th percentile, 99-496 mm2). Rieder et al 25 evaluated four healthy controls at the time of manometry testing with no anesthetic administered and found a median EGJ DI of 2.7 mm2/mmHg (IQR 2.4-8.3 mm2/mmHg). Fukazawa et al 26 also evaluated healthy awake patients undergoing manometry, and found the mean DI in their cohort of nine to be 7.1 mm2/mmHg (±SE 0.0 mm2/mmHg). Pandolfino et al 27 evaluated 20 healthy controls at time of EGD with moderate sedation and found a median DI of 8.2 mm2/mmHg (5th-95th, 1.7-18.7 mm2/mmHg). Smeets et al 28 evaluated 15 healthy controls during EGD without sedation and found a median DI of 3.0 mm2/mmHg (IQR 2.7-4.2 mm2/mmHg). In 2016, Mikami et al 29 evaluated eight healthy, awake controls undergoing FLIP evaluation at time of manometry and found a mean DI of 3.5 mm2/mmHg (±SE 0.4 mm2/mmHg). In 2018, Mikami et al 30 again evaluated a new cohort of eight healthy, awake controls undergoing FLIP evaluation at time of manometry and found a mean DI of 4.5 mm2/mmHg (±SE 0.5 mm2/mmHg). Of note, in both studies, Mikami et al modified the DI calculation by using the equation DI = CSA/(P + intragastric pressure offset) rather than the value displayed on the FLIP monitor. Bredenoord et al 31 published a meta-analysis on FLIP measurements in normal controls with a mix of EF-322 and EF-325 catheter data demonstrating 5th to 95th percentile EGJ-DI at a 40 mL fill 1.96 to 10.95 mm2/mmHg and at a 50 mL fill 2.86 to 10.66 mm2/mmHg.
FLIP measurements of the EGJ in normal control using a 16 cm (EF-322) catheter are published primarily in the GI literature. A cohort of 35 asymptomatic controls was described using panometry and measurements across the EGJ with a 60 mL fill during a sedated upper endoscopy. In their study, median EGJ-DI was 5.8 mm2/mmHg (5th-95th percentiles 3.2-8.4 mm2/mmHg), and all participants had a DI >2.8 mm2/mmHg. Of note, asymptomatic was defined as lack of symptoms including dysphagia, heartburn, and chest pain as well as no history of antiacid medications, BMI <30 kg/m2, and no tobacco or alcohol use disorder. 14
There is variation in reported FLIP values of the EGJ in patients affected by GERD, hiatal hernia, or paraesophageal hernia. However, patients with GERD tend to have less competent lower esophageal sphincters resulting in lower P and higher Dmin, CSA, and DI of the EGJ. Hiatal and paraesophageal hernias will often displace the EGJ into the thoracic cavity, away from the diaphragmatic crura, leading to lower P and higher Dmin, CSA, and DI of the EGJ.
Protocol for Intraoperative Intermittent Measurement
• Measurements with an 8 cm catheter at a 30 or 40 mL(preferred for the EGJ) fill volume to maintain consistency with measurements obtained during other procedures and to ensure intra-bag pressure is always >15 mmHg. Note: if using a 30 mL fill, increase to a 40 mL fill as needed when intra-bag pressure does not reach >15 mmHg, and use caution when interpreting FLIP values where the intra-bag pressure reaches 15 to 20 mmHg. If a 16 cm catheter is used, 60 mL is recommended for EGJ-DI measures with the same pressure concerns noted above, with additional filling to 70 mL to assess maximum EGJ diameter if FLIP is being used to evaluate esophageal motility.
• Timepoints to obtain measurements: ○ If motility and/or EGJ morphology has not been previously assessed, or has been questioned, and is desired, a 16 cm catheter can be placed after intubation and before insufflation of the abdomen. In this case, fills of 40-50-60-70 mLs are performed and panometry is assessed (note that filter must be off during panometry assessment). Generally the EGJ-DI is assessed with the filter on high at 60 or 70 mLs. If an 8 cm catheter is used, panometry cannot be assessed but EGJ DI can be assessed without capnoperitoneum with the filter on high and 40 mL fill. ○ After crural dissection and hernia reduction is complete (without insufflation) ■ If DI <1.0 mm2/mmHg in a patient with an EGJ outflow disorder on preoperative manometry and global clinical and radiographic impression of achalasia, consider lower esophageal myotomy,32,33 utilizing FLIP to confirm if a true outflow disorder exists or if elevated IRP is solely due to present of a hernia, and resolves after hernia reduction. ○ After crural closure (ideal range is 2.5-3.5 mm2/mmHg) ○ After fundoplication or MSA placement
• When obtaining measurements: ○ Patient position: reverse Trendelenburg (approximately 30 degrees) to minimize the need to reposition the patient. Positioning does not affect the EGJ DI, however. ○ Capnoperitoneum: no capnoperitoneum (ie, release insufflation) to maintain consistency with FLIP data from the GI literature and allow for comparative follow-up in the GI suite.
Place catheter trans-orally into the stomach (typically 45-50 cm) immediately after zeroing to atmospheric pressure. a) If resistance is encountered when advancing the catheter, utilize jaw thrust and consider placement under endoscopic guidance. Remove the endoscope and any gastric/esophageal insufflation prior to initiating the following protocol.
Inflate the probe while slowly withdrawing the catheter until the hourglass shape is seen on the FLIP monitor. Utilize the low-weighted filter if there is any difficulty identifying the EGJ. Additional maneuvers to help in EGJ identification include palpation with a blunt laparoscopic instrument, direct visualization of the catheter within the stomach on the laparoscopic monitor or direct endoscopic visualization.
Center the hourglass in the middle of the electrodes. If a compliance output is not displaying, further center the hourglass on the monitor. a) Change back to a high-weighted filter.
Leave catheter in place for 30 seconds to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, DI, and C.
Deflate probe and remove or unplug catheter for intermittent use. The catheter will malfunction if exposed to any energy device other than pure bipolar energy whether inflated or not. The probe should either be removed or it can be unplugged when energy is used in the vicinity of the catheter.
Protocol for Use as a Continuous Bougie
• Measurements at a 30 mL and/or 40 mL fill volume. The authors have found 30 mL to be the ideal volume for use as a continuous bougie, and taking measurements at 30 mL fill will minimize the need for changes in volume of fill throughout the procedure. Note: if intra-bag pressure does not reach >15 mmHg, consider increasing the fill to 40 mL. Use caution when interpreting FLIP values where the intra-bag pressure reaches only 15 to 20 mmHg.
• Timepoints to obtain measurements: ○ After crural dissection ± hernia reduction is complete ○ After crural closure ○ After fundoplication or MSA placement
• When obtaining measurements: ○ Patient position: reverse Trendelenburg (approximately 30 degrees) to minimize the need to reposition the patient. ○ Pneumoperitoneum: no pneumoperitoneum (ie, release insufflation) to maintain consistency with FLIP data from the GI literature and allow for comparative follow-up in the GI suite.
Place catheter trans-orally into the stomach (typically 45-50 cm) immediately after zeroing to atmospheric pressure. a) If resistance is encountered when advancing the catheter, utilize jaw thrust and consider placement under endoscopic guidance. Remove the endoscope and any gastric/esophageal insufflation prior to initiating the following protocol.
Inflate the probe to 30 mL or 40 mL while slowly withdrawing the catheter until the hourglass shape is seen on the FLIP monitor. Utilize the low-weighted filter if there is any difficulty identifying the EGJ. Additional maneuvers to help in EGJ identification include palpation with a blunt laparoscopic instrument, direct visualization of the catheter within the stomach on the laparoscopic monitor or direct endoscopic visualization.
Center the hourglass in the middle of the electrodes. If a compliance output is not displaying, further center the hourglass on the monitor. a) Change back to a high-weighted filter.
Leave catheter in place for 30 seconds to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, DI, and C.
If the volume of fill was increased to 40 mL for measurements, deflate the probe to a 30 mL fill and leave across the EGJ to function as a bougie.
With the EndoFLIP across the EGJ and inflated to 30 mL, perform the crural closure and fundoplication. a) For MSA, deflate the EndoFLIP and pull the catheter above the EGJ. Follow manufacturer’s recommendation for device placement. Replace the catheter across the EGJ and obtain measurement at both 30 and 40 mL fill.
After hernia reduction in patients with elevated IRP and concern for possible EGJ outflow disorder on preoperative HRM, FLIP is particularly useful. If a normal or elevated DI is measured after hernia reduction, an unnecessary myotomy can be avoided. If, after hernia reduction, FLIP measurements are consistent with an outflow disorder, myotomy can be performed, eliminating the need for a second operation at a later date.
If the target DI range is not reached after crural closure, additional crural closure sutures can be placed or existing sutures can be removed to adjust the tightness of the repair. Likewise, after fundoplication adjustments to the wrap can be made to adjust the tightness of the repair.
Differences from previously published recommendations for use during hiatal hernia repair and fundoplication or MSA include additional protocol for use as a continuous bougie and a recommended intra-bag pressure of >20 mmHg. It is up to the discretion of the operating surgeon whether to utilize the FLIP catheter for intermittent measurements or as a continuous bougie.
The DI at crural closure may be as important or even more important than the final post fundoplication or MSA placement DI. Attaar et al 36 found that cruroplasty contributed significantly more to decrease in DI during anti-reflux surgery than fundoplication, and postulate that it may be the most important contributor to restoration of a competent lower esophageal sphincter. Stefanova et al 37 also found approximation of the crural diaphragm to contribute more significantly to overall change in DI than fundoplication.
Several studies have reported FLIP measurements during hiatal hernia repair with fundoplication38-44 or MSA,34,43,45,46 with notable heterogeneity in volume of fill, patient positioning, presence of capnoperitoneum, type of fundoplication, and choice of bougie in the protocols used for data collection to date. FLIP measurements taken after hiatal dissection and hernia reduction, as well as after crural closure and fundoplication can guide intraoperative decision making, such as how tight to close the crura, choice and tightness of fundoplication, and the need for lower esophageal myotomy.32,33,35 Of note, patients undergoing hiatal hernia repair who are found to have esophagogastric junction outflow obstruction (EGJOO) on preoperative high-resolution manometry, with symptoms and radiographic findings supporting achalasia, should be considered for possible lower esophageal sphincter myotomy at the time of anti-reflux surgery. The decision to possibly perform myotomy should be directed by intra-operative FLIP, and pre-operative consent should include the possibility of myotomy. If intra-operative DI
Three recent studies report on intraoperative FLIP use during MSA, with Ayazi et al noting a final DI of ≥0.8 mm2/mmHg with a 30 mL fill at time of postoperative EGD to correlate with no significant dysphagia or GERD symptoms.34,45,46
As previously noted, FLIP measurements taken during anti-reflux surgery help to determine the location and competence of the lower esophageal sphincter, as well as the need to tighten or loosen the crural closure. While FLIP can be used during MSA placement, there are limited outcome data to inform targeted FLIP measurements. As opposed to FLIP measurements following fundoplication, which have been shown to persist in the long term, DI after MSA placement has been shown to decrease over time, possibly related to the foreign-body fibrotic reaction.45,46 There are not yet enough data related to postoperative changes in FLIP impedance planimetry following MSA to be able to predict which patients will undergo significant postoperative scarring.
FLIP Protocol During Laparoscopic Heller Myotomy (LHM)
The ability of FLIP technology, in conjunction with upper endoscopy, to correctly identify the EGJ and provide real-time evaluation of adequacy of myotomy make it an ideal adjunct for use during LHM. Probe distension of the esophagus during myotomy also assists in separating muscle fibers and improves visualization of the myotomy plane. FLIP both guides surgeons during planned lower esophageal myotomy and aids in determining the need to perform a myotomy in patients with ambiguous high-resolution manometry. If the DI after hiatal dissection and hernia reduction remains low, <1.0 mm2/mmHg with associated low Dmin <16 mm, a lower esophageal sphincter myotomy should be performed at the time of hiatal hernia repair assuming that the patient has clinical and radiographic findings also consistent with EGJ outflow obstruction. 33
As previously discussed, there are minimal published data on FLIP values for the EGJ in healthy controls, with reported median DI at a 40 mL fill measured with an 8 cm (EF-325) catheter ranging from 2.7 to 8.2 mm2/mmHg.23,25-30 In contrast, derangements of EGJ geometry measured with impedance planimetry have been well described in achalasia, with increased P and decreased Dmin, CSA, DI, and C seen. With an 8 cm (EF-325) catheter at a 40 mL fill, median DI in cohorts of achalasia patients have been found to range from 0.7 to 1.0 mm2/mmHg.25,27,28
Protocol
• Measurements at a 40 mL fill volume to maintain consistency with measurements obtained during other procedures and to ensure intra-bag pressure is always >15 mmHg. Note: use caution when interpreting FLIP values where the intra-bag pressure reaches only 15 to 20 mmHg.
• Timepoints to obtain measurements: ○ After crural dissection and reduction of any concurrent hiatal hernia ○ After myotomy is complete ■ Consider leaving the catheter in place with 20 to 40 mL volume fill during myotomy. This can provide real-time feedback about the change in EGJ DI and assists in separating muscle fibers to improves visualization of the myotomy plane. ■ After myotomy, the catheter should always be placed under visual guidance (endoscopic and/or laparoscopic) to decrease the risk of perforation: ○ After crural closure (ideal EGJ DI range is 2.5 to 3.5 mm2/mmHg) ○ After fundoplication (ideal EGJ DI range is 2.5 to 3.5 mm2/mmHg)
• When obtaining measurements: ○ Patient positioning: reverse Trendelenburg (approximately 30 degrees) to minimize the need to reposition the patient. Positioning does not affect the EGJ DI, however. ○ Capnoperitoneum: no capnoperitoneum (ie, release insufflation) to maintain consistency with FLIP data from the GI literature.
Place catheter trans-orally into the stomach (typically 45-50 cm) immediately after zeroing to atmospheric pressure. a) If resistance is encountered when advancing the catheter for initial measurements or after crural dissection, utilize jaw thrust and consider placement under endoscopic guidance. Remove the endoscope and any gastric/esophageal insufflation prior to initiating the following protocol.
Inflate the probe to 40 mL and slowly withdraw the catheter until the hourglass shape is seen on the FLIP monitor. Utilize the low-weighted filter if there is any difficulty identifying the EGJ. Additional maneuvers to help in EGJ identification including palpation with a blunt laparoscopic grasper, direct visualization of the catheter within the stomach on the laparoscopic screens or direct endoscopic visualization.
Center the hourglass in the middle of the electrodes. a) Change back to a high-weighted filter
Leave catheter in place for 30 second to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, DI, and C.
Deflate probe and remove catheter for intermittent use. Alternatively, the catheter can be left across the EGJ, but use of energy devices other than bipolar energy may cause catheter malfunction and the need for catheter replacement. If energy other than bipolar energy is used, we recommend unplugging the catheter.
If the target DI range is not reached after crural closure, additional crural closure sutures can be placed or existing sutures can be removed to adjust the tightness of the repair. Likewise, after fundoplication adjustments to the wrap can be made to adjust the tightness of the repair.
These recommendations differ from previously published recommendations for use during laparoscopic Heller myotomy in only recording values at a 40 mL fill to more consistently reach an intra-bag pressure of >20 mmHg. 1
Several studies have reported FLIP measurements during LHM. Those that utilized a 30 or 40 mL probe fill and also reported on outcomes are summarized below.
FLIP Protocol During Peroral Endoscopic Myotomy (POEM)
FLIP technology offers similar benefits during POEM as during laparoscopic Heller myotomy. Identification of the EGJ and determining adequacy of myotomy being the two main uses. In addition, EGJ DI after lower esophageal myotomy has been shown to correlate better with treatment failure than the integrated relaxation pressure (IRP) measured on high-resolution manometry.47-49
Protocol
• Measurements at a 40 mL fill volume to maintain consistency with measurements obtained during other procedures and to ensure intra-bag pressure is always >15 mmHg. Note: use caution when interpreting FLIP values where the intra-bag pressure only reaches 15 to 20 mmHg.
• Timepoints to obtain measurements: ○ After intubation, prior to mucosotomy. ○ After myotomy is complete, prior to closure of mucosotomy (ideal EGJ DI range is 2.5-3.5 mm2/mmHg). ■ Once the mucosotomy has been made, the FLIP catheter should always be placed under endoscopic guidance to avoid cannulation of the submucosal tunnel. Advancing the catheter blindly can result in esophageal perforation.
• When obtaining measurements: ○ Patient positioning: flat
Place catheter trans-orally into the stomach (typically 45-50 cm) immediately after zeroing to atmospheric pressure. a) If resistance is encountered when advancing the catheter for initial measurements, utilize jaw thrust and consider placement under endoscopic guidance.
Inflate the probe to 40 mL while slowly withdrawing the catheter until the hourglass shape is seen on the FLIP monitor. Utilize the low-weighted filter if there is any difficulty identifying the EGJ. Direct endoscopic visualization can also aid in EGJ identification.
Center the hourglass in the middle of the electrodes and remove the endoscope and any gastric/esophageal insufflation. a) Change back to a high-weighted filter
Leave catheter in place for 30 second to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, DI, and C.
Deflate probe and remove catheter for intermittent use.
Differences from previously published recommendations for use during POEM include only recording values at a 40 mL fill to more consistently reach an intra-bag pressure of >20 mmHg. 1
If the target DI range is not reached after myotomy, additional myotomy can be considered within the same myotomy, or a second myotomy can be considered, on a case-by-case basis.
Several studies to date have reported on intraoperative and postoperative FLIP measurements for patients undergoing either LHM or POEM and their correlation with outcomes. Those that utilize a 30 or 40 mL fill are summarized. Pandolfino et al 27 found that a DI of <2.8 mm2/mmHg using a 40 mL fill at time of follow up endoscopy following either pneumatic dilation, LHM, or POEM correlated with poor treatment response, defined as Eckardt Score ≥3. Teitelbaum et al 50 found that final DI between 4.5 and 8.5 mm2/mmHg at a 40 mL fill was associated with optimal outcomes, defined as Eckardt score ≤1 and GERDQ score ≤7. These outcomes were based, however, on subjective GERD symptoms which may not always be present, or can be confounded by regurgitation due to esophageal retention, in a patient with achalasia. Ngamruengphong et al 51 found that final CSA and DI at 30 mL fill after POEM were both significantly higher in those with reflux at time of postoperative EGD (99.5 mm2 vs 79.3 mm2, and 11.2 mm vs 10.1 mm, P < .05), CSA ≥80 mm2 for better Eckardt score (<3), <95 mm2 for less reflux esophagitis (not defined well as upper limit). Ilczyszyn et al 52 found that both DI and Urbach Acahalasia Disease Specific Quality of Life Survey scores did not differ between FLIP guided myotomy and gastroscope guided myotomy, but length of myotomy performed was shorter with FLIP (average of 6.0 cm vs standard 11 cm). Yoo et al 53 found that a final DI after POEM of <7 mm2/mmHg (at either a 30 or 40 mL fill), DI <6.6 mm2/mmHg at 30 mL fill, DI <9.9 mm2/mmHg at 40 mL fill correlated with incomplete response, defined as post-operative Eckardt Score >3. Su et al 54 found worse outcomes after LHM or POEM, defined as post-operative Eckardt Score ≥3, with final DI ≤3.1 mm2/mmHg (P = .001) or change in DI ≤3.0 mm2/mmHg (P = .01) at a 30 mL fill. A final CSA >96 mm2 or Dmin >11.0 mm was predictive of worse reflux at 2 years based on RSI scores. Attaar et al 55 found CSA ≥83 mm2 or DI ≥2.7 mm2/mmHg at 30 mL fill post myotomy during POEM correlated with significantly higher rates of esophagitis, without significant differences in GERD-HRQL or RSI responses between groups. Holmstrom et al 56 found higher rates of esophagitis on follow up EGD after Heller with final DI of 9.3 mm2/mmHg versus 4.8 mm2/mmHg. Amundson et al 57 found a post POEM compliance of ≥125 mm3/mmHg at a 40 mL fill to be associated with normal Eckardt scores at 1st and 2nd follow-up.
FLIP Protocol During Peroral Pyloromyotomy (POP)
Gastric peroral endoscopic myotomy (GPOEM), also known as peroral pyloromyotomy (POP), is an endoscopic pylorus-directed treatment for gastroparesis that was first described in humans by Khashab et al. 58 Since that time, it has gained acceptance as an alternative to other pylorus-directed therapies such as endoscopic dilation, botox injection, gastric stimulator implantation, laparoscopic pyloromyotomy, or partial gastrectomy due to its excellent results and decreased periprocedural risk. 59
Normal distensibility of the pylorus in control subjects has been reported as 7.56 mm2/mmHg (SD 6.33 mm2/mmHg) at a 50 mL fill with a 16 cm (EF-322) catheter, with notable discrepancy between men 9.98 mm2/mmHg (SD 7.53 mm2/mmHg) and women 5.23 mm2/mmHg (SD 3.97 mm2/mmHg). 60 Using a 40 mL fill and an 8 cm (EF-325) catheter, normal control pyloric DI has been measured at 25.2 ± 2.3 mm2/mmHg. 61 In comparison, FLIP measurements of the pylorus in roughly 50% of patients with gastroparesis, defined as >10% retention at 4 hours on a gastric emptying study, are abnormal. Namely, gastroparetic patients will have a lower Dmin, higher P, lower CSA and lower DI than controls.62,63
While the anatomy of the pylorus is significantly different than that of the EGJ, it is possible to obtain reproducible measurements with an 8 cm (EF-325) catheter. Due to the acute angulation of the duodenum, and short length of the pylorus, the authors believe that a shorter probe would be ideal, but such a probe does not yet exist. Thus, it is difficult to interpret FLIP data generated with a 16 cm (EF-322) catheter, as the longer probe may be affected more by the acute angulation of the duodenum. 61
Protocol
• Measurements with an 8 cm (EF-325) catheter at a 50 mL fill volume to ensure intra-bag pressure is always >15 mmHg. Note: use caution when interpreting FLIP values where the intra-bag pressure reaches only 15 to 20 mmHg.
• Timepoints to obtain measurements: ○ After intubation, prior to mucosotomy. ○ After pyloromyotomy is complete, prior to closure of mucosotomy. ■ Once the mucosotomy has been made, the FLIP catheter should always be placed under endoscopic guidance to avoid cannulation of the submucosal tunnel. Advancing the catheter blindly can result in gastric perforation ■ Technical failure can result if the measurements are taken after clips are placed to close the mucosotomy.
55
• When obtaining measurements: ○ Patient positioning: flat ○ Insufflation: remove all intraluminal insufflation from the stomach and esophagus.
Under endoscopic guidance, with use of an endoscopic snare, place the catheter trans-orally into the duodenum immediately after zeroing to atmospheric pressure.
Inflate the probe to 50 mL while slowly withdrawing the catheter until the hourglass shape is seen on the FLIP monitor and the probe is directly visualized with the endoscope to be straddling the pylorus.
Center the hourglass in the middle of the electrodes.
Remove all intraluminal insufflation from the stomach and esophagus and withdraw the endoscope into the proximal stomach.
Leave catheter in place for 30 second to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, DI, and C.
Deflate probe and remove catheter.
Altered FLIP measurements seen in gastroparetic patients, representing a stiff, less compliant pylorus, do not differ based on the etiology of the gastroparesis. 63 Treatment of gastroparesis with pyloric intervention such as POP has been shown to increase Dmin, CSA, and DI, with an associated decrease in P.63-71 While POP has been shown to improve pyloric opening, based on improved gastric emptying and changes in FLIP measurements, pylorus closure does not seem to be negatively impacted by POP. 70 Several studies report on FLIP measurements and correlation with patient outcomes following POP. While Gregor, Attaar, and Conchillio did not define target values, they demonstrated that clinical response correlates to changes in FLIP metrics post-pyloromyotomy.64,70,71 Vosoughi et al 67 defined a target CSA at 50 mL fill of >247.6 mm2 for clinical success, defined as ≥1 point decreased in Gastroparesis Cardinal Symptom Index (GCSI) at 1-year post-op, though their recommendations include mixed data of 8 cm (EF-325) and 16 cm (EF-322) catheters. Jacques et al 68 found a post-myotomy DI (EF-325) of >9.2 mm2/mmHg to be associated with clinical success, defined as ≥0.75 point improvement on GCSI. While these studies do not provide us with definitive target values, they provide a starting range of values to shoot for associated with positive clinical response.
If the target DI range is not reached after myotomy, additional myotomy can be considered within the same myotomy, or a second myotomy can be considered, on a case-by-case basis.
Over time, improvements in pyloric CSA and DI following POP appear to wane, with a 13% loss of response per year in one study. 70 Conchillo et al 71 found that a higher DI post POP correlates with 6-month outcomes, and that antral motility does not matter, as long as the pylorus remains open enough to pass a food bolus.
FLIP Protocol During Peroral Endoscopic Cricopharyngeal Myotomy for Zenker’s Diverticulum (Z-POEM)
Flexible endoscopic cricopharyngeal myotomy for Zenker’s diverticulum is a treatment modality that was first described in 1995.72,73 The addition of submucosal tunneling, similar to that performed during POEM for lower esophageal myotomy, was then introduced by Li et al, 74 and the procedure known as Z-POEM was created. Since that time, Z-POEM has gained traction as an excellent alternative to standard open diverticulectomy, rigid endoscopic diverticulostomy, and traditional flexible endoscopic diverticulosotomy. 75 There are the most limited data on normal impedance planimetry values of the upper esophageal sphincter (UES) of any sphincter in the foregut. One small study performed in 13 awake subjects found a mean resting UES CSA at a 20 mL fill of 23.5 mm2 (SD 2.8 mm2) and correlating mean P of 31.2 mmHg (SD 10.2 mmHg). Distensibility index was not reported. 76 A separate study reporting on FLIP values in Zenkers patients also contained a control group where the UES was evaluated with a 40 mL fill; mean CSA 227.5 mm2 (95% CI, 199.7-255.2) and mean DI 7.6 mm2/mmHg (95% CI, 6.1-9.0). Both were significantly larger than in the patients with Zenkers’ diverticula, although confounded by different anesthesia administered between the control and study population. 77
Protocol
• Measurements at a 30 mL fill volume to ensure intra-bag pressure is always >15 mmHg and to minimize compromise of the airway. Note: use caution when interpreting FLIP values where the intra-bag pressure reaches only 15 to 20 mmHg.
• Must be performed under general endotracheal anesthesia to decrease the risk of aspiration.
• Timepoints to obtain measurements: ○ After intubation, prior to mucosotomy. ○ After myotomy is complete, prior to closure of mucosotomy. ■ The FLIP catheter should always be placed under endoscopic guidance to avoid cannulation of the submucosal tunnel or placement into the diverticulum. Advancing the catheter blindly can result in esophageal perforation.
• When obtaining measurements: ○ Patient positioning: flat ○ Insufflation: remove all intraluminal insufflation from the esophagus
Place the catheter trans-orally under direct endoscopic visualization into the esophagus (typically 15-20 cm) immediately after zeroing to atmospheric pressure. The catheter will often preferentially enter the Zenker’s diverticulum. a) Use the low-weighted filter to identify the UES. b) Withdraw the endoscope beyond the proximal extent of the probe and remove any esophageal insufflation prior to initiating the following protocol.
Inflate probe to 30 mL while slowly withdrawing the catheter until the hourglass shape is seen on the FLIP monitor and the probe is directly visualized with the endoscope to be straddling the UES. a) Switch to the high-weighted filter.
Center the hourglass in the middle of the electrodes.
Leave catheter in place for 30 second to allow for stabilization.
After 30 seconds, once the Dmin reaches its peak, press “pause live image” and record Dmin, P, CSA, DI, and C.
Deflate probe and remove catheter.
Three studies have reported FLIP measurements during Z-POEM and associated outcomes, including a significantly increased Dmin, CSA and DI with a parallel decrease in intra-bag pressure.66,78,79 Sondhi et al 79 described clinical improvement, defined by Eating Assessment Tool-10 scores, in all patients with a DI improvement of ≥26.7% at a 50 mL fill. All studies evaluating FLIP parameters in patients with Zenker’s diverticula or undergoing Z-POEM are limited by small sample sizes and heterogeneity in fill volume. Regardless of the differences in study design, we see an increase in Dmin, CSA, and DI, and the authors postulate that percent change from pre-myotomy to post-myotomy may be more important in Z-POEM than definitive target values. 79 The optimal role for FLIP technology in treatment of Zenker’s diverticula remains undefined and may be the ability of FLIP technology to rule out Zenker’s as an etiology of a patient’s symptoms.
Unlike other uses of the FLIP catheter, you must be very cognizant of the risk of causing airway compromise if the intrabag pressure is too great. Therefore, UES FLIP should only be performed when the patient is under general endotracheal anesthesia.
Expert Consensus Recommend Target Values
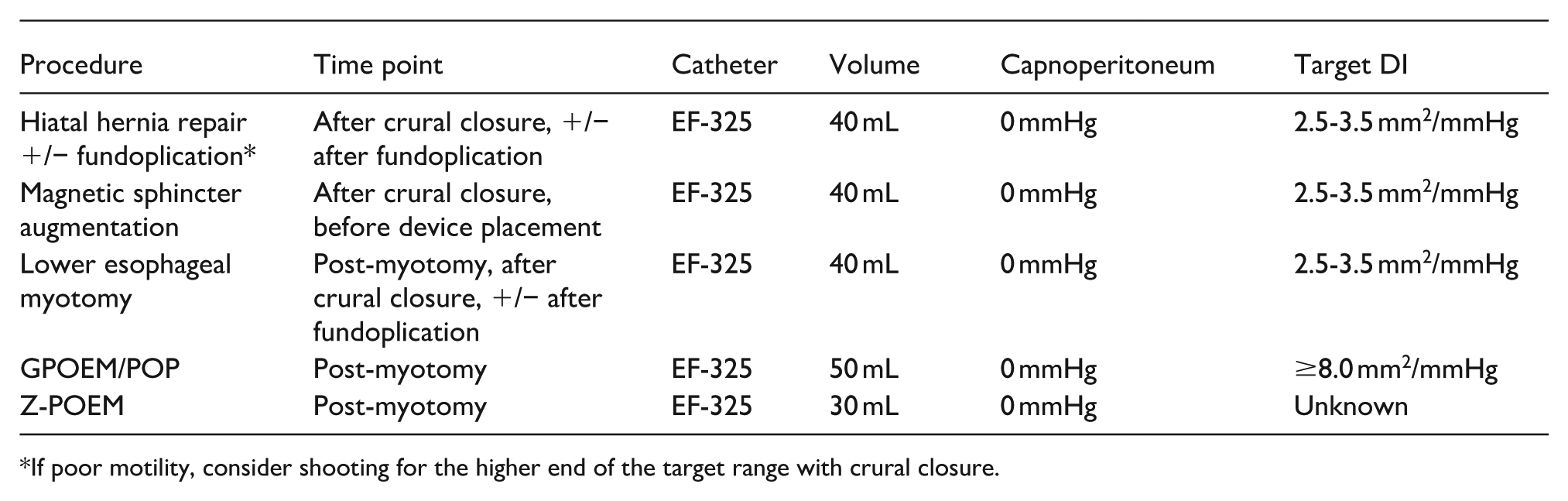
If poor motility, consider shooting for the higher end of the target range with crural closure.
FLIP Protocol for Panometry
• Can be performed in the GI suite or in the operating room; must avoid halogenated inhaled anesthetic agents such as sevoflurane. 13
• Timepoints to obtain measurements: ○ At any time during upper endoscopy in the GI suite, generally after performing an initial diagnostic upper endoscopy ○ After intubation, prior to start of procedure in the operating room
• When obtaining measurements: ○ Patient positioning: flat; left-lateral ○ Insufflation: remove all intraluminal insufflation from the esophagus and stomach ○ Filter: off ○ Probe configuration: 16 cm, maximum fill 70 mL
Place the catheter trans-orally into the esophagus and then fill to 30-40 mL to confirm positioning. If there is a known hiatal hernia, or if any resistance is encountered, place under direct endoscopic visualization to minimize damage to the catheter and to ensure appropriate placement across the EGJ. a) Allow 1 to 3 sensors to be within the stomach, beyond the lower esophageal sphincter. b) Check placement with endoscopy if unable to visualize the hourglass of the lower esophageal sphincter on the monitor.
Stepwise inflation to 50, 60, and 70 mL. Allow for 30 seconds of recording at each volume fill prior to progressing to the next volume fill.
Deflate probe and remove catheter.
Troubleshooting
It is important to remember that the impedance catheter is a device that gives readings on the pressure and cross sectional area of a sphincter but does not replace good clinical judgment.
If a reading does not seem consistent with the clinical picture, always be sure to check the probe for a leak; if no leak is present, a new catheter could be used to confirm the readings
If there are two areas of narrowing seen on the monitor image, this could represent a damaged sensor and the probe should be replaced.
Discussion
There remains a dearth of data published for use of impedance planimetry in foregut surgery obtained with a standardized protocol. This severely limits conclusions on target values and pooling of data for meta-analyses. The purpose of creating this consensus and recommending target value for specific foregut procedures that utilize impedance planimetry is to further standardize protocols for FLIP use for foregut surgery. By doing so, the authors hope to dramatically increase the comparability of published studies and further optimize foregut surgery through the use of impedance planimetry.
One significant update to expert recommendations in this publication from previous expert consensus published in 2020 1 is different volumes of fill recommended for the majority of the listed procedures, including a 30 mL fill at the upper esophageal sphincter, 30 mL or 40 mL fill at the lower esophageal sphincter, and 50 mL fill at the pylorus to routinely reach a minimum probe pressure of >15 mmHg, and ideally reach a probe pressure of >20 mmHg. In the intervening years, several studies have been published following expert protocol recommendations. However, repeat iterations of these protocol and target values will remain necessary as more data becomes available with longer term follow-up, larger cohort sizes, and prospective randomized trials. Additionally, procedures such as fundoplication encompass a wide array of techniques, including multiple types of fundoplication and variations in type of bougie utilized.
Panometry may be able to reduce the need for manometry in the preoperative workup of GERD patients with normal motility, when performed at the time of preoperative upper endoscopy. 13
Conclusion
The FLIP impedance planimetry system is a valuable tool that provides real-time, objective feedback to foregut surgeons in the operating room. While the tightness of a crural closure, fundoplication, and adequacy of a myotomy have been relatively subjective measures to date, the authors believe that patient outcomes can be optimized with this technology. Updated standardized protocols for FLIP impedance planimetry use in the operating room during foregut procedures, as well as expanded practice use, allow for investigators to publish comparable data. We have learned that patient positioning does not affect measured impedance planimetry values, while the presence or absence of pneumoperitoneum does. Therefore, obtaining measurements without pneumoperitoneum is highly important to allow for correlation to measurements taken in the GI lab pre- and post-operatively. Ultimately, the goal is to identify the FLIP parameters that most closely correlate to outcomes and define ranges to optimize outcomes.
Furthermore, FLIP panometry is gaining traction as a promising preoperative and intraoperative screening tool that can be used to rule out major motility disorders in patients undergoing paraesophageal hernia repair. Although panometry at time of endoscopy is best for preoperative planning, intraoperative panometry is ideal for patients who cannot receive preoperative high resolution manometry due to poor tolerance or emergent presentation. Intraoperative impedance planimetry and panometry have the potential to identify missed achalasia diagnoses, and therefore change operative treatment. If panometry is performed, inhaled anesthetic should not be used as it inhibits esophageal motility.
Limitations
Target ranges have been generated based on expert consensus. The authors anticipate these will change in the future as more data utilizing standard protocols for FLIP during foregut surgery is published.
Supplemental Material
sj-docx-1-gut-10.1177_26345161251393103 – Supplemental material for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery
Supplemental material, sj-docx-1-gut-10.1177_26345161251393103 for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery by Julia R. Amundson, Vanessa N. VanDruff, Shahin Ayazi, Christy M. Dunst, Jon Gould, Min P. Kim, Dustin A. Carlson, Kirsten Newhams, Aaron N. Sachs, Paul Severson and Michael B. Ujiki in Foregut
Supplemental Material
sj-docx-2-gut-10.1177_26345161251393103 – Supplemental material for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery
Supplemental material, sj-docx-2-gut-10.1177_26345161251393103 for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery by Julia R. Amundson, Vanessa N. VanDruff, Shahin Ayazi, Christy M. Dunst, Jon Gould, Min P. Kim, Dustin A. Carlson, Kirsten Newhams, Aaron N. Sachs, Paul Severson and Michael B. Ujiki in Foregut
Supplemental Material
sj-docx-3-gut-10.1177_26345161251393103 – Supplemental material for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery
Supplemental material, sj-docx-3-gut-10.1177_26345161251393103 for TULIPS Class 2.0: The Use of Functional Luminal Imaging Probe During Surgery by Julia R. Amundson, Vanessa N. VanDruff, Shahin Ayazi, Christy M. Dunst, Jon Gould, Min P. Kim, Dustin A. Carlson, Kirsten Newhams, Aaron N. Sachs, Paul Severson and Michael B. Ujiki in Foregut
Footnotes
Ethical Considerations
Ethical approval is not required for this manuscript.
Author Contributions
Julia R. Amundson – conception, consensus meeting participation, manuscript preparation; Vanessa N. VanDruff – conception, manuscript preparation; Shahin Ayazi – consensus meeting participation, manuscript preparation; Christy M. Dunst – consensus meeting participation, manuscript preparation; Jon Gould – consensus meeting participation, manuscript preparation; Min P. Kim – consensus meeting participation, manuscript preparation; Dustin A. Carlson – conception, manuscript preparation; Kirsten Newhams – consensus meeting participation, manuscript preparation; Aaron N. Sachs – consensus meeting participation, manuscript preparation; Paul Severson – consensus meeting participation, manuscript preparation; Michael B. Ujiki – conception, consensus meeting participation, manuscript preparation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medtronic provided logistical support for the meeting of this expert consensus panel. Members of Medtronic were present during the meeting, but uninvolved in the development of the consensus statement or creation of this manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Julia R. Amundson MD, MPH: No COI; Vanessa N. VanDruff MD: No COI; Shahin Ayazi MD: No COI; Christy M. Dunst MD, FACS: BD (consultant), Implantica (consultant), Merrit (consultant), Enterra (consultant); Jon Gould MD, MBA: No COI; Min P. Kim MD: Intuitive (teaching); Dustin A. Carlson MD: Medtronic (speaking, consulting, license), Diversatek (consulting); Phantom Pharmaceuticals (speaking, advisory board), Braintree (consulting), Medspace (consulting), Regeneron/Sanofi (speaking, consulting); Kirsten Newhams MD, MPH: Castle Bioscience (speaker, advisory board); Aaron N. Sachs MD, DABS, FACS, FASMBS: No COI; Paul Severson MD, FACS, FASMBS: No COI; Michael B. Ujiki MD, FACS: Boston Scientific (scientific advisory board), Olympus (consultant), Medtronic (speaker), Gore (speaker).
Use of Artificial Intelligence
No artificial intelligence (AI) was used in the preparation of this manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
