Abstract
Minimally invasive esophageal cellular collection devices such as the Capsule Sponge may offer the potential for targeted screening of high-risk patients. The capsule sponge has been shown to be a safe, accurate and effective means of detecting Barrett’s esophagus. Here, we explore two potential real-life scenarios that could occur in a future screening or case finding programme. Firstly, when the capsule sponge test result is positive but confirmatory endoscopy demonstrates <1cm of columnar epithelium and secondly when subsequent endoscopy identifies gastric intestinal neoplasia. We discuss the clinical implication of the result and suggest how these patients could potentially be followed up in the future.
We discuss two potential scenarios:
(a) The capsule sponge test result is positive and suggestive of BE. However, the patient subsequently undergoes endoscopy which is normal and has less than 1 cm of columnar epithelium.
(b) The capsule sponge test result is positive and suggestive of BE. However, the patient subsequently undergoes endoscopy which identifies gastric intestinal metaplasia.
How should these patients be managed?
Introduction
Esophageal cell collection devices enable sampling of superficial cells from the esophageal mucosa that can subsequently be analyzed for evidence of intestinal metaplasia. Following new approaches to couple cell collection with molecular and protein biomarkers 1 there are several Barrett’s tests now available for clinical use. These include the capsule sponge device, which is a pan-esophageal cell collection device coupled with an immunohistochemical marker Trefoil Factor 3 (TFF3) (Figure 1). Taking on board this concept, the Cleveland clinic separately developed an expandable balloon (EsoCheck) to sample the distal esophagus and coupled this with methylation biomarkers including a methylated DNA biomarker panel (EsoGuard) which assays levels of methylated vimentin and methylated cyclin A1. 2 Additionally, the team from John Hopkins have identified novel methylation marker panels which were able to accurately classify BE. 3 These and other similar office-based cell collection tests have the potential to revolutionize screening for esophageal cancer by enabling clinicians to identify patients with BE early.
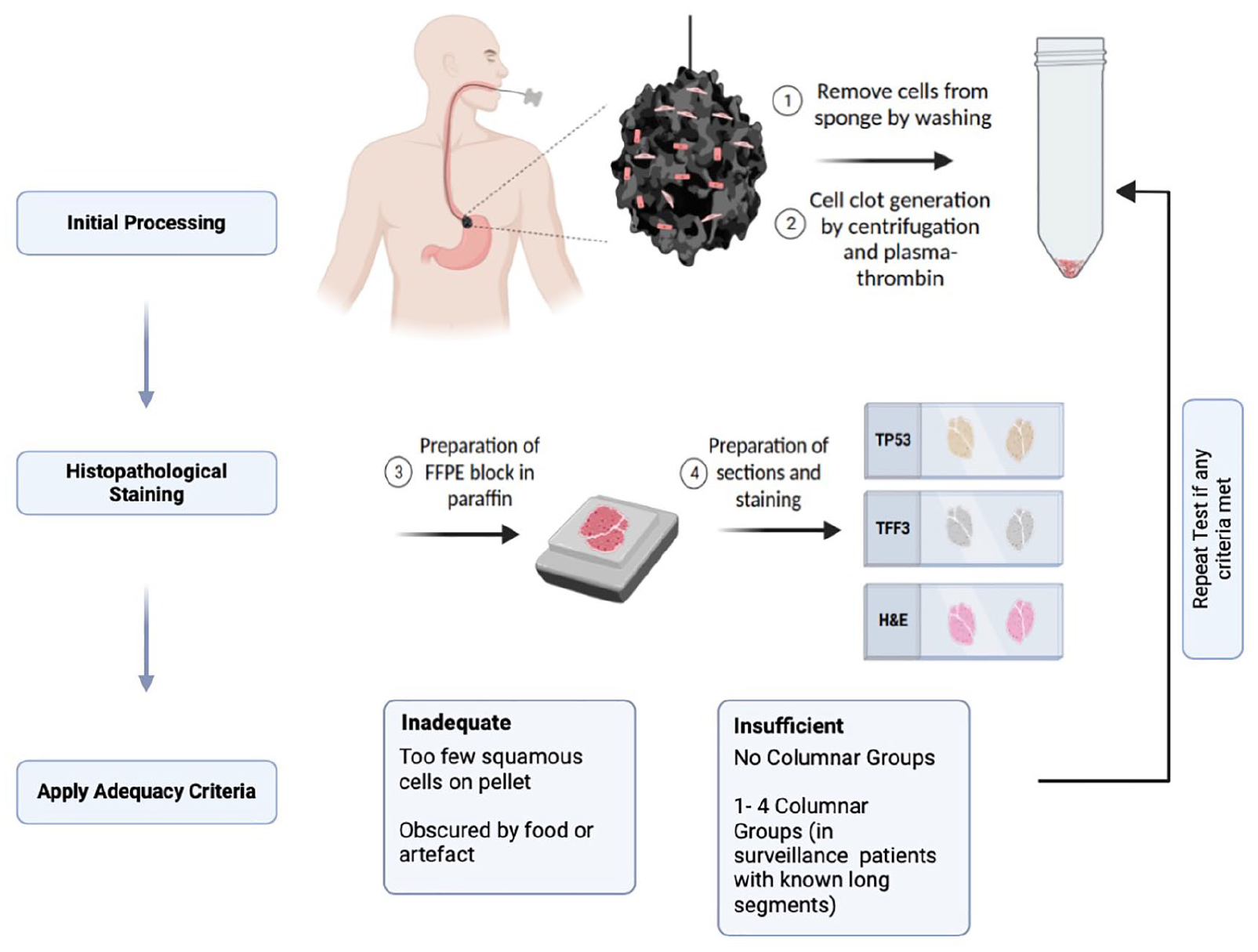
Sampling and initial processing workflow of the capsule sponge.
Here, we focus specifically on the capsule sponge device coupled with immunohistochemical biomarkers. This device is composed of a polyurethane sponge attached to a string, compressed into a vegetable gelatin-coated capsule. This is swallowed by the patient in a routine clinic setting and once the capsule reaches the stomach, the capsule dissolves from the heat of the stomach and the sponge expands. After several minutes, a health care provider then withdraws the sponge which collects the cells from the gastric cardia and the entire length of the esophagus for analysis. Capsule sponge samples are suspended in preservative buffer after sampling, washed and centrifuged to form a cell pellet which when formed into a clot with artificial plasma-thrombin resembles a pseudo-biopsy with over 1 million cells. The pseudo-biopsy is processed into a formalin fixed paraffin embedded (FFPE) block and stained for TP53, TFF3 and with a standard hematoxylin and eosin stain (H&E) (Figure 2). Finally, the H&E slides are reviewed initially to ensure an adequate number of columnar cells are present as a marker of gastric adequacy. 4 This process ensures that only samples which have adequate number of squamous and columnar cells from the esophagus and gastric or Barrett’s mucosa, respectively, are processed to provide certainty that the sponge has reached the stomach, and that the entire esophagus has been sampled by the sponge. The slides are reviewed by a trained pathologist for the presence or absence of intestinal metaplasia and dysplastic cells which may be supplemented by machine learning algorithms. 5
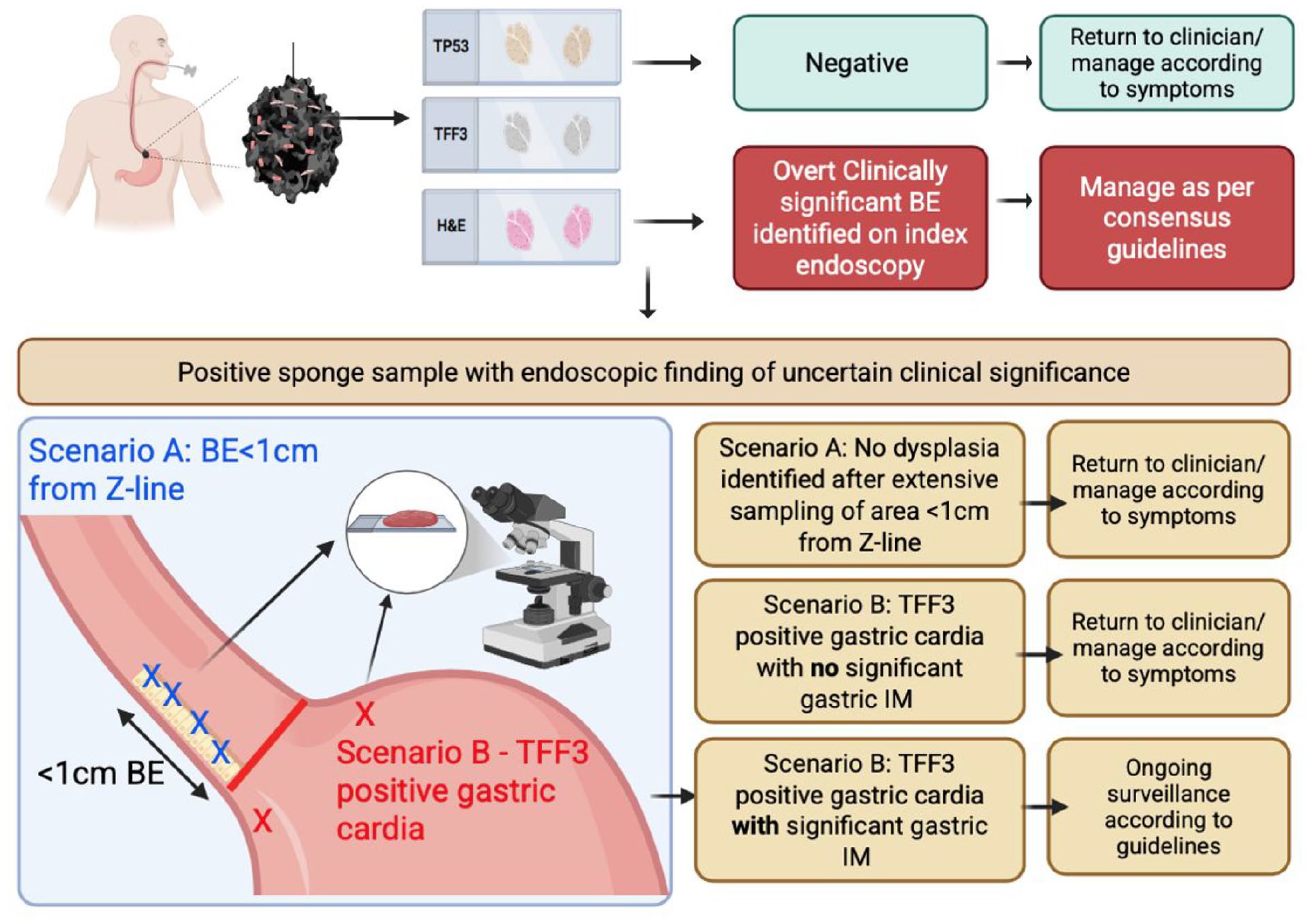
Flow chart of suggested management for scenarios A and B.
The Barrett’s Esophagus Screening Trials (BEST) have proven feasibility, safety and patient acceptability 6 (BEST1) and demonstrated accuracy (including all patients who swallowed the device) comparable to the endoscopic gold standard with a sensitivity of 79.9% (95% CI 76.4%-83.0%) and specificity 92.4% (95% CI 89.5%-94.7%) for the capsule sponge-TFF3 test 7 (BEST2). Most recently the BEST3 trial has demonstrated that screening high risk patients (>50 years, on >6 months acid-suppressant medication for reflux) in the primary care setting identifies over 10 times more BE cases than usual care of a family doctor in which endoscopy is triggered if deemed necessary 8 including some patients with dysplasia. A recent analysis based on 100% adoption of capsule sponge testing across NHS Scotland, indicates that the cost of providing capsule sponge testing to the total eligible population in Scotland is £10.4 million compared with £17.0 million for usual care, giving an incremental cost saving of £6.6 million per year. 9 This is in addition to the ease of performance, opportunity for widespread use in primary care and shortened times to undergo clinically indicated screening. The latest iteration of this trial series, BEST 4, seeks to determine if a sponge-based cytology device will reduce esophageal cancer specific morbidity (earlier stage) and mortality.
Trefoil Factor 3 as a Goblet Cell Marker
To understand the clinical scenarios presented to us we need to understand the biology underlying the biomarker and how it performs. TFF3 is a goblet cell marker that was identified by performing a gene expression profiling study to compare the expression of markers in biopsies from Barrett’s esophagus compared with the other tissues which are sampled by the capsule sponge device – namely the gastric cardia and squamous epithelium. 1 TFF3 was one of several biomarkers selected based not only on the fold change in expression of RNA in BE tissue compared to squamous and cardiac mucosa, but the ability to show a clean immunohistochemical stain. This is because we wished to pursue a marker that would be stable in the clinically routine histopathological setting, compared with the RNA transcript alone. 1
The TFF3 positivity is assessed in combination with the H&E stain and p53 by a histopathologist. 6 On the capsule sponge sample respiratory epithelium can also be collected as the sponge is extracted through the oro-pharynx. Therefore, the sample can also contain columnar cells from respiratory epithelium. Combining both H&E and TFF3 enables accurate identification of TFF3 positive intestinal metaplasia. 4 Therefore, a positive H&E and TFF3 result suggests intestinal metaplasia somewhere in the sample, or a sample may be labelled as equivocal if the staining was poor quality with a low signal to background. Additionally, we can predict the likely length of the Barrett’s from the proportion of TFF3 positive gland groups. 10
Scenario A: Positive Capsule Sponge Test with Columnar Mucosa <1 cm Identified in the Distal Esophagus on Endoscopy
The classification of Barrett’s is controversial and varies in different guidelines. In Barrett’s guidelines from the U.S. 11 and U.K., 12 BE is defined endoscopically by at least 1 cm of circumferential columnar appearing mucosa in the distal esophagus, with the AGA requiring the presence of goblet cells on histopathology for diagnosis. 11 This implies that the presence of intestinal metaplasia in the distal 1 cm of the esophagus lacks an increased risk for development of adenocarcinoma and retrospective studies have shown that the so called “ultra-short” segments have negligible cancer risk. 13 Further to this, recent work has demonstrated that short segment gastric metaplasia (GM) of the esophagus is genomically distinct from IM, with gastric metaplasia having a substantially lower risk of progression. 14
Some studies question the assumption that short segment BE is not clinically relevant. A recent two center study of a relatively large population of patients diagnosed with esophageal adenocarcinoma (EAC) demonstrated that up to half of these patients lack visible Barrett’s at the time of the initial diagnosis. 15 This could suggest that EAC has developed via a Barrett’s independent pathway, or carcinoma develops in a small segment of intestinalized mucosa close to the Z line that would not qualify as Barrett’s or it could highlight rapid tumor overgrowth of a segment >1 cm in length.
Furthermore, although patients with esophagogastric junctional (EGJ) adenocarcinomas have a presumed gastric origin in most patients, 16 carcinoma of the EGJ shares many similarities to BE derived EAC and both are markedly increasing in incidence. 17 Additionally, we have shown that IM of the gastric and esophageal epithelium are remarkably similar from a molecular view 18 and evidence from The Cancer Genome Atlas research network has demonstrated a complex overlap between anatomical location and the molecular characteristics of the tumor sequenced, 19 suggesting that location of the lesions does not necessarily reflect the genomic make-up.
It is also intuitive to wonder if there is a meaningful distinction between circumferential Barrett’s esophagus that is 0.9 versus 1.0 cm in length? The endoscopes we use have 10 mm markings and clinicians tend to round up the distance. Indeed, despite many attempts to rigorously measure Barrett’s length relative to the esophagogastric junction, 20 the definition of a normal Z line used to estimate the length of Barrett’s mucosa remains vague. 21 Furthermore, although an increased risk of EAC is associated with Barrett’s length, carcinoma may theoretically evolve from a genomic catastrophe in a single small focus of short segment metaplasia. Thus, if the false positive test in this scenario is comprised of intestinal metaplasia within 1 cm of the gastroesophageal junction, it may not be a false positive from a biologic standpoint, although the clinical significance may generally be low in a Barrett’s surveillance population. 13
It is informative to consider the evidence from the BEST3 trial regarding the positive predictive value for this test in diagnosing clinically relevant Barrett’s. In BEST3 the diagnosis of BE on endoscopy was based on a clinical decision following British Society Guidelines. In the intervention arm where 1654 patients received a Capsule Sponge test, 221 received a positive result. Of these, 131 had clinically significant BE or EAC on follow up endoscopy and the positive predictive value (PPV) was 59% (131/221). An additional 33 samples were demonstrated to have IM of the EGJ or gastric cardia on follow up endoscopy, so overall the PPV of confirmed IM in the esophagus, EGJ or GC was 74% (164/221).
Recommendation in Scenario A
For this patient, we would recommend the following. If the false positive is intestinal metaplasia that could be present within 1 cm of the gastroesophageal junction, we would carefully visualize including using narrow band imaging and take multiple biopsies of this area (including the cardia) to rule out prevalent dysplasia and carcinoma at index endoscopy or within a year of index endoscopy. If neither is present, despite the capsule sponge sample being positive, we would counsel the patients that their risks are extremely low and that this doesn’t require any follow-up.
Therefore, these samples technically represent a true positive result, as the test has correctly identified the presence of IM. However, assuming biopsy does not identify dysplasia, we would deem the clinical significance to be low.
Scenario B: Positive Capsule Sponge Test with IM Identified in the Gastric Cardia
In this scenario, the TFF3 positive sample appears to have been derived from the gastric cardia. If the IM has come from the gastric cardia then this could represent a diffuse intestinal metaplasia of the stomach which is also a risk factor for cancer.
As part of the BEST3 study protocol, a biopsy of the cardia was included. 8 A recent post-hoc analysis of the BEST3 trial data was performed to determine the rate of detection of gastric intestinal metaplasia (GIM) or gastric atrophy (GA) in this data set. 22 Patients with endoscopic or histologic evidence of BE or IM at the gastroesophageal junction were excluded. 57 patients were included in the final analysis (sponge sample TFF3+, n = 44; TFF3−, n = 13). In the TFF3+ group, 34.1% of patients (n = 15) were diagnosed with GIM/GA and one of these patients had an early gastric cancer. None of the patients in the TFF3− group were diagnosed with GA/GIM. In patients with no evidence of BE, TFF3 positivity was significantly associated with the presence of GA/GIM in gastric biopsies (P = .013). 22
This could mean in our clinical scenario in patients who have a TFF3 positive sponge sample but no esophageal IM identified on endoscopy, it could be useful to ensure the gastric cardia has been adequately sampled. It has been previously demonstrated that gastric IM is patchy in nature and so could be difficult to identify on index endoscopy 23 and so this may account for sponge samples initially labelled as false positives.
Recommendation in Scenario B
Our approach on index endoscopy would include biopsy of the gastric cardia (in the BEST3 trial protocol two biopsies were taken below the Z-line to determine whether gastric IM could account for a positive capsule sponge sample). 8 If the finding is non-goblet cell TFF3 positive cardia mucosa without extensive gastric IM and no dysplasia on biopsy, we would support a patient decision for no further follow up. However, in the case of extensive IM of the stomach we would recommend further surveillance according to established guidelines. 24 In both scenarios A and B an alternative would be to offer a repeat capsule sponge test in 5 years. But we lack evidence on the optimal frequency for non-endoscopic screening tests. Figure 2 depicts a possible future workflow for the scenarios discussed.
Future Directions
In the future, in endoscopy as well as with non-endoscopic tests, we need to move away from a qualitative measure of the length of the segment and the histopathological characteristics of a subjectively targeted biopsy based on perceived Barrett’s length towards a more quantitative measure of the extent and the molecular characteristics of the segment epithelium. New artificial intelligence assisted algorithms may be able to more accurately quantify the area of the Barrett’s epithelium during endoscopy and integrate laboratory tests such as capsule sponge risk stratification biomarkers to assist in risk stratifying patients.
Recent work has demonstrated that p53, cellular atypia, and clinical demographic variables could be used to prioritize patients for endoscopy in those who had a positive capsule sponge test.25,26 Perhaps a similar approach could be used to risk stratify the capsule sponge samples which demonstrated EGJ or GIM on endoscopy in our clinical scenarios and enable a more quantitative approach to managing these patients. Clinicodemographic and histopathological features could be integrated into a scoring system to determine whether patients should undergo further investigation due to a higher risk of developing dysplasia and cancer. These could include p53, 27 copy number profile 28 and multiplexed molecular assays such as the tissue systems pathology [TSP-9] test (TissueCypher). 29 Evidence from ongoing clinical trials is awaited to make firm recommendations on determining follow up intervals or ablative therapy on the basis of these tissue tests.
Footnotes
Abbreviations
BE: Barrett’s esophagus
EGJ: esophagogastric junction
GA: gastric atrophy
GC: gastric cardia
GIM: gastric intestinal metaplasia
HGD: high grade dysplasia
LGD: low grade dysplasia
NDBE: non-dysplastic Barrett’s esophagus
TFF3: trefoil factor 3
Author Contributions
AB/DK: writing – original draft; DK/RCF: conceptualization of manuscript; M O’D/RCF: writing – review and editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RCF: R.C.F. is named on patents related to Cytosponge and related assays which have been licensed by the Medical Research Council to Covidien GI Solutions (now Medtronic) and is a co-founder and share holder (<2%) of CYTED Ltd. The Fitzgerald lab has an ongoing collaboration with AstraZeneca and Natera with provision of expertise and materials. RCF is a member of the SAB for the Astra Zeneca and CRUK Functional Genomics Core and is on an advisory board for 23 and me. RCF is a Trustee Director of: Heartburn Cancer UK; Evelyn Trust; The Lister Institute; The Liver Trust. DK: Advisory board for Sanofi/Regeneron, Bristol Myers Squibb, Cyted, Castle Biosciences. M.O’D: Co-inventor of Cytosponge. Named on some patents relating to Cytosponge. Cofounder, shareholder and lead pathologist at Cyted Ltd. AB: Nothing to disclose.
Ethical Approval
Ethical approval is not required for this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Use of Artificial Intelligence
No artificial intelligence (AI) was used in the preparation of this manuscript.
