Abstract
Background:
Pathologic reflux is a common debilitating digestive condition and anti-reflux surgery is an effective treatment option. Depending on the specifics of the procedure, anti-reflux surgery leads to distinct changes in the endoscopic appearance of the esophagogastric junction (EGJ). This American Foregut Society (AFS) white paper develops nomenclature for describing the endoscopic characteristics of the EGJ after commonly performed laparoscopic antireflux operations.
Methods:
The AFS Anti-Reflux Barrier Collaborative analyzed the anatomic manipulation and corresponding endoscopic appearance of the EGJ after commonly performed anti-reflux surgery including: (1) 360° anterior/posterior (AP) fundoplication (Nissen); (2) posterior partial fundoplication (Toupet); (3) anterior partial fundoplication (Watson, Dor); (4) magnetic sphincter augmentation; and (5) concomitant hiatal hernia repair with transoral fundoplication (cTIF).
Results:
Characteristics common to the anti-reflux surgery procedures include restoration of intra-abdominal esophageal length and crural diaphragm repair such that the hiatal aperture is no longer visible in the retroflexed endoscopic view. With regard to the geometry of anti-reflux procedures, the Collaborative established that when evaluated endoscopically in retroflection, the surgically constructed valve should be described in reference to the angle of His and the extent of anatomic change described in terms of (1) the depth of the anterior and posterior grooves, (2) the apposition of the valve collar to the endoscope, (3) the length of the valve, and (4) the position of the squamocolumnar junction relative to the lip of the valve.
Conclusions:
Current anti-reflux operations share much in common but employ varying degrees of valvular reconstruction leading to distinct endoscopic characteristics. Developing a consistent nomenclature for describing the appearance of the post-surgical valve is essential to develop a better understanding of how variables such as the tightness of the hiatal repair, tightness of the valve collar, and length of the valve can impact the outcome of anti-reflux surgery and predict reflux recurrence.
Keywords
Introduction
Gastroesophageal reflux disease (GERD) is among the most common digestive diagnoses in the US. Among patients being treated for GERD, a certain fraction will have pathological reflux, defined as either abnormal esophageal acid exposure on pH-metry or Los Angeles B, C, or D esophagitis on endoscopy. Anti-reflux surgery can be an effective management option in patients with pathologic reflux and a compromised anti-reflux barrier.1,2 The primary goal of anti-reflux surgery is to restore an effective anti-reflux barrier to reduce or eliminate pathologic reflux resulting in an improved quality of life and decreased GERD complications. This can be achieved with a variety of procedures in conjunction with hiatal hernia reduction and restoration of the crural diaphragm sphincter mechanism. A common element of these procedures is anatomical modification of the esophagogastric junction (EGJ) resulting in distinct changes in the endoscopic appearance of the EGJ viewed in the retroflection.3-6 Figure 1 illustrates a defective anti-reflux barrier and how the appearance of the EGJ changes after hiatal hernia repair and then after construction of a Nissen fundoplication. Anatomic modification of the EGJ after anti-reflux surgery correlates with reflux control and the deterioration of these modifications over time (recurrent hiatal hernia, disruption or malposition of the fundoplication) correlates with recurrent reflux. 7 Despite this, surgeons have not routinely analyzed the endoscopic appearance of the EGJ during or after anti-reflux surgery. Additionally, there is currently no standardized reporting method for describing the endoscopic evaluation of the EGJ after anti-reflux surgery.7,8 Figure 2 exemplifies the utility of the endoscopic assessment of the gastroesophageal valve in the evaluation of a patient with new or recurrent symptoms during and after anti-reflux procedures.
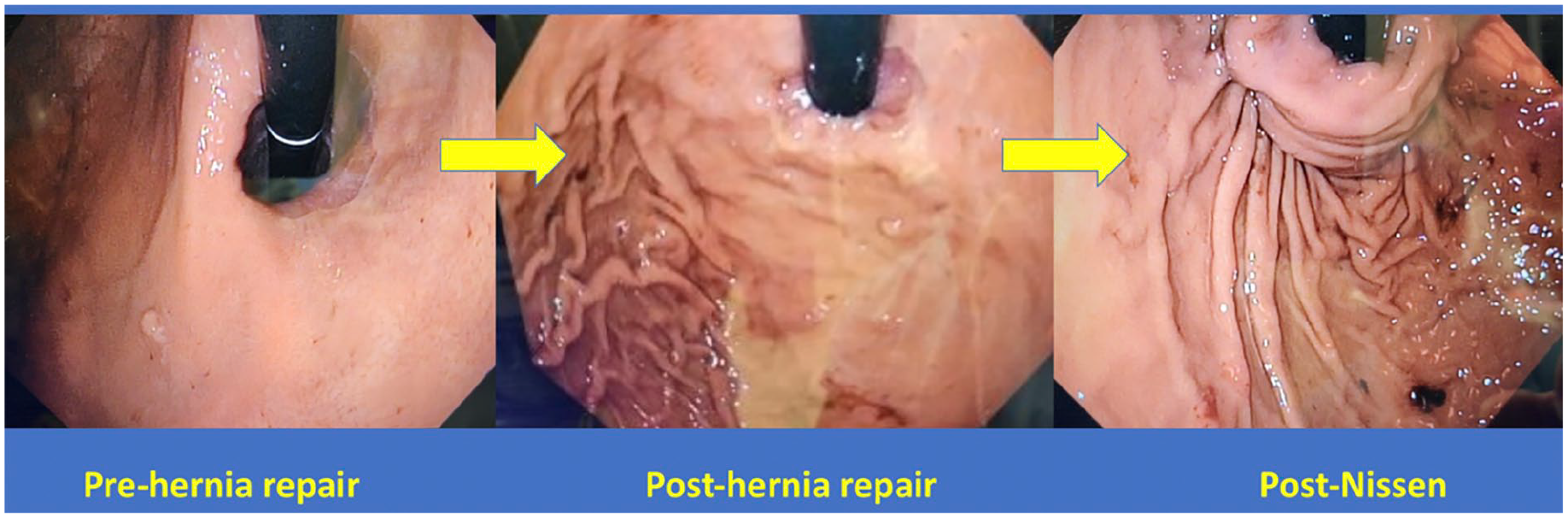
Transition from AFS hiatus grade III (L0 cm, D2.5 cm, FV−) in the left panel to post-Nissen fundoplication. Note the change in gastroesophageal junction anatomy after crural diaphragmatic reapproximation (center) and Nissen fundoplication (right). A surgically formed gastroesophageal valve is evident after fundoplication. Use of this figure courtesy of the AFS, copyright 2024.
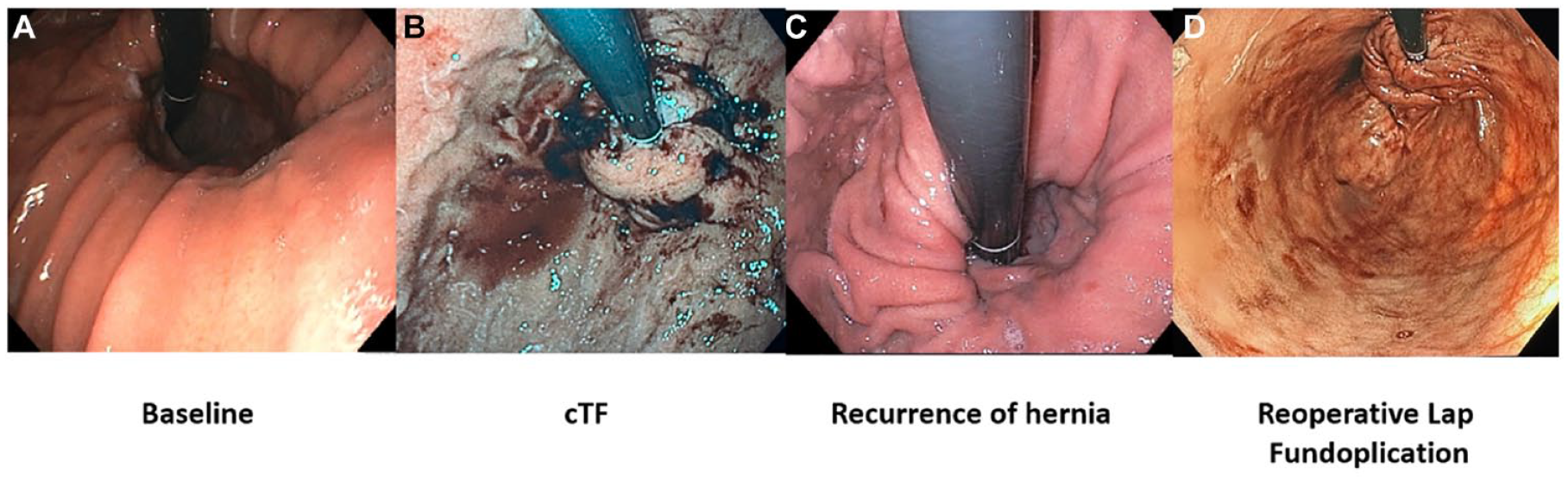
Changes of the esophagogastric junction (EGJ) in a patient over time. Panel A shows the baseline endoscopy with an AFS hiatus grade IV EGJ. This patient underwent cTIF and the immediate postoperative endoscopic view of the EGJ is shown in panel B. Panel C shows the endoscopic appearance of the EGJ in this same patient with recurrent reflux symptoms 2 years after the cTIF, now with a wide open hiatus and complete loss of the gastroesophageal valve. Panel D shows restoration of the antireflux barrier with laparoscopic repair of hiatal hernia and partial fundoplication. Use of this figure courtesy of the AFS, copyright 2024.
Recognizing the importance of the endoscopic appearance of the post-operative gastroesophageal valve, Jobe et al 9 hypothesized that this was a major determinant of the quality and integrity of an anti-reflux operation. Their 2004 study described the endoscopic appearance of the gastroesophageal valve after 5 common anti-reflux surgery procedures: Nissen (complete/total) fundoplication, Collis-Nissen fundoplication (Nissen after esophageal lengthening), Toupet (partial) fundoplication, Dor (partial) fundoplication, and the Hill repair. 9 For each procedure the 7 participating surgeons and 2 participating gastroenterologists evaluated endoscopic photographs of 7 surgical “successes” to define the ideal endoscopic appearance of the post-surgical valve using 11 criteria (lip, body, anterior groove, posterior groove, lesser curvature, adherence to scope, respiration effect, valve type, intra-abdominal location, proper repair position, and unique characteristics) and, by consensus, described how each should appear. Although the drawings from that manuscript are superb, the 11-domain endoscopic evaluation is somewhat cumbersome. Furthermore, because the evaluators were aware of what procedures had been done and that these were carefully selected cases with “ideal” outcomes, the applicability of the proposed schema in clinical practice was tenuous and never widely adopted.
More recently, magnetic sphincter augmentation and laparoscopic hiatal hernia repair with combined transoral fundoplication (cTIF) have been added to the list of common effective anti-reflux operations.10,11 The cTIF adheres to the same principles as conventional “surgical” fundoplications: initial laparoscopic esophageal mobilization to gain intra-abdominal esophageal length, crural diaphragm repair, and creation of a gastroesophageal valve by fundoplication, albeit using a transoral rather than a laparoscopic technique. Similarly, although initially proposed as a means to augment LES yield pressure with an implanted device, magnetic sphincter augmentation has subsequently been combined with formal hiatal dissection, hernia reduction, and crural repair to enhance its effectiveness. 12 Within this context, the American Foregut Society (AFS) convened an 11-member working group of gastroenterologists and foregut surgeons (the Anti-Reflux Barrier Collaborative) to analyze key aspects of the external (surgical) and internal (endoscopic) features of commonly performed anti-reflux operations, including magnetic sphincter augmentation and cTIF. The 11-point description in the Jobe et al. paper and the AFS hiatus grade white paper provided the foundation for this process.9,13 The goals of the Collaborative were to simplify the nomenclature for describing the endoscopic appearance of the anti-reflux valve and broaden it to include these newer technologies thereby making it more adaptable to clinical practice. This document summarizes the deliberations of the Collaborative.
Endoscopic Nomenclature and Technique for Evaluating the Gastroesophageal Valve
Endoscopic inspection of the post-intervention gastroesophageal valve has parallels to the inspection of the presurgical valve with some added considerations. However, the technique utilized is quite similar and a key to obtaining optimal EGJ visualization emphasized in the AFS hiatus grade white paper (and still pertinent here) is obtaining adequate gastric insufflation. Suggested methods include endoscopic insufflation for approximately 30 to 45 seconds or insufflation until there is flattening of the fundic rugal folds to inspect the tightness of the valve “collar” to the endoscope in retroflection, be that at the lip or along the valve’s length. Additionally, endoscopic rotation in the retroflexed position and maneuvers to induce potential axial hiatal herniation are important. This is initiated by gently pulling up the tip of the endoscope in the retroflexed position along the depths of the grooves. This maneuver provides the most accurate assessment of the hiatal opening and extent of hiatal herniation, if present. Without these maneuvers, the valve can often be “under-graded” contributing to interobserver variability. In documenting the endoscopic appearance of the EGJ, the Collaborative recommends that the endoscope be positioned toward the greater curvature with about 2 cm of the endoscope visible. Ideally, the image includes the entire gastroesophageal valve, but if that is not feasible, separate images should be obtained focused on the anterior and posterior groove.
The normal gastroesophageal valve develops as the esophagus obliquely enters the lesser curve side of the stomach. The Collaborative defined the gastroesophageal valve as an anatomic valve composed of the segment of the gastric fundus in direct apposition to the intra-abdominal esophagus. The end-to-side geometry of this complex appears endoscopically as a semi-circumferential “flap” of gastric fundus covering the distal esophagus, accounting for the acute angle of His. Degradation of the anti-reflux barrier is a key mechanism for the development of pathological reflux and is evident by loss of intra-abdominal esophageal length, loss of the gastroesophageal valve, development of a hiatal hernia, and dilatation of the hiatus as stratified by the AFS white paper on the endoscopic classification of the EGJ integrity. 13 With regard to endoscopic orientation, Figure 3 illustrates radial nomenclature including anterior (12 o’clock), posterior (6 o’clock), greater curve (3 o’clock), and lesser curve (9 o’clock) positions. This nomenclature correlates with the anatomic transverse plane. 13 With an intact gastroesophageal valve, categorized as the AFS hiatus grade 1, the gastric fundus envelops the intra-abdominal esophagus semi-circumferentially. 13 The native position of the squamocolumnar junction (SCJ) is at the lip of the valve, corresponding to the apex of the angle of His. Because the apex of the angle of His is positioned at approximate 3 to 4 o’clock in the transverse plane, all fundoplications begin at that point with the gastric fundus enveloping the esophagus in both the anterior and posterior directions for approximately 90°. Based on this geometry, the collaborative adopted the same nomenclature and established the radial point of endoscopic reference for fundoplication to be at apex of the angle of His rather than anatomic anterior or posterior as is often described in laparoscopic fundoplication.
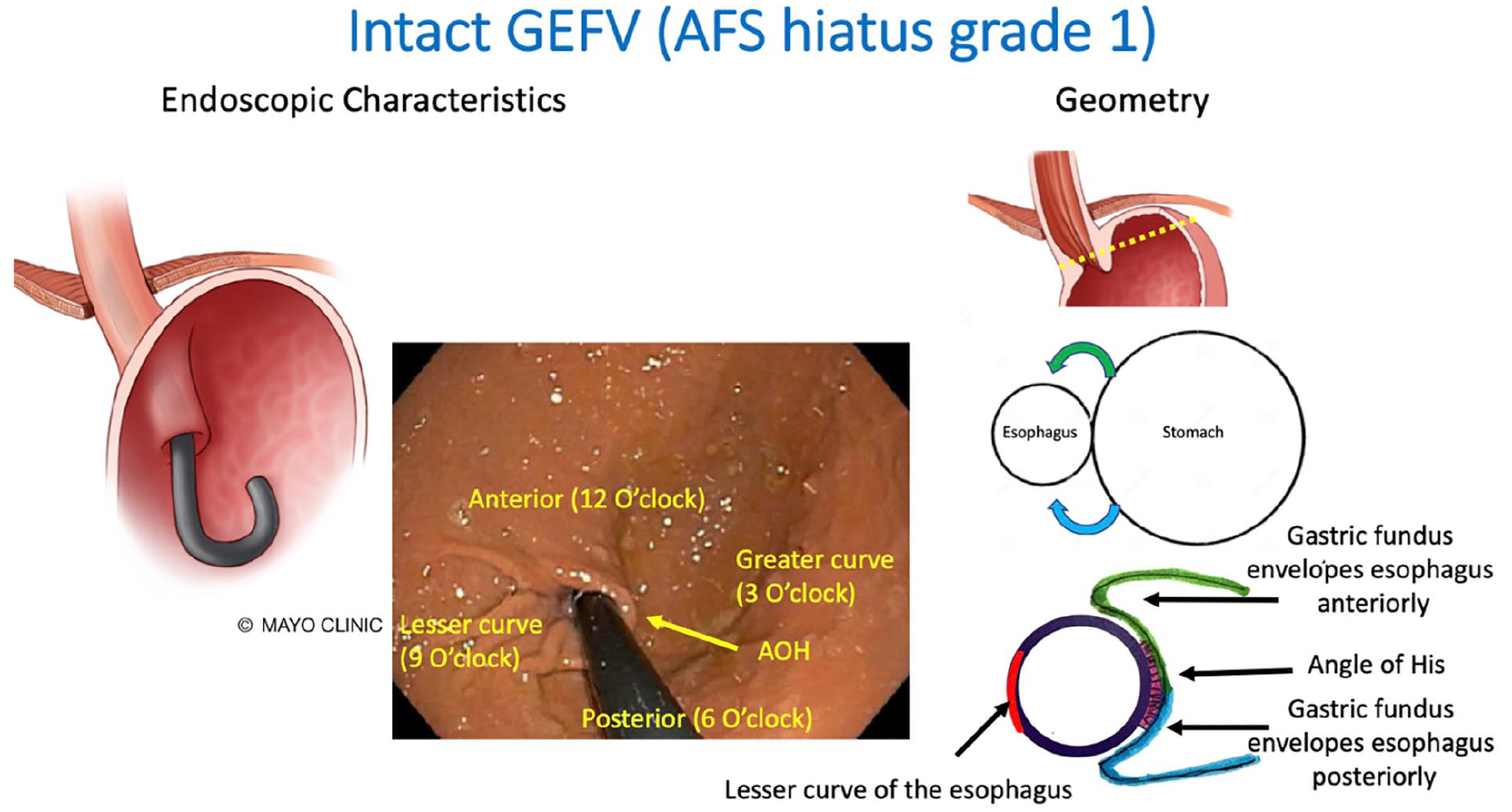
Endoscopic orientation and geometry of an intact gastroesophageal valve (GEFV). A semi-circumferential flap of gastric fundus envelops the intra-abdominal esophagus starting at the angle of His (AOH) in equal proportion anteriorly (green) and posteriorly (blue). The clock orientations correlate with the anatomic transverse plane. Note that the diaphragmatic hiatus is not visible endoscopically with an intact esophagogastric junction anatomy (AFS hiatus grade 1). Use of this figure courtesy of the AFS, copyright 2024.
Principles of Anti-Reflux Surgery
The key components to successful anti-reflux surgery include: (1) restoration of an intra-abdominal esophageal length by reduction and repair of the hiatal hernia; (2) reapproximating the crural diaphragm to decrease hernia recurrence and potentiate the crural diaphragm sphincter mechanism; and (3) modifying gastroesophageal anatomy to restore gastroesophageal valve function (except in the MSA procedure wherein this function is left to the implant). The net effect is to both reduce the occurrence of reflux and to restrict the diameter of EGJ opening when reflux does occur. The most time-tested procedure is a fundoplication and there are several variations of fundoplication which will be discussed. 2
A fundoplication is constructed with reference to the external anatomy of the gastroesophageal junction such that the fundus surrounds the distal tubular esophagus to varying degrees. Viewed endoscopically, this appears as a “collar” around the distal esophagus. With the native position of the SCJ at the gastroesophageal junction, this positions the SCJ at the lip of the surgically reconstructed valve. However, with AFS hiatus grades II, III, or IV there is anatomical change to the gastroesophageal junction such that it has migrated cephalad either closer to the hiatus or above the hiatus. This can be associated with dilatation of the distal esophagus, evident by intraoperative measurement of the circumference, endoscopic measurement in retroflection or intraluminal measurement of the distensibility of the gastroesophageal junction.14-16 Furthermore, there can be associated migration of the SCJ into this dilated esophageal tube along with the appearance of metaplasia. 17 Histologically, this has been called “cardiac mucosa” as biopsies show glandular tissue without intestinal metaplasia or parietal cells, variably with submucosal esophageal glands and squamous islands. 18 However, this is a very controversial topic because as the distal esophagus progressively dilates, it increasingly takes on characteristics that make it appear like the stomach endoscopically with what appear to be “rugal folds” and glandular mucosa while laparoscopically it appears cephalad to the angle of His suggesting it to be part of the esophageal tube. This controversy is very relevant to the current discussion because it impacts on the endoscopic appearance of the post-surgical valve. Rather than extending to the lip of the valve, the SCJ is seen proximally in the distal tubular esophagus closely mimicking the appearance of a distally placed fundoplication. The collaborative opined that the best approach to this circumstance of distorted gastroesophageal anatomy is to document it intraoperatively after dissection of the esophagus and prior to creation of the fundoplication. At that point, the end of the esophageal tube is readily compared to the location of the SCJ, information that then becomes invaluable in clarifying future examinations assessing whether a fundoplication is improperly positioned on the proximal stomach or is properly placed on the distal esophagus.
Endoscopic Features Pertinent to All Anti-Reflux Surgery Procedures
As described above, laparoscopic circumferential dissection of the esophagus and mediastinal mobilization to allow reduction of a hiatal hernia and reapproximation of the crural diaphragm are common to all of the anti-reflux surgery procedures to be discussed. This results in visibly tightening the hiatus and creating intra-abdominal esophageal length as evident in the center panel of Figure 1. Consequently, after any anti-reflux procedures that involved hiatal hernia repair, the hiatal aperture should not be visible in the retroflexed endoscopic view and the valve collar should be relatively snug around the endoscope. Similarly, reflective of whether intra-abdominal esophageal length was effectively restored, visualization of the SCJ from the retroflexed view (or the position of SCJ relative to either the lip of the gastroesophageal valve viewed from below or the caudal end of the esophagus/fundoplication when viewed antegrade) should be noted.
Procedure-Specific Features of the Gastroesophageal Valve
Common among all of the anti-reflux operations considered in our analysis are the laparoscopic reduction of the hiatal hernia with restoration of intra-abdominal esophageal length and crural diaphragm repair. The main variability among the procedures is in the domain of how the gastroesophageal anatomy is modified to restore gastroesophageal valve function. This results in variability among procedures in the appearance of the valve as detailed below for each procedure. The exception is the magnetic sphincter augmentation procedure which does not involve construction of a fundoplication. Consequently, in the case of the MSA procedure, the assessment is essentially the same as in the case of the non-operated patient.
Anterior-Posterior Fundoplication (Original Nissen)
With the Nissen anterior-posterior fundoplication, the gastric fundus is mobilized by dividing the short gastric vessels and a fundoplication is constructed with the anterior and posterior fundic walls wrapped circumferentially around the distal esophagus (Figure 4). The plications are sutured at approximately the 10 o’clock position using 3 sutures resulting in a 2 cm long fundoplication. Unlike the native semi-circumferential gastroesophageal valve, the Nissen anterior-posterior fundoplication completely encircles the esophagus resulting in an endoscopic appearance with deep anterior and posterior grooves. The gastroesophageal valve is generally snug around the shaft of the endoscope both anteriorly and posteriorly, but the collar of fundus may not be of similar diameter along its entire length. However, it is the narrowest part of the collar that is functionally most important and this may be at the proximal end. Consequently, although there may be a gap at the lip of the valve, you should not be able to see through the fundoplication into the esophagus; that would be indicative of an overly loose valve. The intragastric length of the fundoplication is gaged endoscopically by measuring the distance from the lip to the base of the gastroesophageal valve.
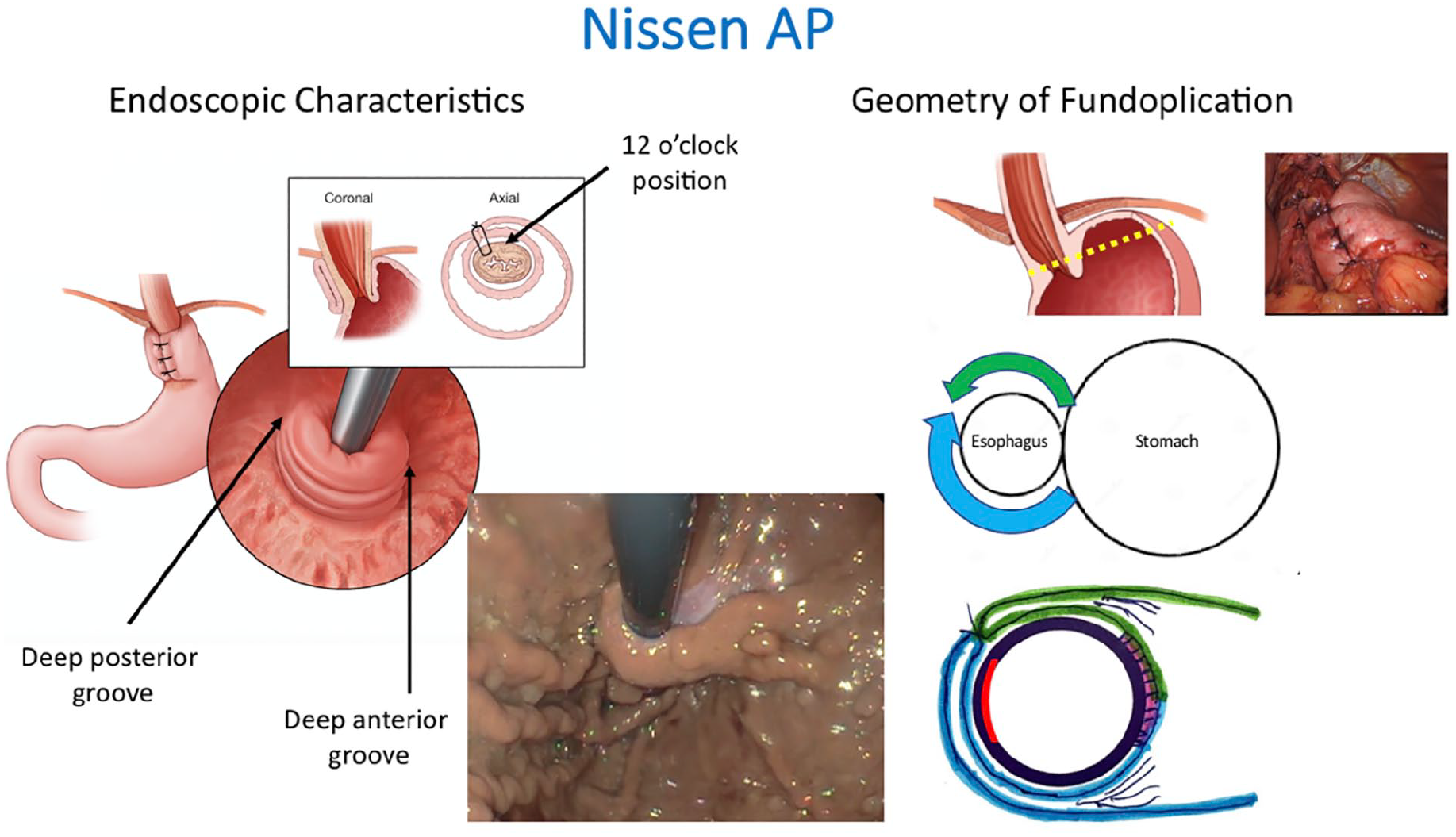
Endoscopic features and geometry of a Nissen 360° anterior/posterior (AP) fundoplication. Note that the portion of the fundoplication covering the lesser curve aspect of the esophagus (red area) will not be visible endoscopically. Also note that in this case esophageal mucosa is visible at the lip of the valve verifying that the valve surrounds the distal esophagus. Use of this figure courtesy of the AFS, copyright 2024.
“Posterior” Partial Fundoplication (Toupet)
The most frequently performed laparoscopic posterior partial fundoplication is the Toupet procedure, albeit with numerous variations in the precise surgical technique. Most commonly, it consists of mobilizing the gastric fundus by dividing the short gastric vessels and creating a fundoplication by wrapping the gastric fundus posteriorly around the distal aspect of the esophagus in a 270° fashion secured at the 10 and 2 o’clock position as illustrated in Figure 5. Referenced to the angle of His, this results in a greater posterior compared to anterior wrap. Endoscopically, there is a deep posterior groove and a shallow anterior groove.
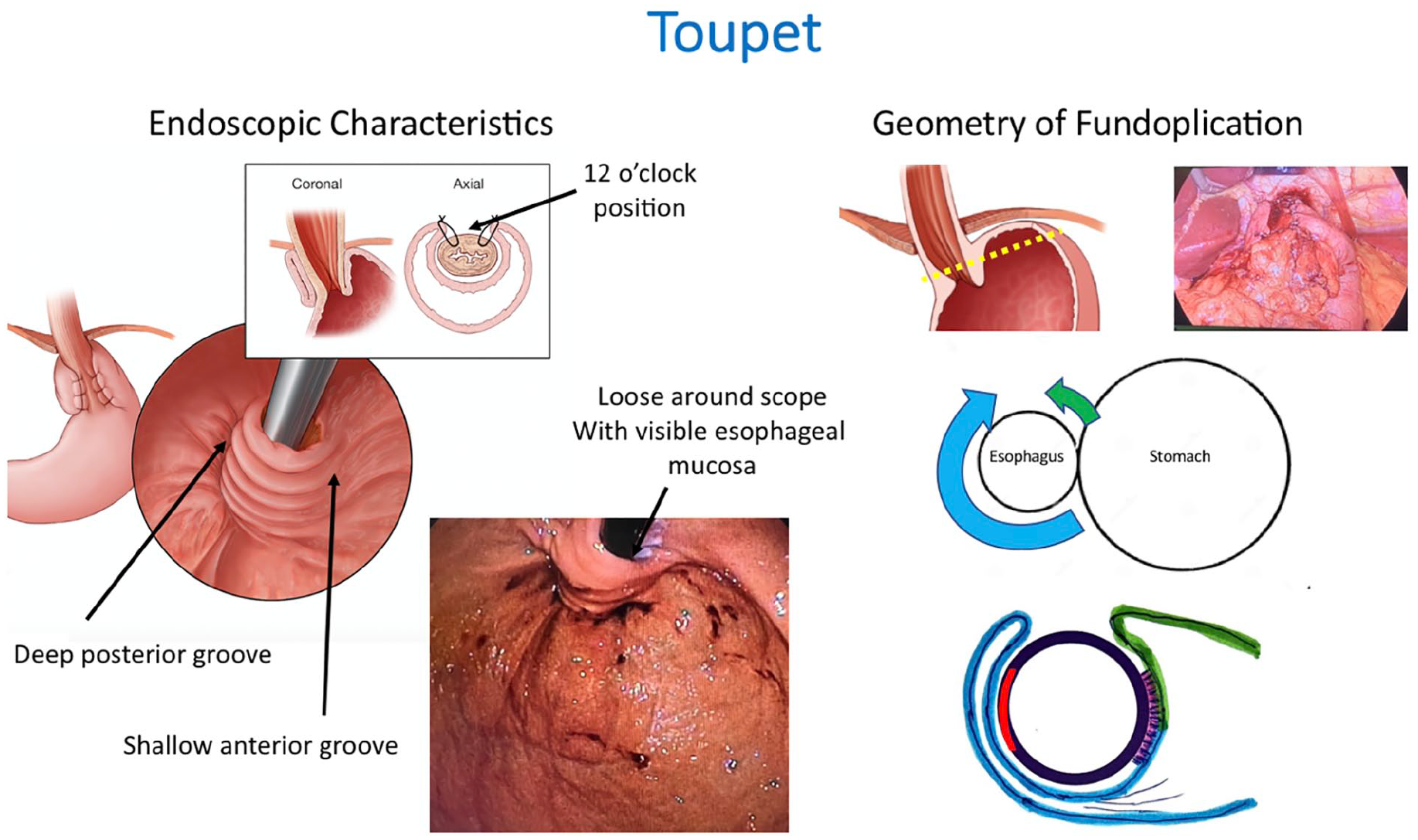
Endoscopic features and geometry of a Toupet fundoplication. There is asymmetry with more posterior gastric fundus wrap (blue) compared to the anterior gastric fundus wrap (green). The fundoplication is considered “partial” because of the exposed area of the esophagus between the 10 and 2 o’clock position (anterior). The asymmetric wrap leads to a deep posterior groove and a shallow anterior groove in the retroflexed endoscopic image. Use of this figure courtesy of the AFS, copyright 2024.
Anterior Partial Fundoplications (Dor, Watson)
The two commonly performed laparoscopic anterior fundoplications carry the eponyms of the “Dor” and the “Watson” procedures. These entail the laparoscopic plication of the gastric fundus anteriorly around the distal aspect of the esophagus to approximately the 9 to 10 o’clock position (Dor) or the 7 o’clock position (Watson), with 12 o’clock being the anterior midline of the esophagus. Hence, with the fundoplication extending from approximately the 3 o’clock position, the geometry of the anterior fundoplication is roughly 180° to 230° wrap from an external perspective (Figure 6a and b). The corresponding endoscopic appearance of an anterior fundoplication is of a deep anterior groove and a shallow posterior groove since there is no posterior component to the wrap. Consequently, the lip of the valve may be loose around the shaft of the endoscope along the posterior aspect of the valve.
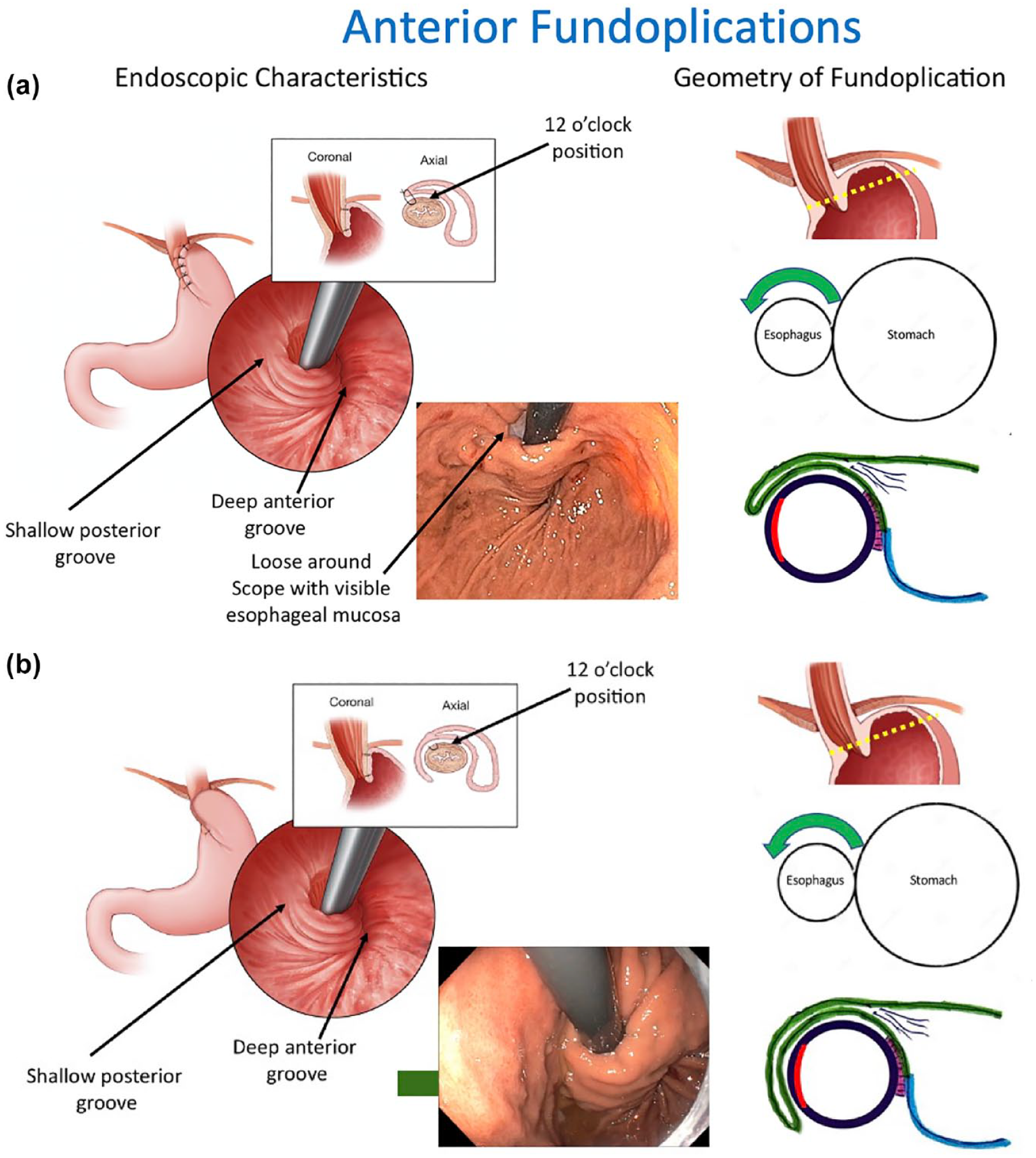
Endoscopic features and geometry of two common anterior fundoplications, the Dor 120° to 180° anterior fundoplication (a) and the Watson 230° anterior fundoplication (b). There is asymmetry with only anterior gastric fundus wrap (green). The posterior gastric fundus (blue) is not plicated. Note the loose area around the scope at the posterior side of the Dor valve with visible esophageal mucosa where there is no fundoplication. Endoscopically, there is a deep anterior groove and shallow posterior groove. Use of this figure courtesy of the AFS, copyright 2024.
Magnetic Sphincter Augmentation
With laparoscopic magnetic sphincter augmentation, no fundoplication is created. After the crural diaphragm repair, the device is sized and placed around the distal esophagus (Figure 7). The sizing of the implant is such that at rest it exerts no force on the esophagus, but with esophageal distention the magnetic beads separate and attract each other thereby augmenting closure. Since there was no reconfiguration of the gastric fundus, the endoscopic appearance closely resembles the native flap valve with shallow anterior and posterior grooves. The implant itself is difficult to appreciate endoscopically, but can be evident as a “bulked-up” lip of the valve.
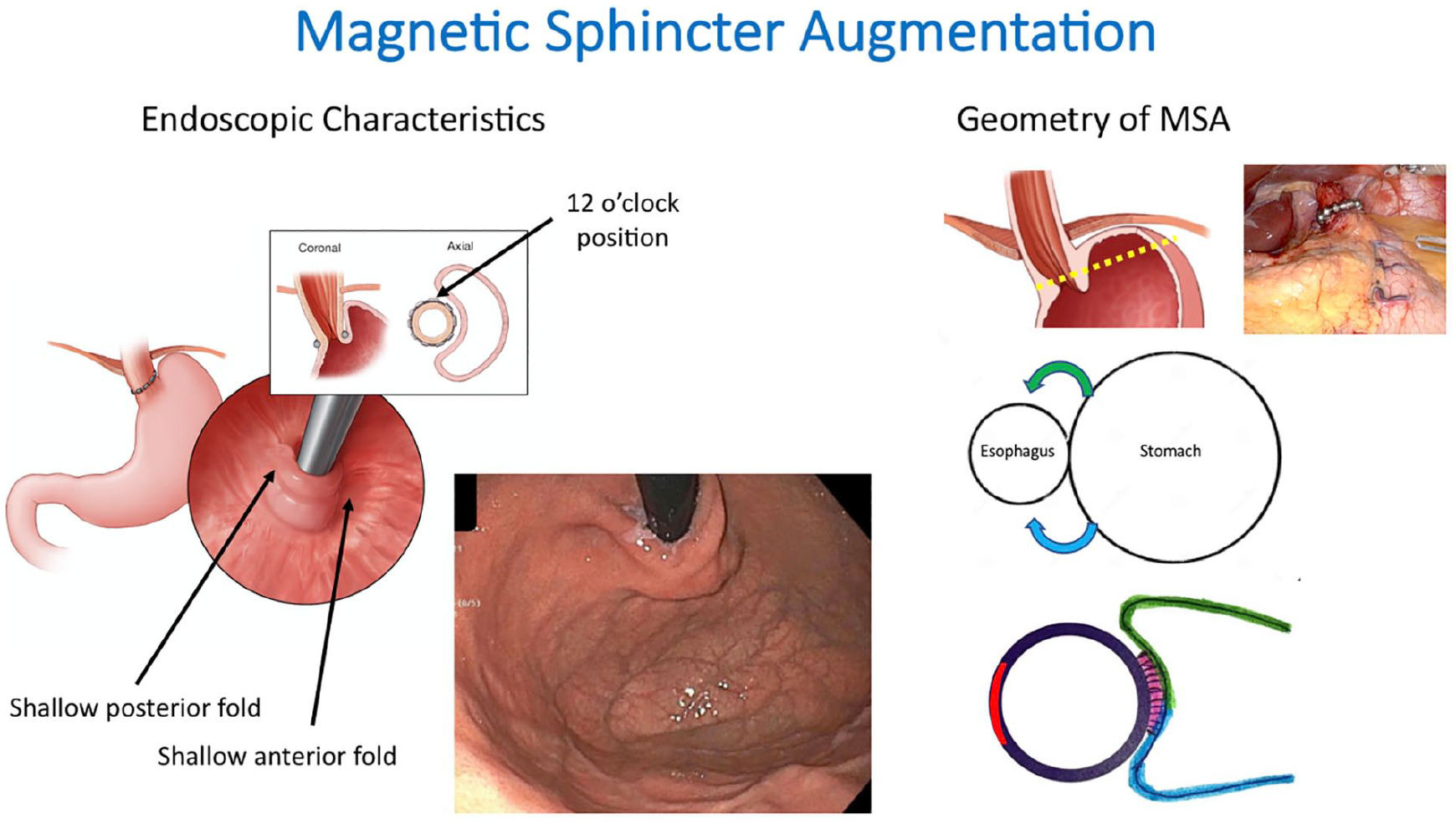
Endoscopic features and geometry of magnetic sphincter augmentation. Both posterior and anterior grooves are shallow as there is no fundoplication. Note that esophageal mucosa is visible at the lip of the valve in the endoscopic photo verifying restored intra-abdominal esophageal length. Use of this figure courtesy of the AFS, copyright 2024.
Laparoscopic Hiatal Hernia Repair With Transoral Fundoplication
In addition to laparoscopic hernia repair, the cTIF procedure creates a fundoplication transorally using the Esophyx® device (Endogastric Solutions, Redmond WA) mounted over the endoscope. With the fundoplication initiation point at the angle of His (3-4 o’clock), the geometry of the cTIF procedure consists of equal anterior and posterior wraps achieved by rotating the Esophyx® device approximately 135° posteriorly and 135° anteriorly with placement of multiple full thickness fasteners to secure it. Although the fundoplication externally extends more anteriorly than posteriorly, the immediate postoperative endoscopic appearance is of an omega shape with approximately 270° of fundoplication with a moderate to deep anterior and posterior grooves. (Figure 8). The valve lip closely approximates the shaft of the endoscope (in part due to edema). Valve length can be clearly visualized as can a variable number of the fasteners used to form the valve. Transmural fasteners will also be visible antegrade along the distal 2 to 3 cm of the esophageal mucosa as the distal esophagus is now within the restored valve.
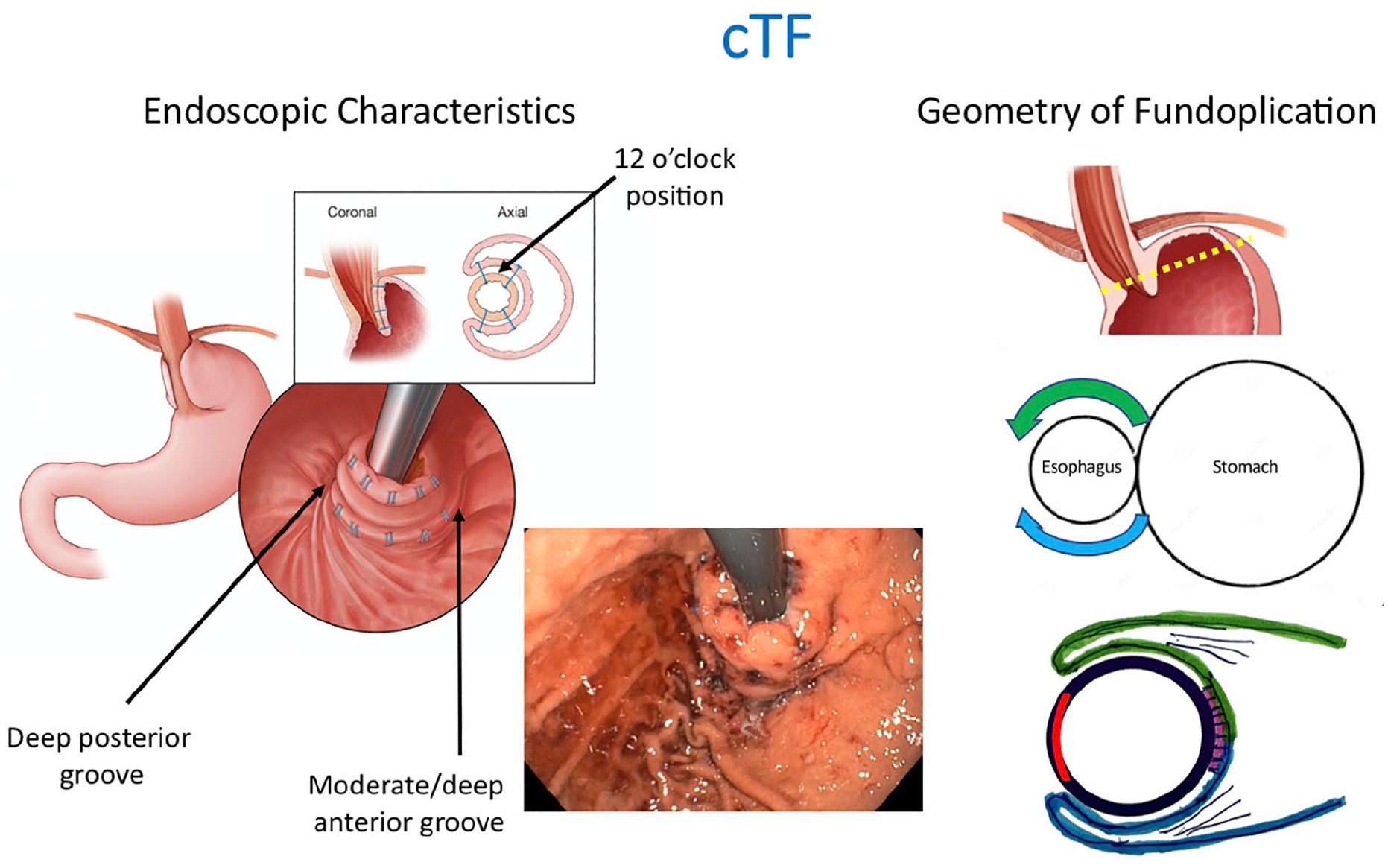
Endoscopic features and geometry of laparoscopic hiatal hernia repair with transoral fundoplication (cTIF). There is equal anterior (green) and posterior (blue) wrap with the exposed area of the esophagus at 9 o’clock. Based on the transverse view, the geometry of a cTIF is a reverse C configuration. Endoscopically, there is a deep posterior grove and a moderate-to-deep anterior groove. Use of this figure courtesy of the AFS, copyright 2024.
Endoscopic Nomenclature for Characterizing the Gastroesophageal Valve
Using the foundation of the AFS hiatus grade classification, the Collaborative concluded that successful anti-reflux surgery would necessarily be equivalent to an AFS grade 1 as there would be no hiatal hernia and a snug fit to the endoscope. In that case, the presence of a gastroesophageal valve should be noted and it can be characterized by the appearance of its anterior and posterior grooves, degree of apposition to the endoscope along the length of the reconstructed gastroesophageal valve, the length of the valve measured from the apex of the angle of His to the diaphragm, and the position of the SCJ relative to the lip of the valve. As with the AFS hiatus grade, the apposition or diameter of the valve is gaged relative to the diameter of the endoscope and may be tightest at the lip of the valve or anywhere along its length. If there is resistance to passage of the endoscope this should be noted. In the case of magnetic sphincter augmentation, one would revert to the AFS hiatus grade and simply denote FV+ or FV− with the added potential to note that the implant caused the appearance of a “bulked-up” lip of the valve.
This algorithmic approach to the endoscopic assessment of the post-surgical valve is summarized in Figure 9. Although there can always be variations in the endoscopic appearance of a post-surgical valve that warrant additional description these basic attributes are fundamental in the assessment of patients who present with recurrent GERD symptoms or de novo symptoms such as late dysphagia after anti-reflux surgery. Hopefully, this will also encourage surgeons to routinely perform intraoperative endoscopy to document the baseline postoperative appearance of the valve to serve as a comparator for such future evaluations.
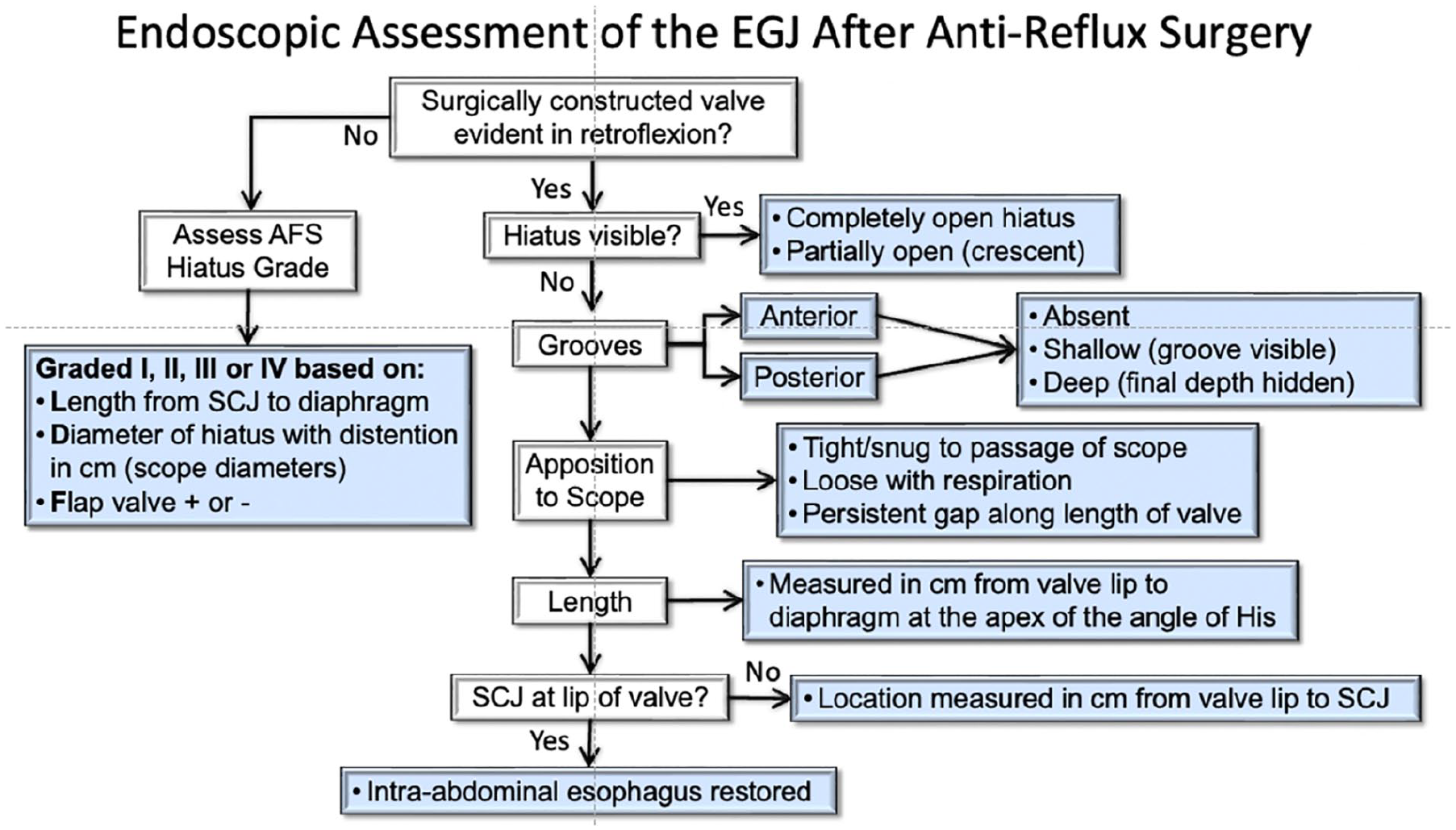
Endoscopic assessment of the gastroesophageal junction after anti-reflux surgery. If there is no history of surgically constructed valve, this reverts to the AFS hiatus grade classification. For patients with a surgically constructed valve, it is important to note hiatal disruption, characteristics of the anterior and posterior grooves, apposition to the endoscope with gastric distention, valve length, and the position of the squamocolumnar junction (SCJ) relative to the lip of the valve. The bulleted options in the blue boxes indicate the nomenclature to be used. Use of this figure courtesy of the AFS, copyright 2024.
Future Directions
The Collaborative recognized the need to further understand the degree to which the endoscopic characteristics of the EGJ after anti-reflux surgery correlate with the effective control of pathologic reflux. Establishing a consistent nomenclature to describe the post-surgical gastroesophageal valve should improve communication among clinicians and aid this process. We have described the “classic” and typical immediate postoperative appearance of commonly performed anti-reflux procedures. We did not examine the Hill or Collis esophageal lengthening procedures as these procedures are being performed only at highly specialized centers. A future direction include correlation of variations in the endoscopic appearance after anti-reflux procedures and the degree to which these variations may be associated with failure of reflux control, dysphagia, or de novo symptoms.
Conclusions
Pathologic reflux is largely attributable to progressive anatomic and physiologic deterioration of the EGJ resulting in incompetence of the anti-reflux barrier. The objectives of anti-reflux surgery are to restore an effective anti-reflux barrier by surgically correcting the EGJ disruption and constructing an anatomical gastroesophageal barrier, often by creation of a fundoplication. Current nomenclature references the degree and location of the fundoplication to the laparoscopic rather than the endoscopic appearance. The Collaborative proposes that this change to an endoscopic orientation be evaluated by the depth (shallow, moderate, deep) of the resultant anterior and posterior grooves, the closest approximation of the reconstructed gastroesophageal valve collar to the endoscope, the valve length, and the location of the SCJ relative to the lip of the valve. We hope that understanding the endoscopic geometry of the post-surgical gastroesophageal valve with a common nomenclature may help clinicians optimize current and future anti-reflux operations and better understand patterns of failure. Future research efforts should correlate these endoscopic characteristics after anti-reflux surgery with short-term and long-term reflux control and quality of life.
Footnotes
Author Contributions
Ninh T. Nguyen, MD (study design, acquisition and analysis of data, draft of paper, review of article, revision of article). Barham Abu Dayyeh,
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This paper does not involve human subjects and/or animals.
Use of Artificial Intelligence
This paper does not involve the use of AI software.
