Abstract
Background:
Magnetic sphincter augmentation (MSA) is an alternative treatment option to laparoscopic fundoplication (LF) for patients with gastroesophageal reflux disease. To date, over 40,000 devices have been implanted worldwide since first approval in Europe in 2010 and the USA in 2012. Despite this clinical reality, the long-term safety and effectiveness of the procedure continues to be questioned. This study aims to systematically summarize and appraise the currently available evidence for MSA relative to effectiveness, safety, and healthcare resource use.
Methods:
A systematic literature search was carried out to identify all clinical studies published in English, as of February 15, 2023. Required endpoints were safety, effectiveness, and cost effectiveness.
Results:
The systematic search identified 212 publications and 14 entries in study registries. After screening and full text analysis, 82 publications were included in qualitative synthesis. One RCT established superiority of MSA compared to twice daily proton-pump inhibitors with respect to the elimination of moderate to severe regurgitation (89% vs 10%, RR 0.11, 95% CI 0.06-0.20, P < 0.001). Eleven cohort studies comparing MSA to LF showed no statistical difference in safety profile and effectiveness measured by post-operative GERD-HRQL score. In addition, patients undergoing MSA significantly retained the ability to belch and vomit when compared to LF. These results were consistent in follow-up out to 7 years.
Conclusions:
LINX has been shown to provide long lasting relief to patients suffering from persistent GERD while maintaining an acceptable safety profile. As an outpatient day-procedure, MSA is cost effective with short recovery.
Key Learning Points
MSA is a viable treatment option for patients with GERD, with comparable safety and effectiveness as fundoplication.
MSA is technically less demanding with shorter recovery time than fundoplication.
MSA device cost is offset by lower hospital and medical expenses in the year following intervention compared to fundoplication.
Introduction
Gastroesophageal Reflux Disease (GERD) is primarily caused by a mechanical weakness or incompetence of the lower esophageal sphincter (LES) resulting in symptoms impairing the quality of life of affected individuals, including heartburn, belching, regurgitation of gastric contents, and coughing.1,2 The estimated lifetime prevalence of GERD is between 10% and 20% in industrialized nations. 3
Standard first-line treatments consist of life-style changes and medication to suppress gastric acid secretion, typically with proton-pump inhibitors (PPI). However, up to 40% of patients taking PPIs report persistent symptoms of GERD. 1 Anti-reflux surgery (ARS) is an option for patients who do not respond to or who are not comfortable with lifelong PPI therapy. Fundoplication, specifically laparoscopic Nissen fundoplication (LF) is currently considered the standard-of-care in ARS. However, LF is associated with a 17.7% risk of recurrence of GERD (if defined as being back on PPI for at least 6 months) and a 4.1% risk of complications.4,5
Magnetic Esophageal Sphincter Augmentation (MSA) utilizes the LINX® Reflux Management System (Ethicon, Johnson & Johnson), which is a “bracelet” consisting of titanium beads with magnetic cores connected with independent titanium alloy wires to form an annular shape. The diameter of a single bead is 5.8 mm, and the ultimate—size (diameter) is determined by the number of beads (13-17) making up each device. The LINX device is placed around the distal esophagus in a laparoscopic procedure intended to augment the LES opening pressure. The specific magnetic attraction between the magnets is such that normal swallowing generates sufficient pressure to separate the magnets. Most gastroesophageal reflux events occur at a pressure gradient insufficient to separate the beads, resulting in a reflux barrier that mimics normal physiology by keeping acid reflux in the stomach. The surgical placement of LINX, including the steps of surgical access, esophageal sizing and implant placement has been described in detail in previous publications. 6 Unlike surgical fundoplication, which uses the gastric fundus to create a one-way valve, the MSA procedure is reversible, does not alter gastric anatomy, and allows venting of gastric contents by virtue of its functioning as a pressure-release valve. 7
This paper reviews the most current published evidence on the safety and clinical effectiveness of MSA with regards to Patient Reported Outcome Measures (PROMs), safety including reoperations and device removals, and healthcare resource use.
Materials and Methods
To understand the current state of scientific knowledge for MSA supported by clinical studies, a systematic literature review was conducted. As this study only included published literature, no IRB approval or written consent was necessary.
Search Strategy
The systematic literature search was carried out in PubMed/MEDLINE, EMBASE, the Cochrane Library, and the study registers ClinicalTrials.gov, ISRCTN and WHO (For search strategy, see Table S1 of the Supplemental Material). The initial search was done January 21, 2021 as part of an application to the German Joint Federal Commission governing healthcare in Germany to evaluate MSA with LINX® and unequivocally establish reimbursement. 8 The search was then updated by rerunning the search strategies again on February 15, 2023. Specific search terms included Magnetic Sphincter Augmentation, LINX, reflux management system, GERD, GORD, gastroesophageal reflux, hiatal hernia, heartburn and related terms. The target of this review was research studies and meta-analyses. The references of review articles were scanned, and the manufacturer contacted to identified additional studies.
Study Selection
All clinical studies published as of February 15, 2023 in English language reporting effectiveness by PROMs and safety results relative to reinterventions, removals and other adverse events were included. Healthcare resource utilization measures which included procedure time, length of hospital stay and any readmissions, were also included. The PICO scheme (Patient-Intervention-Comparator-Outcome) for study selection criteria is provided in Supplemental Table S2 of the Supplemental Material. Letters, conference abstracts and presentations were excluded. Studies reporting only on technical aspects of the procedure were also excluded. After removal of duplicates, titles and abstracts were screened and full text versions of publications which passed screening were further evaluated for inclusion.
Data Extraction and Synthesis
After detailed tabulation of main study parameters (Supplemental Table S3) and outcome data of interest according to PICO criteria (Supplemental Tables S5-S8), recruitment times and study centers were compared to identify possible patient data crossover. Clinical studies identified in study registries were matched to respective publications where possible and published reports compared to database entries for missing data.
Results were synthesized looking at the highest level of evidence available for the respective patient groups. Results were summarized qualitatively with descriptive statistics where reasonable. The quality of comparative studies was assessed by evaluating patient inclusion and baseline characteristics, consistency of follow-up, and possible patient data crossover. However, a formal quality assessment, for example, according to methodological index for non-randomized studies (MINORS) criteria 9 was not performed since a meta-analysis was not the aim of this study.
Results
The systematic search identified 237 publications and 14 entries in study registries for screening. Full text analysis of 115 records for eligibility left 91 publications for inclusion in qualitative synthesis. See PRISMA summary for details of excluded studies in Figure 1.
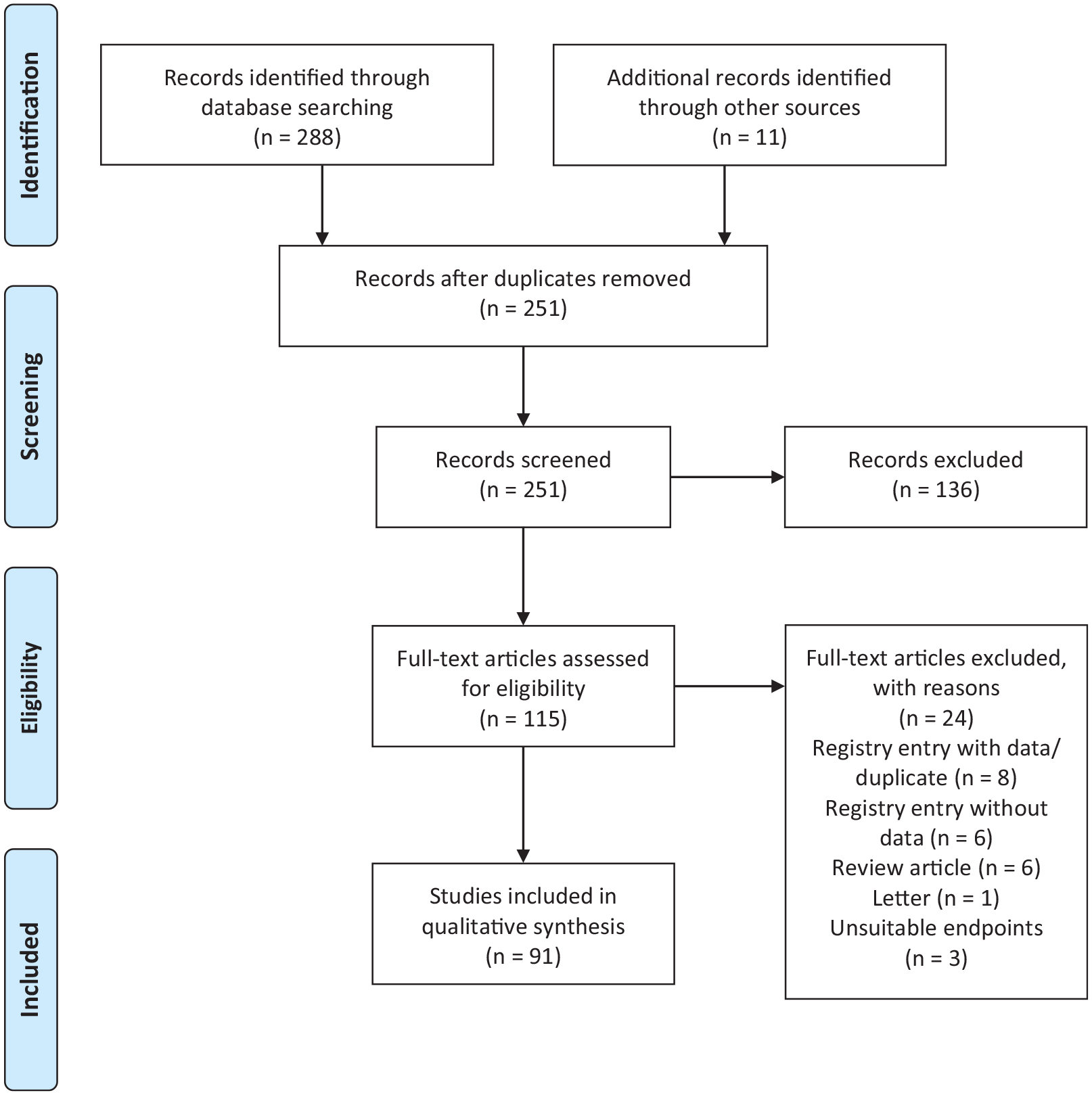
PRISMA summary of systematic review.
Characteristics of Included Studies
The characteristics of the included publications are as follows: 3 publications from 1 randomized controlled trial comparing MSA with PPI therapy; 35 comparative cohort studies, 15 comparing MSA to LF, and 20 comparing MSA between different patient groups. Thirty-eight single arm studies were identified, of which 17 were prospective single or multi-center studies. Four reports on device safety resulted from database searches involving thousands of patients each. Eleven meta-analyses were identified. Detailed characteristics of the 91 publications included are reported in Supplemental Table S3, and an overview of comparative studies MSA versus LF included in meta-analyses is presented in Table S4 of the Supplemental Material.
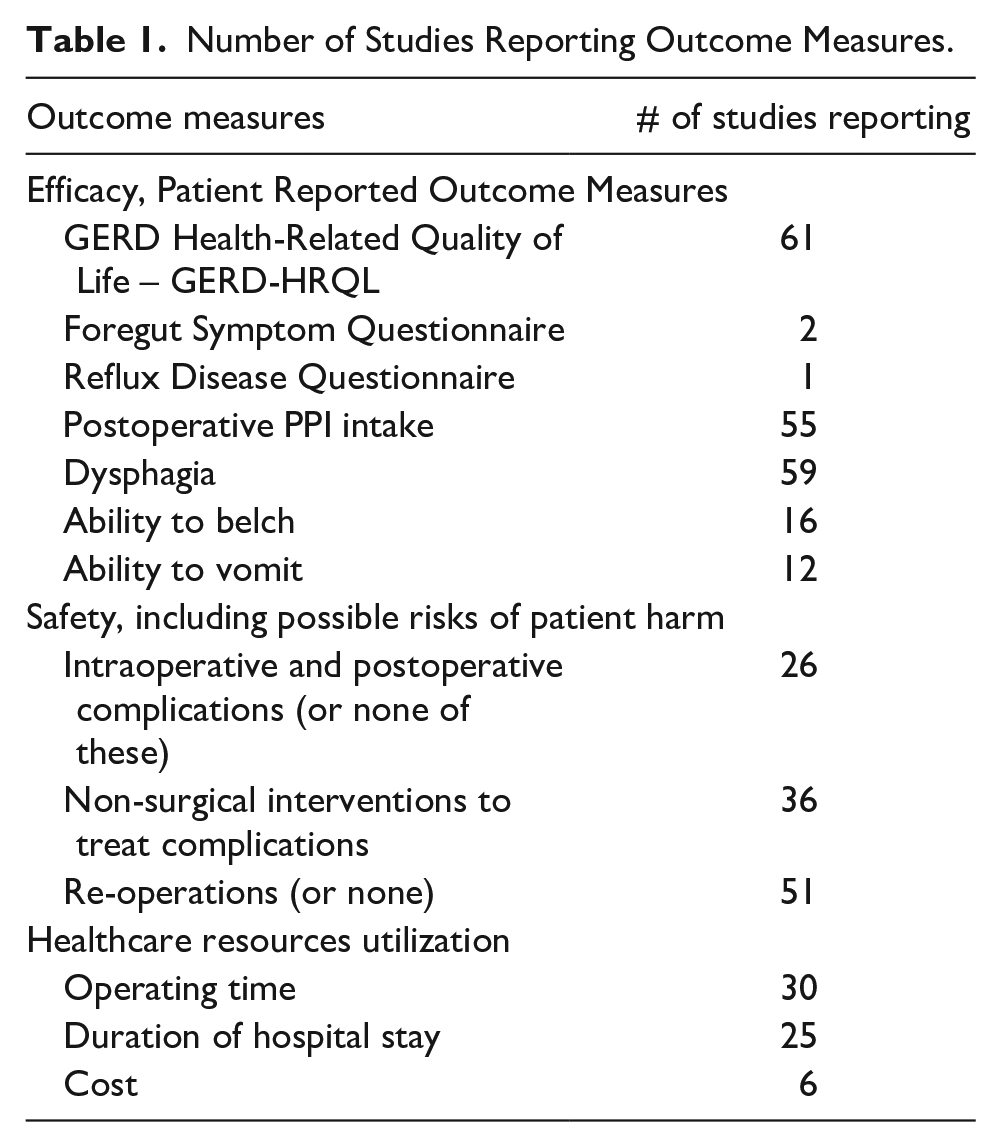
The outcomes of interest reported in these studies are summarized in Table 1.
Number of Studies Reporting Outcome Measures.
Clinical Effectiveness
Most studies report clinical effectiveness outcomes, which are provided in Table S5 (GERD Health Related Quality of Life and PPI use) and Table S6 (retaining ability to belch or vomit) of the Supplemental Material for respective studies.
Results of single-arm MSA studies have been synthesized in 3 meta-analyses.10-12 Baseline GERD Health Related Quality of Life (GERD-HRQL) score ranged from 24 to 27, postoperative GERD-HRQL score ranged from 2 to 6 and complete discontinuation of PPIs was achieved by 79% to 87.8% of patients with 60 months follow-up. For more details of single arm studies and early results see Supplemental Material.
Long term sustained symptom relief was reported by Ferrari et al, 13 who published results at 6 to 12 years follow-up (median 9 years) of 124 patient who underwent MSA surgery at a single center between 2007 and 2014. The mean GERD-HRQL total score improved from 19.9 at baseline to 4.01 at the latest follow-up (P < .001), 89% met the long-term success criteria defined as a ≥50% improvement in the GERD-HRQL and complete PPI cessation. Complete cessation or at least 50% reduction in the average daily dose of PPIs was reported by 79% and 89.5% of patients respectively.
Randomized Controlled Trials
One randomized controlled trial compared MSA with double-dose PPI therapy in patients with moderate to severe regurgitation despite once-daily PPI use. 14 The study included 152 GERD patients from 21 centers in the United States between July 2015 and February 2017. Patients were randomized to twice-daily (BID) PPI therapy (omeprazole 2 × 20 mg/d, n = 102) or to laparoscopic MSA with the LINX® System (n = 50). The primary endpoint of the study was elimination of moderate to severe regurgitation based on Foregut Symptom Questionnaire (FSQ) 15 scoring at 6 months.
After 6 months MSA was superior to BID PPI therapy with respect to the primary endpoint (89% vs 10%, Risk Ratio (RR) 0.11, 95% Confidence Interval (CI) 0.06 to 0.20, P < .001). A greater than 50% improvement in GERD-HRQL was 81% in the MSA group versus 8% in the double-dose PPI group (P < .001). 91% of the MSA group were completely free of PPI use after 6 months. 14 A multivariate analysis showed improvement in regurgitation score and GERD-HRQL remained independent predictors of satisfaction with therapy (P ≤ .001 for each). 16
The 12-month study results confirmed the efficacy and safety of MSA. At 6 months, patients in the BID PPI group could elect to cross-over to MSA. At 1 year, 98% of the MSA patients reported elimination of moderate to high regurgitation, and a ≥ 50% improvement in GERD-HRQL score was achieved in 93% of patients. Instances of dysphagia were reported by 39% of patients, but at 12 months dysphagia scores of >3 were reported by 7% of MSA patients. Patients in the crossover to MSA group reported similar outcomes at 6 months post MSA. Of note, dysphagia and bloating scores were higher (worse) in patients on PPIs throughout the study. 17
Cohort Studies Comparing MSA to Laparoscopic Fundoplication
The literature search identified 14 studies and 15 publications on single or multicenter cohort studies comparing MSA with different variants of laparoscopic fundoplication (LF).18-32 Table 2 summarizes study characteristics and key outcome measures of the ten studies reporting a minimum of 6 months follow up.
Studies Comparing MSA to LF Reporting at Least 6 months of Follow-up.
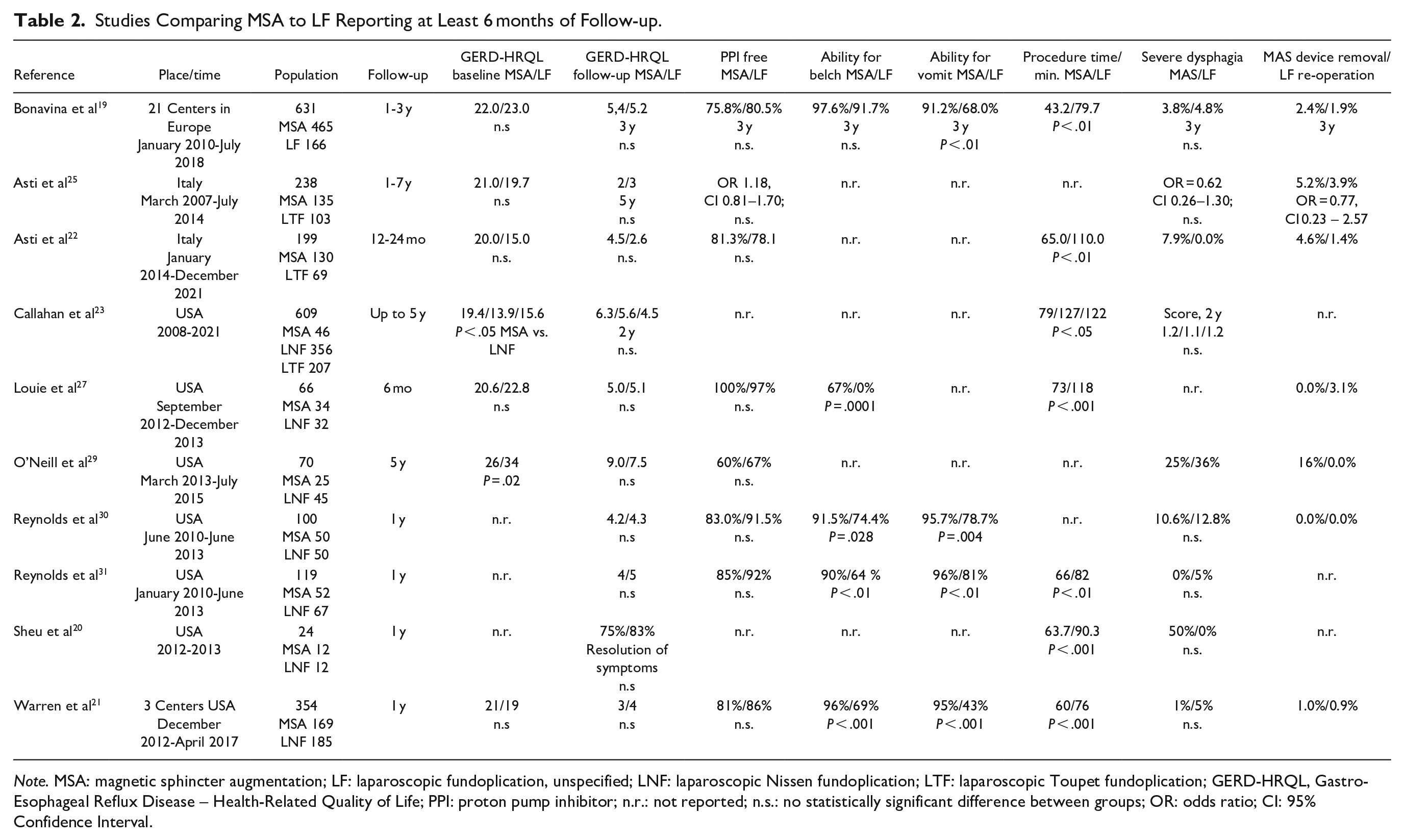
Note. MSA: magnetic sphincter augmentation; LF: laparoscopic fundoplication, unspecified; LNF: laparoscopic Nissen fundoplication; LTF: laparoscopic Toupet fundoplication; GERD-HRQL, Gastro-Esophageal Reflux Disease – Health-Related Quality of Life; PPI: proton pump inhibitor; n.r.: not reported; n.s.: no statistically significant difference between groups; OR: odds ratio; CI: 95% Confidence Interval.
These studies report on a total of 2410 patients, 1118 treated with MSA and 1292 with LF (166 LF unspecified, 747 laparoscopic Nissen (LNF), and 379 laparoscopic Toupet fundoplication (LTF)), but there is a significant cross-over of patients to multiple publications (See Supplemental Material for details).
Bonavina et al 19 reported the final data and 3-year follow-up of 465 MSA and 166 LF patients from a prospective, multicenter, observational registry study that treated patients in 22 centers from 4 European countries between 2010 and 2014. Both procedures resulted in significant improvements in total GERD-HRQL score and a significant decline in PPI usage from baseline to 3 years after surgery, as shown in Table 2. There was no statistically significant difference between the 2 groups for these outcome measures. MSA enabled a significantly higher percentage of patients to vomit when needed at each postoperative time point, with 91.2% of patients still reporting the ability to vomit at 36 months. At the same time point, only 68% of LF patients were able to vomit when needed. 19 Riegler et al 18 had reported on a preliminary subset of data with 1-year follow-up of this European registry.
The MSA and LF cohorts in the prospective, observational study by Asti et al. 2016 were largely similar and the outcomes were in line with those of the European registry. The results also confirmed that symptom improvements are maintained over time for up to 7 years with similar trend in both the MSA and LF groups. 25
O’Neill et al reported 5-year outcomes of a prospective, single center cohort study including 25 patients treated with MSA and 45 with LF. At a median follow-up of more than 5 years, both groups reported a sustained significant improvement from baseline for GERD-HRQL and a decrease in PPI use, but the difference in results between the 2 patient groups were not statistically significant. 29
To circumvent allocation bias, the studies by Reynolds et al 30 and Warren et al 21 compared propensity score-matched pairs of patients who either received MSA or underwent Nissen LF. Reynolds et al. reported on 50 matched pairs and Warren on 114 matched pairs. At 1 year of follow up, there were no significant differences between the MSA and LF groups with respect to GERD-HRQL and severe dysphagia. Both studies report more MSA patients were able to belch or vomit if needed (Table 2) and there were more patients in the LF group suffering from severe gas and bloating. Warren et al 21 reported a higher number of patients required daily PPI postoperatively in the propensity matches subgroup analysis (24% in the MSA cohort vs. 12% in the LF cohort, P = .02), but there was no significant difference in the overall study population at 1 year.
Meta-analyses Comparing MSA to Laparoscopic Fundoplication
The literature search identified 6 meta-analyses including both prospective and retrospective studies comparing MSA to LF10-12,33-35 and 4 comparing MSA with other techniques36,37 or collecting MSA data for special indications.38,39
Of the meta-analyses comparing MSA to LF, only Skubleny et al 34 from 2017 included studies with independent datasets.18,20,21 This meta-analysis includes a combined total of 627 patients with follow-up data, 244 Nissen LF and 383 MSA. The median duration of follow-up ranged from 7 to 16 months for LF and from 7 to 12 months for MSA. The cohorts were largely similar in their baseline parameters.
The GERD-HRQL score showed a significant reduction in symptom severity in both the MSA (from 20.5 pre- to 3.0 post-operatively) and LF groups (from 19.7 to 3.2), but no significant difference between groups. The MSA group was statistically superior to the LF group with respect to the ability to belch (95.2 vs 65.9%, P < .00001) and the ability to vomit (93.5 vs 49.5%, P < .0001). Other efficacy results were comparable for the 2 procedures. 34
A recent randomized trials network meta-analysis aimed at comparing various endoscopic and surgical procedures for GERD treatment and provides an indirect comparison of MSA to the other treatment options via comparison to PPI. This comparison was only possible for the outcomes dysphagia and bloating, and the differences were all not statistically significant. 40
Developing Indications for MSA
Expanding on the standard indication for LINX® implantation, there are recent studies showing that LINX® placement after bariatric surgery in patients with ongoing or de novo GERD is not only a safe and effective method,41,42 but also a viable rescue therapy for refractory reflux symptoms. 43
Recently published data suggested there is regression of Barrett’s esophagus following placement of LINX®. In the retrospective analysis of Alicuben et al. of 443 patients undergoing MSA, 86 patients had preoperative Barrett’s. Of these patients, 71.6% showed regression at a median of 1.2 years of follow-up. 44
The effect of MSA on predominantly atypical symptoms had been discussed recently, 45 but it is clear that more studies will be needed to validate these developing indications of LINX® implantation.
Safety
Besides the safety aspects discussed in the reports of each study (see Supplemental Material Table S8 for details), the literature search identified 6 studies looking specifically at the safety profile of MSA device with respect to device removal and their causes.46-51 Four of these studies evaluated the Manufacturer and User Facility Device Experience (MAUDE) database and had access to the manufacturer’s complaint database. Table 3 summarizes the results together with those from studies with large patient cohorts.
Removal and Erosion of the LINX MSA Device in Large Patient Cohorts.
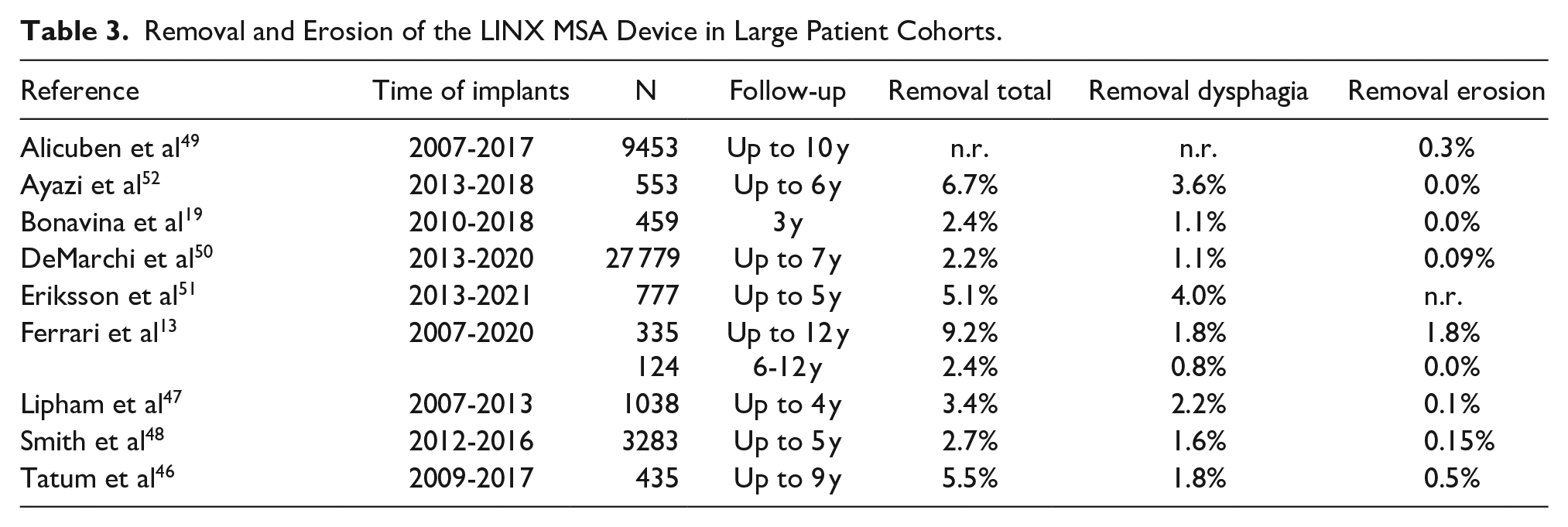
Some long-term complications may require the removal of the implant. Dysphagia is the most common reason for removal accounting for approximately 50% of cases. Other reasons for removal include persistent or recurrent GERD, vomiting/regurgitation, gastric pain, or the need for Magnetic Resonance Imaging (MRI). In contrast, reported removals due to device erosion through the esophageal wall and into the lumen are rather low and the cumulative risk of erosion at 7 years was 0.28% (95% CI: 0.17-0.46%). 50 Over 50% of removals seem to occur within 1 year and over 80% within 2 years of implantation.48,19
A summary of the worldwide experience of 9453 LINX System implantations from February 2007 to July 2017 regarding the rate of erosion identified 29 cases (0.3%) of erosion. 49 90% of these patients presented with a recurrence of dysphagia and median time to erosion was 26 months. All interventions were without complications, and after a median follow-up of 58 days, 24 patients were symptom-free, 4 patients showed mild dysphagia, and 2 patients showed mild odynophagia. Smaller devices were more commonly associated with erosion with 12-bead devices having a 4.93% erosion rate. 49 This device is no longer available on the market.
A later study including 27 779 implantations from 2013 to 2020 did not include the 12-bead device nor the original device design that was secured by sutures instead of the currently used clasp closure. 50 The authors also identified device size is likely to have a correlation to erosion with the smaller sizes (13 and 14 beads) making up 65% of the total. They also observed a change in clinical practice with an increase in average device size from 14.2 ± 1.0 in 2013 to 15.3 ± 1.2 in 2019 (P < .001). 50
In the largest observational study with 465 MSA and 166 LF patients evaluating the two methods in clinical practice, intraoperative and surgery-related complication rates were similar. 19 Intraoperative complication rate was 1.8% for MSA and 1.2% for LF. The procedure-related complication rate was 2.0% for MSA and 1.8% for LF, respectively. The proportion of patients with outpatient clinic visits for GERD symptoms or due to surgery-related complications was similar for MSA and LF patients over the 3 years. Dysphagia rates were also similar in both groups across all follow-up time points with 3.8% of MSA and 4.8% of LF patients scoring their dysphagia as “bothersome.” 19 Additionally, all the other studies comparing MSA to LF did not find a statistically significant difference in dysphagia between the two groups (Table 2).
Healthcare Resource Use
All studies with fundoplication as the comparator showed a statistically significant shorter procedure time for MSA. In two studies with the largest independent data sets, the procedure times were 43 and 60 minutes for MSA and 80 and 76 minutes for LF, respectively (P < .001).19,21 Warren et al, 21 who measured hospital length of stay in hours, found a statistically significant shorter length of stay for MSA of 13 hours versus 32 hours for LF (P < .001). Callahan et al 23 report a length of stay for MSA of 7 hours and for LNF and LTF 31 and 24 hours, respectively (P < .001). From the European registry study, for a hospital length of stay < 24 hours, 36.1% for MSA and 11.4% for LF were reported, and for a hospital length of stay > 48 hours, 50.8% for MSA and 72.3% for LF. 19
At a single institution study, 514 of 553 MSA patients (93%) were discharged home on the day of operation. A total of 39 patients required at least 1 overnight stay, with a mean of 1.5 (1.0) nights. 52 The 1-year results from a post approval study with 200 patients showed 91% of patients were discharged within 24 hours after the procedure. 53
A study performed at a Western Pennsylvania health network compared the cost of 180 MSA patients vs. 1131 LF for a period of 12 months before and after surgery. 26 The median reimbursement for surgery was $13 522 (13 195-14 439) for patients who underwent MSA and $13 388 (9951-16 261) for patients who underwent LF (P = .02). The cost per member per month related to the upper gastrointestinal disease decreased post surgery 66% ($305-$104) for the MSA group and 46% ($233-$126) for the LF group (P = .0001). 26 A study in Southern California comparing procedure cost for 52 MSA and 67 LF patients also showed similar total charges with a mean charge of $48 491 for MSA and $50 111 for LF (P = .506) with the higher cost for the LINX device offset by higher charges for LF patients due to pharmacy/drugs, laboratories/tests/radiology, operating room services, and room and board. 31
Discussion
This systematic review of the clinical evidence available establishes the clinical effectiveness, safety, and cost effectiveness of MSA for the treatment of GERD. Most importantly, patient-reported outcome measures have consistently found significant improvements in quality-of-life metrics as well as cessation or reduction in PPI use. Long-term studies confirm durability for 5 years and beyond.13,54,55
The RCT comparing MSA to twice daily PPI provides the highest level of evidence available. MSA demonstrated superiority regarding patient relevant symptom control of regurgitation (89% after MSA vs 10% with PPI), improvement in GERD-HRQL (81% vs 8%), and satisfaction with the situation after MSA of 81% versus 2% after PPI therapy.14,17 As PPI therapy has been the “gold standard” medical therapy, the results of this study are reflected in recent American College of Gastroentereology (ACG) guidelines recommending consideration of MSA as an alternative to laparoscopic fundoplication for patients with regurgitation who fail medical management. 1
We identified 15 publications reporting PROM outcomes from single center and multicenter cohort studies comparing MSA with different variants of LF (Nissen, Toupet or unspecified). These publications show that both LF and MSA are safe and effective for more than 5 years with quality-of-life scores and rates of post operative PPI usage, as well as the rate of endoscopic dilation for dysphagia and rate of reoperation related to complications being similar in both patient groups. Preservation of the patient’s ability to belch and vomit show a statistically significant advantage of MSA over LF (Table 2). The majority of identified comparative studies compare MSA to LNF and only more recent studied include the partial fundoplication LTF. However, preservation of the patient’s ability to belch and vomit had not been reported from studies comparing MSA to LTF. There would be a need for a study comparing MSA to partial fundoplication reporting on the full scope of outcomes.
Implantation of the LINX® System seems to be a safe procedure as intraoperative complications are reported only in exceptional cases.47,19
Temporary postoperative dysphagia is common to both MSA and LF (see Supplemental Table S7), The long-term incidence of dysphagia in MSA patients is similar to LF. In more severe cases of dysphagia or persistent symptoms, removal of the implant or revision of the fundoplication is indicated, with approximately equal incidence for MSA and LF (Table 2). Removal of the implant occurred predominantly within 1 year after the MSA index procedure and rarely after more than 2 years, indicating that over time rate of removals or problems with the device does not appear to escalate. 48
The cumulative risk of erosion at 7 years was estimated at 0.28%. It is fair to anticipate that after introduction of a new sizing tool by the manufacturer in 2013 and a discontinuation of the smallest size LINX® system with 12 beads, 50 the risk for device erosion can be considered very low.
Laparoscopic removal of the LINX® device can be safely performed as a 1-stage procedure and in conjunction with fundoplication even in patients presenting with device erosion. 7 In comparison, expert opinion suggests it is both more difficult and more traumatic to repair a failed fundoplication than to remove a LINX® System and create a new fundoplication without the need for anatomic reconstruction.48,56
All comparative studies assessing fundoplication and LINX® show a statistically significant shorter operation time for MSA. This is likely a result of the MSA procedure being highly standardized, needing less instrumentation, and can be completed with less tissue disruption and coagulation as compared to LF. With a less traumatic procedure, MSA patients are also more likely to be discharged the same day (93%) or within 24 hours of the procedure (91%).52,53 In contrast, LF patients are most likely treated as in-patients. According to the American College of Surgeons National Surgery Quality Improvement Program (ACS NSQIP), only 7.3% left the hospital the same day. 57 Despite the higher cost for the LINX® implantation compared to procedure cost of LF, total charges for the index procedure seem to be comparable to those for fundoplication whereas MSA results in a reduction of disease-related expenses for the payer in the year following surgery.26,31 As reoperation rates observed over more than 4 years are comparable for the 2 procedures, this is unlikely to shift the cost-balance to either side.
The American College of Gastroentereology, the American Foregut Society and the Society of American Gastrointestinal and Endoscopic Surgeons issued position statements or guidelines about the role of MSA in the broader context of ARS.1,58,59 They see the dynamic nature, objective sizing, and procedural standardization of the device to offer several potential advantages over traditional ARS. 58 They also recognize the growing body of evidence confirming the initial safety profile of MSA that led to FDA approval and has demonstrated to result in long-term GERD control based on symptomatic outcomes. 59 The recently published ACG Clinical Guideline for the Diagnosis and Management of GERD 1 recognizes the minimal surgical dissection required for MSA resulting in greater technical ease, shorter operative times, and shorter durations of hospital stays than for fundoplication. The guideline recommends consideration of MSA as an alternative to laparoscopic fundoplication for patients with regurgitation who fail medical management (strong recommendation, moderate level of evidence). 1 The recently published American Gastroenterological Association guidelines and the outcome of the multi-society consensus conference state that, in patients with proven GERD, fundoplication and magnetic sphincter augmentation are effective surgical options.60,61 In the UK, the National Institute for Health and Care Excellence (NICE) states that the evidence on the safety and efficacy of laparoscopic insertion of a magnetic ring for gastro-oesophageal reflux disease (GORD) is adequate to support using this procedure. 62
This literature review aimed at understanding the complete evidence available for MSA. The sheer number of published studies forced a limitation to higher levels of evidence and therefore this review cannot appreciate all studies. There is a vast number of single arm studies, but the number of studies comparing MSA to standard surgical procedures as LNF is limited or especially to LTF it is minimal. There is no randomized controlled trial (RCT) comparing these surgical procedures. There was no formal quality assessment of included studies and some of the studies may suffer from various forms of bias. Some of the studies comparing MSA with LF have significant differences in baseline characteristics between patient groups. In addition, long term follow-up data may be compromised by higher dropout rates than usually acceptable and small patient numbers in some of these trials led to inconclusive results (no significant difference). In an ideal world, this limitation would be resolved by adequately powered, well-designed, multicenter RCTs comparing the 2 interventions. However, in view of the available data for effectiveness, durability, and safety of MSA with the LINX® device, the added value of such an RCT may be difficult to attain especially given the challenges recruiting the sample size needed to detect smaller differences in outcomes between procedures, which may not be very relevant in clinical practice.63,64
By now it is estimated that over 40 000 LINX® devices have been implanted worldwide. LINX® has been demonstrated to provide long lasting relief to patients suffering from persisting GERD and has a proven safety profile. MSA allows an optimization of healthcare resource with short recovery and can be performed in an outpatient day-procedure setting. This real-world evidence should be recognized among providers, government agencies and payers.
Supplemental Material
sj-docx-1-gut-10.1177_26345161241263051 – Supplemental material for Current Clinical Evidence for Magnetic Sphincter Augmentation: A Scoping Review
Supplemental material, sj-docx-1-gut-10.1177_26345161241263051 for Current Clinical Evidence for Magnetic Sphincter Augmentation: A Scoping Review by Thomas Andreae, Moustafa Elshafei, James A. Gossage, Thomas Kersting and Reginald Bell in Foregut
Footnotes
Acknowledgements
The authors would like to acknowledge the support provided by Christine Eckenweber-Black with setting up the systematic literature review at the various databases and Janet DeMarchi, Nadine Jamous, Ana Dubon Garcia, Vito Paragò and Joerg Tomaszewski for critical review of the manuscript.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Thomas Andreae has a consulting agreement with Johnson & Johnson. Moustafa Elshafei has a consulting agreement with Johnson & Johnson. James A. Gossage has a consulting agreement with Johnson & Johnson. Thomas Kersting has a consulting agreement with Johnson & Johnson. Reginald Bell has a consulting agreement with Johnson & Johnson.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: Ethicon (part of Johnson & Johnson) provided funding for the project.
Ethics Approval
This study only included published literature, no IRB approval or written consent was necessary.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
