Abstract
Transoral outlet reduction (TORe) is an endoscopic intervention for patients who have weight recurrence, dumping syndrome, or post-prandial hypoglycemia following Roux-en-Y gastric bypass (RYGB). TORe offers a minimally invasive alternative to surgical revision, which is often associated with high morbidity rates. This paper will detail the approach to TORe in regard to patient selection, endoscopic technique, and post-TORe outcomes.
Introduction
Obesity, defined as body mass index (BMI) ≥30.0 kg/m2, has become an epidemic in the United States (US). In the National Health and Nutrition Survey (NHANES) from 2017 to 2018, an estimated 42% of US adults met the criteria for obesity and by 2030, it has been projected that 1 in 2 US adults will have obesity.1,2 Obesity can also vary by racial or ethnic background, with an estimated 50.4% of Mexican American populations meeting the criteria for obesity in 2017 to 2018.1,3 While metabolic bariatric surgeries (MBS), such as sleeve gastrectomy and Roux-en-Y gastric bypass (RYGB), have been shown to be effective for weight loss and improvement in metabolic complications associated with obesity, up to 30% of individuals may experience weight recurrence.4,5 Recognition and management of gastro-gastric fistulas is important in the evaluation of post-RYGB weight recurrence, and approach to closure is discussed. In addition, transoral outlet reduction (TORe) has emerged as a safe and effective minimally invasive option in the management of obesity following RYGB. Patient selection, pre-procedural evaluations, technique (including suturing patterns, number of sutures, adjunctive APC, etc.), and evidence of post-revision outcomes are reviewed.
Gastro-gastric Fistula
Gastro-gastric fistulas (GGF) are a well-known cause of weight recurrence after RYGB. GGFs are defined as an abnormal connection between the gastric pouch and excluded remnant stomach or jejunum near the gastrojejunal anastomosis and occur in 1.3% to 6% of operations.6–8 Symptoms of GGFs commonly include epigastric abdominal pain, nausea, vomiting, and gastroesophageal reflux (GERD).6,9 Patients may also present with weight recurrence, inadequate weight loss, or marginal ulceration.6,9 GGF’s are commonly found on UGI contrast study or during diagnostic upper endoscopy (Figure 1). There is no standard approach to treatment of GGFs. Some patients may choose to undergo medical management to treat symptoms of GGFs. However, many patients require closure of their GGFs and options for treatment of GGFs include endoscopic closure or surgery. While a number of different endoscopic options are available, current literature supports closure of GGFs with endoscopic suturing using the Apollo Overstich device (Apollo Endosurgery/Boston Scientific, Austin, TX). Endoscopic suturing of GGFs is safe with a low adverse event rate; however, studies have found the durability of closure after endoscopic suturing can vary, with some studies showing 0% of GGFs >20 mm remain closed after 395 days.10,11 Revisional surgery with resection of fistula and/or revision of the GJ anastomosis is sometimes necessary for the treatment of GGFs.6,10,11 A recent study comparing endoscopic versus surgical treatment of GGFs found that endoscopic treatment of GGF had fewer adverse events (endoscopy 8.9% vs surgery 35.6%, P = .005) but at 12 months, the surgical groups experienced greater total weight loss (endoscopy 1.9% vs surgery 6.2%, P = .007). 10 Given this, the management of GGFs may depend on the size of the GGF, patient symptoms, and the degree of weight recurrence.
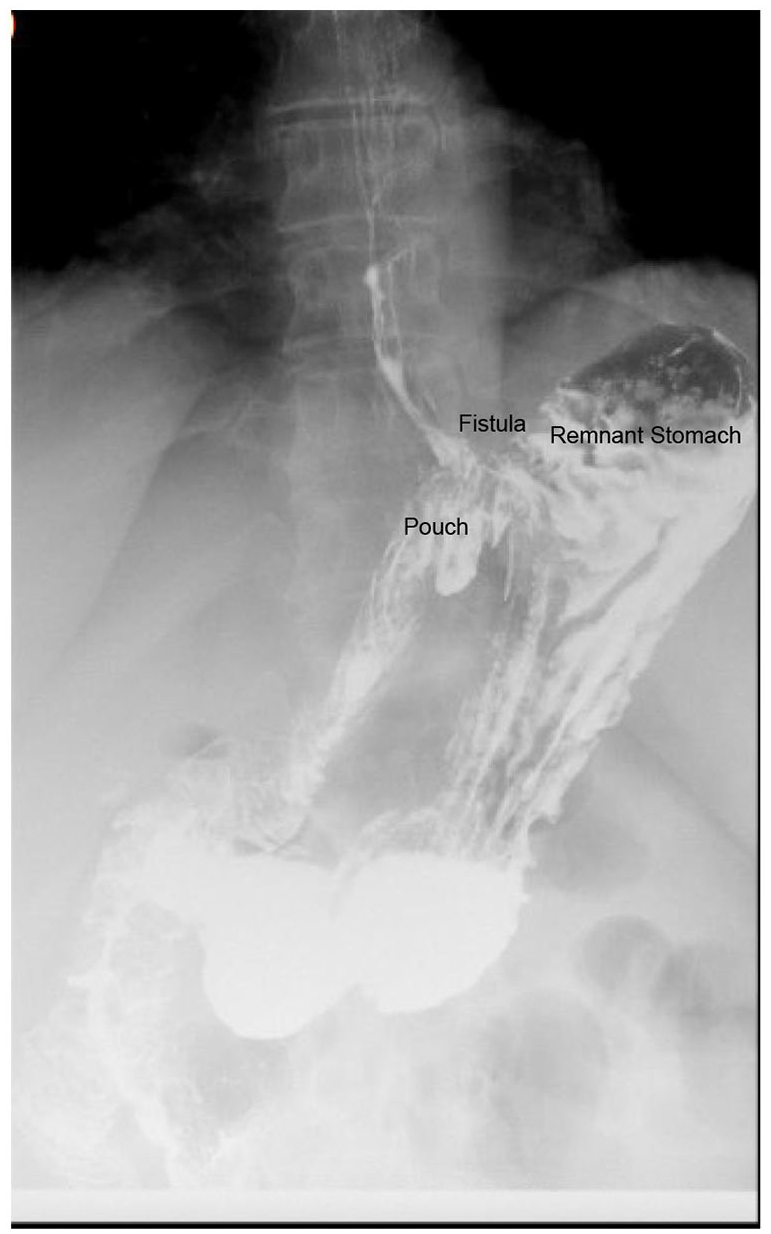
Radiographic representation of a gastro-gastric (GG) fistula.
Transoral Outlet Reduction (TORe)
Introduction
Weight recurrence after RYGB is often complex and multi-factorial. While there are psychosocial, behavioral, and physiological changes that may result in weight recurrence after MBS, studies have found that enlargement of the gastric pouch or gastrojejunostomy (GJ), which leads to a decrease in restriction, may play a role in weight recurrence after RYGB.12–14 Although surgical revision of a dilated GJ has been described, it is associated with an increased risk of surgical adverse events and high risk of morbidity. 15 Endoscopic bariatric therapy (EBT) to treat weight recurrence after RYGB was first described in 2004. 16 Over the next decade, EBTs were refined until the results of a randomized controlled trial comparing TORe to sham procedure provided evidence that TORe is an effective and safe therapy for weight recurrence after RYGB.17–19 Since then, several different approaches to TORe have been described, including using mucosal ablation with argon plasma coagulation (APC) before performing TORe with a full-thickness suturing device, different suturing patterns, and a modified TORe procedure with endoscopic submucosal dissection. 5 In this review, we will focus on mucosal APC-TORe, including patient selection, endoscopic technique, and outcomes.
Patient Selection and Pre-Procedural Evaluations
Patients that qualify for TORe following RYGB include: (1) weight recurrence after RYGB with unsuccessful attempts at weight loss using lifestyle intervention and/or anti-obesity medications (AOM); (2) Minimum BMI of 27 kg/m2; (3) Dumping symptoms, including post-prandial hypoglycemia; (3) Dilated GJ anastomosis of at least 15 mm in diameter.
At the University of Texas Southwestern (UTSW) endobariatrics clinic, all patients are seen in clinic prior to the procedure to discuss their prior weight loss history, TORe procedure, and weight loss expectations. We believe it is important for these patients to be counseled on weight loss expectations since revisional procedures often do not result in similar weight loss as the initial MBS (see Outcomes and Adverse Events). Patients who are actively smoking cigarettes, vaping, or using other tobacco products are not offered a TORe since these products often inhibit tissue healing. Furthermore, we do not require any pre-procedure diet other than remaining without anything to eat or drink after midnight the day before the procedure.
Glucagon-like peptide-1 (GLP-1) receptor agonists, which are increasingly popular AOMs, are often associated with delayed gastric emptying. 20 Consistent with guidance from the American Society of Anesthesiologists (ASA), our anesthesia colleagues recommend holding GLP-1s one week prior to their procedure. 21 However, for patients with severe post-prandial hypoglycemia who have had some benefit in their hypoglycemic events with GLP-1s agonists, we often ask them to stay on their GLP-1 agonists after a multi-disciplinary conversation with our anesthesiologists.
Endoscopic Technique
TORe procedure is performed in the endoscopy unit at UTSW. Patients undergo general anesthesia with endotracheal intubation and the endoscopic procedures are all performed with CO2 insufflation. The procedure can be started with either a standard adult upper endoscope or a double-channel endoscope (GIF-2T160; Olympus America, Center Valley, PA). In my practice, I begin with a double-channel endoscope to minimize time switching scopes; however, it is important to note that applying APC around the GJ with a double-channel scope can be more challenging since the scope is less flexible. A standard upper endoscopy is performed to estimate the diameter of the GJ, pouch length, and to confirm there are no contraindications to the procedure. The most common contraindications I encounter include a small pouch length (~1-2 cm), which can be seen in patients with a history of surgical revision after RYGB, non-dilated GJ anastomosis, and/or large marginal ulceration. Among patients with a large marginal ulceration, treatment with open-capsule PPI can be utilized with a repeat attempt at TORe after treatment has been completed. 22
After a diagnostic upper endoscopy, a 5 to 10 mm area around the GJ is ablated using straight-fire APC. Using the double-channel scope for APC allows sequential suctioning of gas released during APC, which can minimize abdominal bloating and post-procedure discomfort. If a standard adult upper endoscope is used, intermittent suctioning of gas produced by the APC is recommended as this can often cause pain, and may prolong patient recovery and lead to hospital admission.
After APC, the Apollo Overstich device is placed on the double-channel scope and passed into the pouch with or without the use of an Overtube device. The Overtube device can help to facilitate passage into the esophagus and retain CO2 during the procedure but is not mandatory during TORe. Full-thickness sutures are then placed around the GJ, ideally in a purse-string fashion, as a more-bites-per-suture pattern seems to improve durability by reducing tension on the suture-anchor (Figure 2, Video 1).23,24 A full running purse-string usually starts at the 3 o’clock position and continued in a counterclockwise fashion with 8 to 12 bites using a single suture. 24 In some patients with challenging anatomy or small pouches, I sometimes perform a modified purse-string pattern starting at the 3 o’clock position and suturing as much of the outlet that is accessible with the double channel scope, aiming for at least 4 to 5 bites to improve durability of the suture. During fellowship, I was taught to tighten the suture cinch over a 8 to 12 mm hydrostatic balloon passed through the second channel of the endoscope. While this method provides a precise size to the GJ aperture, my typical practice forgoes this method in an attempt to minimize waste and procedural costs. Anecdotally, this has not led to any difference in weight loss outcomes or GJ stenosis rates (see Outcomes and Adverse Events).
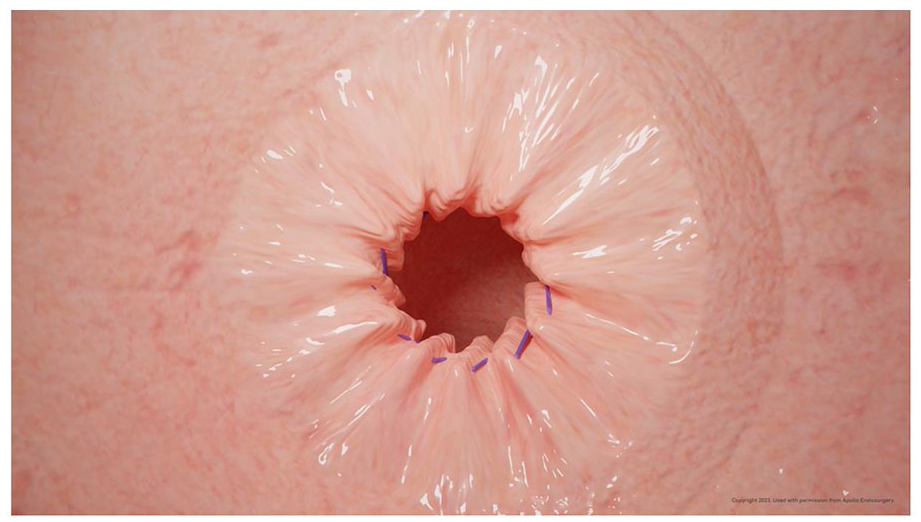
Purse-string suture pattern used in TORe.
Post-Procedure Evaluation and Follow-Up
All patients are observed in post-anesthesia care unit (PACU) for at least 1 h. Patients may receive IV medications to treat abdominal pain and/or nausea. All patients are discharged home the same day as their procedure with a plan for a modified diet, oral anti-emetics and a small amount of oral narcotics. While it would be ideal to provide liquid medications, most insurance plans do not cover liquid narcotics in our area.
The post-TORe diet at UTSW includes clear liquids on the day of their procedure, a week of full liquids, a week of soft foods, and then a slow progression to their regular diet starting on week 3. While there are no guidelines on the type of diet to start after these procedures, published literature varies on the length of time patients remain on a liquid diet, with some centers reporting a full liquid diet for up to 6 weeks after TORe.24,25
Outcomes and Adverse Events
A systematic review and meta-analysis found that total body weight loss (TBWL) at 12-months after TORe have been estimated to be 8.5%. 26 Five year outcomes show similar TBWL among patients who had a TORe. 27 One recent study found that the addition of pharmacotherapy/AOM after TORe can offer greater weight loss than either method alone, with up to 15% TBWL seen after the study period. 28
TORe can also be beneficial to patients with dumping syndrome after RYGB. Dumping syndrome is a constellation of gastrointestinal and vasomotor symptoms, such as diarrhea, syncope, or profound hypoglycemia that occur between 1 and 3 h after consuming a meal.29,30 Studies have found that TORe can be helpful for both classic dumping symptoms and for those patients with post-prandial hypoglycemia.30,31 In one multicenter study, the Sigstad score, which measures dumping symptoms in patients, improved from a mean of 17 to 2.6 three months post-procedure. 30 Further, a recent case-series from UTSW found that TORe can reduce the number of post-prandial hypoglycemic events. 31
Fatal adverse events after TORe have not been reported. The most common adverse events are abdominal pain (4.22%), bleeding (1.16%), and perforation (0.46%).27,32,33 Other reported adverse events include gastrojejunal stenosis (1-4%).26,34
Conclusion
Weight recurrence, dumping syndrome, and post-prandial hypoglycemia are long-term complications of RYGB. Endoscopic interventions to address these complications have been proven to be efficacious across decades of studies with few associated adverse events. TORe is an important intervention which can be offered to patients with complications of RYGB and presents a safe and effective intervention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
