Abstract
Esophageal squamous papilloma (ESP) is an uncommon, normally benign epithelial neoplasm with rare potential for malignant transformation. These lesions are oftentimes found incidentally on upper endoscopy and are typically managed with endoscopic excision, dissection, or ablation. However, there remains a paucity of published management guidelines in the current literature. We present a case of an older female who was found to have an extensive mid-esophageal ESP with recurrence despite endoscopic intervention. We highlight the multi-institutional management approach to her morphologically atypical ESP and the nuance involved in treating recurrent lesions of this type.
Introduction
Esophageal squamous papilloma (ESP) is an uncommon, normally benign epithelial neoplasm with rare potential for malignant transformation.1,2 These lesions are typically found incidentally on endoscopy and are classified morphologically as small, sessile, verrucous nodules. They have been identified throughout the esophagus but appear to be more commonly encountered in the mid to distal esophagus.2-4 The exact pathogenesis of these lesions is currently unknown, but an association with human papillomavirus (HPV) and chronic mucosal irritation and subsequent regeneration are two commonly postulated etiologies.2-5 Anatomic positioning of these lesions appears to depend largely on the identifiable cause of chronic esophageal irritation.3,4 These lesions are typically managed with endoscopic excision, dissection, or ablation, however, there remains a paucity of published management guidelines in the current literature.4,6,7 Here we present a case of an older female who presented with worsening symptoms of acid reflux who was found to have an extensive mid-esophageal lesion with satellite distal esophageal involvement that subsequently returned as recurrent, biopsy-proven esophageal squamous papilloma.
Case Description
A 72-year-old female with a past medical history significant for laryngopharyngeal reflux disease (LPRD), gastroesophageal reflux disease (GERD), fundic gland polyps, as well as unilateral nephrectomy secondary to renal cell carcinoma presented to gastroenterology clinic for a follow-up visit regarding uncontrolled acid reflux. Symptoms had previously been well controlled on daily famotidine with diet and lifestyle modifications, but symptoms recurred during the COVID-19 pandemic because of reported stress and poor diet. She had previously been diagnosed with LPRD by otolaryngology and omeprazole was added to her medication regimen before her office visit; she was previously not taking a proton pump inhibitor to avoid the potential of developing kidney injury given her history of nephrectomy. Despite medication compliance, symptoms persisted. She underwent esophagogastroduodenoscopy (EGD) which revealed a small Schatzki’s ring in the distal esophagus, as well as an area of raised but flat, discolored, and abnormally textured mucosa in the mid-esophagus which was biopsied (Image Panel 1A). Forceps biopsy of the mid-esophageal lesion revealed papillary proliferation of nondysplastic squamous epithelium with fibrovascular cores of lamina propria, consistent with squamous papilloma (Image Panel 1C-D). She underwent repeat EGD where absorptive chromoendoscopy using Lugol’s iodine showed the known lesion involving 80% to 90% of the luminal esophageal circumference (Image Panel 1B). She was referred to an expert in the field of endoscopic submucosal dissection (ESD) due to the extent of the lesion. Between endoscopies, a barium swallow was performed that was consistent with ineffective esophageal motility. ESD was later performed successfully removing 30 mm of abnormal mucosa with total resection of 50 mm of tissue (Image Panel 2A-C).
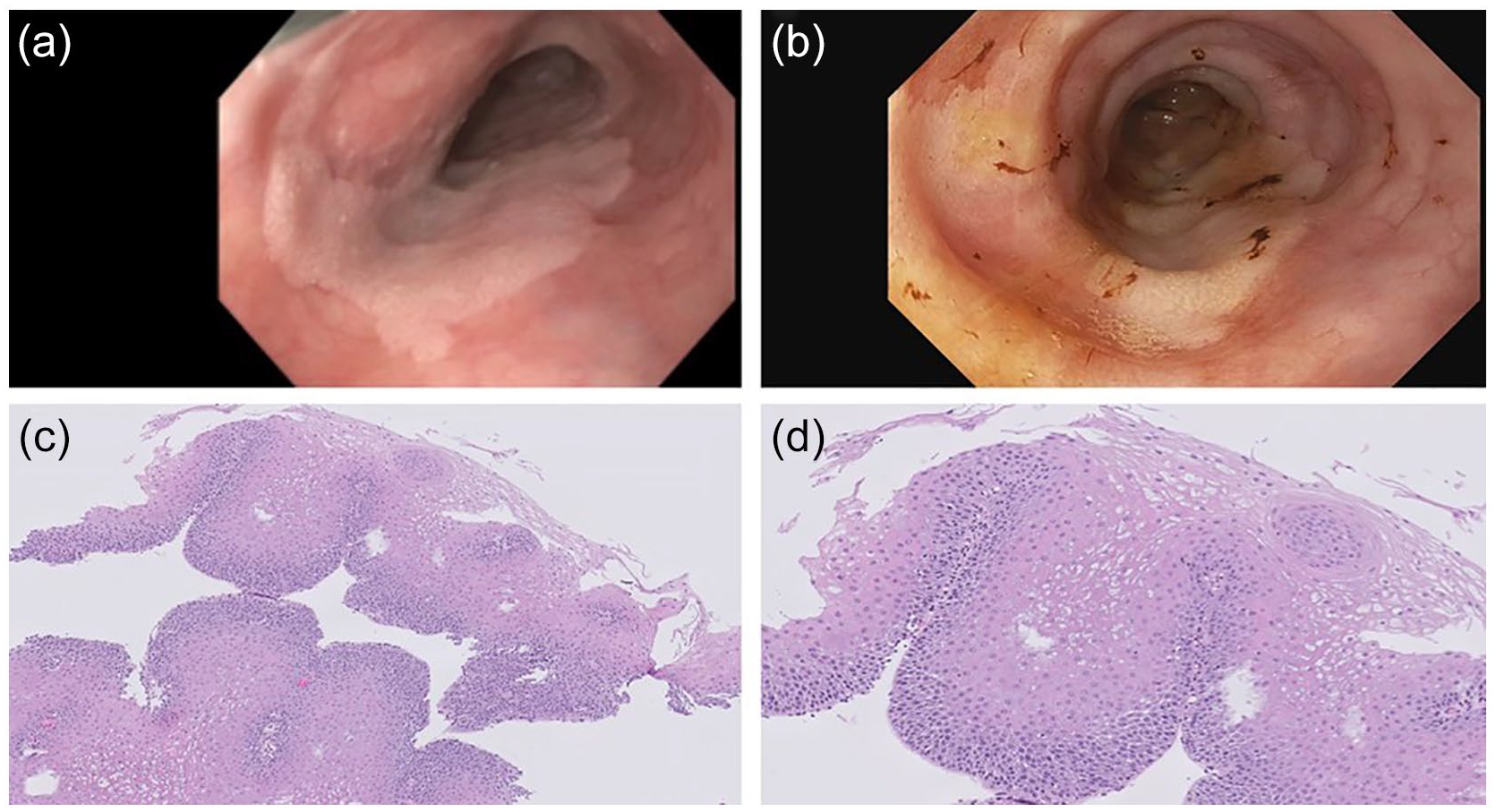
(A) Endoscopic view of ESP on index EGD, (B) endoscopic view of ESP after application of Lugol’s iodine, and (C-D) papillary proliferation of nondysplastic squamous epithelium with fibrovascular cores of lamina propria consistent with squamous papilloma at different magnifications.
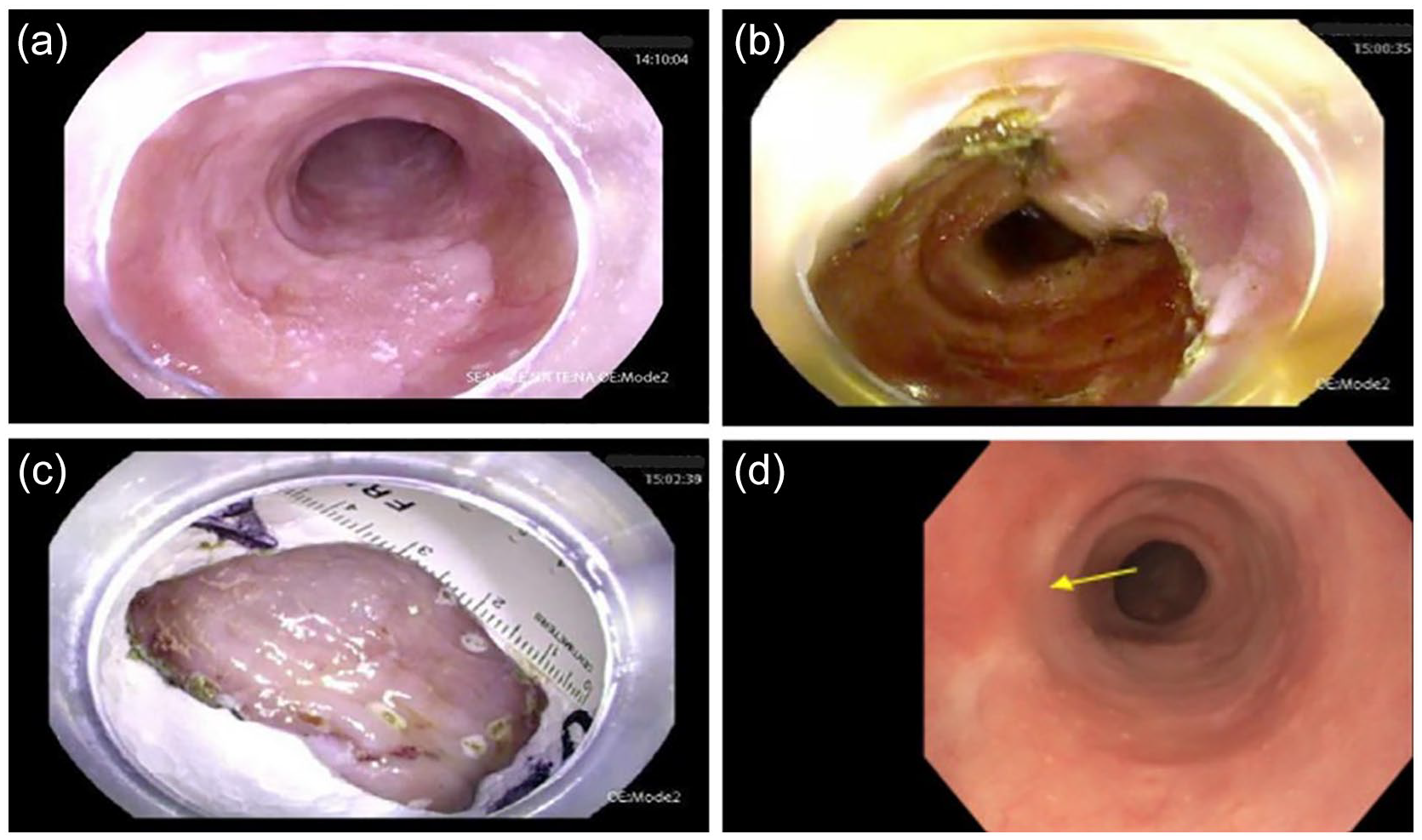
(A) Endoscopic view of ESP before ESD, (B) endoscopic view following ESD, (C) resected section of abnormal esophageal mucosa, and (D) endoscopic view of healthy-appearing ESD scar 6 months after ESD.
Pathology from the resected specimen revealed the known ESP with focal high-grade and low-grade dysplasia, which was not observed on the initial biopsy specimen. Multiple other small, scattered islands of abnormally textured mucosa were also noted in the mid/distal esophagus. Biopsy proved additional areas of squamous papilloma for which radiofrequency ablation (RFA) was performed. Her post-ESD course was complicated by stricture formation near the ESD scar that was treated with two sessions of serial balloon dilation. The most recent repeat endoscopy revealed healthy appearing post-ESD scar tissue with no gross evidence of residual ESP as well as negative biopsies (Image Panel 2D). Repeat biopsies of other small islands of abnormal residual mucosa confirmed ESP on histopathological analysis. RFA was repeated and the lesions were treated successfully without complication. Repeat surveillance endoscopy to monitor response to therapy is planned for 3 months post-intervention given the high-grade dysplastic changes seen on the resected ESD specimen.
Discussion
ESP is a normally benign neoplasm with a prevalence ranging from 0.014 to 0.45% in the general population. 1 There are conflicting reports in the literature regarding its association with HPV, as multiple reports have shown no association.6,7 In contrast, other studies have demonstrated an association with HPV; most commonly high-risk type 16, though a link has also been demonstrated in low-risk HPV strains 6 and 11 in HPV-positive ESP lesions.2,8,9 Risk factors associated with the transmission of HPV are largely related to unprotected sexual intercourse. Transmission from frequently to lesser frequently observed includes genito-oral, perinatal/vertical, auto-inoculation, and oro-oral, though limited data exists to support the latter two modalities. 10 It is hypothesized that a preexisting oropharyngeal HPV infection may travel beyond the oropharynx to infect the esophageal squamous epithelium due to the contiguous nature of the epithelial lining between the two anatomic regions. 8
Current literature has failed to show a true association with one single etiology related to the development of ESP, although chronic mucosal irritation appears to be the more commonly identified inciting factor.1-4 Lesions have been identified throughout the esophagus but appear to be more commonly encountered in the mid to distal esophagus, which is thought to be related to exposure to gastric acid.2-4 Another study on these lesions identified obesity (body mass index > 30) and cigarette smoking as the two most prevalent characteristics in patients found to have ESP on EGD. 11 Interestingly, both obesity and cigarette smoke are strongly correlated with the development of GERD, which is postulated as a major risk factor for the development of ESP.2,12 Despite these findings, our patient was not obese and reported being a lifetime non-smoker. The prevalence of ESP in patients with concurrent eosinophilic esophagitis (EoE) was reported at 4%, which is significantly higher than the general population—this lends more evidence toward mucosal inflammation and irritation as driving mechanisms. 1
A large retrospective study identified 78 ESPs out of over 55 000 EGDs and demonstrated malignant potential in two of these lesions; one patient had low-grade dysplasia in the originally excised ESP without recurrence on follow up endoscopy, and the other developed squamous cell carcinoma in the same location as the previously excised ESP. 6 The malignant potential of ESP, while rare, is important to note given the fact that the patient in our report demonstrated both low and high-grade dysplastic changes, which was not recorded on the initial biopsy specimens. Because of demonstrated malignant potential, endoscopic dissection, excision, RFA, or cryoablation all have utility in management, even when initial biopsies do not demonstrate dysplastic changes. Additionally, there are no definitive guidelines regarding surveillance intervals or first-line modality of removal and most reports depend largely on the size of the lesion(s).4,6 In this specific case, the follow up endoscopic surveillance interval was set at 3 months, similar to guidelines for surveillance of Barrett’s Esophagus with high-grade dysplasia given the high-grade dysplastic changes noted within the large resected specimen. 13 Current plans are for endoscopic surveillance every 3 months until complete eradication of the lesions.
This case demonstrates the need for further studies to better characterize risk factors related to the development of ESPs and their potential progression to malignancy. It is important to report these cases to raise clinician awareness to help better define management in rare cases of recurrence, as well as to consider ESP as a potential cause of resistant symptoms of GERD. Finally, the morphologic appearance of lesions should be categorized with diligence. Although most lesions tend to appear as sessile, exophytic growths, more extensive flat and circumferential lesions, as in this case, may require more advanced endoscopic management including ESD or endoscopic mucosal resection (EMR) to maximize the area of excised abnormal tissue and minimize the potential chance of recurrence. Close surveillance may also be necessary given the apparent tendency for incomplete excision and the potential for malignant transformation.2,4,5
Footnotes
Acknowledgements
Artificial intelligence software was not utilized in the composition of this report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
Ethics statement is not necessary for this report.
Informed Consent
Informed consent was obtained for this report.
Institutional Review Board
This report was deemed IRB exempt by the Institutional Review Board.
