Abstract
Introduction:
Hiatal hernia and abdominal wall hernia (AWH) have a common theme of pathogenesis and etiology. This suggests a potential relationship between these 2 types of hernia. However, no large studies have examined such an association. We designed the current study to characterize this association and identify factors associated with this coexistence.
Methods:
A database of patients seen at a large multi-center health network serving 3 states in the mid-Atlantic region of the United States was queried for patients with a documented diagnosis of hiatal hernia (HH) or paraesophageal hernia (PEH). This population was then divided into groups based on whether or not they had a history of another type of AWH (ventral, inguinal/ femoral, umbilical, or incisional hernia). Demographic and clinical data were compared between groups using univariate, followed by multivariable logistic analysis.
Results:
The final study population consisted of 28 114 patients (66.5% female) with HH/PEH. The prevalence of AWH in this population was 8.2% (n = 2299). Of these 69.1% had only one AWH with the remaining 30.9% having two or more hernias. Multivariable analysis showed male patients with HH/PEH were more likely to have AWH (OR: 1.97; 95% CI: 1.810-2.162, P <0 .0001). The odds of AWH were higher in patients aged 50 to 80 years old compared to younger patients (OR: 1.527; 95% CI: 1.348-1.730, P <0 .0001). Patients with any comorbidity were also more likely to develop AWH (OR: 1.764; 95% CI: 1.566-1.986, P < 0.0001), with obstructive sleep apnea (OSA) being the comorbidity with the highest risk (OR: 1.295; 95% CI: 1.161-1.444, P <0 .0001), followed by obesity (OR: 1.267; 95% CI: 1.156-1.388, P <0 .0001).
Conclusion:
The prevalence of an AWH in patients with a HH/PEH was 8.2%. Although HH/PEH are more prevalent in females, males with HH/PEH are at higher risk for AWH. Obesity, OSA, tobacco use, hypertension, diabetes, and hypothyroidism are independent risk factors for presence of an AWH in patients with HH/PEH.
Introduction
Hernia repair is one of the most ubiquitous classes of surgery performed today. Globally, over 20 million hernia operations are performed each year. 1 The risk factors for hernia development are well established and include comorbidities such as obesity and tobacco use.2 -7 Identifying these risk factors plays an important role in perioperative planning and risk stratification for conditions such as the subclinical contra-lateral groin hernia or the incisional hernia, which may modify surgical decision-making. 8 Studies have shown that many risk factors are common among different types of hernias.5 -7 Additionally, some studies have suggested that the development of 1 type of hernia may be a risk factor for developing other types of hernia. 9 A retrospective study found that the incidence of inguinal hernia was 2.5 times higher in patients with a history of hiatal hernia. 9 Hiatal and abdominal wall hernias are well studied as separate entities.10,11 However, the literature on patients with hiatal hernia and an additional type of hernia is limited, despite the overlap in risk factors.
Studies have suggested that common risk factors between hiatal hernia and abdominal wall hernia indicate a common etiology. 9 The 2 most widely accepted explanations of hiatal hernia development are the “push” and “pull” theories. 9 The “pull” theory suggests that gastroesophageal reflux-induced inflammation causes either fibrotic scarring or vagal stimulation that shortens the esophagus and “pulls” the gastroesophageal junction (GEJ) into the thoracic cavity. The “push” theory suggests that when the thoracoabdominal pressure gradient exceeds the tensile strength of the diaphragm, the GEJ is “pushed” cephalad, forming a hiatal hernia. This “push” is thought to be caused by either weakening the structural integrity of the diaphragmatic musculature or increases in intraabdominal pressures. 9 A combination of weakening of the tensile strength of the abdominal wall and increased intra-abdominal pressure also constitute the prevailing explanation for abdominal wall hernia development. 9 These common etiologies are likely similarly affected by common risk factors, which may explain the relationship between hiatal hernia and abdominal wall hernia.
Despite a shared etiology, there is a paucity of data on the impact of common risk factors among patients with hiatal hernia and abdominal hernia on the risk of developing additional types of hernia. Therefore, we designed this study to characterize the patient population with a history of hiatal hernia and abdominal wall hernias. Additionally, we aimed to identify the risk factors for having one or more abdominal wall hernias in the hiatal hernia or paraesophageal hernia patient population.
Methods
This was a retrospective review of the data obtained from a database of all patients seen at a large multi-center health network serving 3 states in the mid-Atlantic region of the United States from 2015 to 2021. This study was evaluated and approved by our institutional review board (IRB 2022-009-WPH).
Subject Identification and Data Collection
Patients aged 18 to 95 with a BMI of 16 to 80 and either a hiatal hernia or a paraesophageal hernia (HH/PEH) were included for analysis. The patient population was identified by reviewing the medical records of our large multi-center health network using Epic SlicerDicer software (Epic Systems, Verona, WI). SlicerDicer is an electronic medical record (EMR) based self-service tool used for data management. The software allows the operator to query, stratify and examine select data from all patients within the EMR. Queries for hernia diagnoses were based on the International Classification of Disease, 10th version (ICD-10) coding system. 12 Patients with hiatal hernias or paraesophageal hernia were identified using ICD-10-CM K44. The patients identified through this query who met inclusion criteria constituted the total HH/PEH study population. A secondary query of just this patient population was performed for different types of abdominal wall hernia, defined by ICD-10 codes. Specifically, the database was queried for ICD-10-CM K43 (Ventral Hernia), K42 (Umbilical Hernia), K43 (Incisional hernia), K41. (Femoral Hernia), K40. (Inguinal hernia), and K46 (Unspecified abdominal wall hernia). The results of the secondary query separated the total population into 2 groups based on whether or not they had a history of at least 1 additional type of hernia. Demographic information, documented visit diagnoses, and past medical history were reviewed for a history of the following comorbidities: obesity (BMI ≥ 30), tobacco use, chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), hypertension (HTN), obstructive sleep apnea (OSA), diabetes, and benign prostatic hyperplasia (BPH). Demographics and comorbidities were compared between HH/PEH with and without additional hernia groups.
Statistical Analysis
Univariate logistic regression analyses were performed for predicting binary outcomes of AWH and ventral/incisional hernia with respect to each of potential predictors including demographic and clinical factors, respectively. Multivariable logistic regression models for predicting the AWH were fitted for all subjects, male subjects and female subjects, utilizing a stepwise selection that mandated a variable that was statistically significant/borderline significant in the univariate analyses had to have a significance threshold of 0.30 and 0.10 to be opted and retained in the model, respectively. A statistically significant association between a predictor and an outcome is established if p-value < 0.05 for Wald Chi-Square Test or the 95% odds ratio confidence interval did not cross 1.0. Two three-dimension (3D) surface plots that visualized relationship between predicted probability of the AWH and two predictors (age and number of comorbidities; age and BMI), and one effect of age plot stratified by gender and comorbidity with respect to predicted probability of the AWH were generated based on fitted multivariable logistic regression models, respectively. All statistical analyses were performed using SAS software (version 9.4; SAS Institute, Cary, NC). The 3D surface plots were generated using MATLAB software (Mathworks, Natick, MA).
Results
The final study population consisted of 28 114 patients with a sliding or paraesophageal hiatal hernia. The mean (SD) age was 64.3 (15.4) years and BMI was 30.2 (7.0). The population was 66.56% (n = 18 733) female. A total of 2299 (8.2%) patients were found to have at least 1 abdominal wall hernia. Table 1 shows the types and number of abdominal hernias in the study population.
Number and Types of Abdominal Wall Hernias in Patients With Hiatal Hernia (n = 28114).

Univariate analysis comparing demographic and clinical parameters between those with associated abdominal wall hernia and those without is shown in Table 2. Male patients were more likely to have abdominal wall hernia (OR: 2.007; 95% CI: 1.842-2.187, P <0 .0001). The odds of abdominal wall hernia were higher in patients aged 50 to 80 years old compared to younger patients (OR: 1.527; 95% CI: 1.348-1.730, P <0 .0001). The magnitude of this risk was slightly diminished in those >80 years old but remained higher than younger patients (OR: 1.264; 95% CI: 1.074-1.488, P = .0048). Patients with a history of any comorbidity were also more likely to develop abdominal wall hernia (OR: 1.764; 95% CI: 1.566-1.986, P <0 .0001), with sleep apnea being the comorbidity with the highest risk (OR: 1.613; 95% CI: 1.455-1.788, P <0 .0001). The relative contribution of age and number of comorbidities to the probability of abdominal wall hernia is shown in Figure 1.
Univariate Logistic Analysis for Hernia by Potential Predictors for all Patients.
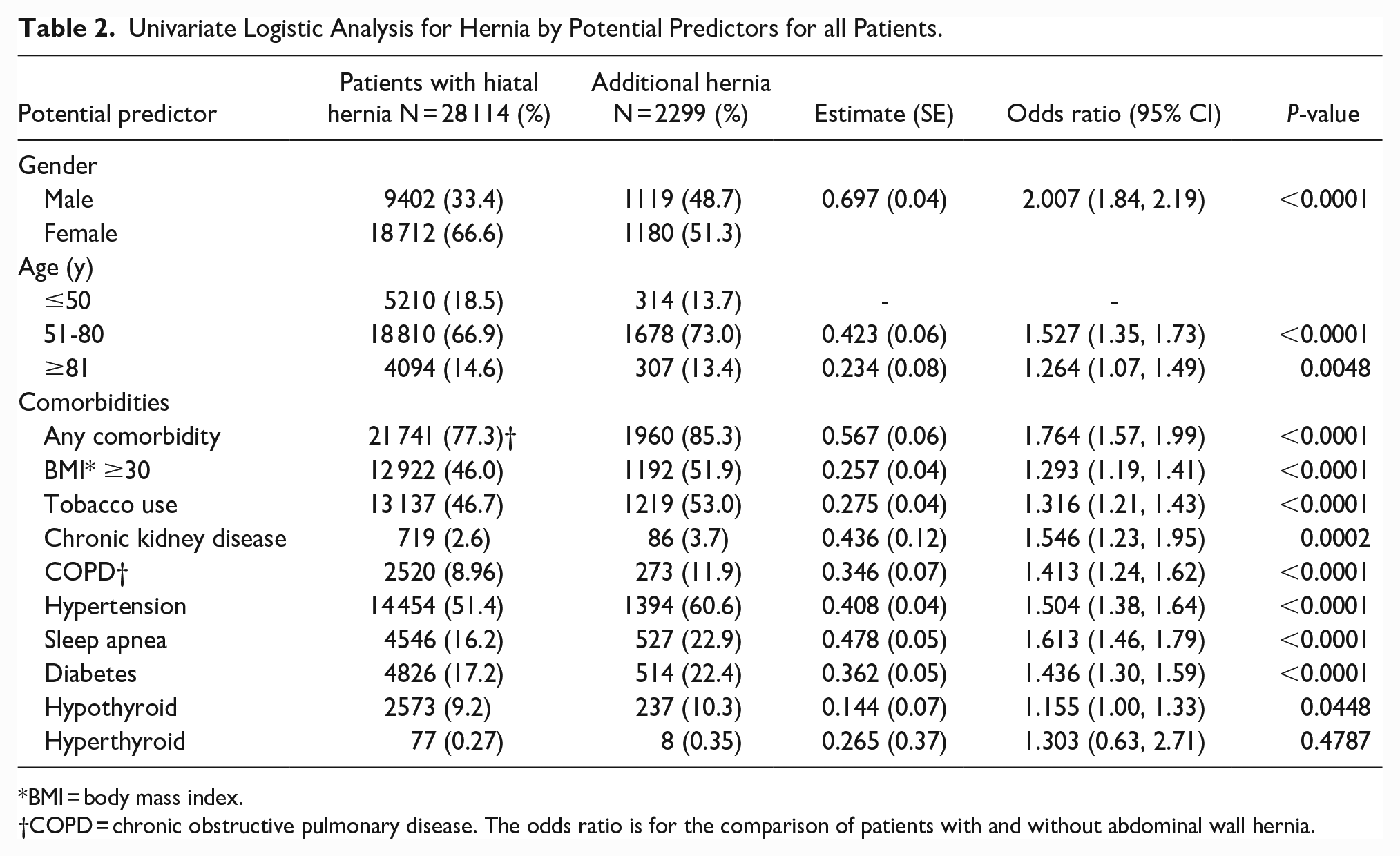
BMI = body mass index.
COPD = chronic obstructive pulmonary disease. The odds ratio is for the comparison of patients with and without abdominal wall hernia.
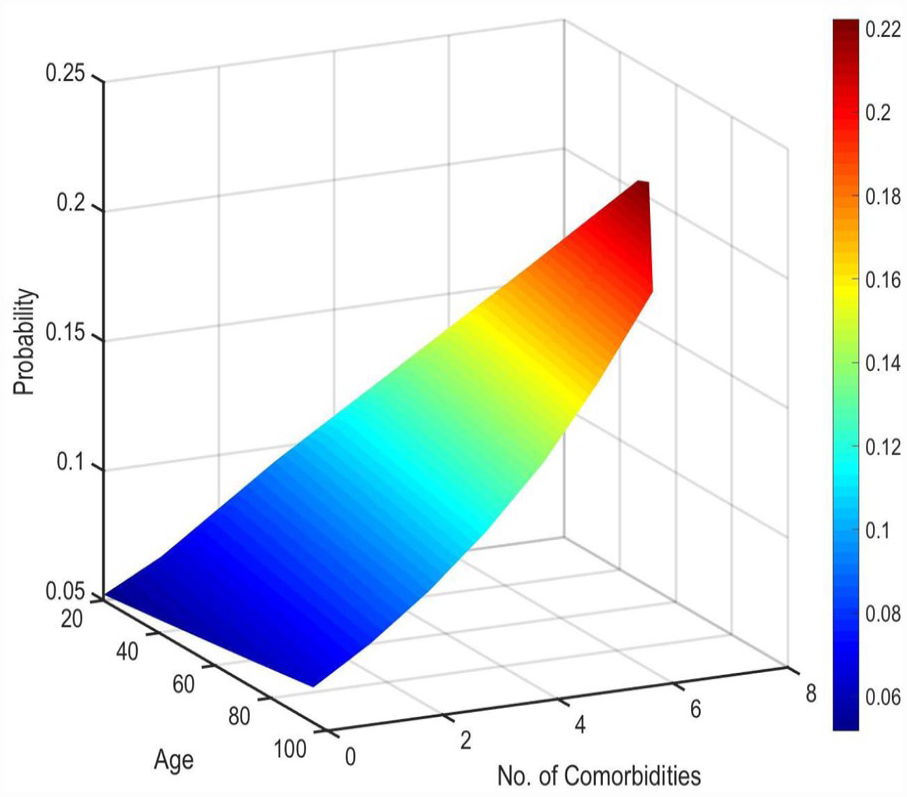
Predicted probability of co-existence of an abdominal wall hernia (z-axis) in patients with hiatal hernia with increasing age (x-axis) and number of comorbidities (y-axis) in all patients (n = 28 114).
The results of the multivariable logistic analysis to identify independent risk factors for the presence of abdominal wall hernia are shown in Table 3. Male sex was the strongest predictor of the presence of abdominal wall hernia (OR: 1.989; 95% CI: 1.819-2.174, P <0 .0001). This raised concern about the possibility of a disproportional effect of sex on other comorbidities. Therefore, univariate and multivariable logistic analyses were repeated separately for male and female populations. Table 4 shows the multivariable analysis of factors associated with abdominal wall hernia in the male population. Sleep apnea was one of the strongest risk factors among men (OR: 1.233; 95% CI: 1.062-1.432, P = 0.0061). Table 5 shows the multivariable analysis of factors associated with abdominal wall hernia in the female population. Body mass index (BMI) was the strongest risk factor among women (OR: 1.417; 95% CI: 1.253-1.603, P < 0.0001). Table 6 shows the univariate analysis for ventral or incisional hernia, excluding inguinal, femoral, and umbilical. A history of any comorbidity (OR: 2.164; 95% CI: 1.812-2.585, P <0 .0001) was the strongest factor, followed by sleep apnea (OR: 1.744; 95% CI: 1.520-2.000, P < 0.0001).
Multivariable Logistic Analysis for Hernia by Predictors for all Patients.
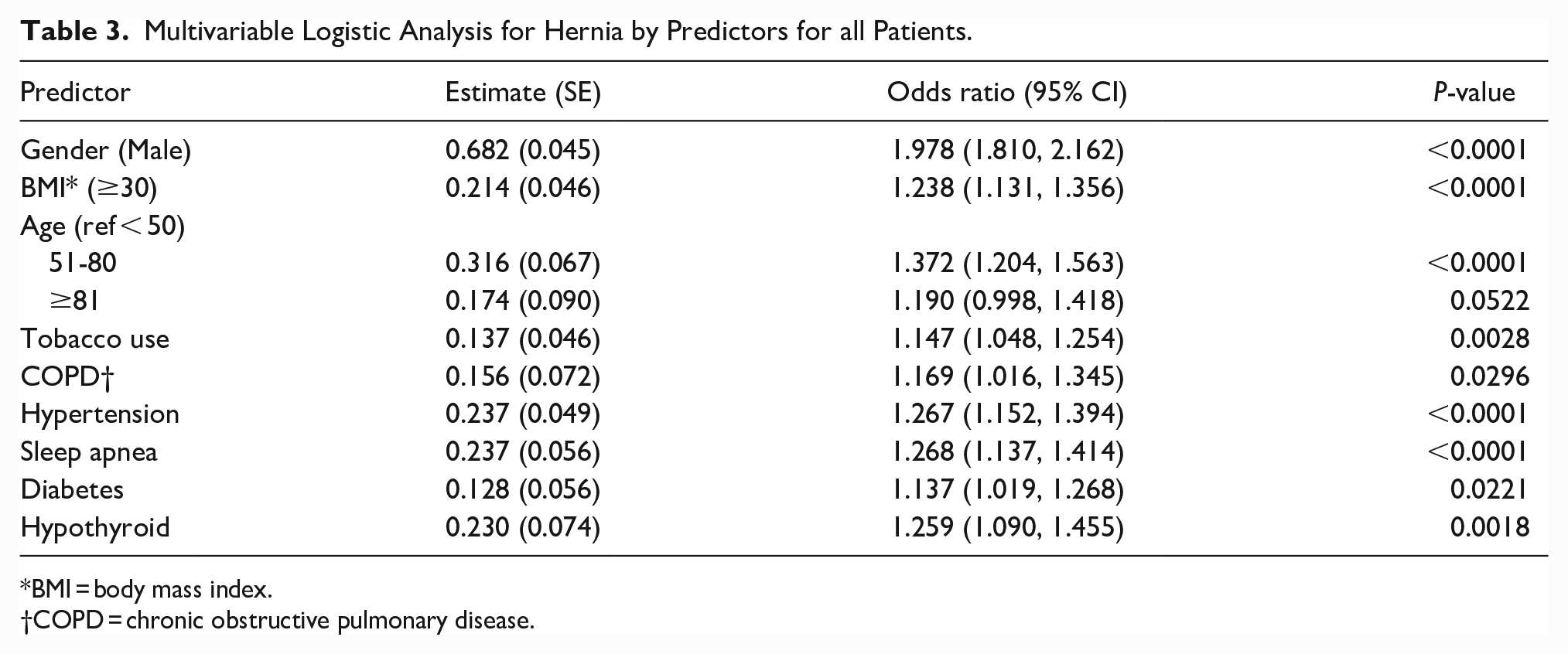
BMI = body mass index.
COPD = chronic obstructive pulmonary disease.
Multivariable Logistic Analysis for Hernia by Predictors for Male Population.
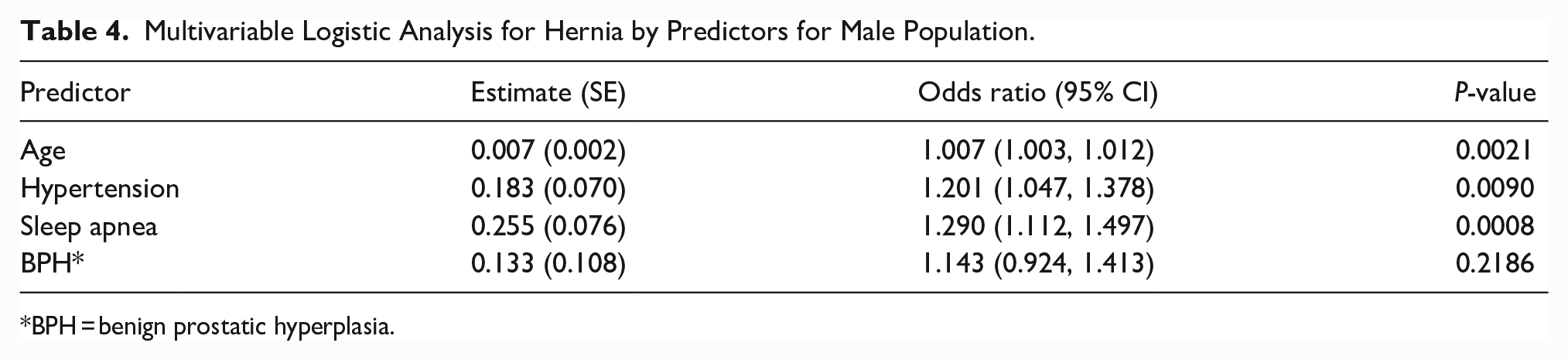
BPH = benign prostatic hyperplasia.
Multivariable Logistic Analysis for Hernia by Predictors for Female Population.

BMI = body mass index.
COPD = chronic obstructive pulmonary disease.
Univariate Logistic Analysis for Ventral/Incisional Hernia by Age and Comorbidity.

BMI = body mass index.
COPD = chronic obstructive pulmonary disease.
The effect of age on the probability of abdominal wall hernia in male and female are shown in Figure 2. The contributions of age and BMI to the probability of abdominal wall hernia is shown in Figure 3.
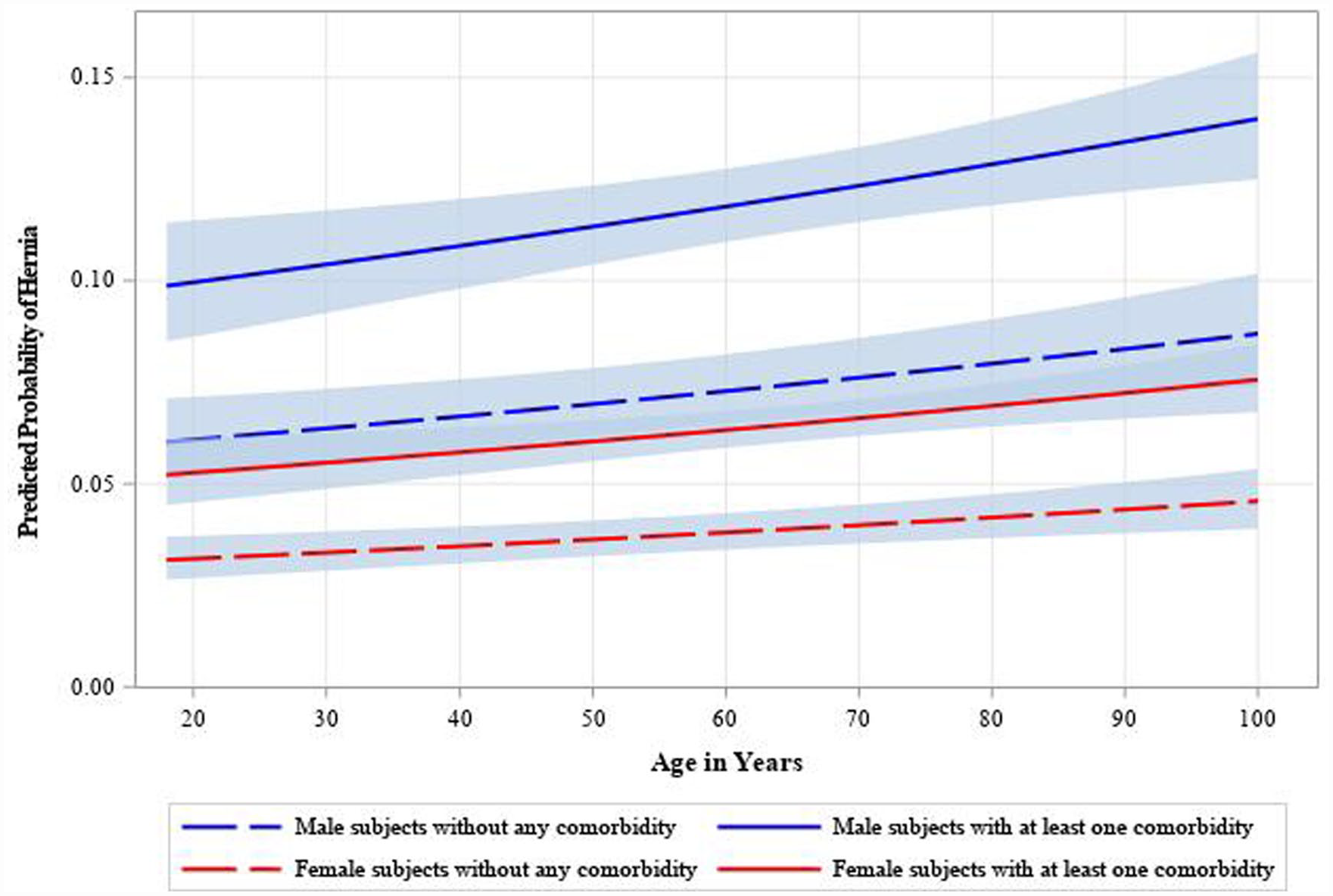
Effect of age on the predicted probability of presence of an abdominal wall hernia in Male (Blue Lines) and Females (Red Lines) patients with hiatal hernia. The continuous curves represent patients with at least 1 comorbidity and the dotted curves represent those with no comorbidities.
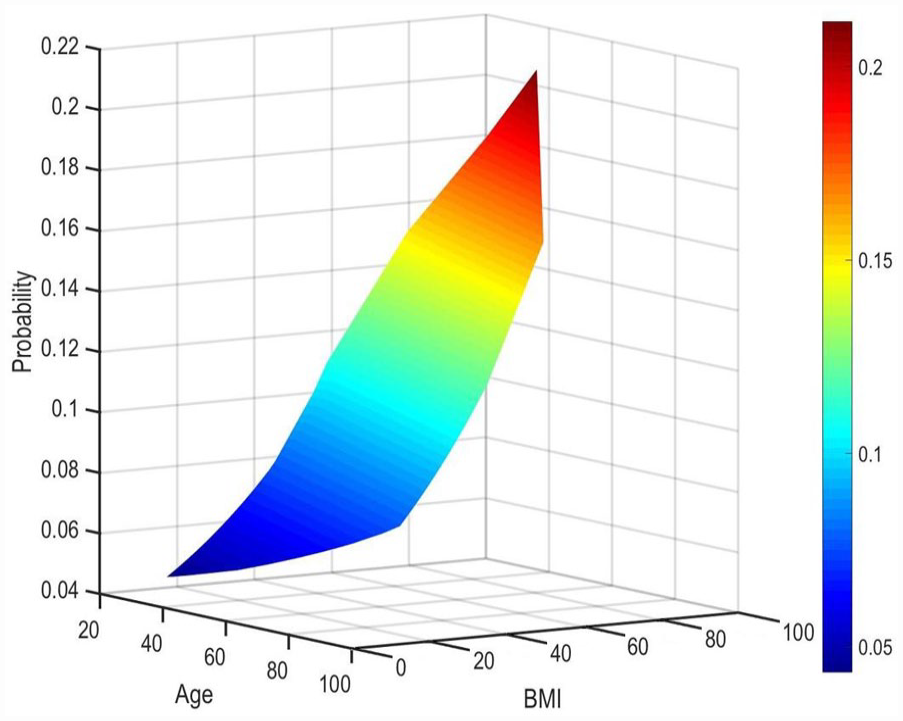
Predicted probability of co-existence of an abdominal wall hernia (z-axis) in patients with hiatal hernia with increasing age (x-axis) and body mass index (BMI) (z-axis) in all patients (n = 28 114).
Discussion
The abdominal wall hernia and its management were first described in the Ancient Egyptian Ebers medical papyrus in 1550 BCE, but hiatal hernia, was not identified as a significant clinical entity until the “Treatise on Diaphragmatic Hernia” by Bowditch in 1853.13,14 Today, abdominal wall and hiatal hernias are well-recognized surgical problems with multiple overlapping risk factors affecting up to 1-in-5 people.1,10,11 Common risk factors imply common mechanisms of development, namely increased intraabdominal pressure and weakness of the abdominal cavity musculature. We found that male sex, advanced age, obesity, tobacco use, hypertension, sleep apnea, diabetes, and chronic kidney disease were independent predictors of abdominal wall hernia in addition to a hiatal hernia. Each of these risk factors contributes to either intraabdominal pressure, weakness of the abdominal wall, or both. This is the first study to evaluate common risk factors for the development of hiatal hernia and another type of hernia.
We found that a BMI ≥30 was an independent risk factor for additional hernia. Obesity and hernia are frequently comorbid.3,4 Meta-analysis of 25 310 patients found that obesity doubled the risk of hiatal hernia. 3 Obesity has also been identified as a risk factor for abdominal wall hernias in both retrospective and prospective studies.4,15 A likely mechanism by which obesity promotes hernia development is increased intraabdominal pressure. Central obesity puts pressure on the abdomen, increasing the thoraco-abdominal pressure gradient and promoting the hiatal hernia formation. Jung et al 16 came to a similar conclusion when discussing their finding that each BMI point gained increased intragastric pressure by 10%. Other studies have found a similar direct correlation between BMI and hernia defect size, with 60% of the patients with a defect size >10 cm reaching a BMI over 30. 17 Obesity also leads to a pro-inflammatory state, which can weaken abdominal wall tensile strength. 18 Excessive macronutrients in adipose tissue promotes the release of inflammatory mediators such as interleukin-6 and tumor necrosis factor alpha, which inhibit type I and III collagen synthesis. 18 Collagen is the primary structural protein that maintains tensile strength. The combination of increased intraabdominal pressure and weakening of the strength of the abdominal wall is the likely explanation for our finding that obesity was a risk factor for having a hiatal hernia and abdominal wall hernias.
Age was another significant risk factor for the development of additional hernias. Similar to obesity, studies have shown that age-related changes in the balance between collagen synthesis and degradation decrease the tensile integrity of the abdominal wall, promoting hernia development. 19 Additionally, CDC data has shown that rates of obesity increase with age. 20 Similarly, we showed that increasing age and BMI increased probability of additional hernias (Figure 3). The highest likelihood of developing an additional hernia was found in the 51 to 80 age group, which corresponded to the highest concentration of central obesity in this patient population, and, therefore, the group with most increased intra-abdominal pressure. 20
Studies have found that complications of obesity, such as sleep apnea, are also risk factors for hernia.21,22 Likewise, we found that patients with OSA were 1.6 times more likely to develop abdominal wall or groin hernia in addition to hiatal hernia. Sugerman et al 22 also found that OSA was an independent risk factor for abdominal wall hernia, while Zenda et al 21 found an association between hiatal hernia and OSA. Apneic inspiration creates a vacuum, amplifying the negative intrathoracic pressure, thereby increasing the thoraco-abdominal pressure gradient. 5 Coupled with the elevated intraabdominal pressure of obesity, the gradient is even further increased, promoting hernia formation, as seen in our patient population.
Distribution of fat may play a larger role in hernia formation than BMI alone. Studies have shown that central obesity and increased visceral fat have a large impact on intra-abdominal pressure. 23 Although females have an overall higher fat content ratio, males have a higher distribution of visceral fat and central obesity. 24 Similarly, despite finding more hiatal hernia in female patients, male patients with hiatal hernia were nearly twice as likely to have additional hernias. This was true at every age and regardless of other comorbidities (Figure 2). The sexually dimorphic distribution of body fat is likely one of many potential factors that contribute to a higher risk of hiatal hernia in male patients. Another uniquely male risk factor that leads to excessive straining and increased intra-abdominal pressure is BPH. 6 Studies have shown that the prevalence of BPH dramatically increases with age, from 8% at 40 to greater than 50% after 50 years old, corresponding with the age group found to have the highest risk for additional hernia. 6
Tobacco use can both increase intraabdominal pressure and weaken abdominal wall tensile strength. Smoking is associated with swallowing excess air, which causes gastric distension and increases abdominal pressure. 2 Frequent episodes of coughing and the need for deep inspiration associated with chronic tobacco use also increase intraabdominal pressure. 2 Studies have found associations between these factors in smokers and hiatal and abdominal wall hernias, consistent with our findings. 2 The impact of coughing was also described in a study by Kim et al 7 that found that 11.6% of COPD patients developed hiatal hernia, compared to only 6.1% of unobstructed smokers. Similarly, we found COPD to be an independent risk factor for additional hernias. The expansion of the lung volume, due to the obstructive nature of the disease, leads to flattening of the diaphragm and indirectly increases intraabdominal pressure. Chronic tobacco use also leads to vascular disease and tissue hypoxia, weakening the integrity of the abdominal wall. 25 Tobacco-induced oxidative stress affects the metabolism of connective tissues by oxidizing collagen, preventing cross-linking, and thereby exacerbating weakness in the abdominal wall. 25
Hypertension, diabetes, and CKD were identified as independent risk factors for additional hernias, likely because these conditions are causes or manifestations of vascular disease and tissue hypoxia.26,27 Chronic kidney disease, in particular, does not manifest until the patient’s vascular disease has become chronic and severe. 28 This progression suggests that comorbidity severity may further increase hernia risk, and may explain why the odds of additional hernia were higher in CKD than in many other significant risk factors. Additionally, CKD may impact hernia risk in a way that diabetes or hypertension may not. Studies have shown that the hyper-albuminuria seen in CKD is associated with impaired collagen organization, a process mediated by matrix metallopeptidase-9 (MMP-9). 29 MMP-9 plays an essential role in the extracellular matrix’s degradation and in collagen organization. 30 Therefore, CKD leads to abnormal MMP-9 levels, which disrupt collagen homeostasis and tensile wall strength, promoting hernia formation.
Diabetes is a known risk factor for poor healing. Chronic hyperglycemia leads to oxidative stress and the formation of advanced glycation end products (AGEs). 31 Collagen is modified by oxidization or AGEs, disrupting normal cross-linking and weakening the tissue, significantly increasing the risk of hernia formation. 31 Additionally, in some patients diabetes-induced delayed gastric emptying can lead to gastric dilation and reduce the angle of His. This altered anatomy plays a very important role in the formation of hiatal hernias and exacerbating reflux disease. 32
This study was not without its limitations, including its retrospective nature and use of SlicerDicer. SlicerDicer is a valuable tool for assessing a snapshot of population-based data, but it lacks the granularity of traditional detailed chart review, such as dates of diagnosis, disease progression, management strategies, or time and cause of mortality. For example, age >80 was not as strong a predictor of additional hernia as age 51 to 80. This may reflect a survivorship bias. Many predictors of hernia are also predictors of cardiac disease, reducing the likelihood that high risk patients would reach the age >80 group. Additionally, as SlicerDicer is dependent on ICD-coded documentation, the true prevalence of hernia is likely underrepresented. Additionally ICD coding obscures clinically relevant granular distinctions, such as K44, which does not distinguish between sliding hiatal hernia and paraesophageal hernia. Despite these limitations, this is the first study to assess the prevalence and risk factors of abdominal hernia in patients with a history of hiatal hernia.
The goal this study was to aid clinicians treating patients with hiatal hernia identify and inform patients of their risk for additional hernias. Many of the identified risk factors, such as obesity and tobacco use, are subject to lifestyle modification. This study can inform discussions of pre-habilitation during preoperative patient counseling. A more specific application of this study is determining the risk of incisional hernia following a hiatal hernia repair. Rates of incisional hernia have been reported to range from 5% to 20%, depending on the surgical type and approach. 33 In high-risk patients, these rates increase to 30%. 33 The risk factors identified in this study can help determine whether to perform an additional facial closure after removing laparoscopic trocars. In our patient population, ventral and incisional hernias were most impacted by obesity, diabetes, and smoking. Similarly, Sørenson et al studied the effect of smoking on the formation of incisional hernia. They found that the rate of incisional hernias in smokers is 4-times higher than in nonsmokers, consistent with our findings. 34 The oxidative stress associated with tobacco use doesn’t just interfere with collagen cross-linking, it also leads to the destruction of neutrophils, and subsequently increases the risk of wound infection and dehiscence. 25
Conclusion
We evaluated the prevalence and risk of having an abdominal wall hernia in patients with a hiatal or paraesophageal hernia and found that 8.2% of patients had multiple hernias. Independent risk factors included male sex, advanced age, obesity, tobacco use, hypertension, sleep apnea, diabetes, and chronic kidney disease. Although hiatal hernias are more prevalent in females, males with a hiatal hernia are at higher risk for additional hernias. There is a stepwise increase in the risk of additional hernias with age, with the highest risk being in the patients aged 51 to 80. Obesity, tobacco use, HTN, and OSA are independent risk factors with the highest risk associated with the presence of additional hernias in the hiatal hernia patient population. About 4.1% of the patients with hiatal hernia developed ventral or incisional hernia. Patients with obesity, sleep apnea, diabetes or chronic kidney disease have a higher risk of developing ventral or incisional hernias. These risk factors can be best used to counsel patients on risk-modification, aid in risk-stratification for incision hernia during pre-operative planning and inform surgical decision-making during wound closure.
Supplemental Material
sj-tif-1-gut-10.1177_26345161231180010 – Supplemental material for Abdominal Wall Hernias in Patients with Hiatal Hernias: Predictors of Association in a Large Cohort of Patients
Supplemental material, sj-tif-1-gut-10.1177_26345161231180010 for Abdominal Wall Hernias in Patients with Hiatal Hernias: Predictors of Association in a Large Cohort of Patients by Nisarg Mehta, Sven E. Eriksson, Ping Zheng, Inanc S. Sarici, Ali H. Zaidi, Blair A. Jobe and Shahin Ayazi in Foregut
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
