Abstract
Background:
Ineffective esophageal motility (IEM) is a commonly identified disorder of peristalsis. Although often asymptomatic, IEM can be associated with dysphagia. Aside from treating co-existing gastroesophageal reflux disease and sources of mechanical obstruction, few options are available for management in this context. We therefore systematically reviewed the literature to identify randomized controlled trials (RCTs) for medical treatments of IEM.
Methods:
MEDLINE, Embase, Cochrane Library, and Web of Science were searched using controlled vocabulary and keywords to identify RCTs from inception through 9/22/2021. Prospective studies evaluating medical therapy to improve dysphagia in adults with IEM were included. The risk of bias was assessed using the revised Cochrane risk-of-bias tool.
Results:
Among 1046 studies identified, 6 (0.58%) met inclusion criteria with a total of 65 patients. Most studies evaluated serotonin receptor agonists (buspirone, mosapride, prucalopride, and sumatriptan), primarily assessing changes on esophageal high-resolution manometry parameters, and only 1 study evaluated patient reported outcomes. Overall, medical therapy improved these pooled outcomes in 5 (83%) studies. Although treatment endpoints varied, the risk of bias in study reporting was low for 4 studies and uncertain for 2 studies.
Conclusions:
There are currently few therapeutic options available for IEM patients with symptomatic non-obstructive dysphagia. Our systematic review identified 6 studies utilizing medical therapy in patients with IEM, and a majority demonstrated an improvement in HRM parameters. Medical therapy may therefore be considered in this context, but additional studies are warranted to assess for similar improvement in patient symptoms.
Key Learning Points
Ineffective esophageal motility (IEM) is a commonly observed pattern that can be associated with dysphagia.
Few randomized control trials exist to guide the medical management of dysphagia in IEM.
In a systematic review of medical therapy for dysphagia in IEM, the findings support a possible benefit with serotonin receptor agonists.
Introduction
Ineffective esophageal motility (IEM) refers to a motility pattern identified on esophageal high-resolution manometry (HRM) characterized by weak and failed swallows. 1 It is frequently associated with gastroesophageal reflux disease (GERD), but IEM is also observed in asymptomatic controls as well as in patients with dysphagia.2-4 Across patient presentations, it remains a frequently identified motility pattern, with studies revealing a prevalence of IEM in approximately 30% of patients undergoing HRM. 5 While original definitions for IEM predate the Chicago Classification, its current criteria have been refined in the Chicago Classification v4.0.1,6,7 While primarily a description of findings rather than a specific diagnosis, criteria require a normal integrated relaxation pressure (IRP) and a distal contractile integral (DCI) between 100 and 450 mmHg/s/cm in >70% of liquid swallows, or ≥50% and <100% of liquid swallows being failed (DCI < 100 mmHg/s/cm or fragmented swallows). These criteria acknowledge abnormal peristalsis in the esophageal body with preserved function of the lower esophageal sphincter. Others have identified poor bolus transit among patients with IEM. 8 Nonetheless, the clinical significance of this manometric pattern remains uncertain.
Given IEM physiology is defined by esophageal body hypomotility and is associated with impaired bolus clearance including refluxate, its association with GERD and impaired bolus transit may lead to associated symptoms such as dysphagia. Indeed, it is estimated that patients with IEM will experience dysphagia with nearly 30% of swallows. 9 However, few options exist to reliably improve esophageal function or improve dysphagia in the setting of IEM. This may be due to the inconsistent presence of symptoms in IEM, and because symptoms, if present, may be attributable to alternative etiologies. As with all patients complaining of dysphagia, assessment for mechanical etiologies for obstructive symptoms is paramount. 9 However, treatment options specific to IEM focus on optimizing management of GERD when present. 10 In other instances, patients with IEM may not be candidates for surgical therapy for GERD, due to the increased risk for post-operative dysphagia. 11 Currently, there are no established guidelines for the management of symptomatic IEM. Instead, narrative reviews have mainly presented data from uncontrolled trials that primarily report the use of medications, such as pyridostigmine and bethanechol, whose side effects may provide fortuitous changes in esophageal motility.12,13 Indeed, many medications have been assessed for a potential prokinetic effect in the management of esophageal hypomotility. Metoclopramide has been shown to augment esophageal contraction in healthy volunteers, but this agent has not been beneficial in IEM.10,14 Similarly, domperidone has not demonstrated improvement in esophageal body motility in a randomized-control trial. 15 Erythromycin has been associated with a significant increase in the amplitude, duration, velocity and strength of esophageal peristalsis in patients with GERD, however it has not been formally studied in patients with IEM. 16 Pyridostigmine, an acetylcholinesterase inhibitor, has shown some promise in a small pilot study of patients with esophageal hypomotility, which included 3 patients with IEM based on Chicago Classification v4.0 and 3 patients with type II achalasia. In this study, pyridostigmine demonstrated a statistically significant improvement in DCI and number of peristaltic swallows. 17 While encouraging, subsequent work utilizing a randomized-control trial design and more homogenous patient populations would be warranted, and identifying effective treatments remains a primary gap in knowledge as identified during a recent expert consensus conference on IEM. 10 As a result, further synthesis of the available data to help guide clinicians in treatment of IEM is needed. We therefore performed a systematic review of prospective, experimental studies evaluating the efficacy of pharmacotherapy in the management of IEM.
Materials and Methods
Information Sources and Search Strategy
We performed a systematic review of the literature to identify and determine the effectiveness of different medical therapies in the management of IEM. The MEDLINE (via PubMed), Embase (via Elsevier), Cochrane Library (via Wiley), and Web of Science SCI Index and SSCI Index (via Clarivate) electronic databases were systematically searched on 9/22/21 by a medical librarian (SC) using a mix of database-specific controlled vocabulary terms and keywords related to ineffective esophageal motility to identify randomized, placebo-controlled trials (RCTs) (Supplemental Appendix 1). The strategies were peer reviewed by another medical librarian prior to execution using the PRESS Checklist. 18 Editorials, letters, case reports, and comments were excluded from the search. Upon review of full text articles, manuscripts that were not written in English or that did not have a reliable English translation were excluded as they could not be evaluated by the reviewers. Additional references were identified by hand-searching bibliographies of included articles, relevant reviews, and via a search of the ClinicalTrials.gov website. All studies were uploaded to Covidence (Veritas Health Innovation, Melbourne, Australia) for screening. Systematic reviews are not subject to review by the institutional review board at Duke University Medical Center, and the search strategy was placed in Open Science Framework.
Eligibility Criteria
Prospective studies evaluating a medical intervention for IEM were included. Any study that did not have a randomized controlled design or utilized non-medication interventions were excluded.
Selection Process and Data Collection
All titles, abstracts, and full-text articles were independently reviewed during 2 phases of screening by 2 of the 4 reviewers (DAL, ATR, RAS, EJS) and any disagreements were resolved by a third independent reviewer.
Using a standardized form, the reviewers independently extracted data for inclusion in the review. The accuracy of data was confirmed by a second reviewer. Data collection included author, year of publication, location, study design, number of patients enrolled, definition of IEM, intervention, measured outcomes, results, and duration of follow-up. These results were compiled and presented in a tabular format.
Risk of Bias Assessment
The risk of bias for each study included in the review was evaluated using the revised Cochrane risk-of-bias tool (RoB 2). 19 This tool covers 5 domains of bias: randomization process, deviations from the intended intervention, missing outcome data, measurement of the outcome, and selection of the reported result. The studies were independently reviewed and any disagreements were resolved via consensus amongst all reviewers.
Results
The search strategy yielded 1046 studies and 6 (0.58%) met inclusion criteria yielding a total population of 65 patients (Figure 1).20-25 The majority of studies (4, 67%) defined IEM using the definition of “weak peristalsis,” which was established in the Chicago Classification v2.0. 26 The remaining studies defined IEM based on the definition described in the Chicago Classification v3.0. 27 The follow-up period after each medical intervention varied from 90 minutes to 2 weeks based on study design, with shorter intervals assessing manometric changes. Primary endpoints also varied by study, including validated patient reported outcome tools such as the Gastroesophageal Reflux Disease Health-Related Quality of Life Questionnaire (GERD-HRQL) and the Mayo Dysphagia Questionnaire (MDQ). Others evaluated observable changes on esophageal manometry parameters, such as final HRM diagnosis, number of primary esophageal motor waves, number of swallows, and threshold volume for generating secondary peristalsis as determined by rapid air infusion via HRM catheter.
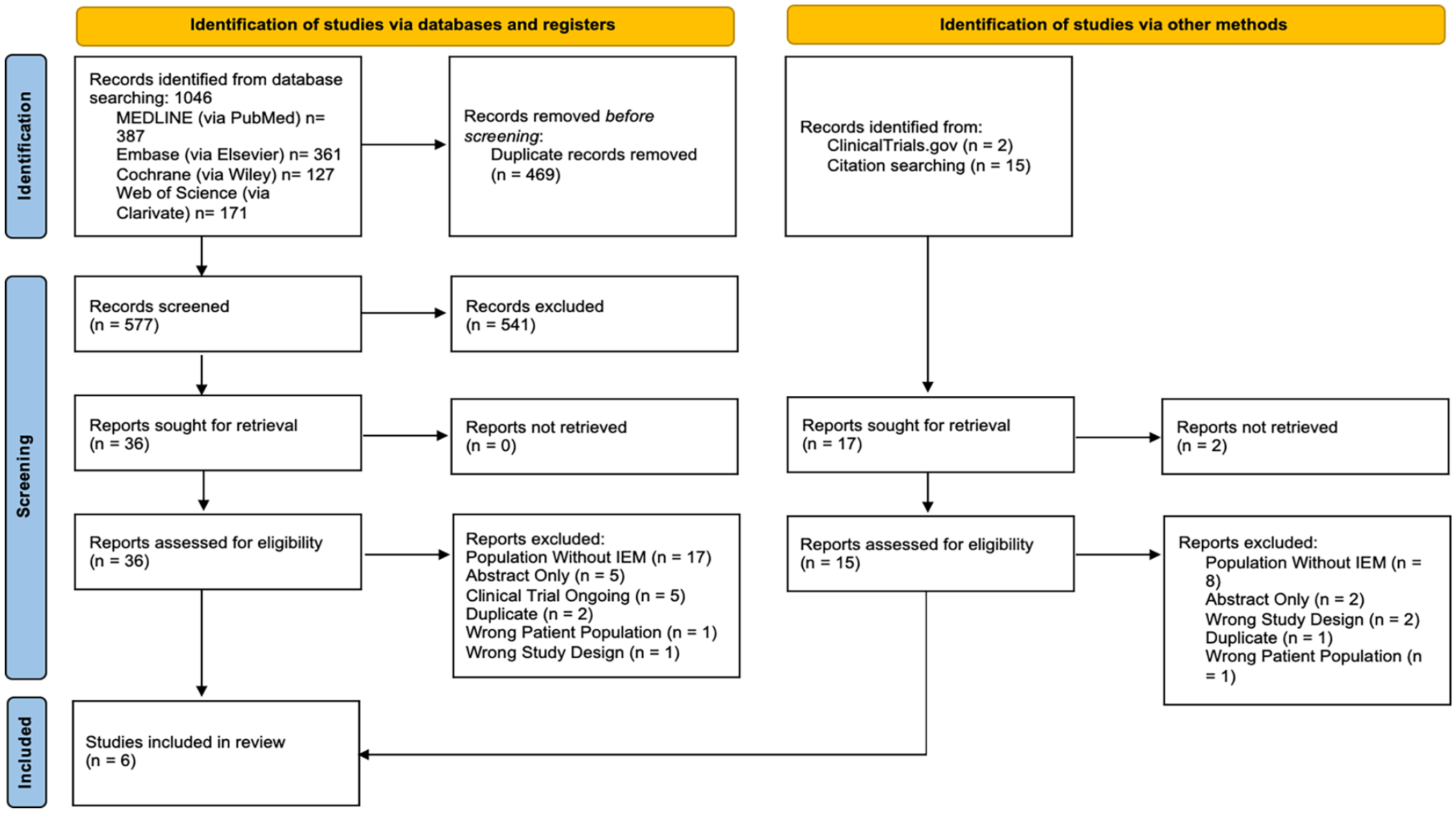
PRISMA flow diagram.
Medical therapies utilized across the 6 included studies consisted of bethanechol, buspirone, mosapride, prucalopride, and sumatriptan, all of which are serotonin receptor agonists (SRAs) except bethanechol which is a muscarinic receptor agonist. Overall, 5/6 (83%) of studies demonstrated improvement in HRM outcomes following medical therapy. SRAs demonstrated improvement in multiple HRM parameters; mosapride and prucalopride administration lowered threshold volumes required to generate secondary peristalsis and increased both the frequency and efficiency of secondary peristalsis while sumatriptan administration improved the total number of swallows and primary esophageal motor waves (Table 1).20-23 One study assessed patient reported outcomes using GERD-HRQL and MDQ, and there was no significant change in these parameters or HRM parameters following buspirone administration. 21 Bethanechol administration increased the percentage of peristaltic swallows, esophageal contraction pressures, and the frequency of complete liquid bolus transit.24,25
Characteristics of studies included in the systematic review.
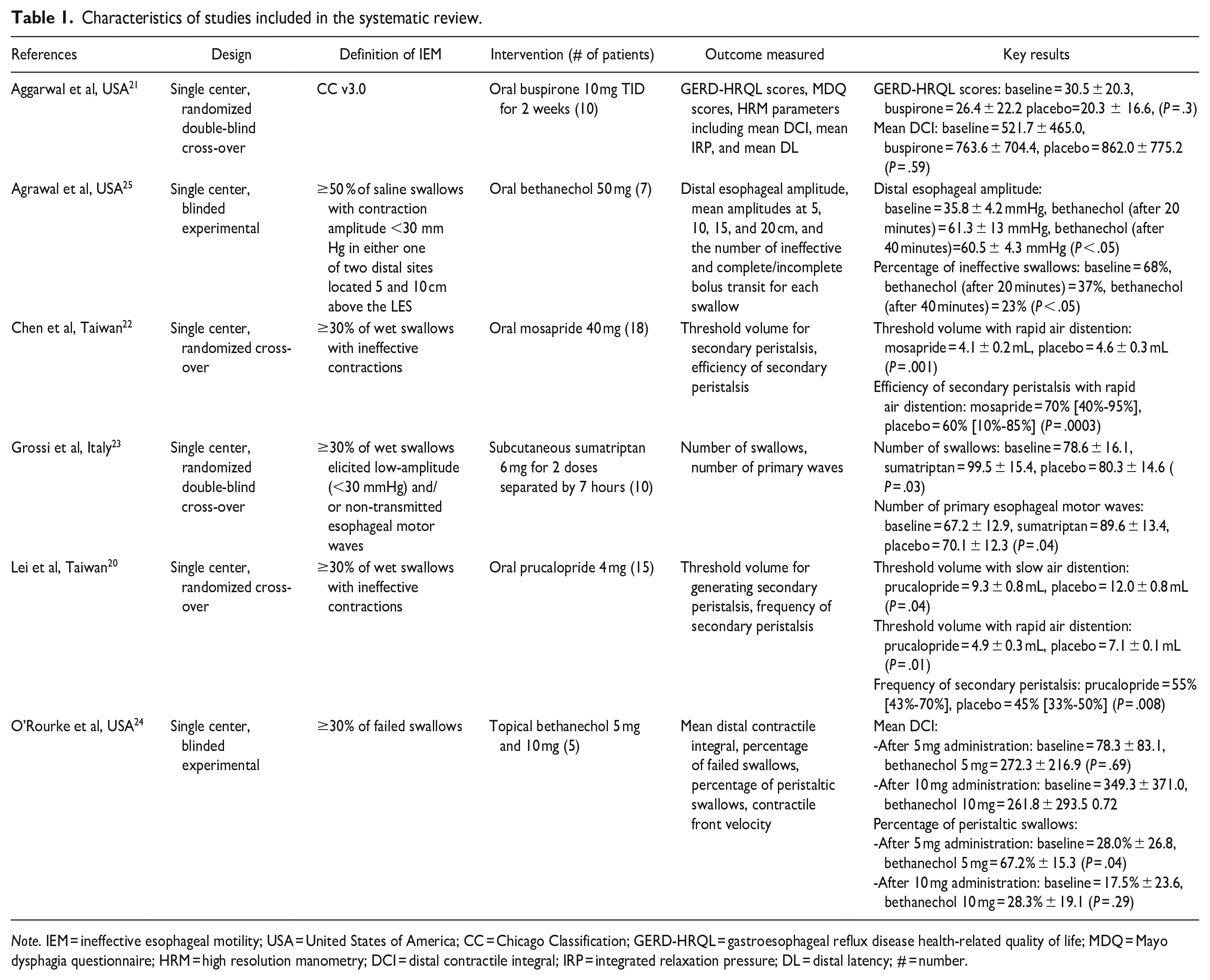
Note. IEM = ineffective esophageal motility; USA = United States of America; CC = Chicago Classification; GERD-HRQL = gastroesophageal reflux disease health-related quality of life; MDQ = Mayo dysphagia questionnaire; HRM = high resolution manometry; DCI = distal contractile integral; IRP = integrated relaxation pressure; DL = distal latency; # = number.
Overall, the risk of bias in conducting and reporting the included RCTs was low for 4/6 (67%) based on the revised Cochrane risk-of-bias tool (Figure 2). There were some concerns regarding the randomization process for 2/6 (33%) of studies, however these studies were otherwise low risk for bias based on all other domains.
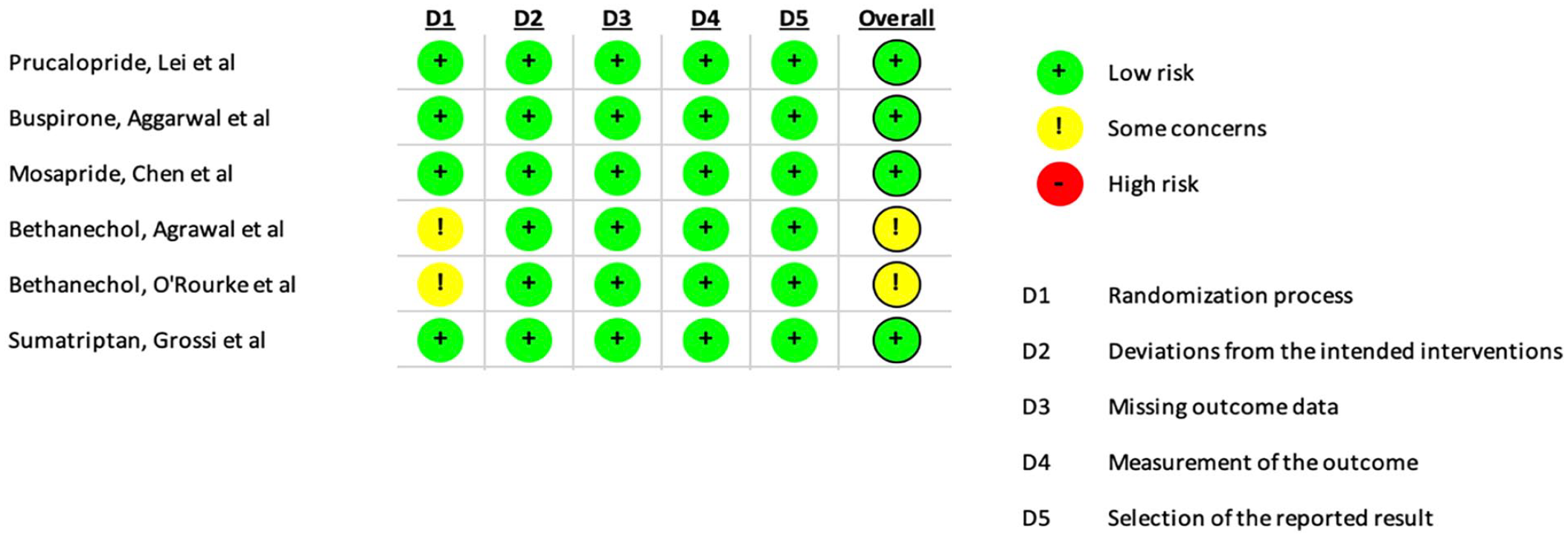
Revised Cochrane risk-of-bias tool.
Conclusions
IEM is a frequently encountered manometric pattern but with a variable symptomatic presentation. It often co-occurs with GERD, though patients with IEM may complain of dysphagia. 28 There are currently no formal guidelines for treatment, but consensus statement recommendations largely relying upon data from uncontrolled studies and narrative reviews exist.12,13 In a systematic review of RCTs that utilized medical pharmacotherapy in the management of IEM to assess for treatment effect, we identified 3 of 4 studies that showed improvement after use of SRAs.
IEM is a manometric description that predates the development of the Chicago Classification and was originally defined by at least 30% ineffective contractions out of 10 liquid swallows. 7 The Chicago Classification has undergone multiple iterations with the goal of identifying clinically meaningful categories or motility patterns, and the definition of IEM has likewise evolved with increasing data. In Chicago Classification v3.0, IEM was defined by a normal IRP and ≥50% ineffective swallows (100 < DCI<450 mmHg/s/cm). This was described as a “minor disorder of peristalsis” and was associated with impaired esophageal bolus clearance. However, a manometric classification of IEM did not always indicate true pathology for patients, and the definition of IEM in Chicago Classification v4.0 captures more severe findings with potential implications for symptoms, especially post-fundoplication dysphagia. 11 In Chicago Classification v4.0, IEM is defined by a normal IRP and >70% ineffective swallows (100 < DCI<450 mmHg/s/cm) or ≥50% failed swallows (DCI < 100 mmHg/s/cm). The newest definition of IEM has successfully become more selective, as shown by Sallette et al who demonstrated that in a cohort of 63 patients diagnosed with IEM per Chicago Classification v3.0, only 39 of these patients (62%) met Chicago Classification v4.0 criteria for IEM. 28
Despite evolving definitions, IEM has consistently been characterized by hypomotility and weak or failed swallows.1,6 Nonetheless, efforts to identify prokinetic therapeutics have generated mixed results, including metoclopramide and domperidone not demonstrating a beneficial effect in IEM.10,15 Medical therapies affecting serotonin receptors such as cisapride, mosapride, prucalopride, and buspirone have previously been suggested for the management of IEM, but early studies either provided conflicting results about the effectiveness of these agents or extrapolated conclusions from other patient populations.29-32 Cholinomimetics, such as pyridostigmine, have also been suggested based on the results of a small pilot study of patients with IEM, but more robust data from RCTs does not currently exist. 17 These suggestions highlight the dearth of available information regarding the medical management of IEM, particularly to augment contractility. Fewer data exist for improvement in symptoms among those with IEM, which may not be directly related to contractile vigor. 9 However, esophageal hypomotility may directly contribute to slow or incomplete bolus transit, resulting in symptoms of dysphagia, as recent data suggest an increasing number of ineffective swallows increases the chances of dysphagia in the setting of IEM. 4 Increasing esophageal contractility may therefore be helpful in the management of patients with IEM experiencing dysphagia without any evidence of outflow obstruction, who currently have few therapeutic options. This may be of particular importance in patients with IEM and GERD who are being considered for anti-reflux surgery, as patients with esophageal hypomotility have been consistently identified as higher risk for post-operative dysphagia after Nissen fundoplication if both IEM and poor esophageal contractile reserve are present. 11 Specifically, in IEM patients with a multiple rapid swallow sequence ratio less than one, dysphagia rates are higher. In this manner, IEM may have important implications for patients with GERD who are being considered for anti-reflux surgery, including proposed surgical approach and expected rate of post-surgical complications.
Our results affirm that few data from RCTs exist to help guide management in IEM. Our systematic review did identify improvement in HRM parameters with medical therapy in 5/6 (83%) of studies, including improvement with ¾ (75%) of SRA medications. However, given the limited number of studies, small patient population included, various HRM endpoints, and lack of patient reported outcomes, these results may be considered exploratory. Indeed, a meta-analysis was not performed due to the heterogeneity of the study drugs and outcomes, despite the related, but distinct, mechanism of action of each SRA identified. We also identified 2 studies assessing the effects of bethanechol in patients with IEM, and their results showed numerical improvement in various manometric outcomes.24,25 However, there was variability in therapeutic targets and the small number of studied patients (n = 12) who received bethanechol may support further investigation rather than definitive clinical conclusions, especially given the lack of well-defined controls in each study design. Long-term follow-up or symptom assessment was not present in most studies included in our review; Aggarwal et al examined buspirone response over 2 weeks, which was the longest follow-up interval among included studies and was also the only study to assess patient reported outcomes. 21 Notably, their results indicate that buspirone failed to improve manometric parameters or patient reported outcomes. Likewise, we were not able to assess for publication bias due to the small number of studies identified.
Similar to prior studies, we found that the studies in our review primarily evaluated real-time manometric outcomes such as DCI and threshold volume for generating secondary peristalsis, and had limited follow-up. Without evaluating adjunctive metrics such as bolus transit, it is difficult to assess whether augmenting DCI or wave amplitude improved esophageal clearance or function. 33 Our findings highlight the importance of objective findings and newer metrics that can be included in outcomes, as well as emphasizing how patient outcomes should be addressed in future research to fill an important gap in our current understanding of interventions on IEM. Additionally, our systematic review search was complete through 9/22/21, which was inclusive of a period after the publication of Chicago Classification v4.0, though no studies using Chicago Classification v4.0 met our inclusion criteria. As a result, studies in our review use older definitions of IEM, which have evolved over time. The definition of IEM is now based on DCI rather than wave amplitude. 7 The threshold for establishing IEM is also more stringent, for example current criteria require a greater number of absent (≥50%) or ineffective (>70%) swallows. 1 While stratifying our results by historical IEM definitions would be of interest, we do not have access to patient-level data to make this assessment. Previous studies have, however, shown that applying different classifications can change diagnostic performance characteristics. 28 Given the focus on ineffective swallows in the current definition of IEM, it is difficult to speculate how the results of prior interventions would perform in patient with IEM by Chicago Classification v4.0. However, this is an important area worthy of future study.
In addition, the potentially intermittent nature of IEM may also limit conclusions regarding therapeutic effectiveness. 34 Based on the diagnostic criteria in Chicago Classification v3.0 and v4.0, a sample of 10 liquid swallows has been established as sufficient for the evaluation of IEM, however the possibility of sampling bias exists. This may be suggested by Aggarwal et al who found that 3 of 10 (30%) of patients diagnosed with IEM had a new diagnosis of normal esophageal motility after administration of a placebo. 21 We acknowledge that individuals’ diagnoses on HRM can change based on medication usage, surgery, or other factors including evolving definitions. 33 In the current systematic review, these effects should be minimized as many studies were conducted on the same patients directly after a medical intervention, including with a catheter still in place. Furthermore, the more stringent definition of IEM in Chicago Classification v4.0 should allow for improved diagnostic confidence and ability to assess treatment effect in future studies.
In conclusion, our systematic review identified 6 high quality studies including 4 studies with a randomized, cross-over design that assessed the efficacy of SRAs in patients with IEM, but results are limited by the lack of symptom outcome data and standardization as well as evolving definitions of IEM. Nonetheless, our findings suggest there may be benefit to augmenting contractility, and these results may provide a framework for further investigation. With the advent of the Chicago Classification v4.0, a more refined and stringent definition has been established to identify patients with clinically relevant IEM, and studying this population may serve as the basis to address a larger need within esophageal diseases pertaining to the improvement and recovery of esophageal motility once it is lost. Larger studies utilizing current IEM criteria, emphasizing patients with dysphagia, and assessing clinically relevant, patient reported outcomes should be performed to assist in the management of this population with esophageal hypomotility for which clear therapeutic options are currently lacking.
Supplemental Material
sj-docx-1-gut-10.1177_26345161231168955 – Supplemental material for Medical Therapy for Ineffective Esophageal Motility: A Systematic Review
Supplemental material, sj-docx-1-gut-10.1177_26345161231168955 for Medical Therapy for Ineffective Esophageal Motility: A Systematic Review by Alexander T. Reddy, Erin J. Song, Rahul A. Shimpi, Sarah Cantrell and David A. Leiman in Foregut: The Journal of the American Foregut Society
Footnotes
Acknowledgements
All authors approved the final version of the article including the authorship list.
Author Contributions
ATR: Project conception/design; data collection; data analysis/interpretation; drafting of manuscript; critical revision; approved final draft. EJS: Project conception/design; data collection; data analysis/interpretation; critical revision; approved final draft. RAS: Project conception/design; data collection; data analysis/interpretation; critical revision; approved final draft. SC: Developing search strategy and conducting systematic search of literature; critical revision; approved final draft. DAL: Project conception/design; data collection; data analysis/interpretation; critical revision; approved final draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
