Abstract
Background:
Celiac disease (CD) is a clinical entity increasingly recognized in the severe obese population and its impact on outcomes following bariatric surgery is not currently understood. We aimed to systematically review the nutritional and clinical outcomes for patients with obesity and CD following bariatric surgery.
Methods:
Systematic search of MEDLINE, Embase, Scopus, and Web of Science was conducted in September 2021. Study followed PRISMA guidelines. Studies evaluating adult patients with CD undergoing bariatric surgery were included. Outcomes were descriptive due to limited studies.
Results:
Our literature review produced 9 studies with 152 patients. The weighted mean age of included patients was 44.2 years (±9.55 years) and 98% (n = 149) were female. Patients diagnosed with CD post-operatively appear to experience substantial perioperative and nutritional complications following bariatric surgery. However, pre-operative diagnosis may allow patient optimization and current studies suggest similar bariatric surgery outcomes can be achieved in these patients when the diagnosis is known. However, this study also highlights a scarcity of evidence evaluating the rate of CD and outcomes for these patients after bariatric surgery.
Conclusion:
Current literature lacks quality, comparative studies investigating the short- and long-term surgical complications and nutritional outcomes of bariatric surgery within patients who have co-diagnoses of obesity and CD. Despite these limitations, our group recommends pre-operative CD screening in patients experiencing either classical GI symptoms of CD including diarrhea, steatorrhea, bloating, or undifferentiated abdominal pain.
Key Learning Points
CD and bariatric surgery can produce similar nutritional deficiencies.
Missed pre-operative CD diagnoses reportedly experience severe GI symptoms post-operatively.
Literature lacks studies on clinical outcomes of bariatric surgery in patients with obesity and CD.
Introduction
Celiac disease (CD) is an autoimmune disorder characterized by a localized pathologic immune response to gliadin at the intestinal mucosa. 1 This results in an inflammatory environment that leads to declines in micro- and macro-nutrient absorption. The identification of subclinical phenotypes of CD has led to an increase in its diagnosis across all weight spectrums, 2 with the prevalence of CD in individuals with obesity ranging from 3% to 13%.3 -5
For patients with a BMI ≥40 kg/m2 without co-morbidities and those with a BMI ≥35 kg/m2 and co-morbidities, metabolic surgery confers the most sustained long-term treatment that not only offers rapid weight loss and glycemic control but improves metabolic outcomes like dyslipidemia, hypertension, and obstructive sleep apnea. 6 While regarded as safe and highly-effective procedures, both malabsorptive and restrictive bariatric procedures can lead to macro- and micronutrient deficiencies in a subset of patients.6,7 The growing need for bariatric surgery worldwide together with the increasing prevalence of subclinical CD poses a challenging dilemma regarding how patients are screened, selected, and managed peri-operatively to avoid the potentially disastrous consequences of malnutrition. Unfortunately, this relationship is currently poorly understood, and represents a potential clinical challenge if not diagnosed pre-operatively.
This study aims to systematically search and summarize the current literature regarding celiac disease in the context of bariatric surgery. Our primary outcomes were to evaluate differences in nutritional deficiencies and post-operative outcomes between CD and non-CD counterparts following bariatric surgery.
Methods
Search Strategy
The medical librarian (JK) developed and executed comprehensive searches in Ovid MEDLINE, Ovid Embase, Scopus, and Cochrane Library (Wiley) on June 4, 2021. The search terms can be found in Appendix 1. The search was updated on September 27, 2021. All relevant keywords pertaining to celiac disease and bariatric surgery were included in the search strategies. No language or date limits were applied. Refer to the appendices for full-text search strategies. In addition to subscription databases, the research team searched Google Scholar and evaluated the first 200 results for inclusion. Bibliographies from included studies were also reviewed. The reporting of this systematic review was guided by the standards of the 2020 Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) Statement with checklist provided as per the Meta-Analysis of Observational Studies in Epidemiology (MOOSE) guidelines (Supplemental Material 1).
Selection Criteria
Titles and abstracts were first screened for inclusion by 2 independent reviewers (LR and VM). Studies meeting inclusion criteria then underwent dual-author full text review. Screening was based on the following inclusion criteria: adult patients (age ≥18 years), patients with a history of bariatric surgery, and patients diagnosed with CD. Diagnosis of CD was defined as having either positive serological findings (at least one of [IgA or IgG]: anti-tissue transglutaminase, anti-endomysial, anti-gliadin antibody), or positive intestinal mucosal biopsy (partial/subtotal/complete villous atrophy, with or without crypt hyperplasia), and if initiated, a response to a gluten-free diet (GFD). 8 Studies which only reported gastric banding procedures were excluded due to the historical nature of this procedure. Other exclusion criteria included studies published solely as abstracts, letters, reviews, or were duplicates. Our key outcomes of interest included nutritional deficiencies and clinical course following bariatric surgery.
Assessment of Risk of Bias
Included studies were assessed for methodological quality and bias utilizing the methodological index for non-randomized studies (MINORS) criteria for non-randomized cohort studies.
Data Analysis
Continuous data was reported as absolute numbers and weighted means, while categorical data was represented as proportions. A meta-analysis was conducted if there were a minimum of 3 studies of high quality (low risk of bias) examining the same outcome.
Results
Study Selection
Search results yielded 562 results, and after eliminating duplicates, 334 unique studies from Ovid MEDLINE, Ovid Embase, Scopus, and Cochrane Library databases, and 200 studies from Google Scholar were screened (Figure 1). Following title and abstract screening, 518 studies were excluded, and 16 studies were included for full-text review. Full-text review further excluded 7 studies; reasons for exclusion can be found in Figure 1. A total of 9 studies9-17 were deemed acceptable for the present review, these included: 1 retrospective case series, 1 case series, 5 case reports, and 2 retrospective cohort study (Table 1).
Characteristics of Included Studies.
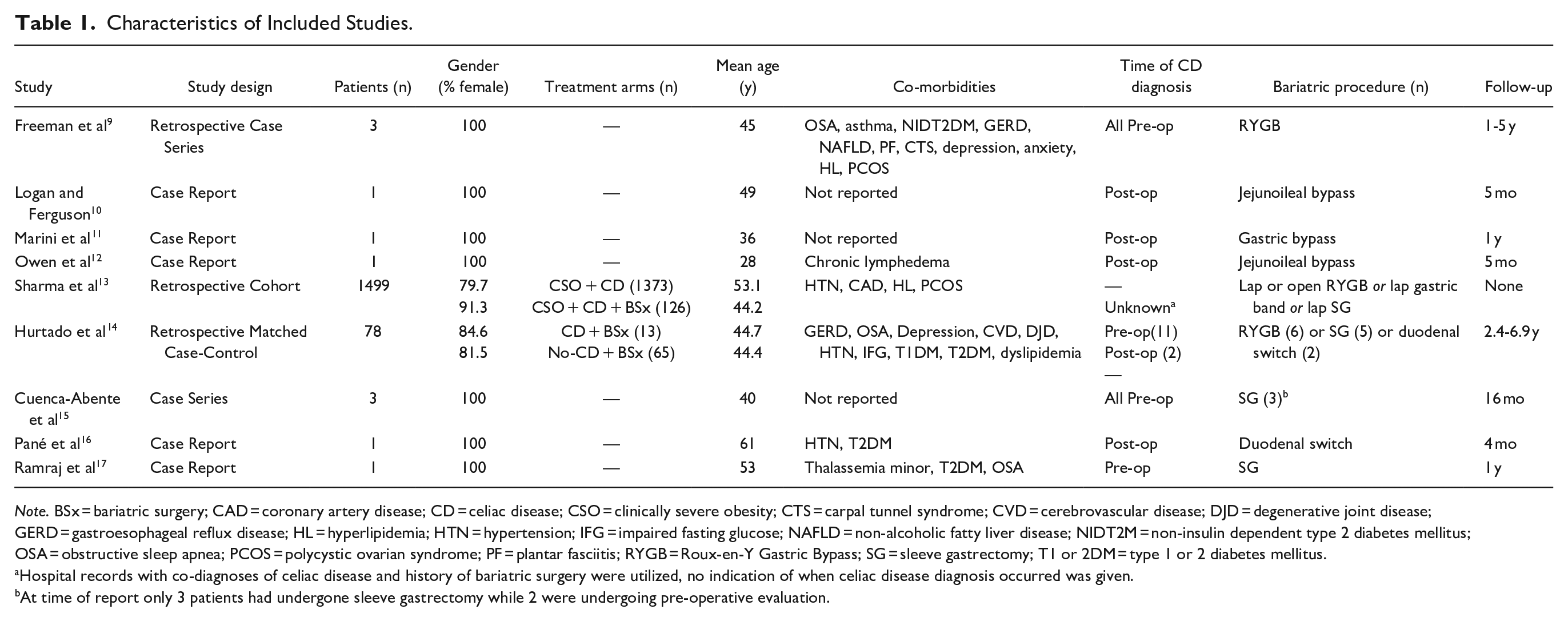
Note. BSx = bariatric surgery; CAD = coronary artery disease; CD = celiac disease; CSO = clinically severe obesity; CTS = carpal tunnel syndrome; CVD = cerebrovascular disease; DJD = degenerative joint disease; GERD = gastroesophageal reflux disease; HL = hyperlipidemia; HTN = hypertension; IFG = impaired fasting glucose; NAFLD = non-alcoholic fatty liver disease; NIDT2M = non-insulin dependent type 2 diabetes mellitus; OSA = obstructive sleep apnea; PCOS = polycystic ovarian syndrome; PF = plantar fasciitis; RYGB = Roux-en-Y Gastric Bypass; SG = sleeve gastrectomy; T1 or 2DM = type 1 or 2 diabetes mellitus.
Hospital records with co-diagnoses of celiac disease and history of bariatric surgery were utilized, no indication of when celiac disease diagnosis occurred was given.
At time of report only 3 patients had undergone sleeve gastrectomy while 2 were undergoing pre-operative evaluation.
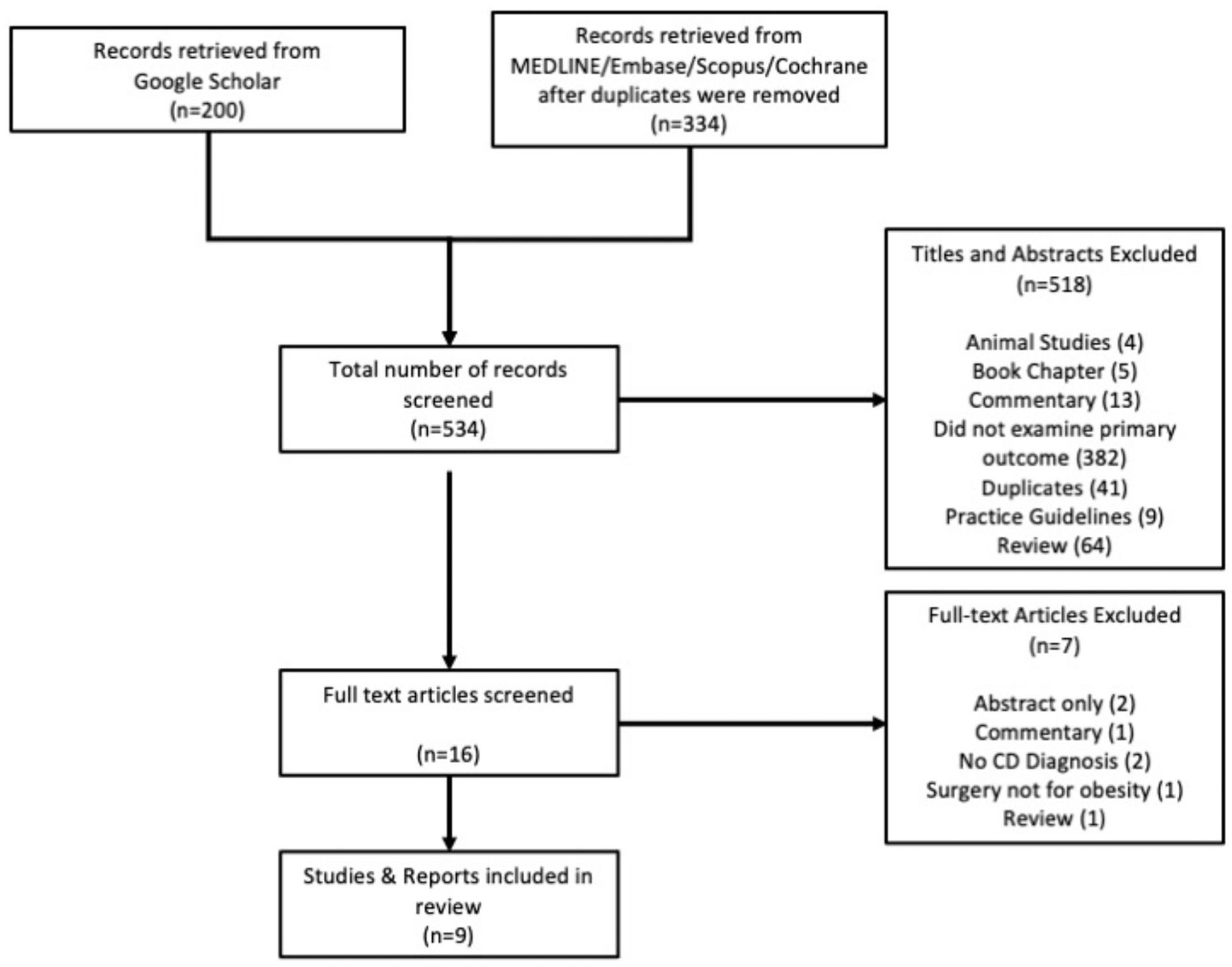
PRISMA diagram of selected studies.
Study Demographics
The 9 studies reported a total of 152 patients who underwent bariatric surgery for obesity management and had a diagnosis of CD (Table 1).9-17 The weighted mean age at time of surgery was 44.2 years (±9.55 years) and 98% of reported patients were female. Reported patient co-morbidities are found in Table 1. Eighteen patients had a pre-operative CD diagnosis, 6 had a post-operative CD diagnosis, and the time of CD diagnosis for the remaining 126 was not reported. Regarding type of bariatric procedure performed, 9 patients underwent RYGB, 2 received a jejunoileal bypass, 9 had a sleeve gastrectomy, 3 underwent duodenal switch, 1 patient was reported to have gastric bypass with no specific indication to type of procedure, 2 patients had yet to undergo bariatric surgery at time of report, and the remaining 126 patients were reported to have either a laparoscopic or open RYGB, gastric band, or SG with no indication to how this was stratified. Follow-up after bariatric surgery ranged from none to 6.9 years.
Patient Outcomes
Freeman et al 9 reported 3 patients with a pre-operative diagnosis of CD via serology and biopsy findings, with whom 2 adhered to a strict GFD prior to surgery. Of these 3 patients who underwent RYGB for treatment of obesity, only 2 developed postoperative vitamin D deficiencies, which were correctable by oral supplementation (Table 2). No other deficiencies were identified in these patients. Similarly, Cuenca-Abenta et al 15 described 5 patients who were diagnosed with CD during pre-operative evaluation due to abnormal endoscopic findings and subsequent confirmation via duodenal biopsy. Three of the 5 patients underwent sleeve gastrectomy and were reported to have an uneventful post-operative course.
Reported Post-Operative Nutritional Deficiencies and Clinical Outcomes Experienced by Patients With Celiac Disease and Their Response to Treatment.
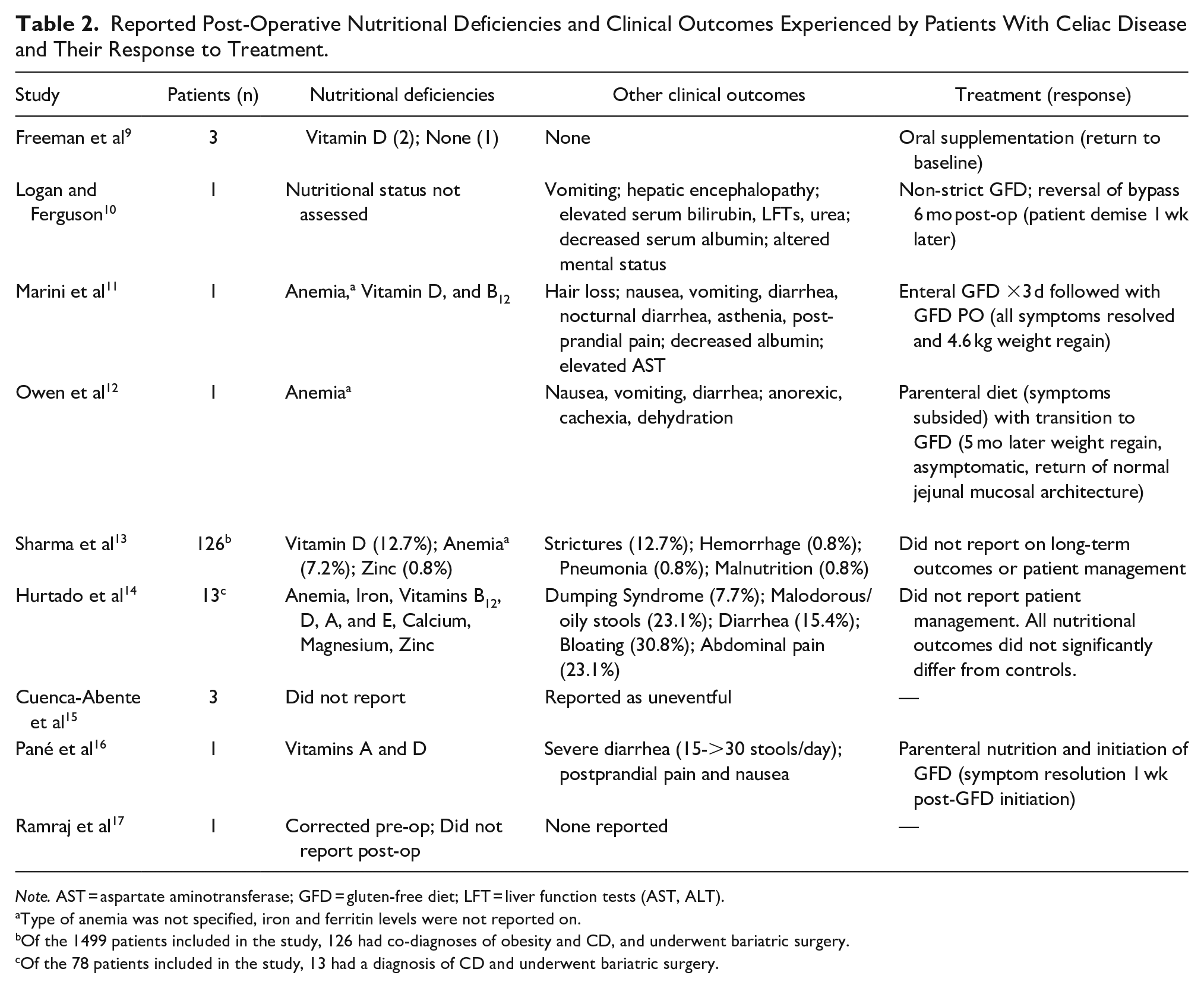
Note. AST = aspartate aminotransferase; GFD = gluten-free diet; LFT = liver function tests (AST, ALT).
Type of anemia was not specified, iron and ferritin levels were not reported on.
Of the 1499 patients included in the study, 126 had co-diagnoses of obesity and CD, and underwent bariatric surgery.
Of the 78 patients included in the study, 13 had a diagnosis of CD and underwent bariatric surgery.
Four case reports10-12,16 outlined patients experiencing either nausea, vomiting, diarrhea, and, in 1 instance, 10 mental status fluctuations and eventual death following bariatric surgery (Table 2). In all cases, post-operative biopsies demonstrated jejunal villous atrophy and crypt hyperplasia, and a diagnosis of CD11,12,16 or subclinical CD 10 were made. Marini et al, 11 Owen et al, 12 and Pané et al 16 reported symptom alleviation following implementation of a GFD. Logan and Ferguson 10 initiated a non-strict GFD and eventual reversal of the jejunoileal bypass; the patient died shortly after these measures. Conversely, Ramraj et al 17 reported an uneventful post-sleeve gastrectomy course in a patient with celiac disease whose nutrient deficiencies were corrected pre-operatively. Nutritional status was not thoroughly assessed in all case reports (Table 2).
Sharma et al 13 reported the impact of bariatric surgery (RYGB, gastric banding, or SG) on clinical, surgical, and nutritional outcomes in patients with obesity and CD. Patients who underwent bariatric surgery did experience vitamin D and zinc deficiencies, and anemia but were statistically only shown to have an increased prevalence and risk of vitamin D deficiency (Table 2). When compared to patients with obesity and CD who did not undergo bariatric surgery, those that received surgery had a reduced risk of renal failure, sepsis, respiratory failure, and urinary tract infections (UTI), and an increased risk of strictures. No differences in anemia, thiamine, or zinc levels were detected between the groups. However, comparisons between type of bariatric surgery and clinical outcomes were not made and several confounding variables, including patient demographics and control groups which are discussed below, raise concerns about the widespread applicability of their results.
Lastly, Hurtado et al 14 was the only cohort study comparing the effects of bariatric surgery (RYGB, SG, and duodenal switch) in patients with CD against a demographically similar control group who did not have CD. Patients with CD did not experience any statistically different nutritional deficiency and clinical outcomes than those without CD (Table 2). Vitamin D and iron were the most common deficiencies amongst both groups. Those with CD were significantly more likely to experience malodorous and oily stools post-operatively and were found to have lower levels of HDL 12-months following surgery. When stratified by procedure there was no difference in the incidence of nutritional deficiencies in patients with CD.
Risk of Bias for Included Study
The MINORS criteria was used to assess the included retrospective cohort studies for risk of bias (Supplemental Material 2, Table S1). 18 Sharma et al 13 did not meet ideal criteria as the compared groups had differences within their demographics, there was a lack of prospective calculation of study size, and there was no follow-up with patients. Similarly, Hurtado et al 14 also did not meet ideal criteria as study design prevented blinding and study size was not calculated.
Discussion
Due to the potential for nutritional deficiency to occur in both CD and bariatric procedures, we aimed to investigate the impact these have together on nutritional status, disease progression, and clinical outcomes. The current review systematically included 2 case series,9,15 5 case reports,10-12,16,17 and 2 retrospective cohort studies13,14 discussing patient outcomes in patients with CD undergoing bariatric surgery. Apparent from the included studies is the potential for adverse outcomes to occur when CD exists following metabolic surgery. We also highlight the sparsity of current studies, highlighting the need for further investigations outlining the prevalence and impact of CD within this population.
A notable finding in this study was the variability of outcomes following bariatric surgery dependent on the timing of CD diagnosis, either being pre-operative or post-operative. In patients with a post-operative CD diagnosis, persistent diarrhea, vomiting, malodorous stools, clinically severe weight loss, abdominal pain, and hospitalization occurred.10-12,16 Although these patients returned to near or fully asymptomatic levels following their post-operative diagnosis and introduction of a GFD, they experienced a myriad of additional symptoms than those with a pre-operative CD diagnosis. Comparatively, patients with pre-operative CD diagnoses appeared to have unremarkable post-operative courses following either RYGB or SG.9,15 This should also raise awareness about the potential for persistent severe nutritional deficiencies following bariatric surgery in patients with CD who are non-adherent to a GFD. Further, these outcomes suggests that awareness of CD diagnosis prior to bariatric intervention may enable diet, procedural, and follow-up optimization to reduce potential malabsorptive complications. To assist with both pre-operative diagnosis and post-operative consideration Table 3 summarizes various nutritional deficiencies that are seen in CD, RYGB, and SG along with possible clinical presentations and recommended nutritional screening procedures.1,19-23
Note. SG = sleeve gastrectomy; WLS = weight-loss surgery.
Females, blacks, patients not attending clinic after surgery, those with thiamine deficiency risk factors, patients with: GI symptoms (intractable nausea, vomiting, constipation, jejunal dilation, megacolon), concomitant diseases, small bowel bacterial overgrowth; other risk factors: excessive alcohol consumptions, rapid weight loss, or malnutrition. 19
Due to the restrictive nature of SG, nutritional deficiencies are far less common than in RYGB.
On the other hand, 2 retrospective cohort studies demonstrated that bariatric surgery can be performed relatively safely in patients with CD when the diagnosis is known pre-operatively. Sharma et al 13 investigated outcomes in patients with CD, comparing those who underwent bariatric surgery with those who did not. While rates of sepsis, UTI, renal failure, and respiratory failure were similar, there appeared to a small be increased risk of vitamin D deficiency and strictures for patients receiving bariatric surgery. However, the comparator group (those receiving bariatric surgery) were predominantly hospitalized non-electively, were younger, and lacked prospective data collection. Furthermore, clinical outcomes assessed were not specifically outlined in their methods, comparisons were not stratified by procedure type, and no follow-up period was assessed, which should be considered when interpreting results. Further investigations evaluating more similar comparator groups should be done to adequately ascertain the safety profile of bariatric surgery in those with pre-operative CD diagnoses. The second cohort study was the only study to compare nutritional, clinical, and comorbid outcomes following a variety of bariatric procedures (RYGB, SG, and duodenal switch) in patients with CD compared to those without CD. 14 No statistical significance in nutritional deficiencies were found, and malodourous and oily stools were the only significant GI symptoms experienced more in those with CD. However, the study only included 13 patients with CD and did not specify timing of CD diagnosis. There continues to be a need for more robust investigations elucidating the safest means of metabolic surgery for those with CD.
Overall, in terms of managing patients with CD who are being considered for bariatric surgery, limited evidence, and guidelines currently exist. Currently, the American Society for Metabolic and Bariatric Surgery (ASMBS) recommends a preprocedural history, physical exam, and nutrient screening in all patients, and suggests that routine upper endoscopy prior to bariatric surgery is justifiable based on the surgeon’s discretion.19,24 However, the ASMBS does not make recommendations on screening for specific enteropathies unless clinically indicated during the pre-procedure work-up. It is noted that if persistent and severe GI symptoms occur ≥5 days post-operatively then a thorough history, physical and upper endoscopy with small bowel biopsies should be done to evaluate for CD. 19 In patients with CD, 3% to 13% are estimated to have a co-diagnosis of obesity,3-5 inclusion of a simple intestinal biopsy during pre-procedure endoscopy may elucidate both the true prevalence of CD in patients with obesity who undergo bariatric surgery, and aid in the prevention of severe post-surgical GI symptoms. Another consideration is that many patients with obesity are prescribed weight-loss medications, like GLP-1 agonists and SGLT-2 inhibitors, that can contribute to symptoms synonymous with those of classical CD; in those patients, having histology available may reveal CD in individuals undergoing surgery in whom medications may mask the diagnosis.
Along with CD screening, consideration should be given for what type of bariatric procedure should be offered for obesity management in the setting of co-existing CD. The malabsorptive state imposed by CD could place a patient at risk for severe nutritional deficiencies if given a procedure that produces similar effects. RYGB has been shown to be a more effective procedure in reducing comorbidities and managing weight in obesity, although more complications have been reported with this procedure.24,25 Similarly, procedures such as single anastomosis duodenal switch may place patients at greater risk of malabsorption, and avoiding these complications may be wise in patients with CD. However, this risk remains only theoretical because studies have yet to comparatively evaluated malabsorptive and restrictive procedures in patients with CD. Similarly, these more complex procedures have higher risk of complications including marginal ulceration, anastomotic stricture and leak, and bowel obstruction. It remains unknown how the inflammatory state imposed by CD interplays with the development of these complications and future studies evaluating these questions would be of interest.
A major limitation of the current review is the paucity of studies investigating the relationship between CD and bariatric surgery, type of bariatric surgery safest in this patient population, and the long-term outcomes of patients with obesity and CD who are treated with metabolic surgery. The low number of patients reported and wide variety of metabolic procedures performed limited the ability for meta-analysis of current literature. Also, lack of and/or inadequate follow-up in several studies may have reduced the number of nutritional deficiencies reported. Another limitation is the risk of bias within the cohort studies13,14 included in our review. As these were the only cohort studies found that investigated this patient population in the context of bariatric surgery, we opted for inclusion and considered their bias when interpreting their results.
Despite these limitations, our work provides the first systematic review demonstrating the large gap of knowledge elucidating the safety and outcomes of bariatric surgery on patients with obesity and CD. Future research should include comparative studies investigating the short- and long-term surgical complications and nutritional outcomes of bariatric surgery within this patient population. Also, comparative evaluation of outcomes following SG versus RYGB would be beneficial to further guide procedure selection. Despite the above limitations, given the potential adverse consequences associated with a missed CD diagnosis in the context of metabolic surgery predisposing for malnutrition we propose that intestinal biopsy during pre-operative upper GI endoscopy and serum anti-TTG should be done in patients who are experiencing symptoms of CD including diarrhea with or without abdominal pain, steatorrhea, bloating, flatulence, or undifferentiated abdominal pain. 8 For symptomatic individuals, serum anti-TTG is both sensitive and specific (>95%), while >4 biopsies offer the best for CD diagnosis, with a duodenal bulb biopsy from either the 9- or 12-o’clock position in addition to biopsies of the distal duodenum having a sensitivity of 96%. 8 The American College of Gastroenterology guidelines provide a review of the diagnostic yield, criteria, and ongoing management of CD, which may be useful for surgeons hoping to implement screening and management strategies. 8 While little evidence exists to guide this screening, this screening approach would be feasible, low risk, and may enable patient optimization with GF diet preoperatively to reduce risks of serious complications. Future investigations should aim to explore the utility of this measure in bariatric surgery work-up and its impact on long-term outcomes following surgery.
Conclusion
With improvements in diagnostic measures, CD is becoming increasingly recognized in patients with obesity. However, the true prevalence of CD in patients who undergo bariatric surgery is not yet known. There is a large gap of knowledge investigating the impact CD has on clinical and nutritional outcomes following bariatric surgery. Further studies should define the value of pre-procedural enteropathy screening in bariatric surgery and delineate the most effective and safe bariatric procedure for patients with obesity and CD co-diagnoses.
Supplemental Material
sj-docx-2-gut-10.1177_26345161221102137 – Supplemental material for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature
Supplemental material, sj-docx-2-gut-10.1177_26345161221102137 for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature by Logan Richard, Valentin Mocanu, Kevin Verhoeff, Ka Man Cheung, Janice Y. Kung, Daniel W. Birch, Noah J. Switzer and Shahzeer Karmali in Foregut: The Journal of the American Foregut Society
Supplemental Material
sj-pdf-1-gut-10.1177_26345161221102137 – Supplemental material for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature
Supplemental material, sj-pdf-1-gut-10.1177_26345161221102137 for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature by Logan Richard, Valentin Mocanu, Kevin Verhoeff, Ka Man Cheung, Janice Y. Kung, Daniel W. Birch, Noah J. Switzer and Shahzeer Karmali in Foregut: The Journal of the American Foregut Society
Supplemental Material
sj-pptx-3-gut-10.1177_26345161221102137 – Supplemental material for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature
Supplemental material, sj-pptx-3-gut-10.1177_26345161221102137 for Celiac Disease and Bariatric Surgery: A Systematic Review of Current Literature by Logan Richard, Valentin Mocanu, Kevin Verhoeff, Ka Man Cheung, Janice Y. Kung, Daniel W. Birch, Noah J. Switzer and Shahzeer Karmali in Foregut: The Journal of the American Foregut Society
Footnotes
Appendix
Database Search Strategies.

| Database | Search strategy |
|---|---|
| MEDLINE Ovid MEDLINE(R) ALL 1946 to September 24, 2021 |
1. bariatric*.mp. or exp Bariatric Surgery/ 2. exp Biliopancreatic Diversion/or (biliopancreatic diversion* or biliopancreatic bypass).mp. 3. duodenal switch*.mp. 4. (gastric bypass* or gastric surger*).mp. 5. exp Gastroenterostomy/or gastroenterostom*.mp. 6. gastrogastrostom*.mp. 7. gastrojejunostom*.mp. 8. gastroileal bypass.mp. 9. gastroplast*.mp. 10. intestin* bypass.mp. or exp Jejunoileal Bypass/ 11. ((jejunoileal or jejuno-ileal or jejunoilial or jejuno-ilial or jejunocolic) adj bypass).mp. 12. mason* procedure*.mp. 13. ((obesity or obese or weight) adj5 surg*).mp. 14. exp Obesity/su [Surgery] 15. restrictive surger*.mp. 16. roux-en-y.mp. or exp Anastomosis, Roux-en-Y/ 17. scopinaro.mp. 18. sleeve gastrectom*.mp. 19. ((stomach or gastric) adj5 stapl*).mp. 20. (SADI or SADI-S or single anastomosis duodeno*).mp. 21. or/1-20 22. exp Celiac Disease/ 23. ((celiac or coeliac) adj1 (disease* or syndrome*)).mp. 24. tissue transglutaminase antibod*.mp. 25. anti-endomysium antibod*.mp. 26. endomysi* antibod*.mp. 27. or/22-26 28. 21 and 27 |
| Embase Ovid Embase 1974 to 2021 September 24 |
1. bariatric*.mp. or exp bariatric surgery/ 2. (biliopancreatic diversion* or biliopancreatic bypass).mp. 3. duodenal switch*.mp. 4. (gastric bypass* or gastric surger*).mp. 5. exp gastroenterostomy/ or gastroenterostom*.mp. 6. gastrogastrostom*.mp. 7. gastrojejunostom*.mp. or gastrojejunostomy/ 8. gastroileal bypass.mp. 9. gastroplast*.mp. or gastroplasty/ 10. intestin* bypass.mp. or exp intestine bypass/ 11. ((jejunoileal or jejuno-ileal or jejunoilial or jejuno-ilial or jejunocolic) adj bypass).mp. 12. mason* procedure*.mp. 13. ((obesity or obese or weight) adj5 surg*).mp. 14. restrictive surger*.mp. 15. roux-en-y.mp. or Roux Y anastomosis/ 16. scopinaro.mp. 17. sleeve gastrectom*.mp. 18. ((stomach or gastric) adj5 stapl*).mp. 19. (SADI or SADI-S or single anastomosis duodeno*).mp. 20. or/1-19 21. exp celiac disease/ 22. ((celiac or coeliac) adj1 (disease* or syndrome*)).mp. 23. tissue transglutaminase antibod*.mp. 24. anti-endomysium antibod*.mp. 25. endomysi* antibod*.mp. 26. exp protein glutamine gamma glutamyltransferase antibody/ 27. exp endomysium antibody/ 28. or/21-27 29. 20 and 28 |
| Cochrane Library (via Wiley) Cochrane Reviews, Trials |
#1 MeSH descriptor: [Bariatric Surgery] 2 tree(s) exploded #2 bariatric* #3 MeSH descriptor: [Biliopancreatic Diversion] explode all trees #4 “biliopancreatic diversion*” or “biliopancreatic bypass” or “duodenal switch*” #5 “gastric bypass*” or “gastric surger*” #6 MeSH descriptor: [Gastroenterostomy] explode all trees #7 gastroenterostom* or gastrogastrostom* or gastrojejunostom* or “gastroileal bypass” #8 gastroplast* or “intestin* bypass” #9 MeSH descriptor: [Jejunoileal Bypass] 4 tree(s) exploded #10 ((jejunoileal or jejuno-ileal or jejunoilial or jejuno-ilial or jejunocolic) near/1 bypass) #11 “mason* procedure*” or “restrictive surger*” #12 ((obesity or obese or weight) near/5 surg*) #13 roux-en-y #14 MeSH descriptor: [Anastomosis, Roux-en-Y] explode all trees #15 scopinaro #16 “sleeve gastrectom*” #17 ((stomach or gastric) near/5 stapl*) #18 SADI or SADI-S or “single anastomosis duodeno*” #19 {OR #1-#18} #20 MeSH descriptor: [Celiac Disease] explode all trees #21 ((celiac or coeliac) near/1 (disease* or syndrome*)) #22 tissue transglutaminase antibod* #23 anti-endomysium antibod* #24 endomysi* antibod* #25 {OR #20-#24} #26 #19 AND #25 |
| Scopus | ( TITLE-ABS-KEY ( bariatric* OR “biliopancreatic diversion*” OR “biliopancreatic bypass” OR “duodenal switch*” OR “gastric bypass*” OR “gastric surger*” OR “gastroileal bypass” OR gastroplast* OR “intestin* bypass” OR “jejunoileal bypass” OR “jejuno-ileal bypass” OR “jejunoilial bypass” OR “jejuno-ilial bypass” OR “jejunocolic bypass” OR “mason* procedure” OR “roux-en-y” OR “sleeve gastrectom*” ) OR TITLE-ABS-KEY ( ( ( stomach OR gastric ) W/5 stapl* ) OR sadi OR sadi-s OR “single anastomosis duodeno*” ) ) AND TITLE-ABS-KEY ( ( ( celiac OR coeliac ) W/1 disease* ) OR ( ( celiac OR coeliac ) W/1 syndrome* ) OR “tissue transglutaminase antibod*” OR “anti-endomysium antibod*” OR “endomysi* antibod*” ) |
| Google Scholar | (“bariatric surgery” OR gastroplasty OR roux-en-y OR sleeve gastrectomy) AND (celiac disease OR coeliac disease) |
| Database | Search strategy |
| MEDLINE Ovid MEDLINE(R) ALL 1946 to June 03, 2021 |
1. bariatric*.mp. or exp Bariatric Surgery/ 2. exp Biliopancreatic Diversion/ or (biliopancreatic diversion* or biliopancreatic bypass).mp. 3. duodenal switch*.mp. 4. (gastric bypass* or gastric surger*).mp. 5. exp Gastroenterostomy/ or gastroenterostom*.mp. 6. gastrogastrostom*.mp. 7. gastrojejunostom*.mp. 8. gastroileal bypass.mp. 9. gastroplast*.mp. 10. intestin* bypass.mp. or exp Jejunoileal Bypass/ 11. ((jejunoileal or jejuno-ileal or jejunoilial or jejuno-ilial or jejunocolic) adj bypass).mp. 12. mason* procedure*.mp. 13. ((obesity or obese or weight) adj5 surg*).mp. 14. exp Obesity/su [Surgery] 15. restrictive surger*.mp. 16. roux-en-y.mp. or exp Anastomosis, Roux-en-Y/ 17. scopinaro.mp. 18. sleeve gastrectom*.mp. 19. ((stomach or gastric) adj5 stapl*).mp. 20. (SADI or SADI-S or single anastomosis duodeno*).mp. |
| 21. or/1-20 |
|
| Embase |
1. bariatric*.mp. or exp bariatric surgery/ |
| Cochrane Library |
#1 MeSH descriptor: [Bariatric Surgery] 2 tree(s) exploded |
| #21 ((celiac or coeliac) near/1 (disease* or syndrome*)) |
|
| Scopus | ( TITLE-ABS-KEY ( bariatric* OR “biliopancreatic diversion*” OR “biliopancreatic bypass” OR “duodenal switch*” OR “gastric bypass*” OR “gastric surger*” OR “gastroileal bypass” OR gastroplast* OR “intestin* bypass” OR “jejunoileal bypass” OR “jejuno-ileal bypass” OR “jejunoilial bypass” OR “jejuno-ilial bypass” OR “jejunocolic bypass” OR “mason* procedure” OR “roux-en-y” OR “sleeve gastrectom*” ) OR TITLE-ABS-KEY ( ( ( stomach OR gastric ) W/5 stapl* ) OR sadi OR sadi-s OR “single anastomosis duodeno*” ) ) AND TITLE-ABS-KEY ( ( ( celiac OR coeliac ) W/1 disease* ) OR ( ( celiac OR coeliac ) W/1 syndrome* ) OR “tissue transglutaminase antibod*” OR “anti-endomysium antibod*” OR “endomysi* antibod*” ) |
| Google Scholar | (“bariatric surgery” OR gastroplasty OR roux-en-y OR sleeve gastrectomy) AND (celiac disease OR coeliac disease) |
Correction (May 2024):
Article updated to add Ethical approval statement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is exempt from our institutional review board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

