Abstract
Sickle cell nephropathy (SCN) is one of the complications of sickle cell disease (SCD). It is a chronic and progressive disease that can lead to end-stage kidney disease (ESKD). Anemia is a common complication in SCD and can exacerbate the symptoms of SCN. Anemia in SCN can be due to increased red cell destruction, reduced red cell production, and various non-disease-associated causes. The prolonged anemia in SCN can lead to various complications, which include rapid progression to ESKD, reduced quality of life (QoL), and increased risk of vaso-occlusive complications (VOC). There are many challenges faced by treating nephrologists while managing anemia in SCN, which have been discussed in this review along with possible solutions.
Keywords
Introduction
Sickle cell nephropathy (SCN) is one of the complications of sickle cell disease (SCD) that affects the kidneys. It is a chronic and progressive disease that can lead to end-stage kidney disease (ESKD).1, 2 Anemia is a common complication in SCD and can exacerbate the symptoms of SCN. 3 Anemia and SCN are both independent risk factors for mortality in ESKD patients; hence, it is not surprising that the mortality rate is higher among ESKD patients with SCD compared with those without SCD. 4 Managing anemia in SCN can be challenging, and it requires a comprehensive approach that takes into account the underlying pathophysiology of the disease. 2
Anemia in SCN
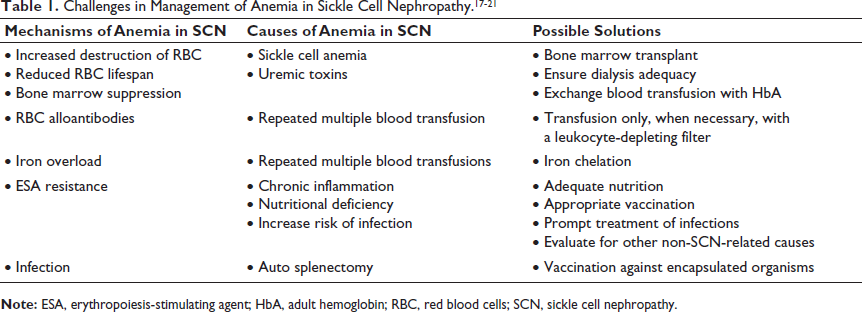
Anemia in SCN can be due to increased red cell destruction, reduced red cell production, and various non-SCD-associated causes, as depicted in Table 1. The prolonged anemia in SCN can lead to various complications, which include rapid progression to ESKD, reduced quality of life (QoL), and increased risk of vaso-occlusive complications (VOC). There are many challenges faced by treating nephrologists while managing anemia in SCN.
Challenges in Managing Anemia in SCN
Refractory anemia is a common complication in SCN and is caused by the destruction of red blood cells and the decreased production of new red blood cells. Repeated red cell transfusion results in the formation of alloantibodies against red cells, which contribute to refractory anemia in patients with SCN. 5 Anemia can exacerbate the symptoms of the disease by decreasing oxygen delivery to the kidneys and other organs. Anemia can also increase the risk of stroke, pulmonary hypertension, and other complications in SCD, which contribute to increased mortality rates among SCN patients. 6
Judicious Use of Erythropoiesis-stimulating Agents (ESAs)
One of the challenges in managing anemia in SCN is the use of ESAs. 7 ESAs are a class of drugs that stimulate the production of red blood cells. They have been shown to be effective in treating anemia in patients with chronic kidney disease (CKD), but their use in patients with SCD is controversial. Evidence shows that ESA can increase the level of fetal hemoglobin (HbF); hence, theoretically, it should reduce sickle cell crises. 8 Some studies, however, have shown that ESAs may increase the risk of vaso-occlusive events in patients with SCD, 9 while others have shown no increased risk. 10 The decision to use ESAs in patients with SCD should be made on a case-by-case basis, taking into account the individual patient’s risks and benefits. They also have relative ESA resistance due to chronic inflammation and chronic hemolysis. 11 Doses of ESA used in SCN are usually 1.5-2.0 times higher compared to non-SCD CKD patients, and this greatly adds to the cost of treatment, especially in a low-resource setting. 12 New therapies like hypoxia-inducible factor–prolyl hydroxylase inhibitors are contraindicated in SCD, as they may precipitate a sickle cell crisis in these cases.
Judicious Blood Transfusions
Another challenge in managing anemia in SCN is the use of blood transfusions. Blood transfusions can be used to increase hemoglobin levels in patients with anemia. 2 However, they are associated with several risks, including the transmission of infections and the development of alloantibodies that can lead to hemolytic transfusion reactions. 5 In addition, chronic transfusion therapy can lead to iron overload, which can cause damage to organs such as the liver and heart. 13 In addition, multiple blood transfusions also increase the risk of pre-transplant sensitization, making them high-risk patients for a kidney transplant. 14 Using a leukocyte-depleting filter can help reduce sensitization, but it comes with a financial implication, that many may not be able to afford. 15 Therefore, the decision to use blood transfusions in patients with SCN should also be made on a case-by-case basis, taking into account the individual patient’s risks and benefits.
Judicious Use of Iron Chelation Therapy
Finally, iron chelation therapy is another challenge in managing anemia in SCN. Iron chelation therapy is used to reduce the accumulation of iron in the body, which can occur as a result of chronic transfusion therapy. However, the use of iron chelation therapy in patients with SCD is limited by its side effects, including gastrointestinal disturbances, skin rashes, and hypersensitivity reactions. The decision to use iron chelation therapy in patients with SCN should also be made on a case-by-case basis, considering the individual patient’s risks and benefits.
Possible Solutions
Various possible solutions to the challenges have been depicted in Table 1. Kidney and bone marrow transplantation are the definitive treatment for managing ESKD patients due to SCN, 16 however, temporary measures like exchange blood transfusion, use of leukocyte filter, adequate dialysis, appropriate vaccination, and judicious use of ESA should be employed. It needs a multidisciplinary approach with combined treatment by a nephrologist and a hematologist. Also, treatment needs to be individualized in each case, as the same treatment does not fit all cases of anemia in SCN.
Conclusions
Anemia is a common complication in SCN and can exacerbate the symptoms of the disease. Managing anemia in SCN requires a comprehensive approach that considers the underlying pathophysiology of the disease. Judicious use of ESAs, blood transfusions, and iron chelation therapy should be made on a case-by-case basis, taking into account the individual patient’s risks and benefits. Further research is needed to understand better the risks and benefits of these therapies in patients with SCD, as still very few patients are able to undergo combined kidney and bone marrow transplantation.
Footnotes
Authors’ Contribution
All authors contributed equally to the conception, design, data acquisition, analysis, and interpretation of the work, as well as the drafting and revising of the manuscript. All authors approved the final version for publication.
Data Available Statement
Data are available with the authors.
Declaration of Conflicting Interests
The authors declared no conflict of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.