Abstract
Background
Multimorbidity is common in patients with atrial fibrillation (AF), yet comorbidity patterns are not well documented.
Methods
The prevalence of 18 chronic conditions (6 cardiometabolic, 7 other somatic, 5 mental health) was obtained in patients with new-onset AF from 2013-2017 from a 27-county region and controls matched 1:1 on age, sex, and county of residence. For AF patients and controls separately, clustering of conditions and co-occurrence beyond chance was estimated (using the asymmetric Somers’ D statistic), overall and for ages <65, 65-74, 75-84, and ≥85 years.
Results
Among 16,509 patients with AF (median age 76 years, 57% men), few (4%) did not have any of the 18 chronic conditions, whereas nearly one-quarter of controls (23%) did not have any chronic conditions. Clustering of cardiometabolic conditions was common in both AF patients and controls, but clustering of other somatic conditions was more common in AF. Although the prevalence of most condition pairs was higher in AF patients, controls had a larger number of condition pairs occurring together beyond chance. In persons aged <65 years, AF patients more frequently exhibited concordance of condition pairs that included either pairs of somatic conditions or a combination of conditions from different condition groups. In persons aged 65-74 years, AF patients more frequently had pairs of other somatic conditions.
Conclusion
Patterns of co-existing conditions differed between patients with AF and controls, particularly in younger ages. A better understanding of the clinical consequences of multimorbidity in AF patients, including those diagnosed at younger ages, is needed.
Introduction
Multimorbidity, defined as the co-occurrence of 2 or more chronic conditions, 1 is common, affecting approximately 1 in 4 adults and more than two-thirds of adults aged 65 years and older in the United States.2–4 Multimorbidity is particularly common in patients with cardiovascular disease, the majority of whom have multiple chronic cardiovascular conditions, but approximately two-thirds also have non-cardiovascular chronic conditions.5,6 Patients with multimorbidity experience poor health outcomes, including reduced quality of life, disability, and increased hospitalizations and death.5,7,8 Other consequences of multimorbidity include challenges with fragmented care and challenges related to disease management due to disease-disease interactions, disease-drug interactions, and drug-drug interactions, which complicates decision-making and burdens patients and their caregivers.5,6
A decade ago, the American Heart Association (AHA), American College of Cardiology (ACC), and US Department of Health and Human Services (HHS) summarized data on common co-occurring conditions in patients with 4 cardiovascular conditions (ischemic heart disease, heart failure (HF), atrial fibrillation (AF), and stroke), and provided recommendations to enhance clinical practice guidelines for patients with cardiovascular disease and multimorbidity. 9 The most common chronic conditions in patients with cardiovascular disease are hypertension (occurring in 81-89% of patients) and hyperlipidemia (63-70% of patients), with diabetes, anemia, arthritis, and chronic kidney disease also among the top most common comorbidities. 9 While clinical practice guidelines have historically focused on single diseases and provided limited recommendations specific to comorbid conditions,9–11 recent guidelines for patients with cardiovascular disease have increasingly incorporated recommendations for patients with comorbid conditions. The AHA/ACC/Heart Rhythm Society (HRS) guidelines for the management of AF include special considerations for management of AF in patients with HF, specific anticoagulation recommendations for AF complicating acute coronary syndromes or percutaneous coronary intervention, chronic coronary disease, peripheral artery disease, chronic kidney disease/kidney failure, and valvular heart disease, as well as management considerations for AF patients with obesity, Wolff-Parkinson-White and preexcitation syndromes, hypertrophic cardiomyopathy, adult congenital heart disease, hyperthyroidism, pulmonary disease, cancer, and in the setting of acute illness or surgery. 12 However, patients with AF may have multiple concomitant chronic conditions which need to be taken into account for their management. A better understanding of the most common combinations of chronic conditions in patients with AF may be used to design future studies targeting treatment strategies based on a patient’s clustering of comorbid conditions. Thus, the goal of the current study was to describe the patterns of clustering of chronic conditions in patients with AF and how these patterns differ from an age- and sex-matched cohort of patients without AF from a large geographically-defined region in the Midwest area of the United States.
Methods
Study population
This study was conducted using the expanded Rochester Epidemiology Project (E-REP) medical records-linkage system. 13 The E-REP captures electronic medical record data from the population residing within a 27-county region of Southern Minnesota (19 counties) and Western Wisconsin (8 counties). Data are available starting in 2010 and include demographics, diagnostic and procedure codes, laboratory results, lifestyle measures such as smoking status, height, weight, body mass index (BMI), blood pressure, outpatient prescriptions, healthcare utilization including hospitalizations and emergency department visits, and dates and causes of death. In 2015, the E-REP captured electronic medical record data for 751,529 persons (65.9% of the population) residing in the 27-county region. In addition, the distributions of age, sex, race, ethnicity, and education are similar for the E-REP compared to the US Census 27-county population as well as the US Census Upper Midwest population. 13 This study was carried out in accordance with the Declaration of Helsinki, and was approved by the Mayo Clinic (22-003812) and Olmsted Medical Center (010-OMC-22) Institutional Review Boards. The study was considered minimal risk by both Institutional Review Boards; therefore, the requirement for informed consent was waived. However, records of any patient who had not provided authorization for their medical records to be used for research, as per Minnesota statute 144.335, were not reviewed.
Atrial fibrillation cases and controls
A cohort of new-onset AF between January 1, 2013 and December 31, 2017 was assembled. Data were available starting in 2010, but we restricted our cohort to 2013 forward to reduce the likelihood of including prevalent AF and to allow sufficient look back to define comorbid chronic conditions. To assemble our cohort, diagnostic codes for AF or atrial flutter (International Classification of Diseases, Ninth Revision (ICD-9) code 427.3 and International Classification of Diseases, Tenth Revision (ICD-10) code I48) from inpatient and outpatient encounters among adults (≥18 years) were obtained. Electronic interpretations of electrocardiograms (ECG) and Holter monitor reports were available from Mayo Clinic and Mayo Clinic Health System. ECG and Holter reports indicating possible, probable, likely, or cannot rule out AF/atrial flutter were considered negative (no AF). Any remaining ECGs or Holter reports with missing or inconclusive interpretations to determine AF status were reviewed by trained nurse abstractors. In addition, Current Procedural Terminology (CPT) codes for ECGs (CPT codes 93000 and 93010) and Holter monitors (CPT codes 93224-93227, 93230-93233, and 93236) were pulled for Olmsted Medical Center because an electronic ECG database was not available. The medical records were reviewed by trained nurse abstractors to determine the rhythm for all ECGs and Holter monitors using the same rules as previously described (excluding possible, probable, likely, and cannot rule out diagnoses).
We used the following rules to validate the diagnosis of AF and assign the incidence date of AF. We required a diagnostic code plus evidence of AF or atrial flutter on either an ECG or Holter monitor to confirm the diagnosis. Patients with ECG or Holter monitor evidence of AF within 30 days prior to 1 year after an AF diagnostic code were included. The diagnostic code date was assigned as the incident AF date. If AF was not confirmed at first diagnosis date, we used subsequent diagnoses and employed the same rules regarding timing and evidence of AF on an ECG or Holter monitor. If the diagnosis of AF was not confirmed by ECG or Holter, the patient was excluded. In addition, persons whose incidence date occurred prior to January 1, 2013 or who were non-residents of our region at the time of AF diagnosis were excluded. A flow diagram describing the AF cohort is provided in Supplemental Figure 1.
For each person with AF in our cohort, a single control from the same population was matched (individual matching, 1:1). Controls were matched to AF cases on age (within 5 years), sex, and county of residence. The case’s date of incident AF was used as the index date for the matched control. The matched controls had no evidence of AF prior to or on their index date, but were not excluded based on development of AF after their index date.
Ascertainment of chronic conditions
A total of 18 chronic conditions were ascertained by electronically retrieving diagnostic codes (ICD-9 and ICD-10) from inpatient and outpatient encounters at all providers indexed in the E-REP. The list of chronic conditions included those identified by the US Department of Health and Human Services (DHHS) for studying multimorbidity.14,15 The list contains 20 chronic conditions, but autism and human immunodeficiency virus were excluded due to low prevalence (<0.5% of the population), along with cardiac arrhythmias because all cases have AF. Although anxiety was not one of the DHHS 20 conditions, it was added because it is prevalent and a potentially important comorbid condition in patients with cardiovascular disease. The diagnostic codes used to define the 18 chronic conditions have been defined previously. 16 Diagnostic codes were retrieved for a 3 year capture frame prior to the date of incident AF (and corresponding index date in controls). A single diagnostic code in the code set for a given chronic condition was used to confirm the presence of the chronic condition.
Statistical analysis
Analyses were completed using SAS software, version 9.4 (SAS Institute) and R software, version 4.0.3 (R Foundation for Statistical Computing). Characteristics of patients with AF and controls without AF were reported as frequencies (percent) for categorical variables and means (standard deviation) or medians (interquartile range; IQR) for continuous variables. Demographic characteristics of patients with AF vs. controls were compared using standardized mean differences, calculated as the difference in means or proportions divided by the standard error. Differences in chronic conditions between patients with AF and controls were compared using conditional logistic regression.
To visualize patterns of chronic conditions in patients with AF and controls, heat maps were produced using the ‘pheatmap’ package in R, each row representing a patient and each column a chronic condition. The rows were ordered according to how patients clustered using agglomerative hierarchical clustering where each patient began as their own cluster, then the two clusters that are most similar were combined, and the process reiterated until one single cluster containing all patients was created. Distance between clusters was assessed using the Jaccard index due to the binary nature of the data, and the similarity of clusters was determined using complete-linkage. 17 For the presentation of results, the chronic conditions were ordered according to their clustering within the groups of cardiometabolic conditions (hypertension, hyperlipidemia, diabetes, coronary artery disease, congestive heart failure, stroke), other somatic conditions (arthritis, cancer, chronic kidney disease, chronic obstructive pulmonary disease, asthma, osteoporosis, hepatitis), and mental health conditions (depression, anxiety, substance abuse disorders, schizophrenia, dementia/delirium).
Next, for AF patients and controls separately, prevalences of each pair of chronic conditions were determined, and heat maps were constructed presenting pairwise prevalences of the chronic conditions. Heat maps were constructed overall and stratified by age group using the age of the AF case for the case and the matched control: <65, 65-74, 75-84, and ≥85 years. In addition, the asymmetric version of Somers’ D statistic was used to measure the degree of association between 2 chronic conditions accounting for coincidental comorbidity,18,19 overall and stratified by age groups. The Somers’ D statistic quantifies the non-random concordance of each pair of chronic conditions beyond chance (ranging from −1 [perfect disagreement] to 1 [perfect agreement] with 0 indicating by chance alone). Accounting for agreement by chance alone when measuring the degree of association is important as coincidental comorbidity by chance increases with higher prevalence. Significance for the Somer’s D statistic was evaluated using a permutation approach with the Benjamini–Hochberg procedure to control the false discovery rate at 0.1%. 20 The same ordering of chronic conditions (clustered by groups of cardiometabolic, other somatic, and mental health conditions) was used for the pairwise prevalence and co-occurrence heat maps.
Results
Distributions of chronic conditions in patients with atrial fibrillation and age- and sex-matched controls without atrial fibrillation.

All results are reported as n (%).
AF, atrial fibrillation; COPD, chronic obstructive pulmonary disease.
Odds ratios were estimated using unadjusted conditional logistic regression models.
The majority of patients with AF had other chronic conditions, with 90.1% having 2 or more of the 18 other chronic conditions and 81.9% having 3 or more chronic conditions. Very few patients with AF (4%) did not have any of the 18 other chronic conditions (Supplemental Figure 2). However, nearly one-quarter of controls (23%) did not have any chronic conditions, 66.8% had 2 or more chronic conditions, and 54.9% had 3 or more chronic conditions. Heat maps for patients with AF and controls were constructed to visualize how chronic conditions clustered together (Figure 1). Cardiometabolic conditions, especially hypertension, hyperlipidemia, and diabetes, clustered together in both patients with AF and matched controls without AF. However, clustering of additional cardiometabolic conditions was more common in patients with AF. In addition, clustering of other somatic conditions was more common in patients with AF. Distribution of chronic conditions in patients with atrial fibrillation and age- and sex-matched controls without atrial fibrillation. Left panel, patients with atrial fibrillation. Right panel, age- and sex-matched controls without atrial fibrillation. Each patient is represented by a row in the heat map. Patients are clustered according to their chronic conditions. Purple indicates presence of the condition while yellow indicates absence of the condition. The chronic conditions were ordered according to their clustering within groups of cardiometabolic, other somatic, and mental health conditions. CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease.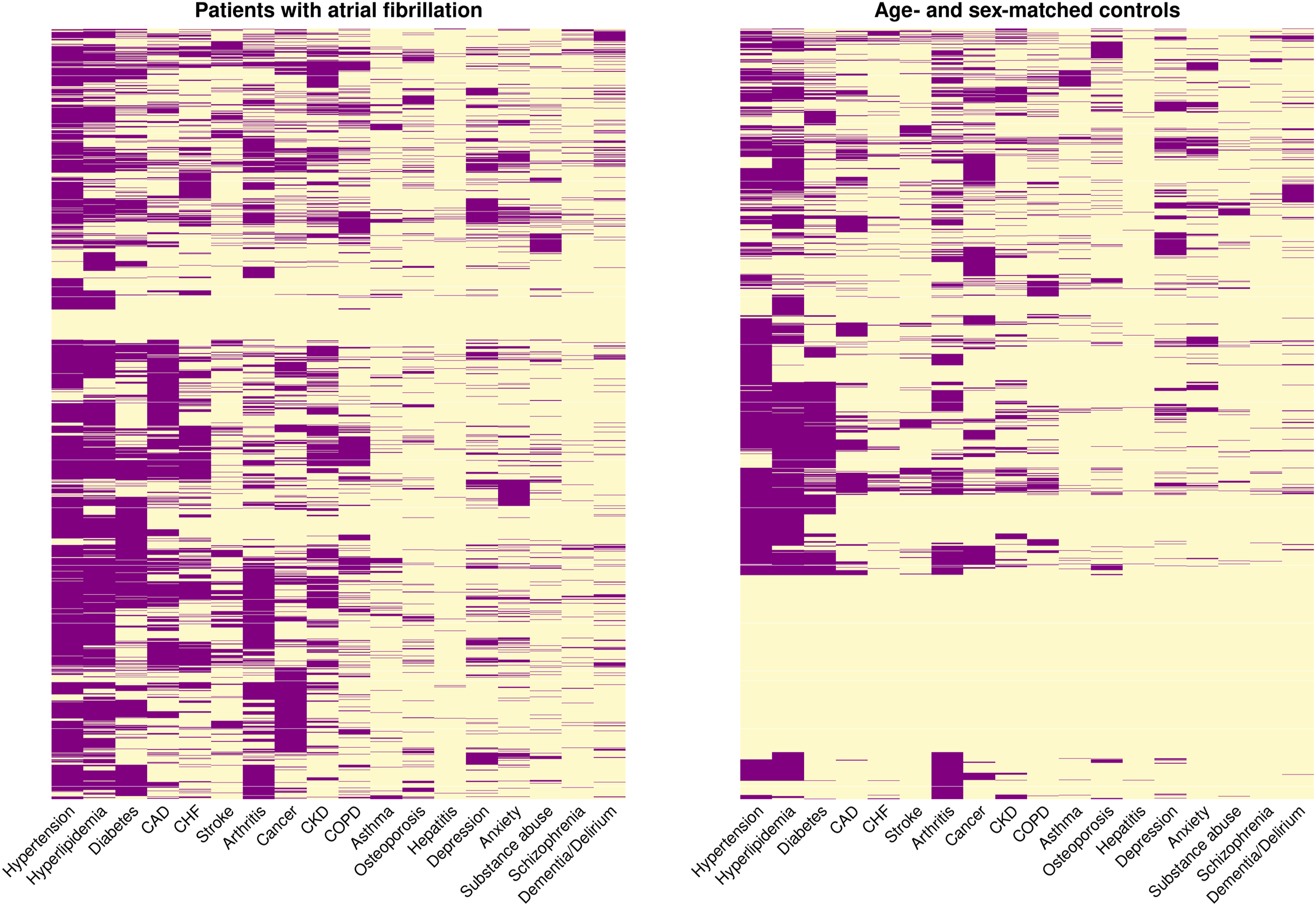
The most common pairs of chronic conditions in both patients with AF and their matched controls included pairs of cardiometabolic conditions such as hypertension/hyperlipidemia (in 60.4% of patients with AF and 40.8% of controls) and hypertension/diabetes (40.4% in patients with AF and 24.8% in controls; Supplemental Figure 3). Arthritis paired with hypertension or hyperlipidemia was also common in both patients with AF and controls, as well as coronary artery disease paired with hypertension or hyperlipidemia. Chronic kidney disease paired with hypertension and hyperlipidemia were also among the top 10 chronic condition pairs in patients with AF, but was not among the top 10 condition pairs in the controls.
The prevalence of most condition pairs was higher in AF patients compared to controls. However, when examining the concordance of condition pairs beyond chance, controls had a larger number of condition pairs occurring together beyond chance compared to patients with AF (Figure 2). Condition pairs where both conditions were cardiometabolic and where both conditions were mental health had highest concordance in both patients with AF and in matched controls without AF. For example, hypertension and hyperlipidemia commonly occurred together beyond chance in both patients with AF and controls. In addition, all other chronic conditions occurred together with hypertension more commonly than chance in controls, whereas in patients with AF, some of the other somatic and mental health conditions did not occur together with hypertension beyond chance. Co-occurrence of chronic conditions in patients with atrial fibrillation and age- and sex-matched controls without atrial fibrillation. Each plot includes patients with atrial fibrillation (upper right side of the plot) compared to age- and sex-matched controls without atrial fibrillation (lower left side of the plot). Left panel, prevalence of condition pairs. Numbers in the boxes correspond to the proportion of patients with both chronic conditions. Right panel, concordance of condition pairs beyond chance. An asterisk (*) indicates the asymmetric Somers’ D statistic met statistical significance controlling for the false discovery rate at 0.1%. CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease.
When stratified by age group (<65, 65-74, 75-48, and ≥85 years), the prevalence of most condition pairs was higher in patients with AF compared to controls without AF for all age groups (Figure 3). However, differences in the concordance of condition pairs beyond chance were observed between patients with AF and controls without AF, with the most pronounced differences in the youngest two age groups (<65 and 65-74 years; Figure 4). In persons aged <65 years, similar concordance of cardiometabolic condition pairs and mental health condition pairs were observed in patients with AF and controls. However, patients with AF more frequently exhibited concordance of conditions pairs that included either pairs of other somatic conditions or a combination of conditions from different condition groups. In persons aged 65-74 years, patients with AF more frequently had pairs of other somatic conditions that occurred together beyond chance compared to controls without AF. In the oldest two age groups (75-84 and ≥85 years), fewer condition pairs occurred together beyond chance and the co-occurrence of condition pairs was generally similar in patients with AF and controls. Prevalence of chronic condition pairs in patients with atrial fibrillation and age- and sex-matched controls without atrial fibrillation by age group. Each plot includes patients with atrial fibrillation (upper right side of the plot) compared to age- and sex-matched controls without atrial fibrillation (lower left side of the plot). Numbers in the boxes correspond to the proportion of patients with both chronic conditions. Top left panel, ages <65 years; top right panel, ages 65-74 years; bottom left panel, ages 75-84 years, bottom right panel, ages ≥85 years. CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease. Concordance of chronic condition pairs in patients with atrial fibrillation and age- and sex-matched controls without atrial fibrillation by age group. Each plot includes patients with atrial fibrillation (upper right side of the plot) compared to age- and sex-matched controls without atrial fibrillation (lower left side of the plot). An asterisk (*) indicates the asymmetric Somers’ D statistic met statistical significance controlling for the false discovery rate at 0.1%. Top left panel, ages <65 years; top right panel, ages 65-74 years; bottom left panel, ages 75-84 years, bottom right panel, ages ≥85 years. CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease.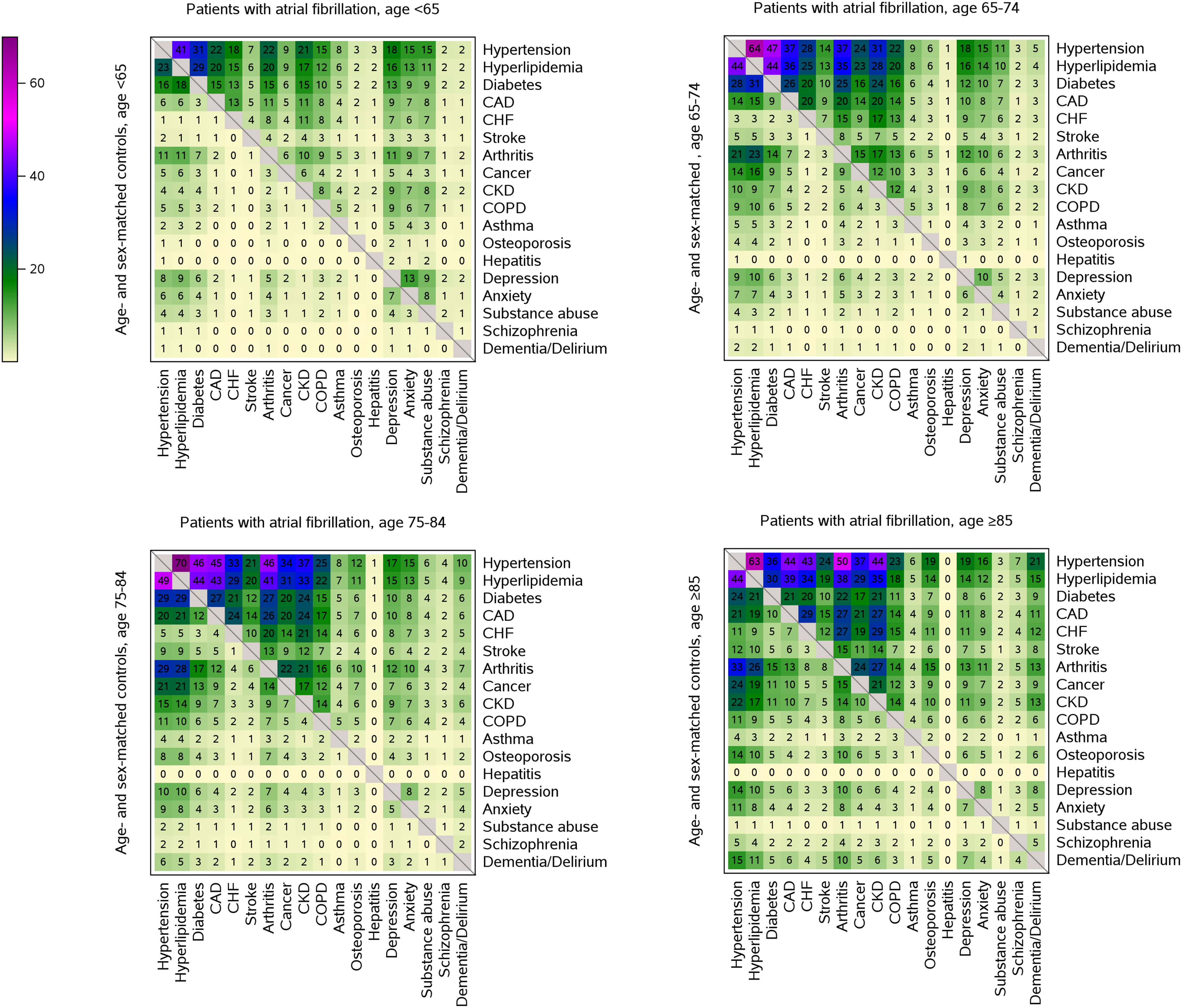

Discussion
In this large study of more than 16,500 patients with new-onset AF matched to patients without AF from the same community, the prevalence of each of the 18 chronic conditions was higher in AF patients compared to matched controls. Only 4% of patients with AF did not have any of the 18 chronic conditions, whereas nearly one-quarter of controls (23%) did not have any chronic conditions. Differences in clustering of chronic conditions were observed between patients with AF and controls, particularly clustering of the cardiometabolic conditions of coronary artery disease, congestive heart failure, and stroke, as well as clustering of other somatic conditions being more common in patients with AF. For pairs of chronic conditions, differences were observed between patients with AF and controls in those <75 years only, with more frequent co-occurrence of pairs of other somatic conditions in patients with AF compared to controls.
Patients with AF experience greater risks of adverse outcomes, including stroke, heart failure, hospitalization, and death, compared with patients without AF.21–28 In addition to AF conferring an excess risk of non-fatal and fatal outcomes, patients with AF are frequently older and have multimorbidity, which may contribute to challenges with disease management and poor outcomes. Indeed, a higher number of comorbidities is associated with lower rates of anticoagulation in patients with AF even though multimorbid patients may potentially benefit most from anticoagulation therapy.29–31 Furthermore, both cardiovascular and non-cardiovascular comorbidities contribute to excess risk of stroke, heart failure, hospitalization, and death in patients with AF.21,25,32–34 In the current study, some of the most frequently occurring chronic conditions which commonly clustered together, such as hypertension, diabetes, and coronary artery disease, are well known risk factors for adverse outcomes in AF including stroke.
The majority of patients with AF have multimorbidity. In nearly 85,000 persons with AF in the United Kingdom captured by the Clinical Practice Research Datalink, 60% of patients had at least 3 comorbidities (out of a list of 18 conditions) with trends of increasing comorbidity burden over time from 48% in 1998 to 68% in 2017. 35 These estimates are lower than the current study, which reported that 82% of patients with AF had 3 or more additional chronic conditions. In a primary care registry network in Belgium including 1830 patients with AF matched to 6622 controls without AF, the number of comorbidities was higher in patients with AF compared to controls (mean modified Charlson Comorbidity Index 5 vs. 4). 36 Patients with AF had a higher prevalence of many chronic conditions. However, similar prevalence of dementia, chronic pulmonary disease, and cancers (history of tumors, leukemia, and lymphoma) were observed in patients with AF and controls, which is discrepant with our reported findings herein of higher prevalence of dementia, chronic obstructive pulmonary disease, and cancer in patients with AF.
Recent studies have also assessed clustering of conditions in patients with AF. In a single center registry of 9401 patients with AF in an intensive care cohort, 4 clusters were observed. 37 A first cluster consisted of the youngest, highest proportion male, and highest proportion with postoperative conditions (74%) and hypertension (64%; postoperative hypertensive cluster), the second was characterized by the highest proportions of chronic obstructive pulmonary disease (17%) and diabetes (26%; non-cardiovascular multimorbid cluster), a third included the oldest with the highest prevalence of heart failure (86%; cardiovascular multimorbidity cluster), and the fourth included the largest proportion of valvulopathies (45%; valvulopathy atrial dilation cluster). In a 1% Medicare sample from 2015-2016, 4 clusters of non-cardiovascular conditions were observed. 38 One cluster had the lowest prevalence of most conditions (minimal cluster), a second was characterized by >40% prevalence of depression and chronic lung disease along with high prevalence of anemia (76%) and musculoskeletal disease (94%; depression-lung cluster), a third included a high proportion of chronic kidney disease (83%) and diabetes (67%; chronic kidney disease-diabetes cluster), and the last included the highest prevalence of all conditions (multi-system cluster). In the Fushimi AF Registry, 6 clusters of AF patients were observed including (1) the youngest with lowest prevalence of comorbidities, (2) the elderly with low prevalence of comorbidities, (3) those with high prevalence of atherosclerotic risk factors but without atherosclerotic disease, (4) those with atherosclerotic comorbidities, (5) those with history of stroke, and (6) the very elderly. 39
However, these aforementioned studies lacked a comparison of patients with AF to controls without AF. The current study provides a unique perspective on multimorbidity in AF by comparing combinations of conditions in patients with AF to controls without AF. Importantly, our study was population-based and did not impose any restrictions based on patient age, setting of diagnosis (outpatient, inpatient), or insurance coverage. In addition, we estimated whether combinations of 2 conditions occurred together more frequently than expected, providing insight as to which combinations of conditions are more likely to occur together in AF compared with controls. Finally, we stratified our results by age and observed that clustering of other somatic conditions was more common in patients with AF, in particular among patients diagnosed at younger ages (<75 years). In contrast, co-occurrence of pairs of conditions was more similar for patients with AF and controls in those 75 years of age and older.
Implications
Our study has characterized multimorbidity in patients with AF, describing clustering of conditions and co-occurrence of pairs of chronic conditions, and how these patterns differ from persons who do not have AF. Although clinical guidelines for AF have increasingly included recommendations for management of AF in patients with comorbid conditions,12,40,41 these recommendations have been generally limited to considerations for patients with AF and a single additional comorbid condition. However, as described herein, patients with AF often have multiple concomitant chronic conditions which may complicate their management. Our study has characterized multimorbidity in patients with AF in a unique, population-based setting providing further information that can be used to design studies targeting treatment approaches based on clustering of comorbid conditions. Clustering of cardiometabolic conditions was common, but in younger patients diagnosed with AF, pairs of other somatic conditions occurred more frequently together beyond chance than in patients without AF. Thus, our findings from a large community cohort emphasize that in addition to cardiometabolic conditions, other non-cardiovascular conditions, which may not be directly related to the pathophysiology of AF, are important to consider for the patients’ management. The results of our findings provide needed context for AF treatment guidelines, which emphasize the importance of comprehensive care of the multiple conditions that increase the risk of AF, are exacerbated by AF, or contribute to outcomes in patients with AF. Furthermore, in order to better inform treatment recommendations for AF patients with multimorbidity, additional research is needed to determine which specific combinations of conditions impart worse prognosis in patients with AF. Such information can guide future studies targeting treatment strategies based on a patient’s clustering of comorbid conditions.
Limitations and strengths
We acknowledge the following limitations. First, the rules we used to identify patients with AF, requiring a diagnostic code plus evidence of AF on an ECG or Holter monitor, likely resulted in missing some patients in our community who have AF. In addition, patients with ECG or Holter reports indicating possible, probable, likely, or cannot rule out AF were not considered as having AF unless there was another ECG or Holter indicating AF. However, these rules were chosen to optimize the likelihood that patients in the AF group truly had AF (high positive predictive value), given results of a prior study that reported code-based algorithms to identify AF are prone to misclassification. 42 Second, because AF can be asymptomatic, symptoms may be non-specific, and some patients may not seek care for their symptoms, it is possible that some matched community controls may have had undiagnosed AF and were misclassified as controls. Third, there may have been some misclassification of the chronic conditions due to errors in coding or assignment of codes for rule out of diagnoses. We required only a single diagnosis code to ascertain the chronic conditions, which while optimizing sensitivity, 43 may have resulted in incorrectly classifying some patients as having chronic conditions who did not. However, it is unlikely differences in the misclassification would have been observed between patients with AF and controls. Fourth, we lacked information on severity of the chronic conditions, duration of the conditions, and treatment for the chronic conditions. Finally, although the E-REP population is similar to the Upper Midwest population, the E-REP includes a lower proportion of non-Whites and Hispanics than the US as a whole. 13 Thus, our results may not be generalizable to all patient populations because our Midwest population included mostly persons of White race. Nevertheless, our study has many strengths including the capture of data from multiple health care providers through the REP records-linkage system, the large sample size allowing for stratification of our results by age, and the ability to compare multimorbidity patterns to similarly aged community controls without AF.
Conclusions
In this matched study of patients with new-onset AF compared to community controls without AF, differences were observed in clustering of chronic conditions between AF and controls. Patients with AF more commonly had clustering of the cardiometabolic conditions of coronary artery disease, congestive heart failure, and stroke, as well as clustering of other somatic conditions. In addition, differences were more pronounced in patients younger than 75 years of age, with more frequent concordance of pairs of other somatic conditions in patients with AF compared to controls. A better understanding of the clinical consequences of multimorbidity in AF patients, including those diagnosed at younger ages, is needed.
Supplemental Material
Supplemental Material - Multimorbidity in patients with atrial fibrillation and community controls: A population-based study
Supplemental Material for Multimorbidity in patients with atrial fibrillation and community controls: A population-based study by Alanna M. Chamberlain, Alvaro Alonso, Peter A. Noseworthy, Konstantinos C. Siontis, Bernard J. Gersh, Jill M. Killian, Susan A. Weston, Lisa E. Vaughan, Sheila M. Manemann, Véronique L. Roger, and Euijung Ryu in Journal of Multimorbidity and Comorbidity
Footnotes
Acknowledgements
We thank Deborah S. Strain for her assistance in formatting the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Aging [grant number AG 062580], and used the resources of the Rochester Epidemiology Project medical records-linkage system, which is supported by the National Institute on Aging [grant number AG 058738], by the Mayo Clinic Research Committee, and by fees paid annually by REP users. The funding sources played no role in the design of the study, the analysis, the interpretation of study results, or the writing of the manuscript.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
