Abstract
Background
Despite the great individual and societal burden associated with multimorbidity, little is known about how to effectively manage it.
Objective
The aim of this multicenter randomized controlled trial (RCT) is to investigate the 12-month effects of a personalized exercise therapy and self-management support program in addition to usual care in people with multimorbidity.
Design
This is a protocol for a pragmatic, parallel-group (1:1 ratio), superiority RCT conducted at five intervention sites (two hospitals, a private practice physiotherapy clinic and two municipal rehabilitation centers) in Region Zealand, Denmark. A total of 228 persons with multimorbidity aged 18 years or older, will be randomly allocated to one of two groups. Both groups will receive usual care, defined as routine care for multimorbidity at the discretion of the treating doctor, while the intervention group will also participate in a 12-week exercise therapy and self-management support program tailored to people with multimorbidity at one of the intervention sites. The primary outcome will be the between-group difference in change in EQ-5D-5L from baseline to the follow-up at 12 months. Secondary outcomes include objectively-measured physical function and physical activity, inflammatory markers, disease and treatment burden, anxiety, depression, stress, sleep, pain and other self-reported parameters. In parallel with the RCT, an observational cohort will follow persons aged ≥18 years with multimorbidity not adhering to all eligibility criteria, as well as people fulfilling all eligibility criteria, but unwilling to participate in the RCT. This study was approved by the Regional Committee on Health Research Ethics for Region Zealand (SJ-857) and results will be communicated in scientific papers, at relevant conferences and to a broader audience.
Discussion
Exercise therapy and self-management support is safe and effective in people with single conditions. However, it is still unclear whether this holds true for individuals with multimorbidity. This pragmatic, multicenter RCT will provide high-quality evidence on the benefits and harms of exercise therapy and self-management support and, if the results support it, lead to the development of a plan for implementation in clinical practice.
Background
Multimorbidity, often defined as the coexistence of two or more conditions in the same individual, 1 affects approximately one-third of the adult population with large variations in the estimated prevalence. 2 Multimorbidity is associated with decreased quality of life, physical and cognitive function and increased health-care utilization.1,3-9 As such, people living with multimorbidity account for 78% of all consultations in primary care 10 and are more likely to be admitted to hospital, have an increased length of stay and risk of premature death as compared to those with only one condition.11,12 Altogether, this highlights the burden of multimorbidity for both the individual and society.
Although more than half of people with any chronic condition have multimorbidity, 13 most research on chronic conditions so far has excluded multimorbidity,14,15 and large parts of the health-care sector treat each individual condition on its own instead of person-centered care, 1 thus highlighting the negative impact of the current disease-based curative models on multimorbidity. 1 With an ageing population, and a significant number of people with multimorbidity younger than 65 years of age, 13 the proportion of people with multimorbidity is increasing rapidly.1,16 This emphasizes the need to take action to deal with the increasing burden of multimorbidity through prevention, treatment and facilitation of self-management.
Unfortunately, despite the considerable burden of multimorbidity, little is known about how to effectively manage it.1,17 A promising management option is exercise therapy, which addresses functional limitations and is able to improve symptoms in people with chronic conditions. 18 Exercise therapy is effective, safe and supported by substantial evidence in at least 26 different chronic conditions. 18 This includes knee or hip osteoarthritis (OA),19,20 chronic obstructive pulmonary disease (COPD), 21 heart failure (HF) or coronary heart disease (CHD),22,23 hypertension, 24 type 2 diabetes mellitus (T2DM), 25 and depression, 26 which are among the leading causes of global disability, affect several hundred million people around the world and often co-exist. 16 Although evidence on the effects of exercise therapy in multimorbidity is limited, a recent systematic review found that exercise appears safe and effective in people with multimorbidity. 27 Furthermore, self-management is increasingly recognized as an essential component of intervention to improve outcomes and reduce health-care utilization.1,28 Self-management support focuses on enabling persons to develop a set of behavioral skills and abilities to help them navigate a range of disease management tasks across different chronic conditions. 28 However, evidence of the effect in multimorbidity is sharse. 17
Overall, this highlights exercise therapy and self-management support as viable treatment options for people with multimorbidity, but the quality of the evidence supporting this is of low quality and high-quality randomized controlled trials (RCT) on different combinations of chronic conditions are needed. 27 This is also reflected in clinical guidelines for multimorbidity that do not have specific recommendations on exercise therapy. 29
The aim of this RCT is to investigate the effects of a personalized exercise therapy and self-management support in addition to usual care, on self-reported, objectively measured and physiological outcomes in people with multimorbidity (i.e. at least two of the following diseases: knee and hip OA, COPD, heart disease (HF or CHD), hypertension, T2DM, depression). We hypothesize that a personalized exercise therapy and self-management program in addition to usual care is superior to usual care in improving quality of life at the 12 months follow-up.
Methods
Study design
This study protocol (version 1.6, 10 August, 2022) describes the design of a pragmatic, assessor-blinded, multicenter, parallel-group RCT (1:1 ratio) conducted at five intervention sites across the health care Region of Zealand, one of five health-care regions in Denmark.
The RCT was approved by the Regional Committee on Health Research Ethics for Region Zealand (SJ-857) on May 28, 2020 and is conducted in agreement with the Helsinki Declaration. Informed consent material is available in Danish with the approved protocol. If a participant sustains any trial-related harm, he or she will be covered by Danish law. Furthermore, the RCT was approved by the Danish Data Protection Agency of the Region of Zealand (REG-015-2020) and was pre-registered at ClinicalTrials.gov (NCT04645732). The current report adheres to the SPIRIT statement. 30
Participants
A total of 228 participants with multimorbidity fulfilling the eligibility criteria listed below will be included in the RCT: Inclusion criteria 1. At least two of the following conditions: knee or hip OA, COPD, heart disease (HF or CHD), hypertension, T2DM, depression (having other comorbidities does not exclude a person from participating) 2. Adults aged 18 years or above 3. Ability to walk 3 meters without assistance 4. A score of at least 3 on the Bayliss Disease Burden: Morbidity Assessment by Self-Report scale
31
for at least one of the conditions listed above and a score of ≥ 2 for at least one of the other listed conditions 5. Willingness and ability to participate in a 12-week supervised exercise therapy- and self-management program twice a week Exclusion criteria 1. Participation in supervised systematic exercise for one of their diseases within the last 3 months 2. Persons with an unstable health condition or at risk of serious adverse events as assessed by a medical specialist 3. Terminal patients and persons with a life expectancy of less than 12 months 4. Persons categorized as Class IV on the New York Heart Association (NYHA) Functional Classification scale 5. Persons with psychosis disorders, post-traumatic stressdisorder, obsessive compulsive disorder, attentiondeficit hyperactivity disorder, autism, anorexianervosa/bulimia nervosa and/or persons with dependency disorders 6. Other reasons for exclusion (unable to understandDanish, mentally unable to participate, etc.)
Recruitment procedure
The overall trial flow is outlined in Figure 1. Trial flow. Abbreviations: OA: osteoarthritis; COPD: chronic obstructive pulmonary disease; HF: heart failure; CHD: coronary heart disease; T2DM; type 2 diabetes mellitus; RCT: Randomized, controlled trial. *aged 18 years or above, at least two of the relevant conditions and not having psychosis disorders, post-traumatic stress disorder, Obsessive Compulsive Disorder, attention deficit hyperactivity disorder, autism, anorexia nervosa/bulimia nervosa or a dependency disorder or Other reasons for exclusion.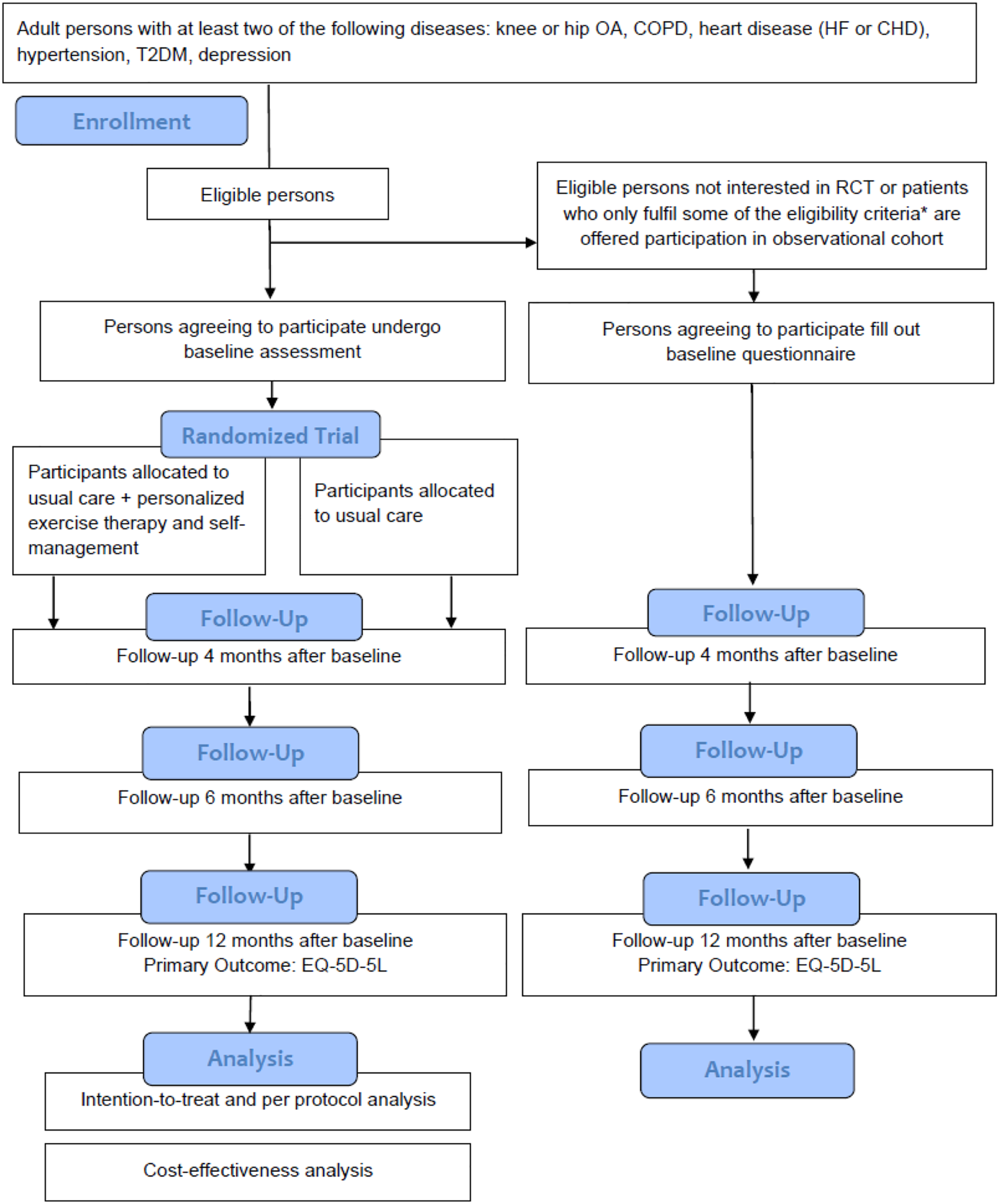
The recruitment procedure was adapted and improved through a systematic review performed in MOBILIZE to identify strategies to improve recruitment and retention in exercise trials for people with multimorbidity. 33 Additionally, we consulted the latest Cochrane systematic review on recruitment and retention.32,34
Participants will be recruited from six general practitioners, three psychiatric facilities, and seven hospital departments in the Region of Zealand, Denmark, through direct consultations, Facebook ads and local newspaper articles as well as other advertisement and posters/handouts. Recruitment began in November 2021.
Persons visiting one of the recruitment sites and who comply with the eligibility criteria are invited to participate in the RCT. Oral information about the study will be provided by a local health professional in an undisturbed room during or right after a usual care consultation or via telephone. Staff at the recruitment sites have been instructed in the recruitment procedure to maximize the recruitment rate, following evidence-based strategies.33–34 Patient lists will also be screened for eligible participants, who will be contacted by phone. Persons showing interest are referred to the MOBILIZE project team and a member of the project team will subsequently contact them to determine final inclusion.
Self-referrals who have seen the Facebook posts or newspaper articles/advertisements will be contacted by a member of the project team by phone with thorough information about the study and to determine eligibility for study enrollment. A MOBILIZE-associated medical specialist will assess the exclusion criterion regarding unstable health conditions and risk of serious adverse events for all self-referrals.
Potential participants will receive written information via e-mail or by post and are recommended to take at least 24 hours to consider and discuss their potential enrolment with others (e.g. spouse, relatives or similar). Following oral agreement to participate, a written informed consent will be obtained by study personnel prior to study enrollment.
Blinding
Only outcome assessors and the statistician will be blinded to randomization. Furthermore, a blinded interpretation of the study results will be conducted. 35
Randomization procedure
People willing to participate and fulfilling the eligibility criteria will be randomized (1:1 allocation ratio) after baseline assessment. A priori, an independent statistician have prepared a computer-generated randomization schedule in randomized, permuted blocks of four or six persons stratified by the number of chronic conditions the individual suffers from (2 or 3+) and recruitment centres (hospitals, general practitioners and self-referrals). Allocation numbers will be concealed in opaque sealed envelopes, which are only accessible by a study coordinator opening them after informed consent and baseline assessment have been completed.
Observational cohort
People who only fulfil inclusion criteria 1 and 2 and do not fulfil exclusion criteria 5 or 6 and those fulfilling all eligibility criteria, but unwilling to participate in the RCT, will be offered participation in an observational cohort study where only the self-reported outcomes are completed. They will be contacted by a member of the project team and receive a separate participant information sheet and consent form. Provided they agree, written consent will be obtained. This group will provide data that can be compared to the more extensively followed participants in the RCT and act as an observational cohort. Recruitment for observational cohort will proceed as long as we are recruiting for the RCT.
Interventions
Participants will be randomized to one of two treatments: a personalized exercise therapy and self-management program on top of usual care or usual care alone. The participants in both groups will continue their current treatment as provided by their general practitioner or specialist, including pharmacological treatment.
Personalized exercise therapy and self-management program
Participants randomized to the personalized exercise therapy and self-management program will twice weekly participate in a 12-week program tailored to people with multimorbidity at one of the intervention sites.
The program will consist of an individual introduction (60 minutes) with a physiotherapist followed by 24 self-management sessions (30 minutes/session) and 24 supervised exercise sessions (60 minutes/session).
There is increasing evidence that effective management of multimorbidity requires the patient´s active participation in the care process. 36 The self-management program in the current study was developed in close collaboration with persons affected by multimorbidity, patient partners, health-care providers, and other relevant stakeholders. It aims to equip the participants with better self-management skills and build motivation and capability to sustain an active life and better quality of life following the 12-week intervention. 37
The content of the exercise therapy program was guided by existing recommendations for exercise in people with chronic diseases and healthy adults and developed in close collaboration with experienced physical therapists, participants from the feasibility study, patient partners and other relevant stakeholders.37,38
Each exercise session includes five phases: • Warm up (8 minutes) • Balance exercises (5 minutes) • Strengthening exercises (20 minutes) • The participant’s choice: The participant can choose between more strengthening exercises, aerobic esxercise, or functional exercises (20 minutes) • Cool down (7 minutes)
The exercises are personalized based on 1-5 levels of difficulty. The strengthening exercises are performed with 10-15 repetitions in 2-3 sets following recommendations from the American College of Sports Medicine, 39 and progressed based on the ONMI scale. 40 The level of difficulty of the aerobic exercises is adjusted by intervals and progressed based on the Borg rating of Perceived Exertion Scale (a rating of 14-15). 41
A draft of the self-management and exercise therapy program was tested in a feasibility study with 20 persons with multimorbidity fulfilling the same eligibility criteria as for the RCT. 38 The feasibility study showed that self-management and exercise therapy was feasible in people with multimorbidity, and the intervention was found acceptable by both participants and physiotherapists. However, some amendments to increase adherence and improvement were done before proceeding to the RCT. 38
Details of the self-management and exercise therapy program*.

Described according to the Template for Intervention Description and Replication (TIDieR). 42
The self-management and exercise therapy program will be delivered at one of the intervention sites at the hospitals of Næstved and Slagelse, a private practice physiotherapy clinic in Holbæk and at rehabilitation centers in the municipalities of Roskilde and Lolland. All MOBILIZE physiotherapists will attend a specific one-day course to be certified to deliver the personalised treatment program
Attendance and progression of the exercises will be registered in a diary for the participants in the group randomized to self-management education and exercise therapy. Adherence is considered satisfactory if participants attend at least 18 out of the 24 (75%) proposed self-management and exercise sessions, respectively. Enrolled persons participating in less than 18 sessions will be included in the intention-to-treat (ITT) analysis, but excluded from the per-protocol analysis due to insufficient adherence.
Usual care
Usual care is the care that the participants would receive had they not participated in the study, i.e. treatments or services that are provided routinely in the settings from which the participants are recruited. Participants will continue their current treatment, if needed, and receive supplementary treatments if their general practitioner or specialist find it relevant for their comorbidities. The Danish healthcare system is mainly tax-financed and based on the egalitarian principle of equal healthcare access for equal healthcare needs. Clinic visits are free of charge and expenditures for prescribed drugs are partly reimbursed and to some degree also expenditures for e.g. physiotherapy. In Denmark, the general practitioner usually provides diagnosis and routine care for people suffering from the most common chronic conditions and functions as a gatekeeper for specialist care.
Data collection procedure and outcomes
Overview of measures and outcomes.

Abbreviations: cm: centimeters; FVC: Forced Expiratory Volume; FEV1: Forced Expired Volume in the first second; FEV1%: Percent predicted of Forced Expired Volume in the first second; Nm: Newton meters; kg: kilograms; HDL: High Density Lipoproteins-cholesterol; LDL: Low Density Lipoprotein-cholesterol; HbA1c: hemoglobin A1c; CRP: C-reactive protein; TNF: tumor necrosis factor; IL-6: Interleukin 6; VAS: Visual Analogue Scale; WHO: World Health Organization.
aEvaluated using combined wearable thigh and wrist accelerometers (Axivity AX3).
Participant characteristics
Participant characteristics such as educational level, work status, marital status, smoking habits, address place of residence and if living alone or with others will be evaluated by self-report., Age and sex will be extracted from the participants social security number (CPR), height will be measured and lung function will be evaluated at baseline using the Medikro Pro spirometer (Medikro Oy, Kuopio, Finland).
Outcomes
Primary outcome
The primary outcome is the descriptive index of the EQ-5D-5L questionnaire (5-level version, ranging from -0.624 to 1, higher is better) at 12 months. Itis a reliable and valid measure of general health/quality of life 44 and will be self-reported at baseline, 4, 6 and 12 months.
Secondary outcomes
Secondary outcomes will also be reported in the primary paper with the 12-month follow-up as the primary endpoint unless stated otherwise. These include objectively measured outcomes as well as self-reported measures (Table 2).
Objectively measured outcomes
Functional performance will be assessed by two frequently used tests, both performed at baseline and at follow-up at 4-and 12 months. The 6-minute walk test is an extensively used valid and reliable measure of functional capacity, 45 also in trials of people with multimorbidity. 27 The 30-second chair-stand test is a commonly used, valid and reliable measure of lower extremity strength and endurance in older adults. 46
Objectively measured physical activity and sleep will be measured at baseline, and at the 4- 12 months follow-up using two Axivity AX3 (Axivity, Newcastle UK) accelerometers placed on the front of the right thigh and the wrist of the non-dominant. Physical activity intensity are generated using ActiGraph counts 47 and step counts using a recently developed algorithm. 48 Sleep parameters are estimated with the GGIR package based on the wrist acceleration using the method proposed by Van Hees et al. 49 Participants are instructed to wear the accelerometers for seven consecutive days (24 hours/day), including during sleep and any land-based or water activity. A threshold of minimum 22 hours of wear time out of 24 hours/day in at least 3 weekdays and 1 weekend will be considered valid. 50 Non-wear is determined using both acceleration and temperature. 51 To calculate total daily physical activity, data will be weighted by 5/7 for weekdays and 2/7 for weekends.
The following physical activity and sleep entities will be evaluated: - Steps per day (Secondary outcome) - Minutes/day spent being physically active with at least light intensity (Secondary outcome) - Sedentary activity: Time spent on sedentary activities (Other outcome) - Light physical activity: Time spent on light intensity activities (Other outcome) - Moderate physical activity: Time spent on moderate intensity activities (Other outcome) - Vigorous physical activity: Time spent on vigorous intensity activities (Other outcome) - Moderate to vigorous physical activity: Time spent on moderate to vigorous intensity activities (Other outcome) - Adherence to the World Health Organization (WHO) physical activity recommendations, i.e. ≥150 min. MVPA or ≥75 min. VPA weekly (Other outcome) - Sleep (Other outcome)
Self-reported measures and other measures
Participants will self-report secondary outcomes via online questionnaires. The outcomes used to collect such data from the participants are listed in Table 2 and will be collected at baseline, 4, 6 and 12 months unless stated otherwise.
Burden of illness and current treatment for the individual conditions will be measured using the Bayliss burden of illness measure that assesses how much illness affects the individual's daily activities (on a 1-5 scale for each individual condition, summed to a total score for all conditions (higher is worse)).
The Personal Health Questionnaire Depression Scale (PHQ-8, ranging from 0-24 points, higher is more severe depression), excluding the ninth PHQ item on self-harm, and the General Anxiety Disorder-7 (GAD-7, ranging from 0-21, higher is more severe anxiety) are valid and reliable measures of depression and anxiety in the population that is sensitive to change.52,53
Self-Efficacy for Managing Chronic Disease is a 6-item scale (ranging from 1 to 10, higher scores indicating higher self-efficacy) that is a reliable and valid measure of self-efficacy. 54
Overall functioning and disability will be measured using the 12-item WHO Disability Assessment Schedule (WHODAS 2.0, ranging from 0 (no disability) to 100 (full disability). The WHODAS 2.0 has been found valid and reliable in the general population and in people with a range of chronic conditions.55,56
Participants will be asked to self-report adverse events (AE) and serious adverse events (SAE). Furthermore, medical records will be inspected for any AEs and SAEs occurring during the follow-up period. An AE is defined as any undesirable experience during follow-up leading to contact with the health-care system. If an AE results in death, hospitalization, prolonged inpatient hospital care, permanent disability or damage, or if an AE is life-threatening, it will be categorized as an SAE. 57 If available, date of healthcare system contact, duration and potential consequences of SAEs will be registered and reported.
The EQ-VAS of the EQ-5D-5L questionnaire (ranging from 0-100, higher is better) will be evaluated alongside the descriptive index as a measure of overall self-rated health. 44
Mortality during the 12-month follow-up period will be recorded, as mortality is one of the core outcomes in multimorbidity. 43
Other outcomes
Other outcomes will be reported in secondary publications and will be collected at baseline, 4, 6 and 12 months unless stated otherwise.
Objectively measured outcomes
Isometric knee-extension strength will be measured as the highest score out of three attempts using the EASYFORCE digital dynamometer (Meloq AB, Stockholm, Sweden) at baseline and at follow-up at 4 months and 12 months. The EasyForce dynamometer can be considered a reliable tool for assessing knee extension strength. 58
Isometric handgrip strength: will be performed using the Jamar Smart digital hand dynamometer (Patterson Medical, Warrenville, IL, USA) at baseline and at follow-up at 4 months and 12 months and will be measured as the highest score out of three attempts. The Jamar Smart digital hand dynamometer has been validated and has proven excellent inter-instrument reliability and strong concurrent validity. 59
Weight in kilograms (kg) will be assessed at baseline and at follow-up at 4 and 12 months using a digital floor scale.
The Guralnik 30-second balance test will be performed at baseline and at follow-up at 4 months and 12 months. It assesses balance in 30 seconds with 10 seconds in each of the 3 different positions (parallel feet, semi-tandem and tandem stand) resulting in a score from 0-30, higher is better. The 30-second balance test is reliable to assess balance. 60
Systolic and diastolic blood pressures after 5 minutes of rest will be measured as the mean of three consecutive measurements using the Rossmax X5 blood pressure monitor (Rossmax, Heerbrugg, Switzerland) at baseline and at follow-up at 4 months and 12 months.
Blood samples will be collected at the hospitals at baseline and follow-up at 4 months for measurement of inflammation, glycated haemoglobin, fasting plasma glucose and fasting insulin levels, cholesterol and triglyceride levels. All participants will provide written informed consent before blood samples are withdrawn. The samples will be stored at the local Departments of Clinical Biochemistry in Holbæk, Roskilde, Næstved, Slagelse, Nykøbing Falster as well as at Rigshospitalet in Copenhagen, prior to analysis.
Self-reported measures and other measures
The participants will self-report whether they have pain in their left or right knee or hip, and, in case of pain in several joint, which joint that is the most affected. Participants who answer yes to knee or hip pain at any assessment, will self-report pain intensity using the Visual Analogue Scale (VAS; ranging from 0-100, higher is more pain) for the most affected joint. The VAS scale is generally well accepted by patients and is reliable and validated. 61
Pain from the musculoskeletal system within the previous 24 hours will be self-reported on a region-divided body chart of the front and back sides of the body. Classification of pain using the number of self-reported body sites with pain is a well-known and supported approach to assess bodily pain. 62
Perceived Stress Scale (PSS) 4-item version is an easy-to-use questionnaire with established acceptable psychometric properties. 63 Scores on the PSS range from 0-16 with higher scores indicating higher perceived stress. 64
Adapted Fried Frailty and pre-frailty criteria is assessed on five domains (weight loss, exhaustion, grip strength, low physical activity, slow walking pace). 65 The adapted version of the Fried Frailty questionnaire has shown good validity.65,66 People with multimorbidity are deemed frail if they meet at least three criteria; pre-frail, if they fulfill one or two criteria, and not frail if no criteria are met.
Sleep quality will be assessed at baseline, 4, 6 and 12 months with the Modified Karolinska Sleep Questionnaire (4 items), 67 ranging from 4 to 24, lower is worse sleep quality. In addition to the accelerometer-based measurement, Sleep quantity will also be measured at baseline, 4, 6 and 12 months asking the patients “on a normal week, how many hours do you sleep on average per night in the weekdays? And “On a normal week, how many hours do you sleep on average per night in the weekend?”. These questions have been used in previous studies to assess sleep quantity. 68
Fatigue will be self-reported by one-item from the PROMIS Global Health short form. 69 The participants rate their fatigue on a 5-point scale from None (1) to Very severe (5) in response to the question “How would you rate your fatigue on average in the past 7 days?”
Global perceived effect (GPE) for physical activity level, activities of daily living and quality of life will also be reported. GPE has excellent test-retest reliability. 70 GPE will be assessed on a 7-point Likert scale ranging from (1) “Much better, an important improvement” to (7) “Much worse, an important deterioration”, higher score is worse. 70 The GPE scales provide reliable assessments of health transition. 70
Patient acceptable symptom state (PASS) will be assessed by asking the person “Thinking about your quality of life, would you consider your current health as satisfying? With quality of life, you should consider your activities of daily living, sport and recreational activities, personal care, level of pain, and any anxiety or depression.”. Answered by “yes” or “no”. The PASS has shown good validity and reliability. 71
Treatment failure will be assessed at the 4- and 12-months follow-up only by persons answering “no” to PASS.72,73 Treatment failure will be assessed with the question “Would you consider your current state as being so unsatisfactory that you consider the treatment to have failed?”. Answered by “yes” or “no”. 72
Adherence to intervention will be assessed at the 4-month follow-up as the number of exercise and self-management support sessions attended out of the total number of sessions available. Satisfactory adherence will be attained when completing 18 out of 24 sessions (75%).
Other treatment(s) during follow-up will be reported by asking the person about changes in their pharmacological treatment as well as any other treatments that they have received outside the study since the last assessment in the study. If the person answers yes, elaborative questions will be asked.
Furthermore, the Multimorbidity Treatment Burden Questionnaire (MTBQ 10-item version) will be collected. It has proven content validity, construct validity, reliability and responsiveness. 74 Answers range from 0-100, the higher scores being worse. 74
Also, the Short form of Patient Activation Measure (PAM) will be reported. The measure is both reliable and valid. 75 Answers range from 0-100, higher scores indicate higher levels of patient activation. PAM assesses patient self-reported knowledge, skills, and confidence in self-management of one's health or chronic condition and can provide insight into behavioural change. 75
One item on sedentary behavior (hours spent sitting per day) and two items on physical activity (minutes a week spent on physical activity causing shortness of breath and physical activity during activities of daily living) will be self-reported. 76
Individual items from the 12-item WHODAS 2.0 will be assessed with answers ranging from 1 to 5, higher score is worse.55,56
Falls will be assessed by asking the person “Have you fallen in the last 4 months? “2 months” at 6-month follow-up and “6 months” at the 12-month follow-up. A fall is defined as an event resulting in you inadvertently coming to rest on the ground or another lower level, with or without loss of consciousness or injury. Never; Once; Twice; Three times or more.
Fear of falling will be assessed by asking the person “How concerned are you that you might fall? Response categories include: Not at all concerned, somewhat concerned, fairly concerned, very concerned.”
Patient involvement
Patient involvement is crucial in the MOBILIZE project. Throughout all phases of the project at least two persons with multimorbidity and one partner to a patient with multimorbidity have been involved in important meetings and decisions in the project sharing their experiences, needs and preferences, corresponding to Collaborate level on the IAP2 Spectrum of Public Participation. 77
Data management
Data collection and management procedures have been approved by the Danish Data Protection Agency in Region Zealand (REG-015-2020). Secure, protected and approved electronic platforms will be used for data collection and data storage to ensure that missing data are kept to a minimum and that privacy and confidentiality of personal data are maintained.
Data monitoring and auditing
The study will not have a formal data monitoring committee, but the central trial management group (STS, MD, MN, AB) will closely monitor the trial and discuss any unforeseen circumstances or concerns and unexpected SAEs with the rest of the co-authors of the trial and involve external experts if needed. Additionally, the author group will monitor recruitment, treatment, and attrition rates.
The Danish Regional Committees on Health Research Ethics are regularly selecting a number of studies for auditing, a process independent of investigators and sponsors.
Adjudication committee
An independent adjudication committee comprising two to three members with prior adjudication experience will categorize AEs into serious or non-serious, based on the FDA criteria. 57 The committee will receive the adverse events in raw format after all participants have undergone the 12-month follow-up and independently conduct the adjudication followed by a consensus process within the committee. If needed, additional information will be requested from the hospitals.
Sample size calculation
The RCT study will be powered to detect a difference in change of 0.074 points between the two groups in the primary outcome (EQ-5D) from baseline to the follow-up at 12 months. This difference has previously been found to be the minimally important difference in persons with varying comorbidities. 78 To detect this difference, 95 participants in each intervention group are needed (assuming a common SD of 0.156, power=90%, alpha level=0.05). A total of 228 participants will be recruited to account for a potential loss to follow up of 20%, which has previously been involved in trials involving exercise therapy and self-management support.79,80
Statistical analysis
Between-group comparisons of change from baseline to the follow-up at 12 months in continuous outcomes (including the primary outcome) will be analysed using a repeated measures mixed-effects model with participants as random effect, visit (baseline, 4, 6, and 12 months) and treatment arm (Usual care, personalized exercise therapy and self-management program) as fixed effects, and with adjustment for randomization stratification factor, i.e. number of chronic conditions and recruitment centre.
Between-group comparisons of change from baseline to the follow-up at 4 months in continuous outcomes only evaluated at the 4-month follow-up will be analysed using analysis of covariance with adjustment for baseline scores in the dependent variable and randomization stratification factor, i.e. number of chronic conditions and recruitment centre.
All randomized participants will be included in the ITT analysis. In the per-protocol analysis, the following participants will be excluded: 1) participants in the exercise and self-management group participating in less than 18 out of the 24 self-management and exercise sessions;79,80 2) participants in both groups undergoing major surgery during follow-up affecting their ability to perform activities of daily living (e.g. joint replacement or open heart surgery); and 3) participants in the usual care group participating in 12 or more supervised exercise therapy sessions for one of their diseases during follow-up.
A detailed statistical analysis plan will be published online before unblinding the data and before any analyses are performed.
Furthermore, an analysis of predictors of responders and non-responders to the personalized program, and of the working mechanisms behind the effects from the program, will be conducted and published in a separate publication based on measures such as physical activity level, glycated haemoglobin, fasting plasma glucose and fasting insulin levels, cholesterol and triglyceride, inflammation, depression, socioeconomic status, and combinations of physical and mental health conditions. The definition of a responder will be based on the global perceived effect outcome by subtracting the mean score on quality of life and physical function of participants reporting ‘unchanged’ symptom status from participants reporting ‘better’ symptom status. 81
Finally, a cost-effectiveness analysis will be conducted following recommended methods as previously applied by the authors.82,83
Dissemination and protocol amendments
The results, regardless of whether they are in favor of the intervention or comparator group, or inconclusive in relation to the study hypothesis, will be communicated in scientific papers, at relevant conferences, to a broader audience in the news and social media, through online support tools including multimedia and print education resources, in oral presentations and popular science magazines. Authorship eligibility will be based on the recommendations from the International Committees of Medical Journal Editors (ICMJE). All authors will have access to the data.
Any important protocol amendments will be reported to the Committee on Health Research Ethics for Region Zealand, registered at ClinicalTrials.gov and presented in the primary RCT report.
Discussion
Given the small and often negligible effects on important outcomes for people with multimorbidity, such as health-related quality of life and function, demonstrated in prior RCTs of interventions for people with multimorbidity,1,17 it is important to identify effective and cost-effective treatments to address the major burden of multimorbidity. 1
Substantial evidence highlights exercise therapy as a safe and effective treatment of at least 26 different chronic conditions, 18 and while it also appears safe and effective in people with multimorbidity, 27 high-quality evidence supporting it is sparse. Furthermore, evidence on the effects of self-management in multimorbidity is limited. 17 Interestingly, evidence on effective, cost-effective, and acceptable forms of exercise therapy for people with multimorbidity was identified as being in the top 10 most important research priorities in a recent James Lind Alliance Priority Setting Partnership. 84 Our feasibility study highlighted that the current MOBILIZE intervention was feasible and acceptable, 38 while the ongoing RCT described in this protocol will hopefully provide evidence on the effectiveness and cost-effectiveness of the exercise therapy and self-management support program.
In a systematic review of the few existing multimorbidity guidelines, 29 regular physical activity as part of a healthy lifestyle is encouraged, but the guidelines do not include specific recommendations for exercise therapy. 29 Therefore, the findings of our RCT will either provide high-quality evidence supporting the continuation of current practice (i.e. with limited use of exercise therapy) or provide the foundation for integrating self-management and exercise therapy into clinical guidelines and treatment plans of people with multimorbidity.
Supplemental Material
Supplemental Material - Study protocol for a multicenter randomized controlled trial of personalized exercise therapy and self-management support for people with multimorbidity: The MOBILIZE study
Supplemental Material for Study protocol for a multicenter randomized controlled trial of personalized exercise therapy and self-management support for people with multimorbidity: The MOBILIZE study by Søren T Skou, Mette Nyberg, Mette Dideriksen, Jan A Overgaard, Christine Bodilsen
Footnotes
Acknowledgements
We would like to thank Gregers Aagaard and Margit Dybkjær, two people living with multiple chronic conditions, and Tue Dybkjær a partner of a person with multimorbidity, for their feedback on MOBILIZE, including study design, methods and conduct. We would also like to thank the recruitment centres, health-care providers and persons involved in the study conduct. Finally, we owe gratitude to the MOBILIZE scientific advisory board consisting of Prof. Susan Smith, Prof. Sallie Lamb, Prof. Alan Silman, Prof. Bente Klarlund Pedersen, Prof. Ewa M. Roos and Prof. Rod Taylor.
Declaration of conflicting interests
STS is associate editor of the Journal of Orthopaedic & Sports Physical Therapy and has received personal fees from Munksgaard, TrustMe-Ed and Nestlé Health Science, all of which are outside the submitted work. He is co-founder of Good Life with Osteoarthritis in Denmark (GLA:D®), a not-for profit initiative hosted at University of Southern Denmark aiming at implementing clinical guidelines for persons with osteoarthritis in clinical practice. The authors declare that there is no other conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (MOBILIZE; grant agreement No 801790), a research program grant from Region Zealand (Exercise First), Næstved, Slagelse and Ringsted Hospitals’ Research Fund, The Danish Regions and The Danish Health Confederation through the Development and Research Fund (project no. 2703) and The Association of Danish Physiotherapists Research Fund. Furthermore, STS is the recipient of an ongoing, unrelated grant from the European Union’s Horizon 2020 research and innovation programme (ESCAPE; grant agreement No 945377). The funders did not have any role in the study other than to provide funding and they were not involved in the decision to submit the report for publication, nor do they have ultimate authority over any study activities.
ClinicalTrials.gov registration
NCT04645732.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
