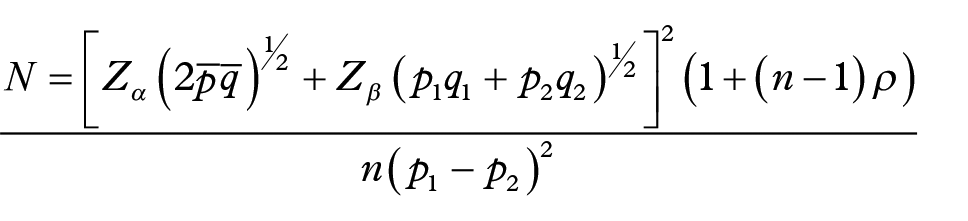Abstract
Background:
Preeclampsia is a leading cause of maternal morbidity and mortality. Various predictive methods exist, but none are without flaws. Risk factors vary in different women and at different gestational ages; hence, investigating non-modifiable factors, such as ABO and Rhesus blood groups, may aid prediction and early detection of preeclampsia.
Objectives:
This study aimed to evaluate the association between maternal ABO blood groups and preeclampsia.
Design:
This was a prospective cohort study.
Methods:
This study included singleton, normotensive, Rhesus-positive primigravidae with different ABO blood groups at <20 weeks of gestation. The primary outcome was the incidence of preeclampsia in mothers with Rhesus (D)-positive ABO blood groups (O, A, B and AB), while the secondary outcomes were the overall preeclampsia incidence and the odds ratio for preeclampsia between blood group O and non-O (A, B and AB) participants.
Results:
A total of 264 participants were enrolled, with 244 followed up until delivery. Eighteen participants developed preeclampsia with an incidence of 7.4% (95% confidence interval: 3.9–10.7). Most participants had blood group O (57.0%), followed by A (29.5%), B (12.7%) and AB (0.8%). The mean gestational age at preeclampsia onset was 37.8 ± 2 weeks. Blood group AB had the highest incidence of preeclampsia (50%), followed by B (16.1%), O (5.8%) and A (5.6%). Among 105 participants with non-O blood groups, 10 developed preeclampsia (p = 0.265). The adjusted odds ratio for the non-O blood group who developed preeclampsia was 1.65.
Conclusion:
Maternal ABO blood group was not significantly associated with preeclampsia incidence. However, blood group AB exhibited the highest preeclampsia incidence compared with the other blood groups.
Plain language summary
Preeclampsia is a condition that affects pregnant women and is characterised by high blood pressure (hypertension) and high protein levels in the urine (proteinuria). It is a leading cause of pregnancy complications and death worldwide. Several methods have been used to predict the development of preeclampsia; however, these are not always reliable because preeclampsia develops differently in each pregnancy. This study aimed to determine the relationship between hypertension during pregnancy and maternal blood type (A, B, O, or AB) with Rhesus factor, which is a type of protein found in red blood cells. In the present study, the incidence of hypertension and significant proteinuria during pregnancy was 7.4%. Maternal blood group AB had the highest risk of developing preeclampsia and may be a risk factor for disease occurrence. However, no significant relationship between maternal ABO blood groups and hypertension during pregnancy were found. Maternal ABO and Rhesus blood types may be of great value for the prediction and early detection of hypertension with significant proteinuria during pregnancy.
Introduction
Preeclampsia is a multifactorial pregnancy-specific disorder and a leading cause of maternal mortality worldwide. 1 Most deaths from preeclampsia are due to the progression to severe preeclampsia or eclampsia.2,3 Globally, preeclampsia and eclampsia account for 10%–15% of maternal deaths.4,5 Developing countries, including Nigeria, are the most affected. In Port Harcourt, Nigeria, 35.6% of maternal deaths were due to preeclampsia and eclampsia. 6 In Nnewi, south-east Nigeria, two separate studies on preeclampsia reported mortality rates of 25% and 27%.7,8 Given the significant burden of preeclampsia-related maternal and perinatal complications, better diagnostic capacity for preeclampsia is essential.
Despite extensive research, the exact cause of preeclampsia remains unknown. Several theories have been proposed, including poor trophoblastic invasion of the endometrium, abnormal immunologic adaptation, imbalances in prostaglandin production and abnormal vascular responses to vasoactive substances.9–11 Researchers have attempted to find a test for predicting preeclampsia, but there is no widely accepted screening test due to potential shortcomings. Improvements in diagnosis and good access to antenatal care have led to early diagnosis; however, this has not brought a commensurate reduction in case fatality rates. 12 Factors such as blood group and genotype do not change during pregnancy, and have not been extensively studied to determine their association with preeclampsia. Extensive predictive studies for preeclampsia using Doppler ultrasound of the uterine artery and assays of soluble endoglin, placental protein-13, soluble tyrosine kinase-1 and free foetal DNA in the maternal serum have been done. However, these tests are costly, not readily available and yield inconsistent results.
It is suspected that preeclampsia may result from changes in vascular haemostatic function, which may be influenced by the ABO blood group.13,14 ABO blood group antigens are oligosaccharides conjugated to cell-surface glycoproteins and glycolipids. 15 These antigens may alter the presentation of cell-surface glycans and modulate their interactions with pathogens. 16 Hence, ABO antigens have been associated with various diseases, potentially including preeclampsia. A recent review suggests that preeclampsia involves widespread maternal immune system activation and coagulation changes, with differences in the ABO blood group potentially affecting disease risk based on inherited antigens. 4 Inherited thrombophilia has been investigated as a preeclampsia risk factor. Studies have found that ABO blood groups may influence haemostatic balance, wherein non-O blood groups (A, B or AB) increase the risk of thrombosis as compared to blood group O. 17 ABO blood groups have been associated with several thrombotic conditions, with non-O blood groups increasing the risk of venous thrombosis. 18
Although several studies have evaluated the association between the ABO system and preeclampsia, no consensus has been reached regarding the true association between preeclampsia and blood groups.17,19–26 Studies have demonstrated that preeclampsia could be connected to a single gene or the Rhesus blood group,27–29 but studies on the association between preeclampsia and ABO blood groups have shown conflicting results. Two case–control studies showed that the risk of developing preeclampsia was higher among pregnant women with blood group AB than among those with blood groups A, B or O.20,21 However, a study in Brazil found that the risks for preeclampsia were not significantly different among pregnant women with different ABO blood groups. 30
Since some studies have shown that having blood group O protects pregnant women from preeclampsia, this study aimed to determine the association between preeclampsia and the ABO blood group system in our locality. The ABO blood group does not change during pregnancy, similar to other factors associated with preeclampsia. If an association exists, obstetricians and other health workers who care for pregnant women will have a high index of suspicion for women with non-O blood groups (AB, B and A), which will aid in the prediction and early detection of preeclampsia in women with non-O blood groups. Low-dose aspirin is currently recommended as a preventive strategy for the prevention of preeclampsia in at-risk women. If an association between maternal ABO and preeclampsia is established, this preventive strategy could be extended to relevant blood groups. Other measures, such as regular prenatal examinations, and healthy lifestyle promotion, including regular exercise, stress reduction and a balanced diet, may also be implemented. Health education and early intervention can improve patient outcomes if high-risk women are identified early. Blood group typing is common, inexpensive and does not require any specialised training, unlike other methods for predicting preeclampsia. This study evaluated the association of ABO blood groups with preeclampsia in primigravidae, and determined the overall incidence of preeclampsia among different ABO blood groups and whether there was a difference in the rate of disease development between the O and non-O blood groups.
Methods
Study design
This was a prospective cohort study, which conformed to the Strengthening the Reporting of Observational Studies in Epidemiology statement (Supplemental Material). 31
Study settings
This study was conducted at the Nnamdi Azikiwe University Teaching Hospital (NAUTH) in Nnewi, Anambra State, Nigeria, between 6 March and 5 September 2016.
Study population
Normotensive primigravidae of Nigerian origin between the ages of 20 and 40 years with singleton pregnancies of <20 weeks of gestation were included in the study. Primigravidae with any of the following conditions were excluded from the study: chronic hypertension, diabetes during pregnancy, renal disease during pregnancy, multiple gestations, Rhesus-negative women and those who withheld consent. Rhesus-negative women were excluded to minimise the potential confounding effects related to Rhesus incompatibility, which could independently influence pregnancy outcomes, including preeclampsia.
Sample size
The sample size was determined using the formula for dichotomous outcomes in a prospective cohort study, assuming a consistent difference in proportions between the two groups across time points: 30
where N was the number of subjects in each of two groups; Zα was the percentage point of the normal distribution (standard normal deviation) corresponding to the two-sided significance level of 5% (Zα = 1.96); Zβ was the one-sided percentage point of the normal distribution (standard normal deviation) corresponding to 100% – the power at 80% (Zβ = 0.84); p1 was the incidence of preeclampsia in the O blood group (2.3%) 32 ; p2 was the incidence of preeclampsia in the non-O blood group (14.2%) 32 ; n was the number of time points (seven monthly measurements including baseline and in labour); and ρ was the assumed common correlation across the n observations (0.6). Anticipating an attrition rate of 30%, 32 the adjusted minimum sample size was 77 subjects per group. A total of 244 participants (139 in the O group and 105 in the non-O group) were enrolled in the study, which satisfied the minimum sample size requirement.
Participant recruitment
An antenatal clinic (ANC) was the site of recruitment for the study. Upon arrival at the ANC, the resident doctor on duty scrutinised the antenatal records of the patient to determine eligibility for the study. A purposeful sampling method was used for subject selection, serially following the ANC card numbers until the minimum sample size was achieved. New patients were selected daily, and medical history was obtained from patients who met the inclusion criteria. They were educated about the purpose of the study, and those who provided written consent were recruited for the study. All relevant information was recorded pro forma.
Follow-up and diagnosis
The participants were followed up on their ANC appointment days. Blood pressure and urinalysis results were determined and recorded at each visit, and they were followed up during labour until delivery. Follow-up was discontinued after 24 h postpartum.
Definition of terms
The terms were defined according to the International Society for the Study of Hypertension in Pregnancy. 33 Preeclampsia was defined as hypertension and significant proteinuria after 20 weeks of pregnancy in a previously normotensive, non-proteinuric woman. Significant proteinuria was defined as two clean-catch midstream urine specimens collected ⩾4 h apart or a catheter urine specimen with ⩾2+ on a reagent strip. Hypertension was defined as a systolic blood pressure of ⩾140 mmHg and/or diastolic blood pressure of >90 mmHg measured twice at least 4–6 h apart, or a single measurement of ⩾160 mmHg systolic blood pressure and/or ⩾110 mmHg diastolic blood pressure.
Study procedure
Blood pressure was measured by a researcher or trained assistant using a mercury-in-glass sphygmomanometer with a standard adult cuff size. The patient must have rested for at least 5 min and sitting comfortably on a chair with a backrest while resting her left arm on a desk at the same level as the heart. 34 The first Korotkoff sound was recorded as the systolic pressure, and the fifth (disappearance of the sound) as the diastolic pressure. The fourth Korotkoff sound was used when the fifth was not recordable.
The procedure for blood sample collection was explained to the participants. A prominent vein (the median cubital vein) was selected, and about 2 ml of blood was drawn, which was emptied into a labelled plain bottle. Collected samples were sent to the NAUTH laboratory for blood grouping and typing.
Blood grouping and Rhesus (D) typing were based on agglutination. Red blood cells with antigens agglutinated in the presence of antibodies against these antigens. Individuals who lack A and/or B antigens in their red blood cells naturally have antibodies in their plasma that are directed against the missing antigens. Commercial antisera were used to test the individual cells. The ABO grouping included forward (cell) and reverse (plasma or serum) procedures.
Forward grouping procedure
The following steps were done to group and type the blood of the participants: (i) A 12 mm × 75 mm test tube with proper participant identification was labelled; (ii) a few drops of the red blood cells from the participant was transferred to the labelled test tube; (iii) a 3%–5% suspension of the cells using saline as the diluent was prepared; (iv) three 12 mm × 75 mm test tubes were labelled as A, B and D; (v) a drop of the corresponding commercial antiserum was added to the tubes labelled A (anti-A), B (anti-B) and D (anti-D); (vi) a drop of the 3%–5% suspension of the red blood cells from the participant was added to each test tube; (vii) the test tubes were shaken gently and incubated for up to 15 min at room temperature or immediately centrifuged; (viii) after centrifuging the test tubes at high speed (15–20 s at 3500 rpm), the red blood cells formed a button or pellet at the bottom of each test tube; and (ix) the cells were gently resuspended and examined macroscopically using an illuminated optical instrument for agglutination.
Reporting results
The agglutination results were recorded and reported as follows: Group A, patient cells showed agglutination with anti-A antiserum and no reaction with anti-B antiserum; Group B, patient cells showed agglutination with anti-B antiserum and no reaction with anti-A antiserum; Group AB, patient cells showed agglutination with anti-A and anti-B antisera; Group O, patient cells showed no reaction with anti-A or anti-B antisera; Rhesus (D) positive, patient cells showed agglutination with anti-D antiserum; and Rhesus (D) negative, patient cells showed no reaction with anti-D antiserum.
For urinalysis, participants were instructed to clean the vulva with clean water before collecting a midstream urine sample in a wide-mouthed container without stopping the flow. A catheter urine specimen was used for patients with urinary catheters. The urine specimens were collected from the subjects by the researcher or trained assistant using gloved hands, and a Medi-Test Combi 2 (Neumann-Neonder Str.68952355 Duren) dipstick urinalysis strip was taken from its container, dipped into the urine and read following the procedural instructions of the manufacturer. The colour of the strip pad was compared to the reference chart on the container, and the results were recorded.
Outcome measures
The primary outcome measure was to determine the incidence of preeclampsia in mothers with blood groups O, A, B and AB, while the secondary outcome measures were to determine the overall preeclampsia incidence and to estimate the odds ratio (OR) of developing preeclampsia between blood group O and non-O (A, B and AB) participants.
Statistical analyses
Rhesus (D)-positive patients were classified based on their blood groups (A, B, AB and O). Patients with blood group O were placed in one arm, while those with non-O (A, B, AB) blood groups were placed in the other arm. The generated data were analysed using SPSS version 20.0 (IBM Corp., Chicago, IL, USA). The relationship between maternal ABO blood group and preeclampsia was determined using the chi-square test. Logistic regression was performed to estimate the OR for the likelihood of developing preeclampsia in the non-O blood group compared with the O group, while controlling for relevant confounders such as age, body mass index (BMI) and educational level. Statistical significance was set at p < 0.05.
Results
Of 300 primigravidae that were initially assessed for eligibility, 264 were enrolled in the study, with 244 (92.4%) followed up until delivery (Figure 1). The mean maternal age was 28.0 ± 4.6 years, and the mean BMI was 30.1 ± 3.2 kg/m2. The majority of the participants (69.3%) had secondary education (Table 1).

Flowchart of study recruitment for Rhesus-positive primigravidae followed up to 24 h postpartum for the development of preeclampsia.
Patient demographics.

BMI, body mass index; HLE, highest level of education.
Table 2 shows the overall preeclampsia incidence and its incidence across different blood groups. The majority of the participants belonged to blood group O (n = 139, 57.0%). Of the 244 women analysed, 18 (7.4%) developed preeclampsia. The incidence of preeclampsia was highest in blood group AB (50.0%).
Incidence of preeclampsia in the population and the individual blood groups.

CI, confidence interval.
The distribution of diagnoses according to gestational age and individual blood groups is shown in Figure 2. Most women (66.7%) developed preeclampsia between 37 and 39 weeks, with four cases each in blood groups A, B and O. Four women (22.2%) were diagnosed between 40 and 42 weeks, with two cases in blood group O and one case each in groups B and AB. Only one woman (5.6%) was diagnosed between 31 and 33 weeks, and another between 34 and 36 weeks.

The column chart shows the distribution of preeclampsia by gestational age and blood group.
Table 3 shows the relationship between maternal ABO blood group and preeclampsia. Using blood group O as a reference, the adjusted OR for the non-O blood group developing preeclampsia was 1.65.
Relationship between maternal ABO blood group and preeclampsia.
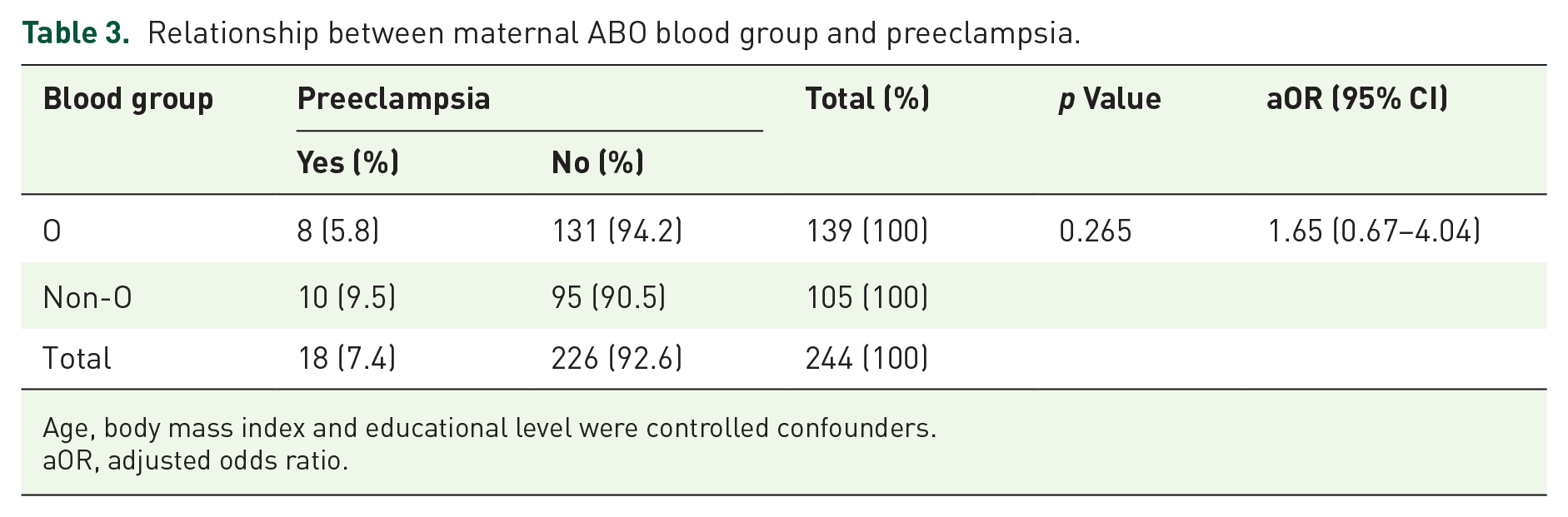
Age, body mass index and educational level were controlled confounders.
aOR, adjusted odds ratio.
Discussion
This study demonstrated that preeclampsia was most common in women with blood type AB, and the overall preeclampsia incidence was 7.4%. No correlation between preeclampsia and ABO blood group was observed. The non-O blood group did not significantly increase the risk of preeclampsia development.
This result is partly consistent with the studies of Alpoim et al., 17 Hiltunen et al. in Finland, 20 Spinillo et al. in Italy, 21 Lee et al. in Sweeden, 22 Rwitusmita et al. in India, 23 Phaloprakarn and Tangjitgamol in Thailand 24 and Reshmarani et al. in India. 25 Blood group O had the lowest preeclampsia incidence in previous studies, contrary to our study, wherein blood group A had the lowest preeclampsia incidence. This difference could be a result of variations in methodology. Reshmarani et al. conducted a cross-sectional study in India, and their work was not specific to preeclampsia but focused on pregnancy-induced hypertension. In addition, maternal age, gestational age upon enrolment, parity and multiple gestations were not considered in their study. Lee et al. conducted a retrospective cohort study in Sweden that examined gestational hypertensive disorders in general rather than focusing specifically on preeclampsia. In Thailand, Phaloprakarn and Tangjitgamol conducted a retrospective study on ABO blood group and adverse pregnancy outcomes including preeclampsia, gestational diabetes, preterm delivery, low birth weight and small gestational age. The retrospective study by Hiltunen et al. in Finland included 248 patients with preeclampsia and 679 controls. Severe preeclampsia, early preeclampsia and preeclampsia with intrauterine growth restriction were analysed separately. The influence of factor V Leiden and anti-beta2-glycoprotein on preeclampsia was also studied. Geographical and ethnic variations in the phenotypes of the different blood groups may have also accounted for the inconsistencies in the present study. These factors make blood type-based predictions difficult, particularly in diverse populations; hence, further studies are required to address these crucial factors.
The present study was similar with a study by Mishra and Pradhan in India, 32 wherein blood group A had a significantly higher risk (63%–81%) of pregnancy-related hypertensive disorder as compared to blood group B and O. Blood group A was significantly associated with hypertensive disorders of pregnancy; however, the incidence of gestational hypertension was highest in blood group O and the incidence of preeclampsia was highest in blood group A. Their study examined the association between maternal ABO blood group and hypertensive disorders of pregnancy rather than focusing solely on preeclampsia. They did not adjust for maternal age and parity, which may be potential confounders and may explain the differences in the results.
In Brazil, Hentschke et al. did not find any association between maternal ABO blood groups and preeclampsia. 35 Their retrospective study included Rhesus-negative women, and they did not consider maternal age and parity. On the other hand, Lawoyin and Ani found opposite results in Saudi Arabia, wherein Rhesus (D)-positive women with blood group O were twice as likely to develop preeclampsia as others. 26 Lawoyin and Ani recruited a small sample size of 20 participants, which may account for the contrasting findings relative to ours.
The gestational age at preeclampsia diagnosis did not vary significantly among the various blood groups. The majority of the diagnoses were made between 37 and 38 weeks (late-onset preeclampsia), while the mean gestational age at the preeclampsia diagnosis was 37.8 ± 2.1 weeks. This finding aligns with the study by Phaloprakarn and Tangjitgamol in Thailand, which reported a mean gestation age at delivery of 38.2 ± 1.7 weeks and no significant difference in gestational age at delivery. 24 Only two patients, both from blood group O, were diagnosed before term at 32 and 34 weeks. This raises the question of whether blood group O increases the risk of the occurrence of early-onset preeclampsia (occurring at or before 34 weeks). This study is not sufficient to draw such a conclusion, and further research is needed to confirm or refute this possibility.
Blood group O had the highest frequency (57.0%) in the current study. Although similar, our findings differ slightly from those reported in other studies. Phaloprakarn and Tangjitgamol reported frequencies of blood group O (36.8%), B (34.4%), A (26.7%) and AB (7.1%). 24 Rwitusmita et al. reported frequencies of blood group A (15.2%), B (23.5%), AB (5.2%) and O (56.1%). 23 Lee et al. reported frequencies of blood group A (45.4%), B (11.6%), AB (5.2%) and O (37.9%). 22 Apart from the study by Lee et al., group O was the most prevalent in other studies. Group AB consistently had the lowest frequency across all studies.
The incidence of preeclampsia among primigravidae in our study was 7.4%, which aligns with the findings by Wagner, Phaloprakarn and Tangjitgamol, and Lee et al., who reported incidences of 3–7%, 6.6% and 5.9%, respectively.22,24,36 For individual blood groups, preeclampsia incidence was most common in blood group AB (50.0%). An incidence of 7.4% is relatively high, which warrants a high index of suspicion, especially for women in the non-O blood group. However, as this was a local study, the results may not be generalisable to the entire population. A multicentre study is appropriate to validate these results. In addition, since only primigravidae, with primigravidity being a known risk factor for preeclampsia, were included, this may have affected the preeclampsia incidence observed.
There was a relationship between maternal ABO blood group and preeclampsia; however, having a non-O blood group did not significantly increase the risk of developing preeclampsia. The adjusted OR for preeclampsia in the non-O blood group, using blood group O as a reference, was 1.65. When individual ORs were calculated for individual blood groups (AB, OR: 8.7; B, OR: 2.8; A, OR: 0.97), women with non-O blood groups (B and AB) had an increased risk of developing preeclampsia, with AB having the highest risk. This finding aligns with the studies by Lee et al., 22 Phaloprakarn and Tangjitgamol, 24 Hiltunen et al. 20 and Spinillo et al. 21 In these studies, the non-O blood groups were found to have an increased risk of developing preeclampsia compared to the O blood group, with AB having the highest risk; however, a protective effect was found in blood group O. Our results partly agree with previous findings; however, we found that group A had a lower incidence of preeclampsia than group O. Nevertheless, the observed preeclampsia incidence in blood group AB should be interpreted with caution because of the small sample size and lack of statistical significance.
This study has several implications. The distribution of blood types in this study is more informative when considered in the context of ethnic and geographical differences. Understanding the prevalence of blood groups in specific populations can help contextualise the risk of preeclampsia and how this might vary across different settings. In some regions, blood group O is more prevalent, whereas other areas show higher frequencies of blood groups A or B.37,38 Differences in the blood group distribution may confound comparisons between our study and those conducted in other regions, highlighting the limitations of generalising the findings to populations with different ABO blood group distributions. Confounding factors, such as other ethnic or environmental risk factors associated with preeclampsia, can vary between populations and affect the risk of preeclampsia. Although the distribution of blood types in our cohort was similar to that of other studies conducted in regions with similar populations, our primary focus was to assess the association between blood type and preeclampsia risk. Therefore, our findings may not be directly applicable to populations with different ABO blood group distributions. For example, in regions where blood group O is less prevalent, the relationship between ABO blood type and preeclampsia may differ; hence, our results should be interpreted in light of these geographic and ethnic variations. Preeclampsia is a multifactorial condition influenced by genetic, environmental and anthropological factors. 14 While blood type, particularly the ABO system, may show some associations with preeclampsia, it cannot explain the development of the disease on its own. Genetic predispositions, maternal age, pre-existing health conditions, such as hypertension, and environmental factors, such as diet and lifestyle, also play a significant role. 14 Acknowledging the complexity of preeclampsia is crucial because it has a broader interplay of various factors that go beyond a single biological marker.
This study also had some limitations. Determining the exact gestational age at which preeclampsia first developed was not always possible (except for those who developed preeclampsia intrapartum) because participants were not seen daily but at least once weekly. As a result, participants may have developed hypertension and/or proteinuria between visits, and the exact time of onset may have been missed. In addition, we could not extend the follow-up period to 6 months to identify those that may develop postpartum preeclampsia. This was a single-centre study, and a multicentre study with a large sample size is required to validate or refute our findings. The findings in the AB blood group should be interpreted with caution because of the small sample size. The present results in the AB blood group may be susceptible to bias and may have limited generalisability. We plan to address this issue in future multicentre studies with larger sample sizes.
Conclusion
There was no statistically significant association between maternal ABO blood groups and preeclampsia incidence in this study population. When the blood groups were considered individually, women with blood group AB exhibited the highest incidence of preeclampsia compared to other maternal blood groups. However, this finding should be interpreted with caution because the small sample size and lack of statistical significance limit its reliability. Nonetheless, our findings highlight the potential role of ABO blood group in identifying women at risk of preeclampsia even during preconception care. Obstetricians should have a high index of suspicion for preeclampsia in women with non-O blood groups, especially groups AB and B, to facilitate early detection of preeclampsia. Pregnant women with blood groups AB and B may benefit from close antenatal surveillance and interventions early in pregnancy. Future studies with larger sample sizes should be conducted to confirm our preliminary findings in these blood group populations.
Supplemental Material
sj-docx-1-reh-10.1177_26334941251338621 – Supplemental material for Association between maternal ABO blood group and preeclampsia: a prospective cohort study
Supplemental material, sj-docx-1-reh-10.1177_26334941251338621 for Association between maternal ABO blood group and preeclampsia: a prospective cohort study by Zebulon C. Okechukwu, George U. Eleje, Joseph I. Adinma, Gerald O. Udigwe, Innocent I. Mbachu, Chukwuemeka O. Ezeama, Nkiru N. Ezeama, Chukwudi A. Ogabido, Chukwunwendu F. Okeke, Chidinma C. Okafor, Stella C. Okechukwu, Chinedu L. Olisa and Chigozie G. Okafor in Therapeutic Advances in Reproductive Health
Footnotes
Acknowledgements
The authors are grateful to the study participants, the NAUTH hospital staff and everyone who contributed to the study.
Declarations
Declaration of publication statement
The authors declare that this manuscript has not been submitted to any other journal.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.