Abstract
Gestational diabetes mellitus (GDM) and thyroid disorders during pregnancy pose significant health concerns, impacting a substantial number of mothers globally. Globally, about 14% of pregnant women develop GDM, while thyroid disorders impact approximately 2%–3%. Both conditions contribute to adverse outcomes, including gestational hypertension, excessive fetal growth, and heightened perinatal morbidity. The central focus of this literature review is to examine the relationship between vitamin A, a crucial fat-soluble micronutrient in fetal development, and the occurrence of GDM and thyroid disorders during pregnancy. The primary research question investigates the association between vitamin A, GDM, and thyroid disorders, analyzing their combined impact on maternal, fetal, and neonatal outcomes. The review underscores the potential of vitamin A to modulate the risk and outcomes of GDM and thyroid disorders during gestation, emphasizing its role in GDM development and resolution and its influence on thyroid function in pregnancy.
Introduction
Vitamin A deficiency (VAD) is a global health concern that disproportionally affects women in socioeconomically disadvantaged areas, including an estimated 19 million pregnant women annually. 1 Pregnant women are susceptible to developing VAD during the last trimester owing to accelerated fetal growth and increased maternal blood volume. 2 Vitamin A plays a crucial role in fetal growth and development and is necessary for maintaining normal vision, gene expression, embryonic development, and immune function. 3 Notably, excessive vitamin A can be harmful, potentially causing teratogenic effects, underscoring the importance of maintaining optimal levels of vitamin A. 4 Using isotretinoin (vitamin A derivative) during pregnancy substantially heightens the likelihood of congenital abnormalities. Metabolic byproducts of retinoids, like 4-oxo-transretinoic acid and retinyl palmitate, are acknowledged for their potential to induce birth defects. 5 Excessive retinoids pose risks of toxicity and teratogenicity to embryos and fetuses, particularly in early pregnancy stages. 6 In a study encompassing 154 pregnancies exposed to isotretinoin, commonly prescribed for severe acne, outcomes included 95 elective terminations, 26 healthy infants, 12 spontaneous miscarriages, and 21 infants with deformities. Focused scrutiny of 36 pregnancies revealed 8 miscarriages, 23 healthy infants, and 5 infants with deformities. The relative risk associated with isotretinoin exposure for severe malformations was 25.6, with a confidence interval of 11.4–57.5. Deformities observed comprised craniofacial, cardiac, thymic, and central nervous system (CNS) defects, aligning with findings from animal studies. Recorded malformations included microtia or anotia in 15 infants, micrognathia in 6, cleft palates in 3, cardiac and aortic arch defects in 8, thymic abnormalities in 7, and CNS and optical issues in 18. 7 Research indicates that pregnant women consuming more than 4500 retinol activity equivalents (RAE) μg/day of total dietary vitamin A intake face an elevated risk of teratogenic effects. 8 Therefore, it is essential to closely monitor vitamin A intake levels during pregnancy to avoid the risk of toxicity.
Recent studies suggest that VAD may be associated with prevalent endocrinopathies during pregnancy, like gestational diabetes mellitus (GDM) and thyroid dysfunction. 9 GDM affects approximately 14% of pregnancies globally and poses substantial risks to both maternal and fetal health. 10 Mothers with GDM are at increased risk for cardiovascular disease and type 2 diabetes postpartum, while infants are at risk for birth complications, macrosomia, and even autism. 10 Vitamin A has a recognized role in glucose homeostasis and insulin sensitivity, as evidenced in animal studies, which suggests that there may be an association between DM and VAD. 11 Further understanding the role of vitamin A in glucose homeostasis and insulin sensitivity during pregnancy is pivotal to identifying modifiable causes, such as VAD, which may mitigate the incidence of GDM.
Thyroid dysfunction, including hypothyroidism, subclinical hypothyroidism (SCH), and hyperthyroidism, is another common endocrinopathy and affects 2%–3% of pregnancies annually. 12 The mother’s thyroid function significantly influences growth patterns in the uterus, playing a pivotal role in early placental development, intrauterine growth, fetal tissue accumulation, and differentiation. 13 Maintaining optimal thyroid function in expectant mothers is crucial for proper fetal growth and neurocognitive development. 14 While numerous studies indicate that hypothyroidism and hyperthyroidism during pregnancy are linked to adverse outcomes like miscarriage and intrauterine fetal demise,15,16 a smaller body of research suggests that there is also an association between SCH and adverse pregnancy outcomes. 17 Notably, subclinical hyperthyroidism does not appear to be associated with adverse pregnancy outcomes.18,19
The overarching objective of this study is to comprehensively review existing literature to explore the association between maternal endocrinopathies (GDM, hypothyroidism, hyperthyroidism, and SCH) and vitamin A nutritional status during pregnancy. By unraveling these complex interconnections, this review seeks to contribute valuable insights into potential preventive strategies and interventions to optimize maternal and fetal health during pregnancy.
Recommended dose of vitamin A during pregnancy
In the human diet, vitamin A is found in two forms: preformed vitamin A (retinol and retinyl ester) comes mostly from animal sources, while another form, provitamin A carotenoids (α-carotene, β-carotene, and β-cryptoxanthin), comes mostly from plant sources. 20 Vitamin A is also available in standalone supplements and is commonly included in most multivitamins. It is often present in forms such as retinyl acetate, retinyl palmitate, provitamin A β-carotene, or a combination of these. 21
Recommended dietary allowance (RDA)
The RDA for vitamin A during pregnancy is higher than that for non-pregnant women. According to the National Institutes of Health, the RDA for pregnant women aged 19–50 years is 770 μg of RAE per day. 21
Food products
Pregnant women or those planning to become pregnant are generally advised to avoid consuming liver and liver-based foods rich in preformed vitamin A. 22 Consequently, β-carotene, a provitamin A carotenoid, becomes their primary source of vitamin A, with key sources being orange and dark green vegetables and fortified beverages. 21 For those unable to meet vitamin A requirements, adequate β-carotene intake is essential to maintain proper vitamin A levels during pregnancy and prevent developmental disorders. However, no upper limit of β-carotene intake has been defined yet in pregnant women. 23
Supplements and multivitamin pills
A study recommends that prenatal supplements should include 1200 μg of preformed vitamin A (as retinol) and 1000 μg of mixed carotenoids. 23 However, commercial prenatal supplements contain preformed vitamin A (retinol) in 35% of prenatal supplements ranging from 500 to 8000 IU and β-carotene in 73% of prenatal supplements ranging from 80 to 10,000 IU. 23 Currently, most prenatal supplements do not likely contain preformed vitamin A (retinol) due to the risk of teratogenic effects from excessive vitamin A and the absence of current prenatal screening recommendations for vitamin A levels in pregnant women.
Effect on maternal, fetal, and neonatal outcomes
Several studies have shown that maternal GDM, hypothyroidism, hyperthyroidism, and subclinical hyperthyroidism can affect maternal, fetal, and neonatal outcomes (Table 1).
Mentions the common effects of GDM and maternal thyroid status on maternal, fetal, and neonatal outcomes.

GDM, gestational diabetes mellitus; IUGR, intrauterine growth restriction; LBW, low birth weight; LGA, large for gestational age; SGA, small for gestational age.
GDM and pregnancy outcome
GDM is divided into class A1, which is effectively managed through nutritional therapy without medication, and class A2, which requires insulin or other medications for effective glycemic control. 27 Class A1 GDM is often linked to factors such as advanced age and higher weight. Women with class A1 GDM face elevated risks of hypertension and cesarean delivery compared to the general obstetric population. 27 Infants born to these women tend to be significantly larger, contributing to an increased risk of shoulder dystocia or other delivery complications. About one in eight women with a class A1 GDM delivers a large for gestational age (LGA) infant. 25
Women with class A2 GDM also face heightened risks, including obstetric complications, the need for higher insulin doses for effective glycemic control, and the development of type 2 diabetes mellitus or hypertension in late puerperium. Infants born to women with class A2 GDM are more likely to have an LBW (<2500 g) or LGA. The combination of maternal obesity and GDM has a more pronounced impact on adverse pregnancy outcomes, including preterm birth, larger newborn size at birth, and an elevated likelihood of cesarean section, compared to the individual effects of either condition.26,28 Regardless of GDM classification, infants born to mothers with GDM are also at an increased risk for macrosomia, neonatal hyperglycemia, IUGR, stillbirths, and neonatal intensive care unit admission. 29 However, it is important to note that the findings lacked conclusiveness. 43
Thyroid disorder and pregnancy outcomes
The interaction between thyroid disorders and pregnancy outcomes is intricate.
Hypothyroidism and pregnancy outcomes
Hypothyroidism is the most common pregnancy-related thyroid disorder, affecting approximately 4% of all pregnant women. 44 Overt maternal hypothyroidism in iodine-deficient areas constitutes a risk factor for abnormal neurobehavioral development of the affected child. A study showed an interesting association between pregnancy outcomes and thyroid-stimulating hormone (TSH) levels. The study revealed that when TSH was less than 10.0 mIU/L, approximately 6.5% of pregnant women had a spontaneous abortion; when TSH was greater than 10.0 mIU/L, spontaneous abortions occurred in 12.5% of pregnancies. In addition, the likelihood of preterm birth increased with increased TSH levels. The rate of preterm birth was 5.4% when TSH was less than 6.0 mIU/L, 7.8% when TSH was between 6.0 and 10.0 mIU/L, and 11.4% when TSH exceeded 10.0 mIU/L. 45
Hyperthyroidism and pregnancy outcomes
Gestational transient thyrotoxicosis stands out as the predominant contributor to hyperthyroidism during pregnancy, impacting approximately 1%–3% of all pregnancies. Gestational transient thyrotoxicosis in mothers typically has minimal impact on the fetus and seldom necessitates intervention. This condition commonly resolves by approximately the 20th week of gestation, coinciding with the decline in human chorionic gonadotrophin (hCG) levels. 46 Graves’ disease is the second most prevalent cause of hyperthyroidism in pregnancy, with an occurrence rate of 0.2% in all pregnancies. 46 Graves’ disease tends to be exacerbated between the 10th and 15th weeks of gestation but often improves in the later stages of pregnancy.47,48 Overt maternal hyperthyroidism is also linked to complications such as heart failure and neonatal mortality. 49 Thyroid storm (TS) is an uncommon complication associated with uncontrolled growth hormone, but its occurrence during pregnancy lacks comprehensive understanding. According to a study by Davis et al., 50 only one instance of a mother experiencing TS was reported among 120,000 deliveries spanning 11 years at a single institution. 35
Identifying signs of hyperthyroidism in the fetus is critical, particularly as a robust predictor of neonatal hyperthyroidism, especially in cases of poorly managed maternal Graves’ disease. 51 Fetal indicators encompass tachycardia, thyroid enlargement, intrauterine growth retardation, polyhydramnios or oligohydramnios, advanced bone age, craniosynostosis with microcephaly, and hydrops. It is crucial to distinguish fetal goiters resulting from hyperthyroidism from those caused by fetal hypothyroidism, with Doppler ultrasonography revealing distinct blood flow patterns associated with each condition. 51
Maternal Graves’ disease during pregnancy may result in transient neonatal thyrotoxicosis, but insufficiently treated neonatal hyperthyroidism could lead to persistent adverse outcomes. If neonatal hyperthyroidism continues, it is linked to a 27% rate of morbidity and a 1.2% rate of mortality. 52 Potential consequences encompass conditions such as heart failure, liver dysfunction, microcephaly, craniostenosis, pulmonary hypertension, coagulopathy, and intellectual disability. 52 However, limited research exists on the neurocognitive effects of infant thyrotoxicosis. An investigation with eight children having a history of neonatal thyrotoxicosis found that six of them faced intellectual impairment and craniosynostosis, while four exhibited intellectual impairment at the age of 2 years or older. 51 Another study involving 17 children born to hyperthyroid mothers who received anti-thyroid drugs (ATD) during pregnancy observed no impact of ATD treatment on thyroid gland size/function or the physical and intellectual development of the children beyond the neonatal period. 51
Subclinical thyroid disorders and pregnancy outcomes
Subclinical hypothyroidism
Traditionally, SCH was thought to occur in 2%–3% of pregnancies, but recent research has revised the acceptable TSH range, setting a new upper limit at 2.5 mIU/L in the first trimester and 3.0–3.5 mIU/L in later trimesters. 53 This updated criterion has significantly increased the identification of SCH, with studies reporting varying prevalence rates, from 6.8% in Belgium to over 15% in the United States. 51 Some research suggests adverse effects of SCH on both mothers and fetuses, while other studies indicate no negative consequences. 54 Whether there is a connection between GDM and SCH is still up for debate. Three previous meta-analyses had conflicting findings, with some suggesting a moderate risk or no significant difference in GDM risk for pregnant women with SCH compared to those with normal thyroid function.32,37,55
Research on cognitive impairment of infants born to women with SCH has produced inconsistent findings. A subgroup analysis of 6 studies within a meta-analysis encompassing 4449 participants revealed that children (aged 2–9 years) born to mothers with SCH scored, on average, significantly lower in intelligence and motor skills compared to controls. 46 However, a prospective cohort study with 4615 mother–child pairs found no clinically significant association between first-trimester TSH levels and standard assessment test scores from children 4–15 years of age. 56 It is important to acknowledge the challenges of direct comparisons due to varied definitions of SCH and different cognitive testing methods across studies. 46
Subclinical hyperthyroidism
Subclinical hyperthyroidism manifests when the TSH is low, while the levels of FT4 and FT3 remain within the normal range. 46 Subclinical hyperthyroidism tends to be well-tolerated during pregnancy, with no reported adverse outcomes. In a study of 25,765 women, the prevalence of subclinical hyperthyroidism was 1.7%, and among these women, there was no observed increase in negative pregnancy outcomes. 46 A meta-analysis investigating the impact of maternal subclinical hyperthyroidism on pregnancy outcomes found no significant differences in the likelihood of hypertensive disorders, preterm delivery, macrosomia/LGA, or pregnancy loss between pregnant women with subclinical hyperthyroidism and euthyroid controls. 19 However, Zhang et al.’s study presented conflicting results. Subclinical hyperthyroidism in weeks 4–8 of pregnancy may reduce the likelihood of abortion but could increase the risk of preeclampsia and placental abruption (Figure 1). 57 Figure 2 illustrates the effects of GDM and thyroid disorders on maternal, fetal, and neonatal outcomes.24–42
Graphical representation of the aim of our study.
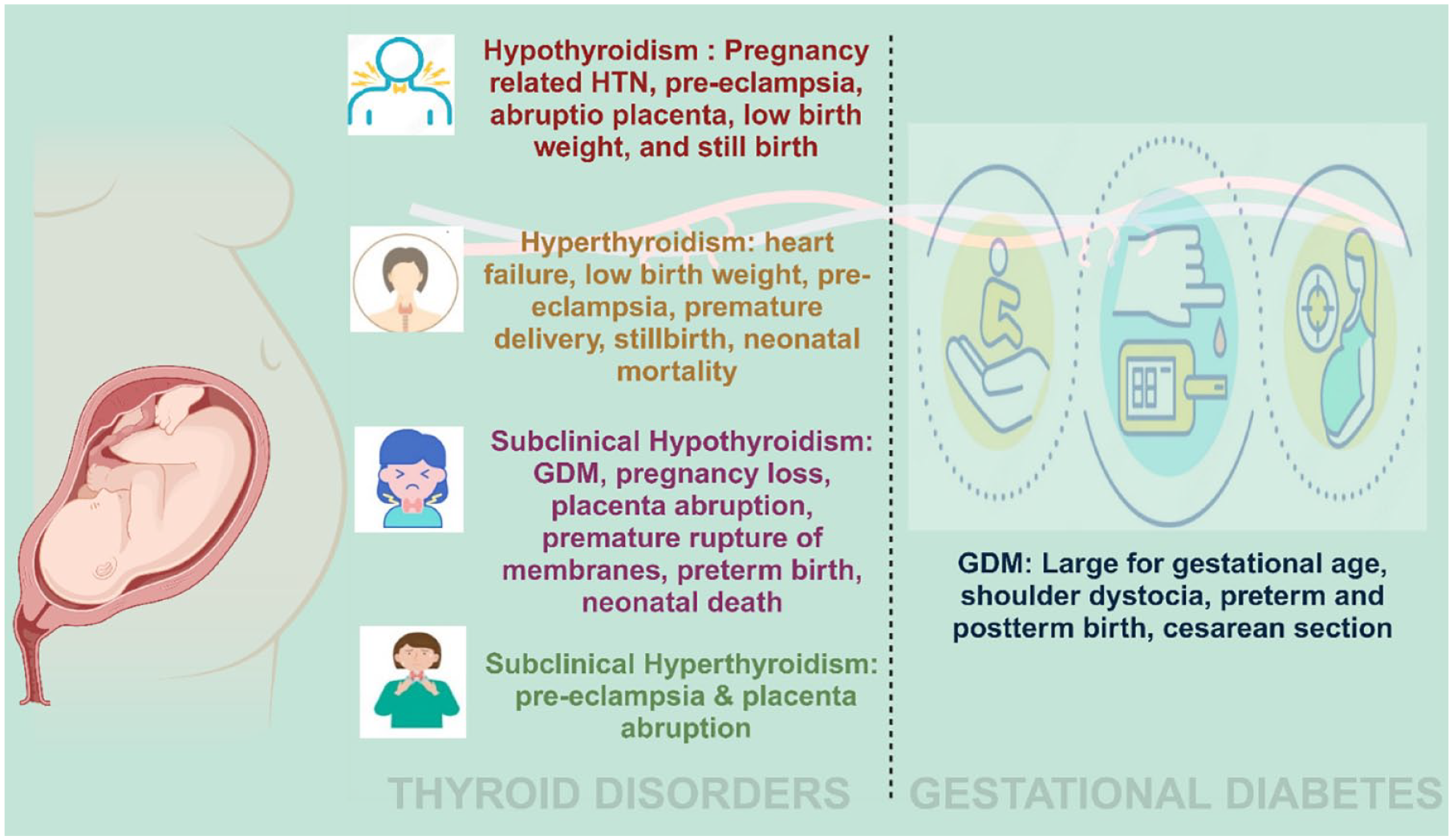
Effect of GDM and thyroid disorder on maternal, fetal, and neonatal outcomes.
Mechanism of action
Vitamin A influence on endocrine changes during pregnancy
During a normal pregnancy, the endocrine system is altered to support fetal growth with changes in multiple hormone levels including progesterone, estrogen, and beta-human chorionic gonadotrophin (beta-hCG) levels. 58 A study conducted by Panth et al. on the effect of vitamin A supplementation on plasma progesterone and estradiol levels during pregnancy revealed that plasma progesterone levels were higher in those who received vitamin A supplements, whereas there was no effect on estradiol levels. 59 Progesterone upregulates the genes involved in thyroid cell growth and differentiation, and from this, we can speculate that vitamin A may have a protective effect on the thyroid gland. 60 However, high progesterone levels also lead to impaired glucose metabolism via decreasing peripheral insulin efficacy and beta-cell proliferation, suggesting that vitamin A status may also contribute to the risk of GDM. 61 Moreover, a study by Hidayat et al. elucidated that vitamin A supplementation helps to decrease trophoblast regression and beta-hCG levels 62 by binding to the retinol-binding protein (RBP) receptors on trophoblasts to control cell proliferation and elicit apoptosis. 63 A study by Yoshimura et al. revealed that the structure of beta-hCG resembles TSH molecules and that beta-hCG is thyrotropic. 64 As far as its effect on glucose metabolism is concerned, a study showed that beta-hCG influences insulin sensitivity in adipose tissue and the gene expression of glucose transporter 4 (GLUT4), 65 which suggests that beta-hCG has a potential role in fostering GDM.
VAD and thyroid hormone synthesis
Pregnant women are more prone to develop VAD during the third trimester of gestation due to an increase in both maternal and fetal metabolic demand. 66 VAD is thought to influence thyroid hormone synthesis and thyroid homeostasis by reducing iodine status. Several proposed mechanisms highlight the association between VAD and the manifestation of thyroid dysfunction. 67
Pregnant women, being particularly susceptible to mineral deficiencies, may experience altered thyroid function due to decreased iodine uptake and sodium iodine transporter activity. VAD in pregnancy further reduces iodine uptake, iodothyronine coupling, and thyroglobulin synthesis with impaired glycosylation of thyroglobulin (Tg). Furthermore, women with VAD also experience decreased T3 binding in tissues and decreased hepatic conversion of T4 to T3. 68 In another study involving pregnant women, results showed that maternal FT4 in early pregnancy was positively associated with optimal fetal growth and higher vitamin A levels. 69 A study by Morley et al. involving rats showed that vitamin A supplementation resulted in increased T3 levels that could be attributed to reduced tissue responsiveness to T3. 70
Effect on TSH production
Retinoids are well known for their involvement in the modulation and moderation of gene expression via their interaction with nuclear receptors (retinoid X receptors (RXR)) and act as a cofactor in cell growth and differentiation. 68 VAD may lead to increased TSH production, despite normal levels of thyroid hormones T3 and T4, as retinol interacts with RXR on pituitary thyrotropes. In addition, in the presence of iodine deficiency, the impact of VAD on the thyroid axis is further heightened, potentially contributing to SCH.71,72
Vitamin A and thyroid hormone regulation
TSH, composed of alpha and beta subunits, is secreted by the anterior pituitary and stimulates the release of T3 and T4 from the thyroid gland. Feedback inhibition of TSH release is regulated by thyroid hormone receptors (TR). 73 T3 binds to TR and forms a heterodimer with RXR gamma 1, a vitamin A receptor. 74 Experiments with RXR gamma-deficient mice demonstrated increased TSH suppression, indicating the significance of both vitamin A in regulating TSH expression. 75
Obesity and thyroid dysfunction
Obesity is characterized by a state of chronic inflammation during which there is a surge of inflammatory cytokines like interleukin-1, tissue necrotic factor-alpha, and leptin. These cytokines inhibit the mRNA expression of the sodium/iodide symporter to reduce thyroid iodine uptake, induce vasodilation of blood vessels supplying the thyroid gland, and modulate the expression of deiodinases. Taken together, these morphological and functional changes in the thyroid can induce hypothyroidism. 76 Research reveals that women with obesity may have reduced vitamin A levels. A study involving school children in China showed that obese people have reduced vitamin A levels as a result of unbalanced nutrient intake with less intake of vegetables and legumes. 77 It has been noticed that VAD leads to a reduction in thyroid hormone levels, so it is possible obesity alters thyroid metabolism through decreasing pituitary thyroid axis responsiveness to vitamin A.
VAD and autoimmunity
Retinoic acid (RA) plays a crucial role in the proliferation, activation, and differentiation of lymphocytes by interacting with retinoid receptors present on the surfaces of regulatory T cells (T-reg). 78 Vitamin A is essential for modulating immune functions, particularly T-cell-mediated immunity. In a study involving non-obese diabetic mice, RA administration was shown to prevent autoimmune destruction of beta cells and thyrocytes, protecting against diabetes and thyroid autoimmune disorders by increasing the levels of immunosuppressive T-reg cells. 79 Consequently, a deficiency in vitamin A during pregnancy may increase the likelihood of developing GDM and/or thyroid dysfunction due to impaired immune regulation.
Vitamin A and gestational diabetes
Studies have indicated that vitamin A may have a significant role in glucose tolerance and insulin sensitivity,11,80 and thus, several investigations have proposed different mechanisms to explain the association between vitamin A and GDM.
Dietary patterns of pregnant women
Some studies have explored the dietary patterns of pregnant women and linked their nutritional status to the development of GDM. One study found that some pregnant women refrained from consuming foods with significant quantities of vitamin A, particularly liver products. This behavior could be due to concerns about potential side effects that excessive vitamin A consumption may cause during pregnancy. As a result, these women may not have been consuming enough vitamin A, putting them at a higher risk of developing GDM. 81
A case–control study conducted in China identified the “vitamin” dietary pattern in early pregnancy leads to a decreased risk of GDM. The most essential nutrients that played a role included vitamin A, carotenoids, vitamins B2 and B6, vitamin C, and calcium, in addition to potassium, dietary fiber, and folate. 82
Wang et al. investigated the relationship between dietary vitamin A intake and its source and GDM in the first trimester. The findings demonstrated that consuming vitamin A from plant-based sources had no impact on the development of GDM. However, adequate consumption of animal-based sources of vitamin A may lower the likelihood of developing GDM. 83 Studies conducted on the dietary patterns of pregnant women have suggested consumption of vitamin A-rich sources like eggs, meat, and vegetables may lower the likelihood of developing GDM.
Imbalance between oxidative and antioxidative state
A study was conducted to investigate the relationship between lower antioxidant vitamins such as vitamin A and GDM and found unexpected results. While gestational diabetes is generally associated with increased oxidative stress, the study found that higher levels of vitamin A were linked to GDM. However, the precise molecular mechanism behind this association is not yet fully understood. 84
Effect of GDM on RBP
RBP is a type of transport protein that plays a crucial role in the transportation of vitamin A in the blood. It is synthesized primarily in the liver and secreted into the bloodstream. RBP has a high affinity for binding to vitamin A and serves to protect it from degradation while it is transported from the liver to other tissues throughout the body. 85
Studies have indicated a positive association between RBP and type 2 diabetes. 86 This association may be due to the increased risk of obesity, which is a significant risk factor for type 2 diabetes. In addition, some studies have suggested that this relationship could also explain the link between vitamin A and GDM.87–89 The elevated levels of RBP could potentially cause a decrease in plasma levels of vitamin A, which might contribute to the development of GDM. However, it is important to note that this correlation may not be causal, as higher RBP levels could also increase the risk of obesity, which is a known risk factor for GDM. Therefore, it is not clear whether the relationship between VAD and GDM can be fully explained by the association between RBP and GDM.
In fact, a different study conducted by Beverly et al. did not find a significant association between RBP and GDM, which supports the idea that the relationship between RBP and GDM may not be straightforward. 90 The conflicting results between these studies could be attributed to various factors, such as differences in research methods, timing of vitamin A measurements, variation in the body mass index of pregnant participants, dietary habits, ethnicity, and inclusion or exclusion criteria.
Literature review
Table 2 provides a synopsis of research investigating the relationship of vitamin A with GDM and thyroid disorders.
A synopsis of research investigating the relationship of Vitamin A with GDM and thyroid disorders.
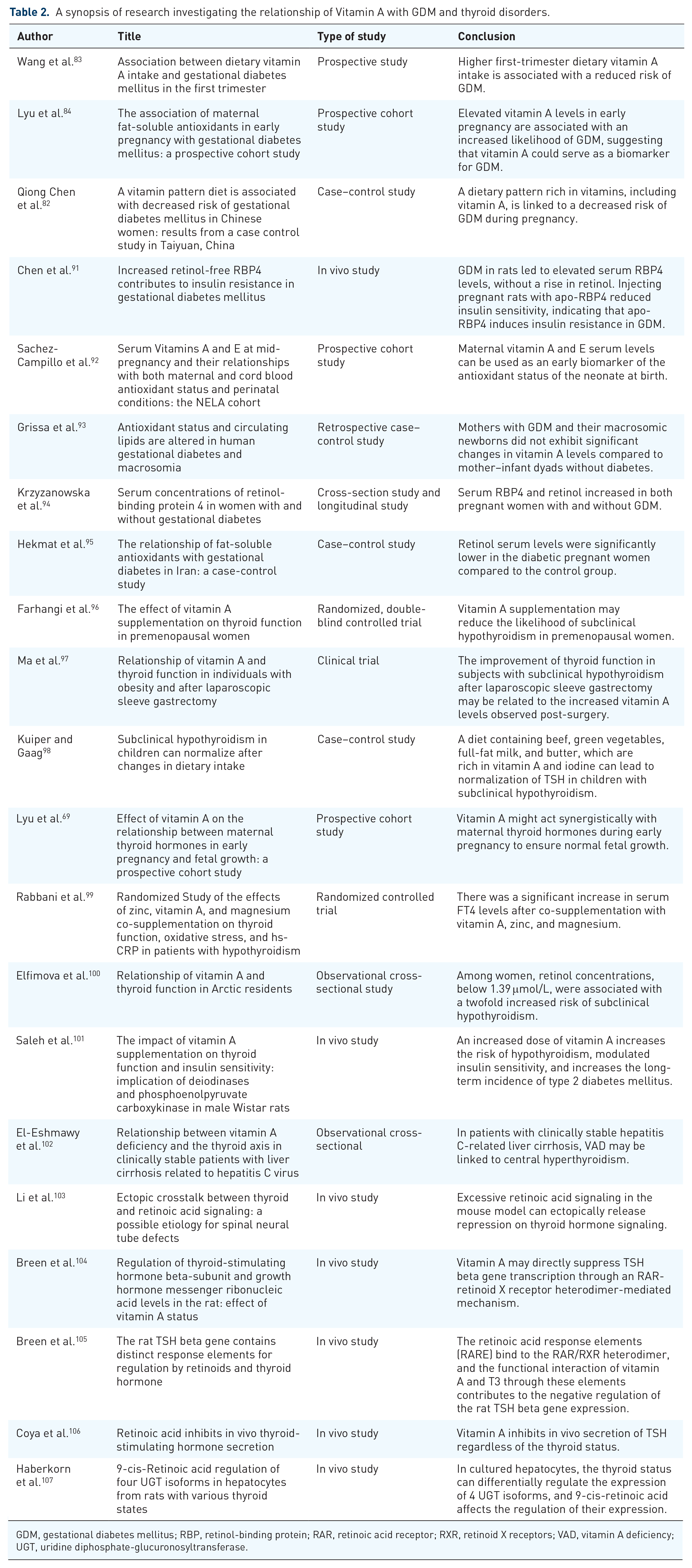
GDM, gestational diabetes mellitus; RBP, retinol-binding protein; RAR, retinoic acid receptor; RXR, retinoid X receptors; VAD, vitamin A deficiency; UGT, uridine diphosphate-glucuronosyltransferase.
Vitamin A appears to exert a multifaceted influence on glucose homeostasis during pregnancy. Its role in modulating insulin signaling pathways and enhancing the capacity of beta cells to secrete insulin underscores its potential impact on glucose regulation. Furthermore, the anti-inflammatory properties of vitamin A are believed to contribute positively to glucose homeostasis, while its role in mitigating oxidative stress is crucial for maintaining overall maternal health during pregnancy. The reported benefits of vitamin A supplementation, including delaying the rise in blood glucose, enhancing antioxidant enzyme activity, suppressing oxidative stress and inflammation, and inhibiting the NF-κB signaling pathway, collectively suggest that vitamin A supplementation can be beneficial in improving blood glucose levels and promoting overall maternal well-being both during and after pregnancy. 108 Although global consensus suggests a potential protective role of adequate vitamin A levels against the development of GDM, a prospective cohort study by Lyu et al. provides evidence that contradicts the positive protective role of vitamin A in GDM. This study concluded that higher concentrations of vitamin A during the early stages of pregnancy are markedly linked to a greater probability of developing GDM. There are many possible reasons for these discrepancies. First, measurements of RBP4 and TTR levels were not conducted. Second, the absence of information on dietary habits and physical activities results in an inability to adjust for confounding factors. Third, the study was conducted in a specialized hospital for women and children, which may have compromised the generalizability of the findings. Finally, the study lacked a predetermined power calculation suggesting there may have been an insufficient sample size. 84 When a similar study was performed after adjusting for confounding factors, results favored the potential protective role of vitamin A. This study found that diminished levels of retinol in the first trimester of pregnancy were associated with a higher likelihood of developing GDM requiring insulin treatment. 109
Studies exploring the association between vitamin A and thyroid disorders during pregnancy present varied mechanisms. For instance, the study by Ma et al. suggests that vitamin A influences thyroid hormone production, potentially offering protection against thyroid dysfunction. 97 This aligns with the findings of Breen et al., who reported that vitamin A modulates TSH beta gene expression, suggesting a direct regulatory role of vitamin A in thyroid function.104,105 However, the evidence remains fragmented, with some studies like that by Kuiper and Gaag highlighting the importance of dietary improvements in thyroid function, indirectly implicating vitamin A. 98 These discrepancies may stem from different study designs, population heterogeneity, and varying methodologies.
Studies in non-pregnant populations indicate a significant association between vitamin A and thyroid function, with evidence suggesting a bidirectional relationship influencing thyroid hormone synthesis and regulation97,106 (Table 2). These findings are crucial for understanding potential impacts during pregnancy, where thyroid hormone demands increase. However, the unique physiological changes of pregnancy, such as altered metabolic rates and hormonal fluctuations, necessitate cautious extrapolation of these results in pregnant women. Pregnancy may present as a state of potential VAD due to increased nutritional demands. 69 This deficiency could have implications for thyroid health, considering vitamin A’s role in thyroid hormone synthesis. 68 However, current research underscores the importance of investigating the association between vitamin A and thyroid disorders specifically in pregnant women. While studies in non-pregnant populations provide valuable insights, they cannot fully capture the unique physiological changes of pregnancy. There is a critical need for observational studies that focus on pregnant women to establish a direct link between vitamin A status and thyroid health in this demographic.99,101 Given the potential implications of VAD on thyroid function during pregnancy, it is essential to propose routine assessments of vitamin A levels in pregnant women with thyroid disorders. 110 This approach should also extend to the newborns of these women, considering the possible intergenerational impacts of maternal vitamin A status on thyroid health. 69
Clinical implications
The absence of clear recommendations on routine evaluation of vitamin A levels in pregnant women with GDM reflects the intricate nature of the vitamin A–GDM relationship. Monitoring vitamin A levels in infants born to mothers with GDM may be crucial, given the potential impact of maternal vitamin A status on neonatal health. 83 However, implementing routine vitamin A level monitoring in newborns poses practical challenges, including the cost and logistical feasibility of such screenings. The long-term benefits and potential interventions following the identification of VADs in these infants have not been fully explored. Developing comprehensive guidelines for vitamin A supplementation and monitoring requires well-designed, large-scale studies to provide conclusive evidence on the safety and efficacy of vitamin A supplementation in this context. Tailoring the approach to routine evaluations based on the specific needs and characteristics of the population is essential. In areas with limited resources, implementing such assessments may be challenging due to infrastructure constraints and cost-effectiveness, and the actual impact of vitamin A supplementation on health outcomes must be carefully evaluated. A balance between the potential benefits and the practicality of implementation is essential to ensure that any proposed routine assessments align with the broader goals of optimizing maternal and neonatal care. 111 Further research with a more comprehensive data collection and analysis approach would contribute to a more robust understanding of the relationship between vitamin A and GDM. 93 Furthermore, monitoring vitamin A levels could be pivotal in understanding and managing thyroid disorders during pregnancy. Exploring the benefits of vitamin A supplementation in pregnant women with thyroid disorders is another crucial area of research. 112 Controlled studies are necessary to determine if vitamin A supplementation can positively influence outcomes, such as reducing the prevalence of thyroid disorders or mitigating their severity during pregnancy. Such research would provide valuable insights into the therapeutic potential of vitamin A in managing thyroid health during pregnancy and could guide clinical recommendations for monitoring vitamin A levels and supplementation in pregnant women with common thyroid disorders.
Conclusion
In conclusion, the intricate relationship among vitamin A, GDM, and thyroid disorders in pregnancy demands thorough investigation. While some studies suggest a protective role of vitamin A against GDM, conflicting evidence underscores the need for comprehensive research. The nuanced association between vitamin A and thyroid disorders, along with proposed mechanisms, requires further exploration. Routine assessments of vitamin A levels in pregnant women with GDM and thyroid disorders, coupled with the investigation of potential supplementation benefits, merit consideration for optimizing maternal and neonatal health. Additional research in pregnant populations is crucial for establishing a direct link and informing clinical recommendations.
