Abstract
Background:
Medical management of adenomyosis is an emerging perspective in modern gynecology. Though levonorgestrel intrauterine system (LNG-IUS) and dienogest (DNG) effectively relieve symptoms in adenomyosis, neither has been approved for the same indication. Our study aims to compare the efficacy and safety of these progestins in treating adenomyosis.
Objective:
To study the efficacy and safety of LNG-IUS versus DNG in patients with symptomatic adenomyosis.
Design:
Open-labeled, parallel, single-centered, randomized clinical trial.
Methods:
Patients with adenomyosis-associated pain with or without abnormal uterine bleeding were randomly allocated to either LNG-IUS group or DNG group. The primary outcome was a reduction in painful symptoms after 12 weeks of treatment measured by visual analog scale (VAS) score. Changes in menstrual blood loss (MBL), improvement in quality of life (QoL), and adverse drug reactions were also analyzed.
Results:
The VAS score significantly decreased from baseline in both groups. The baseline and post-treatment VAS scores in the LNG-IUS group were 6.41 ± 1.07 and 3.41 ± 1.04 (p = <0.001) and in the DNG group, were 6.41 ± 0.95 and 3.12 ± 1.40 (p = <0.001), respectively. A significantly greater proportion of patients in the LNG-IUS group experienced lighter MBL as compared to the DNG group [27/30 (90%) in the LNG-IUS group versus 17/22 (77.2%) in the DNG group (p = 0.006)]. Both the groups had improvement in QOL scores calculated by the World Heath Organisation QOL scale (WHOQOL BREF) questionnaire; however, it was more pronounced in the DNG group [(28.76 ± 30.47 in the LNG-IUS group versus 48.26 ± 44.91 in the DNG group (p = 0.04)]. Both the agents were safe as there were no reported major adverse drug reactions.
Conclusion:
DNG can be an effective and safe alternative to LNG-IUS for the medical management of adenomyosis.
Trial registration:
The trial was prospectively registered at the clinical trial registry – India (CTRI) vide CTRI number CTRI/2020/05/025186.
Plain language summary
Adenomyosis is a condition affecting women, typically aged 40–50, but its incidence is rising in younger women, impacting fertility. It causes painful symptoms like dysmenorrhea, dyspareunia, chronic pelvic pain, and heavy menstrual bleeding. Managing symptoms is crucial, and medical approaches include levonorgestrel intrauterine system (LNG-IUS) and dienogest (DNG). LNG-IUS is reversible contraception, approved for eight years, effectively treating symptoms. DNG, a newer progestin, is effective for endometriosis, but evidence for adenomyosis is limited. This single-center, open-label randomized clinical trial compared LNG-IUS and DNG in treating adenomyosis. Women over 20 with pelvic pain were diagnosed using ultrasound and met specific criteria. After informed consent, participants were assigned randomly to LNG-IUS or DNG groups. Treatment outcomes, including pelvic pain, quality of life (QoL), and adverse effects, were assessed over 12 weeks. Out of 84 assessed, 74 women were recruited, with 34 in each group analyzed. After 12 weeks, both groups showed significantly reduced pelvic pain (VAS scores), but no significant difference was found between the groups. LNG-IUS resulted in a significantly greater reduction in heavy menstrual bleeding (HMB), whereas DNG showed better improvement in overall QOL. Adverse effects were similar in both groups, with hot flushes reported in the DNG group. This study is one of the few comparing LNG-IUS and DNG for adenomyosis, finding both effective for symptom relief. Although LNG-IUS was superior in reducing HMB, DNG showed better overall improvement in QoL. Safety profiles were similar. Previous studies support the efficacy of DNG in reducing adenomyosis symptoms. To conclude, both LNG-IUS and DNG effectively alleviate adenomyosis symptoms, with LNG-IUS superior in reducing heavy menstrual bleeding and DNG showing better overall improvement in QOL. DNG is a viable and effective alternative to LNG-IUS.
Introduction
Adenomyosis is an estrogen-dependent disease in women aged 40–50 years, though its incidence is increasing gradually in younger women, affecting their fertility. It is a significant cause of various painful symptoms like dysmenorrhea, dyspareunia, chronic pelvic pain (CPP), and heavy menstrual bleeding (HMB). 1 Painful adenomyosis symptoms negatively impact daily activities and quality of life (QoL). Hence, symptomatic relief is considered the cornerstone in the management of adenomyosis.
Medical management of adenomyosis is an evolving perspective that includes levonorgestrel intrauterine system (LNG-IUS), gonadotrophin-releasing hormone agonist (GnRH-a), combined oral contraceptives (COCs), aromatase inhibitor, and danazol. There are a few other newer therapeutic agents available, like dienogest (DNG), selective progesterone receptor modulators (ulipristal, mifepristone), and dopamine agonists (bromocriptine), which have demonstrated efficacy in the treatment of symptomatic adenomyosis.2–5 Medical methods can effectively relieve symptoms, are safe in terms of long-term use, and avoid hysterectomy, a major surgery with its own surgical and anesthesia-related complications.
The LNG-IUS is a reversible method of contraception which is approved for 8 years by the U.S. Food and Drug Administration in 2022. It is also effective in treating dysmenorrhea and HMB.5–7 It has been recommended as a treatment option for adenomyosis based on following mechanisms: (a) downregulation of estrogen receptors in both eutopic and ectopic endometrial glands; (b) decidualization of endometrium which subsequently leads to atrophy of adenomyotic ectopic endometrium; and (c) reduction of prostaglandin production within the endometrium thereby reducing painful symptoms in adenomyosis.6,7 LNG-IUS is associated with a more significant reduction in painful symptoms, uterine volume, and HMB than COCs.2–5 It is also used after adenomyomectomy or other conservative surgical procedures to prevent recurrence. 8
On the other hand, DNG, a progestational 19-norsteroid derivative, is a synthetic oral progestin with a highly selective affinity to progesterone receptors. The mechanism of action is multifaceted, involving antiovulatory, mild hypoestrogenic, anti-proliferative, and antiangiogenic effects. 4 It is used as a treatment option for painful symptoms of endometriosis; hence, it is expected to be effective for adenomyosis as both disorders have similar pathogenetic mechanisms. DNG is more effective in reducing painful adenomyosis symptoms than placebo, 4 COCs, 9 and GnRH-a (triptorelin acetate). 3 It also decreased HMB, uterine volume, and uterine artery blood flow compared to COCs. 9 DNG is a relatively newer drug than other progestins; hence, robust evidence regarding its efficacy and safety profile on long-term use for the treatment of adenomyosis is lacking in the literature.
The earlier studies evaluating the role of different medical methods had objective outcomes like uterine volume, uterine artery Doppler, and number of days with HMB. However, subjective improvement in terms of reduction of painful symptoms, change in menstrual pattern, or patient satisfaction has not been emphasized as essential outcomes despite the known detrimental effect of adenomyosis on QOL. Though both LNG-IUS and DNG effectively relieve painful symptoms and HMB in adenomyosis, neither of these agents has been approved for the same indication. Strong evidence from randomized clinical trials (RCTs) supporting the superiority of one agent over another is lacking. In a recent clinical trial, Ota et al. concluded that DNG is superior to LNG-IUS for the management of pain and uterine bleeding in adenomyosis. However, the allocation method in this study is not accurately recognized as a RCT. 10 Our study, to the best of our knowledge, is the first properly designed prospective RCT to compare the efficacy of these progestins in treating adenomyosis with pelvic pain with or without HMB.
Materials and methods
This study was a single-center, open-labeled parallel RCT that was prospectively registered at the clinical trial registry – India (CTRI) vide CTRI number CTRI/2020/05/025186. It included women attending outpatient gynecologic clinics at All India Institute of Medical Sciences, Bhubaneswar, India. The Institute Ethics Committee approved the study protocol. The trial was activated on 15 May 2020. Study participant recruitment started on 1 June 2020, and the last participant was recruited on 29 August 2021. The database was locked on 30 November 2021 for analysis. This study was reported according to the Consolidated Standards of Reporting Trials guidelines.
A detailed history and physical examination were carried out in women with age >20 years who presented with pelvic pain (dysmenorrhea or CPP) with or without uterine bleeding. The same investigator performed two-dimensional ultrasonography and color Doppler on all eligible participants. Transvaginal sonography (TVS) was performed using the Mindray Ultrasound equipment model no: DC-N3 (2013) or UMP 150 (2018). Diagnosis of adenomyosis was based on the Morphological Uterus Sonographic Assessment (MUSA) criteria, which includes (a) asymmetrical thickening of myometrial wall, (b) myometrial cysts, (c) hyperechoic islands, (d) fan-shaped shadowing, (e) echogenic sub-endometrial lines and buds, (f) translesional vascularity, (g) irregular junctional zone, and (h) interrupted junctional zone. Adenomyosis was diagnosed when any two of the above-mentioned features were found to be present. In cases of inconclusive diagnosis, a magnetic resonance imaging (MRI) of the pelvis was performed.
We included women of age >20 years who presented with pelvic pain and were diagnosed with adenomyosis by imaging. The exclusion criteria included women with ovarian endometrioma or uterine fibroids diagnosed by imaging, those planned for hysterectomy or any other modality of treatment, those who wished for pregnancy in the future, those with any of the contraindications to the use of LNG-IUS 11 or DNG 12 and baseline hemoglobin (Hb) level less than 8 g/dL. Patients with Hb levels <8 g/dL received treatment for anemia before being recruited into the trial.
After written informed consent, women were assessed for baseline measures like degree of pelvic pain (dysmenorrhea or CPP) and QOL. CPP was defined as non-menstrual pelvic pain lasting for ⩾6 months of duration. The degree of pelvic pain was measured by the visual analog scale (VAS) score. VAS is a visual assessment method in which a score 0 indicates no pain, and 10 indicates intolerable pain. The participants were asked how they evaluated their pain in their last menstrual cycle. QOL was assessed by the WHO-QOL BREF questionnaire in English or local language as preferred by the patient. WHO-QOL BREF questionnaire contains 26 questions with 4 domains: physical health, psychological health, social relationship, and environment domain. Participants were asked to answer the questionnaire by thinking about their lives in the last 2 weeks. A score of each domain was calculated and converted to another score with a range of 0–100 for final interpretation as per the instructions provided in the questionnaire manual. For overall QOL, scores of all the domains were added to get the total score. The greater the score, the better the QOL.
Participants were allocated to one of two groups after randomization using a computer-generated block randomization scheme (block size = 10). Allocation concealment was done by serially numbered sealed opaque envelopes. Preparation and sorting of the serially numbered envelopes were performed by an investigator who did not participate in evaluating patients either before recruitment or in the follow-up stage. The participants selected and opened the envelope in front of the investigators. Each participant was allocated to one of two groups. In the LNG-IUS group, LNG-IUS (Mirena 52 mg; Bayer Yakuhin, Osaka, Japan) was inserted according to the manufacturer’s instructions, whereas the DNG group received tablet DNG 2 mg once daily orally for 12 weeks starting between the second and fifth day of the menstrual cycle.
Participants were scheduled for outpatient follow-up after 12 weeks, and outcome measures were assessed. Compliance with DNG administration was ensured by asking to show the empty medicine packet. The degree of pelvic pain (dysmenorrhea and/or CPP) and QOL were assessed in similar ways as in the pre-intervention visit. Menstrual blood loss (MBL) was assessed subjectively as to whether it was light, heavy, or normal as before. This is per the International Federation of Gynecology and Obstetrics recommendation. 13 Patients were asked regarding any adverse drug reactions like hot flushes, breast tenderness, vaginal spotting, amenorrhea, or any other unusual symptoms.
The primary outcome was the change in the level of adenomyosis-associated pelvic pain (dysmenorrhea or CPP) from before treatment to 12 weeks after treatment, measured by VAS. The secondary outcomes were a change in menstrual pattern, a change in QOL, and reported adverse drug reactions.
The sample size calculation was based on the primary outcome (improvement in pain as indicated by VAS score after treatment). Using a two-sided chi-square test with an α of 0.05, the total sample size was calculated to be 106 patients in the 2 groups (i.e. 53 in each arm) with 80% power to detect a 30% difference in VAS score between LNG-IUS and DNG. Assuming a dropout rate of 10%, the study was supposed to recruit 120 participants (i.e. 60 in each arm).
Data were analyzed using IBM–SPSS version 23 (Statistical Package for the Social Sciences; International Business Machines Corporation, New York, United States) software. Continuous variables such as age, weight, height, body mass index, VAS, and QOL score were expressed as mean ± standard deviation and categorical values such as presence of symptoms and subjective assessment of bleeding were expressed in percentage (%). A paired t test was performed to compare continuous variables before and after treatment within groups, whereas unpaired t test was used for comparison between groups. For dichotomous variables, chi-square was used to estimate the significance value. A p-value of <0.05 was considered statistically significant.
Results
Eighty-four eligible women were assessed for eligibility, but 74 women could be recruited (i.e. 37 in each group). Ten patients were excluded, out of which 4 did not consent to study participation, and 6 opted for surgical treatment for adenomyosis. Thirty-four patients in each group were analyzed as per protocol (Figure 1). The groups were similar in baseline characteristics except for the prevalence of HMB, which was significantly greater in the LNG-IUS group (p = 0.022) (Table 1). MRI pelvis was performed in three patients in the LNG-IUS group and four patients in the DNG group because of indeterminate findings on USG. However, these small subgroups of patients had minor discrepancies between USG and MRI imaging; the MRI finding was considered for final diagnosis.

CONSORT flow chart.
Baseline characteristics.

Data represented as mean ± standard deviation, rest as frequency (percentage).
BMI, body mass index; CPP, chronic pelvic pain; DNG, dienogest; Hb, hemoglobin; HMB, heavy menstrual bleeding; LNG-IUS, levonorgestrel intrauterine system; VAS, visual analog scale.
After 12 weeks of treatment, there was a significantly lower VAS score for pain in both groups (Table 2). The baseline and post-treatment VAS scores in LNG-IUS were 6.41 ± 1.07 and 3.41 ± 1.04 (p < 0.001) and in the DNG group were 6.41 ± 0.95 and 3.12 ± 1.40 (p < 0.001), respectively. However, the reduction in VAS score was not significant between both groups [3.00 ± 1.32 in the LNG-IUS group and 3.29 ± 1.26 in the DNG group (p = 0.389)]. A significantly greater number of patients had lighter MBL in the LNG-IUS group [27/30 (90%) in the LNG-IUS group versus 17/22 (77.2%) in the DNG group (p = 0.006)]. The overall and domain-wise QOL improved significantly after treatment in both the groups (p < 0.001) except the social relationship domain in the LNG-IUS group (p = 0.062). The overall QOL and environment domain improvement of QOL was significant in the DNG group (p = 0.04 and 0.03, respectively) (Table 2).
Change in VAS and QOL score.
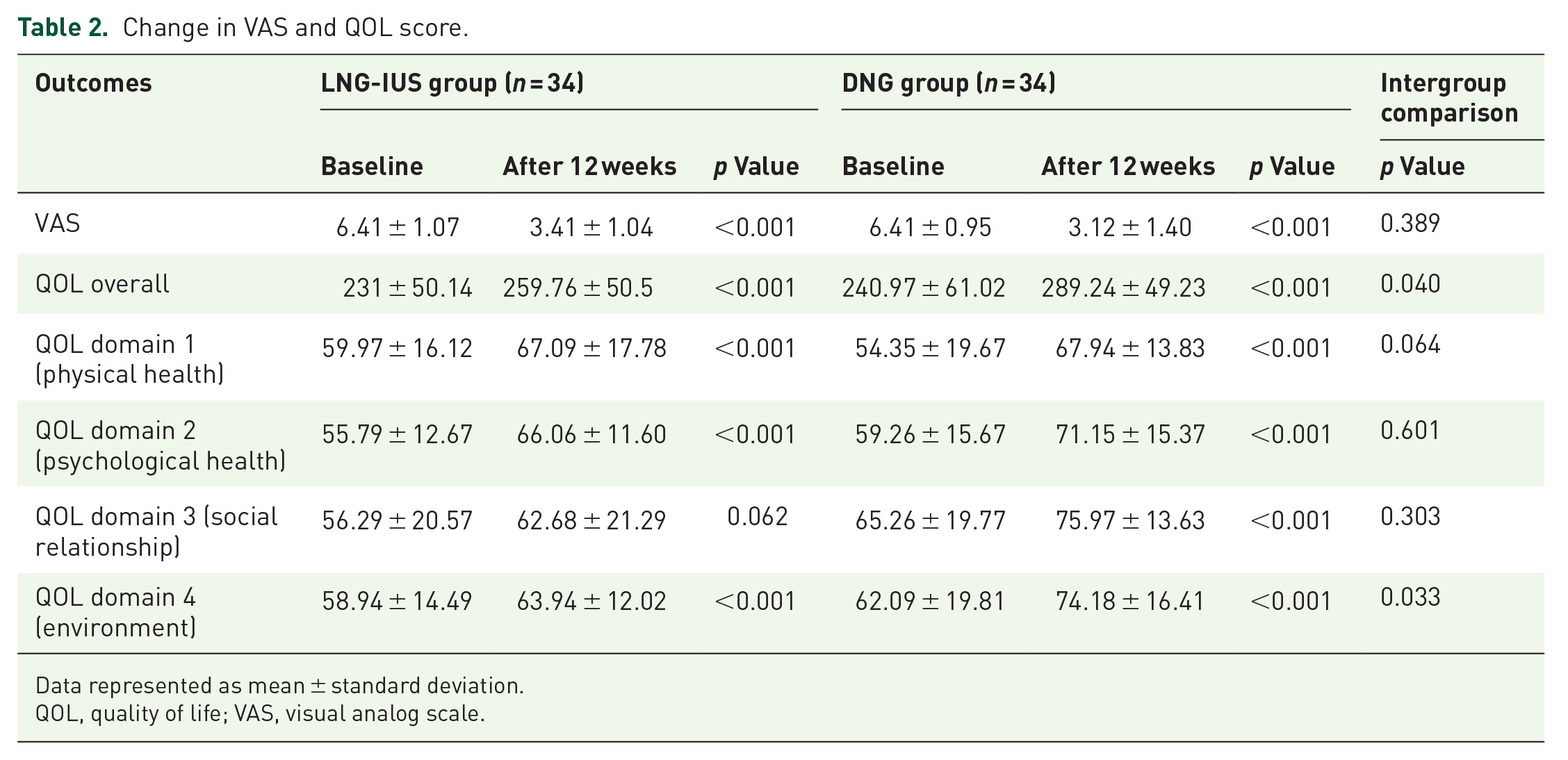
Data represented as mean ± standard deviation.
QOL, quality of life; VAS, visual analog scale.
A common adverse event of both LNG-IUS and DNG is vaginal spotting, and all the reported adverse events were similar in both groups (Table 3).
Adverse drug reactions in both groups.
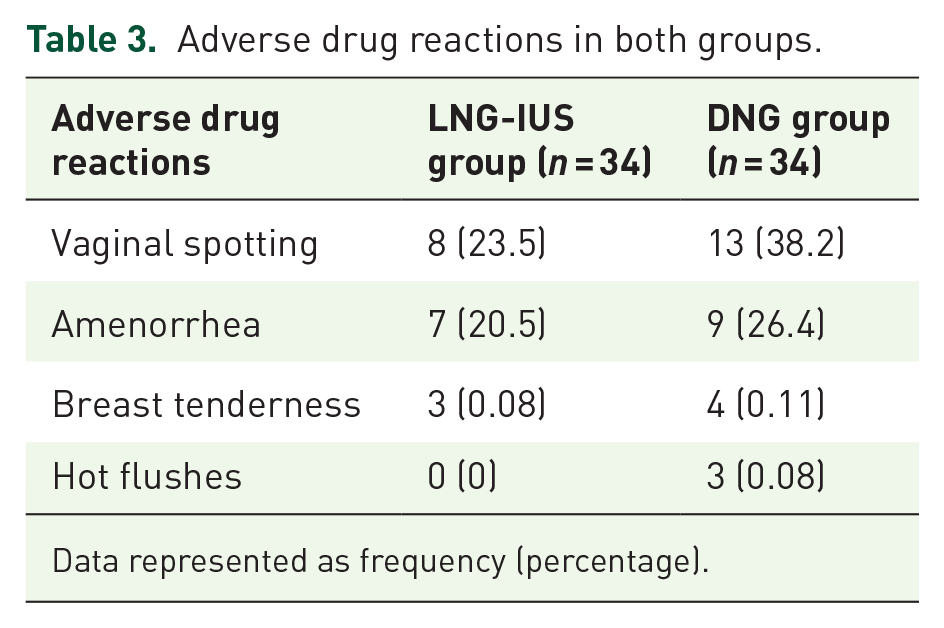
Data represented as frequency (percentage).
Discussion
To the best of our knowledge, it is one of the few randomized studies comparing the efficacy of LNG-IUS and DNG in alleviating symptoms in adenomyosis and affecting QOL. Both treatments are effective in the treatment of pelvic pain and bleeding associated with adenomyosis. However, the superiority of either intervention drug in alleviating pelvic pain could not be established. A greater number of patients had improvement in MBL in the LNG-IUS group compared to the DNG group. Patients who received DNG had better improvement in overall QOL than those who received LNG-IUS. Regarding safety, there were no major adverse drug reactions of both the progestins.
Our study found a significantly lower VAS score for pain after treatment in both groups. Though LNG-IUS has been studied extensively before for pain and bleeding control in adenomyosis, studies addressing the efficacy of DNG in adenomyosis-associated symptoms are scarce in the literature.
In a controlled clinical trial, Ota et al. 10 concluded that the reduction in VAS was greater in the DNG group than in the LNG-IUS group after 3 months. Osuga et al. 4 followed up 67 adenomyotic patients for 16 weeks and reported a similar reduction in pain score in the DNG group compared to the placebo group. Hirata et al. 14 observed that in 17 patients with painful symptoms of adenomyosis, dysmenorrhea, CPP, and dyspareunia decreased significantly (from 7.96 ± 1.37 to <1) at 8, 16, and 24 weeks follow up after treatment with DNG 2 mg/day. Longer duration of treatment with DNG also resulted in a significant reduction in VAS score. The mean VAS score decreased from the baseline value of 6.59 ± 2.03 to 5.27 ± 2.57 at 24 weeks and 5.67 ± 2.42 at 52 weeks of DNG treatment. Mean VAS score for dysmenorrhea was significantly decreased with DNG use for >80 months in a retrospective cohort study by Neriishi et al. 15 In an RCT comparing DNG with COCs, Hassanin et al. 9 found that DNG brought about a significantly greater reduction in VAS score as compared to COCs (from 6.27 ± 1.21 to 3.21 ± 1.18 in the DNG group and from 6.11 ± 1.13 to 4.92 ± 1.22 in COCs group). On comparison with triptorelin acetate, Fawzy et al. 3 concluded that both DNG and triptorelin acetate are equally efficient in reducing VAS score as a measure of dyspareunia and CPP. Matsushima et al. 16 demonstrated that improvement of adenomyosis symptoms like dysmenorrhea, CPP, and menorrhagia was significant when DNG was started after 6 months of GnRH-a therapy. Ota et al. 17 observed that the VAS score decreased significantly when DNG was added postoperatively after microwave endometrial ablation in adenomyosis. All these studies support our results and prove the efficacy of DNG in reducing painful symptoms of adenomyosis.
In our study, a significantly greater number of patients in the LNG-IUS group had a reduction in MBL compared to the DNG group. A significantly higher prevalence of HMB in the LNG-IUS group may be a contributory factor in the reduction of HMB in a higher number of patients in the IUS group.
This is supported by a prospective observational study which concluded that abnormal uterine bleeding improved after 6 months of LNG-IUS insertion in 29 patients in whom adenomyosis was diagnosed by MRI. 18 LNG-IUS has shown its efficacy in reducing menstrual bleeding after 6 months of use as compared to low-dose oral contraceptive pills (OCPs) in an open-label RCT. 2 In a controlled clinical trial, a significant decrease in days of uterine bleeding was observed after 12 months of DNG administration compared to those with LNG-IUS. 10 So, studies with more extended follow-up periods can add evidence regarding the efficacy of DNG in reducing MBL in adenomyosis.
In our study, we used patient-determined subjective assessment of the volume or flow of menstrual bleeding as a measure of MBL, which has not been used in previous studies. Studies that have been conducted to assess MBL in adenomyosis have used objective assessment methods like the Pictorial Blood Assessment Chart, the number of sanitary pads used, maintaining a menstrual diary, or changes in Hb level. As per the National Institute for Health and Clinical Excellence, HMB is defined as ‘excessive MBL, which interferes with a woman’s physical, social, emotional and/or material quality of life.’ 13 We consider the patient determined subjective assessment of MBL as a superior method as it involves every aspect of QOL, which are affected by menstrual bleeding.
In our study, the overall QOL significantly improved in both the study groups when analyzed from baseline to 12 weeks after treatment, and there was significant improvement in QOL in the DNG group compared to the LNG-IUS group. On intra-group comparison, three out of four domains (physical health, psychological, and environment) improved significantly in the LNG-IUS group. In contrast, all four domains of QOL improved significantly in the DNG group after treatment.
Ozdegirmenci et al. 19 concluded that LNG-IUS is at least as effective as hysterectomy in improving overall QOL and is superior to it in improving psychological and social domains of QOL score after 12 weeks of treatment. In our study, we suppose that instead of three out of four domains, all domains might have improved QOL if the follow-up period was longer enough. Improvement of QOL in the DNG group is also supported by a placebo-controlled RCT by Osuga et al., 4 who stated that QOL improved significantly in 35 patients diagnosed with adenomyosis concerning pain symptoms. Out of eight QOL domains of the Medical Outcomes Study Short Form (MOS SF36) Health Survey used in the above-stated study, only the score in the bodily pain domain increased significantly (p < 0.001). Contrary to it, in our study, all domains of QOL improved significantly after 12 weeks of DNG treatment, though the questionnaires used to assess the QOL differed in both studies. So, DNG can be a better progestin for the overall improvement of QOL involving physical health, psychological, social relationships, and environment domains.
The primary reason for treatment discontinuation of DNG is irregular uterine bleeding, leading to severe anemia. The risk factors for irregular uterine bleeding in patients receiving DNG are young age, anemia before DNG initiation, changes in serum estradiol level after DNG treatment, and type I adenomyosis (intrinsic adenomyosis). 20 We did not report severe anemia due to uterine bleeding in the DNG group due to correction of severe anemia in two patients before DNG initiation.
In our study, hot flushes occurred in three (8.8%) patients receiving DNG, whereas no LNG-IUS inserted patient developed it. The incidence of hot flushes after DNG treatment varies from 6% to 22.9%.4,9 The possible explanation for this can be the variation in the duration of follow-up periods in different studies. A comparable number of patients [7/34 (20.5%) in LNG-IUS versus 9/34 (26.4%) in the DNG group] had amenorrhea after 12 weeks of treatment, whose incidence was expected to increase on long-term follow-up. Incidence of breast tenderness was also comparable in both groups [3/34 (0.08%) in LNG-IUS versus 4/34 (0.11%) in the DNG group].
The main strength of our study is its randomized design. To the best of our knowledge, it is the first prospective RCT comparing the efficacy between LNG-IUS and DNG in adenomyosis. The use of USG for diagnosis of adenomyosis was as per recommendation of MUSA, which is the standardized protocol for describing and reporting various USG features of adenomyosis. This helped in reducing subjective variation among sonologists. We consider the patient-determined assessment of menstrual bleeding a superior method as it involves every aspect of QOL affected by menstrual bleeding. We used the WHO-QOL BREF questionnaire in English and the local language, which most patients could read and understand. This ensured that the communication bias was minimal.
However, our study had limitations. The major limitation of our study is the limited number of study participants, which decreases the generalizability of the conclusions. The study had a short follow-up duration, so long-term efficacy and safety profiles could not be analyzed. Uterine volume and uterine artery blood flow were not included as secondary outcomes, considering the short follow-up period of 12 weeks. The questionnaire used to assess the QOL in our study is not specific to adenomyosis. There is no validated disease-specific QOL measure for adenomyosis, unlike uterine fibroids and endometriosis, which can be considered an important research priority.
Conclusion
In conclusion, both LNG-IUS and DNG effectively reduce painful symptoms in adenomyosis, such as dysmenorrhea and CPP. LNG-IUS is superior to DNG for the decrease in MBL. Improvement in QOL is better with the administration of DNG. DNG can be an effective and safe alternative to LNG-IUS for the medical management of adenomyosis. However, further multicenter RCTs with larger study participants and longer follow-up periods need to be conducted to compare the efficacy and safety of DNG with LNG-IUS.
