Abstract
Background:
Fertility preservation is an important quality of life issue for women of reproductive age undergoing gonadotoxic treatment. The possibility of administering an adjuvant long-acting gonadotropin-releasing hormone agonist (GnRHa) with the aim of reducing the number of follicles susceptible to the effects of chemotherapy and thus reducing the risk of ovarian damage is considered in some international society guidelines, particularly in certain cancers such as breast cancer. Nowadays, the administration of long-acting GnRHa after controlled ovarian hyperstimulation (COH) for fertility preservation by cryopreservation of oocytes or embryos is increasingly used. However, cases of ovarian hyperstimulation syndrome (OHSS) have been reported following the use of long-acting GnRHa after COH for fertility preservation, indicating that the potential adverse effects of this treatment need to be further investigated.
Objectives:
The aim of this systematic review was to comprehensively characterize patients who developed OHSS after treatment with long-acting GnRHa following COH for fertility preservation.
Methods:
A comprehensive search of major electronic databases through January 2023 was performed. Studies reporting the use of long-acting GnRHa after COH for fertility preservation and the development of OHSS were included. Risk of bias was assessed using a modified version of the Newcastle-Ottawa scale. Results were synthesized qualitatively.
Results:
Three studies with five patients met the eligibility criteria. The majority of patients were diagnosed with breast cancer and all patients underwent COH for oocyte cryopreservation. OHSS occurred in all patients after administration of long-acting GnRHa. The interval between ovulation induction and administration of long-acting GnRHa thereafter ranged from 3 to 5 days. All patients were treated conservatively and recovered without complications.
Conclusion:
Current evidence suggests that the use of long-acting GnRHa after COH for fertility preservation may be associated with OHSS. Healthcare providers should thoroughly discuss the benefits and risks of this intervention with their patients before making a decision. Further studies are needed to fully elucidate the causal relationship between long-acting GnRHa and OHSS in this population.
Keywords
Introduction
Women of reproductive age are susceptible to developing several cancers, including breast, colorectal, and cervical. These cancers can have a significant impact on women’s reproductive health. 1 Fertility preservation is an important aspect of quality of life because many oncologic treatments, such as chemotherapy and radiation, can lead to ovarian insufficiency and infertility. 2 To address this issue, major scientific societies such as the American Society of Clinical Oncology, 3 the European Society for Medical Oncology, 4 the American Society for Reproductive Medicine, 5 and the European Society of Human Reproduction and Embryology, 6 have created specific guidelines for fertility preservation in cancer patients. These guidelines provide healthcare professionals with recommendations for optimal reproductive care in this patient population. Oocyte and embryo cryopreservation are established techniques for fertility preservation in women. 7 These techniques require controlled ovarian hyperstimulation (COH) as the first step in the treatment process and have been shown to be effective in preserving fertility in women and achieving successful pregnancy after cancer treatment. 8
Ovarian hyperstimulation syndrome (OHSS) is a potential complication associated with COH for fertility preservation that may pose a significant risk to oncology patients. This serious condition is primarily caused by hyper-response of the ovaries to exogenous gonadotropin stimulation, resulting in increased vascular permeability and subsequent fluid accumulation in the peritoneum and other body cavities. OHSS is characterized by enlarged ovaries, ascites, hemoconcentration, hypercoagulability, electrolyte imbalances, and systemic symptoms such as abdominal pain, nausea, and vomiting. 9 Cancer patients have an increased risk of thromboembolism, which further increases the potential risks associated with OHSS. In addition, the clinical condition of these patients is of critical importance, as prompt initiation of oncologic treatment is crucial for a better prognosis. 10 Therefore, prevention of OHSS in these patients is of paramount importance and should be carefully considered before the use of fertility preservation procedures.
The use of long-acting gonadotropin-releasing hormone agonist (GnRHa) as adjunctive therapy during chemotherapy to reduce the risk of ovarian insufficiency and infertility remains controversial. 11 Some international society guidelines consider its use, particularly in certain cancers such as breast cancer. However, administration of long-acting GnRHa after COH to preserve fertility by cryopreserving oocytes or embryos is becoming more common. Cases of OHSS have been reported following the use of long-acting GnRHa after COH for fertility preservation, indicating that the potential adverse effects of this treatment need to be further investigated. 12
The aim of this systematic review was to comprehensively characterize patients who developed OHSS after treatment with long-acting GnRHa following COH for fertility preservation. It is anticipated that the results of this review will provide valuable insight to healthcare providers and help them make informed decisions regarding the treatment and management of these patients, ultimately improving patient outcomes and quality of life.
Methods
Given the limited number of studies published to date and the fact that the aim of this review is to capture the state of knowledge, the following study designs were considered, taking into account the hierarchy of evidence and methodological quality of the primary studies identified: randomized clinical trials, quasi-randomized clinical trials, non-randomized clinical trials, cohort studies, case-control studies, and case reports. To be included, studies had to report data on incidence, prevalence, or risk of OHSS in cancer patients of reproductive age who underwent COH and received a long-acting GnRHa after oocyte retrieval. This could be based on clinical symptoms, laboratory findings, or any other validated diagnostic criteria for OHSS. The studies could include data on other complications related to fertility preservation procedures if OHSS data were reported separately. Studies that did not include data on OHSS or studies that focused exclusively on other complications unrelated to fertility preservation procedures were excluded. In addition, studies using animal models, in vitro studies, review articles, editorials, commentaries, conference abstracts, and studies with insufficient or incomplete data were excluded from the analysis.
For this review, an extensive search of studies from a variety of sources was conducted. Electronic databases searched included the Cochrane Central Register of Controlled Trials (Central), Database of Abstracts of Reviews of Effects, Base, PsycINFO, Latin American and Caribbean Literature on Health Sciences, and Medical Literature Analysis and Retrieval System Online. In addition, electronic searches were performed in the Gray Literature Database (https://opengrey.eu), Clinical Trials Databases (https://clinicaltrials.gov), and the International Clinical Trials Register Platform. Manual searches were performed in the Medrxiv database (https://www.medrxiv.org) and in the lists of bibliographic references of each of the included articles to search for information potentially relevant to the study. All sources were searched through January 2023, with no restriction on publication date.
A comprehensive search strategy was developed to identify relevant studies using appropriate keywords and medical subject headings, which were adapted to each database as needed. The search terms used were [(gonadotropin-releasing hormone) or (ovulation induction)] and [(fertility preservation) or (oncofertility) or (cryopreservation)] and [(OHSS) or (complications)]. No language or date restrictions were applied, and both full texts and abstracts were searched (PROSPERO registration number CRD42023401492).
Studies were selected using COVIDENCE web-based software and reviewed by two independent reviewers, CI and GB. The process involved two stages. In the first stage, the reviewers reviewed the titles and abstracts of studies identified by the search strategy. Potentially eligible studies were preselected, and studies that clearly did not meet the eligibility criteria were excluded. Any disagreements were resolved through discussion and consensus. In the second phase, the full text of the studies was reviewed by the same two independent reviewers, and any discrepancies were again resolved by discussion and consensus. For data collection, the same independent reviewers extracted data from the included studies using a standardized data collection form. Disagreements were resolved by discussion and consensus.
Outcomes for which data were sought in this review included the occurrence of OHSS and complications related to OHSS in female cancer patients. In addition, we collected data on study design, participant characteristics (e.g., age, cancer type, and treatment), and intervention characteristics (e.g., type and dose of long-acting GnRHa). No assumptions were made about missing or unclear information; any unclear or missing information was noted in the review. Case reports included in this study were assessed for risk of bias using a modified version of the Newcastle-Ottawa scale developed by Murad et al. 13 which consists of eight selection, ascertainment, causality, and reporting questions (Table 1).
Methodological quality assessment tool for case reports and case series. Adapted from Murad et al. 13 Reproduced with permission. Questions 4, 5, and 6 are particularly relevant to adverse drug events.

A narrative synthesis of results was conducted for this systematic review. The synthesis included a systematic summary of the characteristics of the included studies, followed by a descriptive analysis of the results. Data from the studies were tabulated and the similarities and differences between the studies were discussed. A qualitative synthesis was also conducted by summarizing the findings of the studies in a narrative format. This synthesis included a discussion of the strengths and limitations of the studies and an assessment of the overall quality of the evidence. The synthesis was presented in a comprehensive and structured manner that allows the reader to understand the findings of the included studies and the implications for clinical practice.
Results
A comprehensive search of the relevant databases was performed, resulting in a total of 1111 entries, of which 192 were duplicates and were removed. After screening the remaining 919 records by title and abstract, 10 studies were selected for full-text review. Of these, three studies met the inclusion criteria and were considered for data extraction and analysis (Figure 1).

Flow diagram of study selection process according to PRISMA guidelines.
Seven studies were excluded from data extraction and review. The studies by Oishi et al., 14 Oliveira et al. 15 and Hanada et al. 16 were excluded because of the wrong intervention, as they did not use long-acting GnRHa after ovulation induction. The study by Massarotti et al. 17 was excluded because of the wrong intervention, as the authors used the long-acting GnRHa to trigger ovulation after COH and did not use the medication after the ovulation induction. The study by von Wolff et al. 18 was excluded because of the wrong outcome, as it did not consider the OHSS risk in patients using long-acting GnRHa. The study by Gurbuz et al. 19 was excluded because of the wrong population, as it did not include patients diagnosed with cancer. Finally, the study by Tsafrir et al. 20 was excluded because of the study design, as it was a letter to the editor.
The first included study 21 was a case report of three patients with breast cancer, colorectal carcinoma, and medulloblastoma who underwent COH for fertility preservation before gonadotoxic chemotherapy. All patients underwent COH with a GnRH antagonist protocol and oocyte cryopreservation followed by ovarian suppression with long-acting GnRHa. Despite the use of a short-acting GnRHa as an ovulation trigger, patients developed severe, early-onset OHSS immediately after receiving long-acting GnRHa. The study concludes that the risk of severe OHSS may be increased when a long-acting GnRHa is used immediately after oocyte retrieval for ovarian suppression. The authors concluded that in patients at high risk for developing OHSS, delaying the use of long-acting GnRHa after oocyte retrieval should be considered.
The second included study 22 was a case report of a 28-year-old woman diagnosed with breast cancer and polycystic ovary syndrome who developed OHSS after receiving a long-acting GnRHa for ovarian suppression immediately after oocyte retrieval. The objective of the study was to describe the risk of OHSS in patients with cancer who receive a long-acting GnRHa for ovarian suppression after COH with a GnRH antagonist protocol. The study concludes that in patients at high risk of OHSS, delaying the start of treatment with long-acting GnRHa after oocyte retrieval should be considered.
The third included study 23 was a case report of a 28-year-old woman with breast cancer who underwent COH for oocyte cryopreservation before chemotherapy. The patient developed OHSS after receiving a long-acting GnRHa for ovarian suppression, delaying the start of chemotherapy. The study concludes that the timing of administration of long-acting GnRHa should be carefully considered to avoid the risk of OHSS.
The studies included in this systematic review were conducted in Italy and the United States. The age of the patients ranged from 15 to 39 years and the most common cancer type was breast cancer. Chemotherapy was the most common oncological treatment. Regarding fertility preservation, oocyte cryopreservation was the technique used in all patients. All patients underwent ovarian stimulation with concomitant use of the aromatase inhibitor letrozole. The drug dose ranged from 5 to 7.5 mg/day. In all patients, short-acting GnRHa was used as ovulation induction, except for one patient in which a dual trigger consisting of short-acting GnRHa and 1500 IU hCG was used. The peak estradiol levels ranged from 379 to 1747 pg/mL. The total number of retrieved oocytes ranged from 14 to 25 and the total number of mature oocytes ranged from 13 to 16 (no information on the number of mature oocytes is available in three of these cases). The interval between ovulation induction and administration of long-acting GnRHa ranged from 3 to 5 days thereafter. All five patients developed OHSS with characteristics such as ascites, moderate hemoconcentration and leukocytosis, hyponatremia, and oliguria. These results suggest that further studies are needed to determine the optimal use of long-acting GnRHa in patients undergoing COH for fertility preservation (Table 2). The modified Newcastle-Ottawa Scale (NOS) score for case series showed that the included studies were of moderate methodological quality (Table 3).
Included studies in the systematic review.

GnRHa, gonadotropin-releasing hormone agonist; OHSS, Ovarian hyperstimulation syndrome.
Methodological quality evaluation of the included studies.

N/A, Not applicable.
Discussion
The use of long-acting GnRHa for ovarian suppression in cancer patients undergoing gonadotoxic chemotherapy is a topic of great interest. In our systematic review, we found three case reports with a total of five patients reporting cases of OHSS after administration of long-acting GnRHa following COH. Despite the small number of cases, these reports raise concerns about the potential risks associated with the use of long-acting GnRHa in this context.
The mechanism by which long-acting GnRHa may contribute to OHSS in this population is not fully understood, but there is a plausible biological explanation in the form of the flare-up effect. This effect, which is well documented in other contexts, involves a transient increase in gonadotropin secretion by the pituitary gland following administration of a GnRHa. Long-acting GnRHa formulations may maintain the increase in gonadotropin levels for up to 14 days after administration. This increase in gonadotropin levels could stimulate the receptors of LH on several corpora lutea in the ovary after oocyte retrieval, leading to the production of angiogenic factors, such as vascular endothelial growth factor, fibroblast growth factor, and angiopoietin 1, which are responsible for increased angiogenic activity and eventually lead to the development of OHSS (Figure 2).
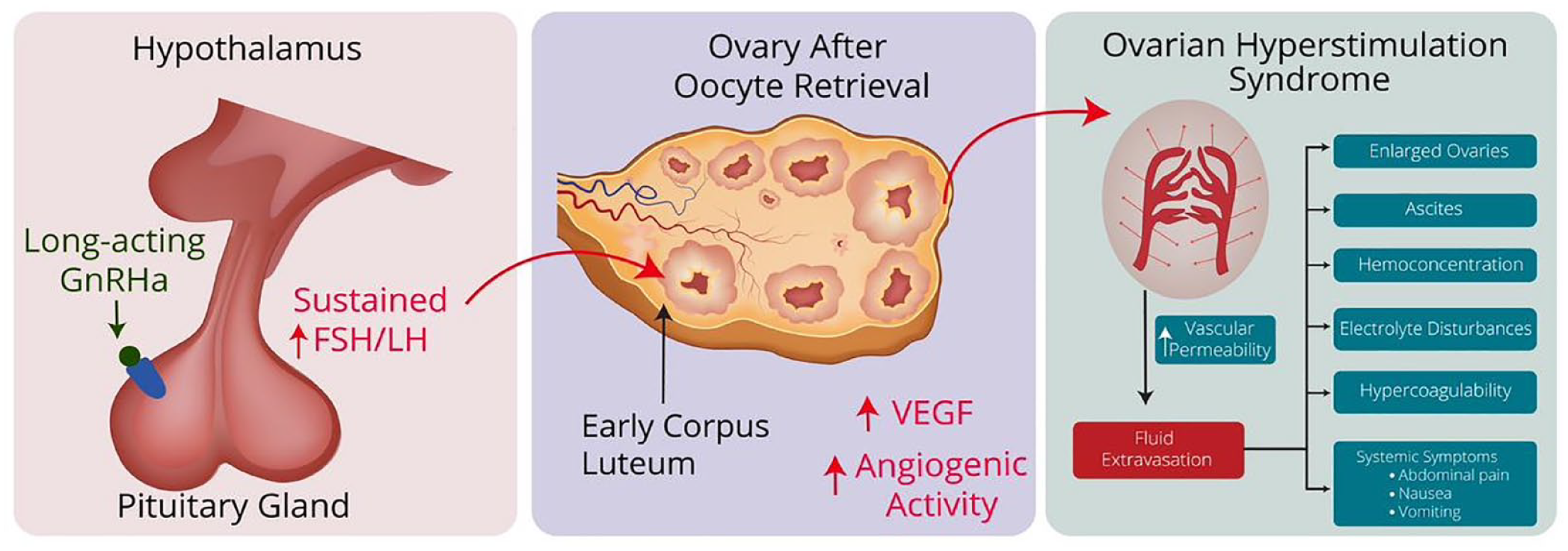
Possible mechanism by which GnRHa may contribute to OHSS. Long-acting GnRHa stimulates pituitary gland receptors resulting in a sustained increase in FSH and LH hormones via the “flare-up” effect. Elevated levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) act on the corpora lutea present in the ovary after oocyte retrieval. Corpora lutea then releases angiogenic factors, such as VEGF, leading to increased angiogenic activity. These factors result in elevated vascular permeability, leading to fluid accumulation in peritoneal and other body cavities, hemoconcentration, hypercoagulability and electrolyte disturbances, which characterizes OHSS.
All patients in these reports were in a clinically compromised state and needed to begin chemotherapy immediately. One report stated that oncologic treatment had to be postponed to allow the patient to recover from OHSS. 23 Although it is possible that this was also the case in the other reports, it was not specifically mentioned. Delaying the start of oncologic treatment because of the development of OHSS could have a negative impact on the patient’s prognosis. Cancer patients need a rapid start of treatment to improve their chances of survival, and any delay in treatment may worsen their prognosis. 10 Although all patients in these case reports eventually recovered after intensive treatment, it is important to note that in severe cases, OHSS can be a life-threatening condition that requires immediate medical attention and intervention to prevent serious complications such as thromboembolism, renal failure, and respiratory distress syndrome.24,25
In this context, it is important to consider the potential risks associated with the use of long-acting GnRHa as an ovulation trigger in patients undergoing COH, particularly in the setting of cancer treatment. One study suggested the use of this approach, 17 but it may not be appropriate for cancer patients who need to start treatment as soon as possible to achieve optimal results. Therefore, caution should be exercised when using long-acting GnRHa as an ovulation trigger, especially in cancer patients who need rapid and effective treatment. Further research is needed to fully evaluate the safety and efficacy of this approach in this patient population. If the use of long-acting GnRHa is included in the patient’s treatment plan after COH has been performed, strategies to reduce the risk of OHSS may include deferring the onset of long-acting GnRHa use or even discussing the use of GnRH antagonists aimed at reducing or blocking GnRH action to rapidly stop the output of FSH and LH without triggering the undesirable “flare-up” effect.
Participants in our study underwent ovarian stimulation cycles with administration of a GnRH antagonist to prevent premature ovulation, and most patients in our study received an ovulation trigger with short-acting GnRHa. Concurrently with ovarian stimulation, letrozole was administered to lower estradiol levels during treatment. These measures were taken with the aim of minimizing the occurrence of OHSS and are consistent with previous studies reporting rare cases of OHSS after a short-acting GnRHa trigger, usually in patients with high ovarian reserve and a large number of retrieved oocytes. 26
However, we cannot attribute with certainty the occurrence of OHSS solely to the use of the long-acting GnRH agonist administered a few days after ovulation was triggered. The development of OHSS could possibly be due to the ovarian stimulation procedure itself. It is important to highlight that in three of the five cases, the trigger was administered at a higher than usual leuprorelin dose for ovulation induction, although it is a short-acting GnRHa, which may have contributed to a higher OHSS risk. Furthermore, in our systematic review, we identified a single study reporting OHSS in two patients diagnosed with cancer who had not received long-acting GnRHa after the ovulation induction. This study, published by Oishi et al. 14 was excluded from the review because of the wrong intervention, as they did not use long-acting GnRHa after ovulation induction. These patients had breast cancer and were diagnosed with polycystic ovary syndrome, which is associated with increased ovarian reserve. In addition, ovulation induction in these patients was performed with high doses (10,000 IU) of human chorionic gonadotropin (hCG), a drug known to significantly increase the risk of ovarian hyperstimulation. It is noteworthy that in both patients a considerable number of oocytes were retrieved after stimulation (20 and 26 oocytes). This study shows how difficult it is to distinguish OHSS due to COH from OHSS following GnRH administration after oocyte retrieval.
It is important to consider the concern that anecdotal reports do not always reflect a true causal relationship between an intervention and an adverse effect. However, in the case of the three studies presented, there is a plausible biological mechanism in which the flare-up effect of long-acting GnRHa use could explain the development of OHSS in patients who underwent COH for fertility preservation. 27 In addition, the three studies with a total of five patients provide sufficient information to allow a detailed evaluation of the OHSS that occurred. Although further studies are needed to fully elucidate the causal relationship between the use of long-acting GnRHa after COH and OHSS, the utility of discussing the actual benefit for these patients should be considered.
The use of long-acting GnRHa for ovarian suppression during chemotherapy is recognized by select international scientific societies as a reliable strategy for preserving ovarian function and fertility, particularly in patients diagnosed with breast cancer.28–30 Recent evidence has provided more clarity on the potential benefits of this procedure. Despite its position as a viable option in clinical practice, the significant benefit of a realistic possibility of fertility preservation through oocyte or embryo cryopreservation is noteworthy. 8
Because the care of women of reproductive age diagnosed with cancer is a multidisciplinary endeavor involving many professionals who may be located in different centers, all health care providers involved in patient care should have a thorough discussion about the benefits and potential risks of using long-acting GnRHa in patients undergoing fertility preservation. 31 This discussion should include an assessment of the patient’s individual risk factors for OHSS, such as age, body mass index, and baseline markers of ovarian reserve, as well as careful monitoring and treatment of OHSS symptoms if they occur. To ensure a comprehensive, coordinated approach for patients requiring fertility preservation treatment, effective communication channels, multidisciplinary team meetings, and adoption of standardized clinical guidelines that address the unique needs of these patients are critical. Future studies with larger samples and rigorous methods are needed to determine the true incidence of OHSS in patients undergoing long-acting GnRHa treatment after COH for fertility preservation.
Given the limitations in the existing literature, further research is needed to better understand the potential risks and benefits of long-acting GnRHa in this context. There is a need for larger, controlled studies that specifically examine the true incidence of OHSS after COH or long-acting GnRHa use after COH in cancer patients. These studies would provide more robust and reliable data on the true incidence and prevalence of OHSS in this population and contribute to a better understanding of the risk factors, treatment strategies, and overall safety profile associated with these interventions. In addition, further investigation of the biological mechanisms underlying the development of OHSS in this population is warranted.
Conclusion
The use of long-acting GnRHa after COH for fertility preservation has been associated with the development of OHSS. Although the number of reported cases is small, it is important to weigh the potential risks and benefits of this approach in patients undergoing fertility preservation. The three studies presented in this review suggest a possible biological mechanism for the development of OHSS in these patients. Further research is needed to fully elucidate the causal relationship between long-acting GnRHa use and OHSS in this particular clinical scenario. In the meantime, healthcare providers should have an in-depth discussion with their patients about the benefits and potential risks of using long-acting GnRHa for fertility preservation, taking into account individual patient characteristics and preferences. It is important to note that fertility preservation is a critical component of quality of life for cancer patients of reproductive age. Therefore, patients should be offered all available options to preserve fertility while minimizing the potential for adverse effects. Future research should aim to gain a more comprehensive understanding of the benefits and risks of using long-acting GnRHa in patients undergoing fertility preservation.
