Abstract
Background:
Twin-to-twin transfusion syndrome (TTTS) is a severe condition causing preterm delivery, fetal death, and neurodevelopmental disorders. This study presents a data-based controlled amnioreduction (AR) protocol composed of sequential amniodrainage in treatment of TTTS.
Methods:
A total of 18 procedures were performed in 11 TTTS pregnancies at 17 to 34 weeks of gestation. The amniotic pressure was measured along with sequential removal of the amniotic fluid, 500 mL each step. The umbilical artery systolic/diastolic (S/D) ratio for each twin was measured pre- and post-AR. Long-term neurodevelopmental outcomes of all TTTS survivors were evaluated from parental answers to a phone survey.
Results:
The amniotic pressure decreased exponentially with the increased volume of removed amniotic fluid until a plateau was obtained. Changes of the S/D ratio between pre- and post-AR procedure did not reveal a clear tendency. The survival rate was 86.4% although 91% of all twins were at Quintero stage III. Long-term neurodevelopment outcomes in the 19 surviving twins were 68.4% optimal, 26.3% suboptimal, and 5.3% abnormal.
Conclusion:
The controlled AR procedure resulted in a relatively high rate of twin survival with favorable long-term neurodevelopment outcomes.
Keywords
Introduction
Twin-to-twin transfusion syndrome (TTTS) is a complication in monochorionic diamniotic (MCDA) twins in which blood is transferred from a donor to a recipient twin via anastomoses in the shared placenta. 1 The TTTS occurs only in 10–15% of MCDA pregnancies; however, if untreated, the expected perinatal morbidity and mortality is extremely high. 2 Historically, the placenta anastomoses have been well described since the mid-19th century, but intervention procedures were reported only in the late 1970’s. 3 The amnioreduction (AR) protocol, in which a large volume of amniotic fluid is removed starting from the 14th week of gestation, was the standard of care until the late 1990’s.2,4 Since the early 2000’s, the fetoscopic laser ablation (FLA) of the superficial placental anastomoses became the recommended management for TTTS.1–4
Numerous reports on the outcome of both AR and FLA interventions revealed very high rates of survival for either one or both twins and significant reduction of postnatal impairments, especially neurodevelopment.5–15 The FLA procedure is the recommended procedure for stage II through IV of TTTS diagnosed before the 26th week of gestation based on clinical trials. 8 However, some concerns were debated regarding the comparison analyses between FLA and AR outcomes.4,16–18
All in all, the AR procedure is still in clinical practice either before the more invasive FLA 12 or at the end of the FLA protocol 2 or when post-FLA repeated intervention is required, and when TTTS is diagnosed beyond the 26th week of gestation.19,20 The mean amniotic fluid volume (with significant variations) in normal singleton pregnancies increases with the gestational age from of about 250 mL in week 18 to a peak of about 1000 mL around weeks 30–35, and then, it descends to about 750 mL in week 40.21–24 Measurements of the amniotic pressure in normal singleton pregnancies demonstrated a wide band of variability with mean values progressively increase from 3 mmHg in week 10 to 6 mmHg in week 34.25,26 A similar pattern was also observed in normal twin pregnancies. 25 It should be noted that the amniotic pressure in most cases of polyhydramnios and TTTS was significantly higher than in uncomplicated pregnancies,27,28 and it significantly fell after draining the excess amniotic fluid.26,28 The treatment strategies of AR are quite subjective and based only on ultrasound imaging without clear guidelines as to how much and at what rate should the amniotic fluid be drained, as well as its relationship to the amniotic pressure. As a result, there is neither a commonly accepted AR protocol, nor indicators for termination of the drainage procedure. In the absence of an objective measures, the decision to terminate the procedure is usually based on the clinical experience of the physician and ultrasound imaging.1,2,29
In this study, we report a data-based controlled AR procedure composed of simultaneous measurement of successive small volumes of amniodrainage along with the amniotic pressure. In addition, we also measured the blood flow in each twin before and at the end of the procedure. We explored the relationship between amniotic pressure and progression of the removed volume of amniotic fluid, and evaluated the long-term neurodevelopmental outcome of the surviving twins.
Materials and methods
Population
We recruited 11 TTTS patients at Quintero stages II and III with MCDA twins during 2004–2015. The study was approved by the hospital IRB committee (No. 04-220; clinical trial identification No. NCT04148859), and informed consents were signed by the participants. The recruited patients were counseled about the two most common treatment procedures – FLA and AR – and the choice for AR treatment was done by the mother. All the surviving twins were recruited for a long-term neurodevelopmental follow-up. The following maternal and perinatal data were collected and summarized in Table 1: maternal age, parity, stage of TTTS at diagnosis, gestational age at the first controlled AR procedure, number of controlled AR procedures, volume removed per one AR procedure, gestational age at delivery and birth weight for each twin.
Clinical characteristics of the patients with TTTS that were intervened with the controlled amnioreduction and the newborn twins.
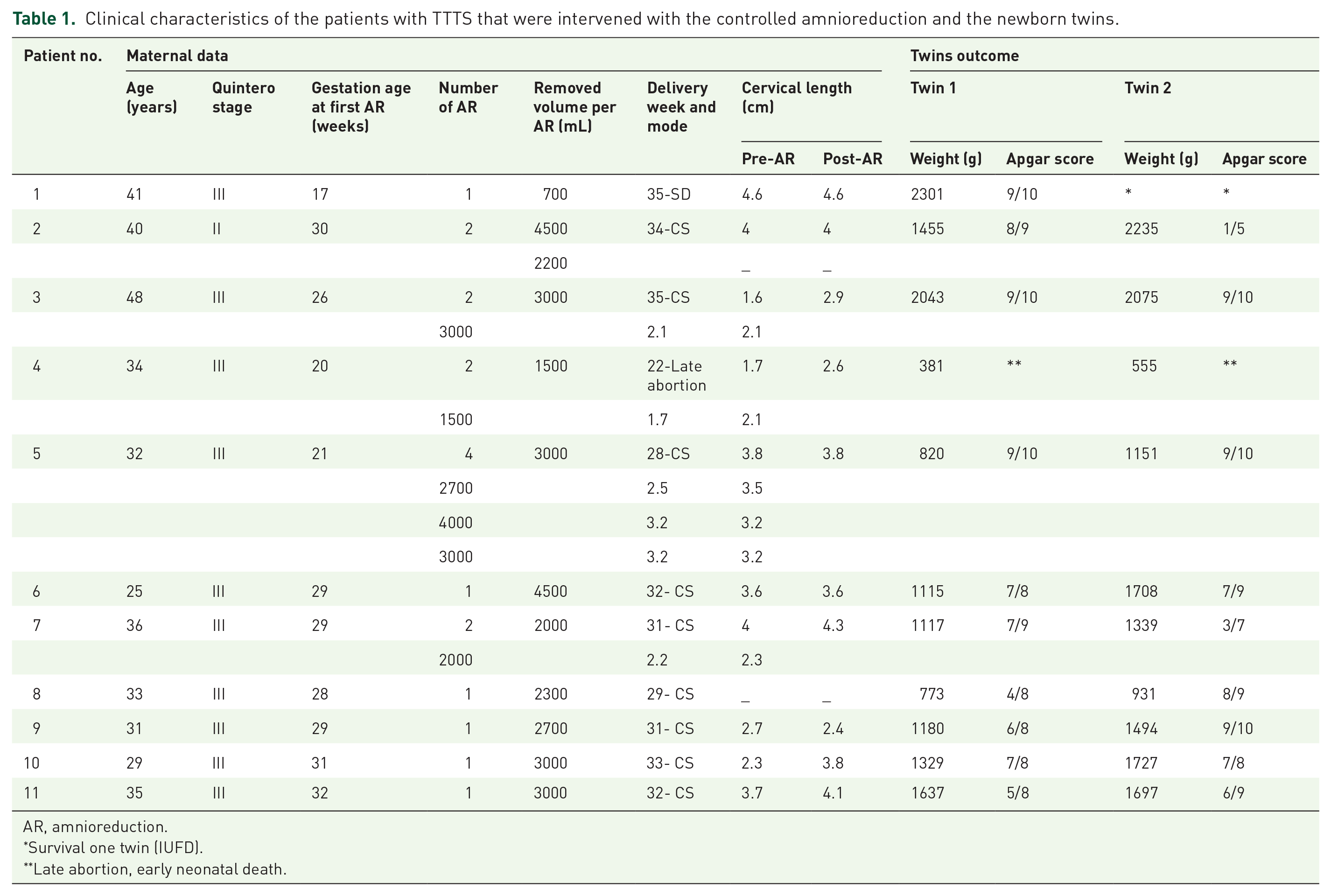
AR, amnioreduction.
Survival one twin (IUFD).
Late abortion, early neonatal death.
Controlled AR procedure
The protocol of the controlled AR included simultaneous recording of the removed volume of amniotic fluid and the amniotic pressure. The cervical length and the flow velocity waveform (FVW) at the twins’ umbilical cords were measured pre-AR and post-AR. The procedure was performed via an epidural 18 G hypodermic needle under continuous transabdominal ultrasound guidance while the patient is in the supine position and local maternal anesthesia (Figure 1). The excess amniotic fluid was drained into a sterile bag through a plastic tube and a three-way stopcock attached to the hub of the needle. The removed volume of fluid was recorded every 500 mL and followed by measurement of the amniotic pressure.

Schematic description of the controlled amnioreduction procedure. The three-way stopcock is in position to measure the amniotic pressure with the manometer. Turning the stopcock 90° counterclockwise allows for amniodrainage into the bag.
The amniotic pressure in the polyhydramniotic sac was measured by a water manometer at the level of the needle tip. The stopcock was open to the atmosphere to allow for the amniotic fluid to flow from the uterus into the manometer tube. After stabilization within a few seconds, the pressure was acquired in cmH2O and converted to mmHg by a factor of 0.74. Immediately after reading the pressure, the tap was closed and the fluid re-directed into the sterile bag. The twins FVWs were measured with a Doppler ultrasound machine (Sonoline Elegra, Siemens and GE Voluson E6) at the free loop of the umbilical cord. At least five sequential normally shaped FVWs were acquired, and the umbilical artery systole/diastole (S/D) ratio was calculated. Cervical lengths were measured before and at the end of the procedure.
In case of uterine contractions prior to the procedure, the patients received Indomethacin (PR, 100 mg). In case of repeated uterine contractions, the amniotic pressure was recorded immediately after complete relaxation. The procedure was terminated when the amniotic pressure did not change after two sequential drainages of 500 mL each or in case of maternal discomfort. The procedure was repeated only if TTTS occurred again.
Long-term neurodevelopmental follow-up
The long-term neurodevelopmental outcomes in the surviving twins were evaluated for 2 up to 13.5 years using a parent survey. The questionnaire for this study was designed as semi-opened questions in Hebrew that covered the following 10 subscales of neurodevelopment: gross motor, fine motor, vision, hearing, speech, self-care, emotional development, memory/learning, cognitive development, pain, or physical discomfort. The parents reported on their twins’ neurodevelopmental status at the time of follow-up and the information was rated on a three-point scale of categorical qualifying variables: optimal (i.e. normal), suboptimal (i.e. mild anomalies without functional deficits) and abnormal (i.e. neurological and/or functional impairment or deficits). The semi-structured phone interview was conducted by co-author AJJ and the content of responses was analyzed by co-author AFV.
Statistical analysis
Descriptive statistic was used for the demographic, clinical, obstetrical, and perinatal parameters. The survival rate and neurodevelopmental outcomes were discussed in comparison to published reports.
Results
We conducted 18 controlled AR procedures due to severe TTTS following the choice of the mother. All the cases were free of maternal infection or complications, as well as premature delivery within 48 h of the intervention. Clinical and perinatal outcome characteristics of the study population are presented in Table 1.
Ten of 11 TTTS cases were categorized as Quintero stage III (91%). The gestational age at the first AR treatment ranged from 17 to 32 weeks, with a mean age of 26.8 weeks. The volume of amniotic fluid removed during a single session varied between 700–4500 mL. The time required to perform the procedure ranged from 20 min to more than 2 h, in accordance with the drained volume. Five patients (45.5%) were treated with the repeated controlled AR, so that the number of amniocenteses ranged from one to four per pregnancy. In all cases, births were premature with delivery at 28–35 gestational weeks. The mean donor weight was 1185 ± 470 g, and the mean recipient weight was 1566 ± 547 g. Nine out of 11 twin pairs were born alive. The outcomes in the other two pregnancies were as follows: a spontaneous delivery at week 35 with one surviving twin and the other macerated stillbirth, and a late abortion in week 22 with intrauterine death of one fetus and the co-twin died soon after birth. Therefore, the total survival rate was 19 out of 22 twins (86.4%). The pre- and post-AR cervical lengths are summarized in Table 1. In six mothers, it increased, while in the rest, it remained unchanged within the normal range.
Amniotic pressures were measured prior, during, and at the end of the AR procedure as depicted in Figure 2. In this figure and the following figures, each participant was assigned the same color. Generally, the amniotic pressure decreased during removal of excess amniotic fluid and reached a plateau toward the end of the procedures, except five patients in which AR was interrupted due to patient discomfort. We did not find a correlation between the amount of drained amniotic fluid, duration of the AR procedure, and the end plateau pressure. The amniotic pressure drop at each AR procedure is shown in Figure 3 along with the reference range for singleton pregnancies. 25 The pre-AR pressures (i.e. squares) for all patients were higher than the normal range. As expected, in most cases (61.1%) the post-AR pressures (i.e. open circles) were lower than the pre-AR pressures (i.e. open squares) and within the reference range while slightly higher in 38.9%.

The amniotic pressure versus the drained volume of amniotic fluid during the controlled AR procedure. Solid curves represent completed procedures; Dash curves represent interrupted procedures. The different colors represent the different patients listed in Table 1.

Pre-AR (squares) and post-AR (circles) amniotic pressures at the gestation week the controlled AR was performed. The arrows represent the direction of change due to the controlled AR. The black dashed curves represent the 5% and 95% percentiles of normal amniotic pressures during singleton pregnancies. The colored symbols and curves represent the same patients as shown in Figure 2.
The umbilical artery S/D values of each fetus were measured pre-AR (i.e. open squares) and post-AR (i.e. open circles) and are depicted in Figure 4 along with the reference range for singleton pregnancies. 30 For each procedure, which is assigned by the week of gestation it was performed, we provided two lines of the S/D ratio change for each of the two fetuses. The initial pre-AR S/D ratio of recipient was higher in three cases (mean percent difference is 29.3%), while in the rest cases, the donor twin had the higher S/D ratio. Absence of end diastolic flow (AEDF) occurred in three donors, and thus, an arbitrary value of 9 was given as the initial one for graphic representation. All these donors showed hemodynamic improvement after the AR (Figure 3). In general, most of the post-AR S/D ratio values (23 of 36, i.e. 63.8%) were within the normal range according to the given weeks of gestation. Comparison of inter-twin differences of S/D ratio values in pre- and post-AR did not show a particular tendency: inter-pair differences were significantly decreased after the procedure in seven twins, increased in five twins and were unchanged in six cases.

Pre-AR (squares) and post-AR (circles) umbilical artery S/D ratios in each of the fetuses at the gestation week the controlled AR was performed. The arrows represent the direction of change due to the controlled AR. The black dashed curves represent the 5% and 95% percentiles of normal S/D ratios during singleton pregnancies. The colored symbols and curves represent the same patients as shown in Figure 2.
The neurodevelopmental status of all the 19 surviving twins was followed for 2–13 years with a mean age of 7 years at the time of the survey. Of the 19 tested children, 13 (68.4%) were reportedly optimal, 5 children (26.3%) were scored as suboptimal and one (5.3%) was rated abnormal due to cerebral palsy. Examples of suboptimal neurological development included mild anomalies in vision, speech, dexterity, learning, and/or remembering. The relatively high survival rate with very little long-term neurodevelopmental impairment demonstrated improved outcomes with the controlled AR treatment of TTTS pregnancies.
Discussion
Drainage of the amniotic fluid was gradually performed while the amniotic pressure was monitored. This controlled procedure of AR was successfully applied to 11 pregnant women diagnosed with severe TTTS up to the 32nd week. Termination of the procedure was based on the amniotic pressure measurements, rather than the residual fluid volume which is estimated from ultrasound imaging. The overall survival rate of both twins was relatively high (86%), while none of the cases was affected by maternal infection, complications, or premature delivery within 48 h post-AR.
The total removed volume of amniotic fluid per AR session in this study varied between 700 and 4500 mL (Figure 2) with an estimated error of 5% for the described methodology. This range is within the published data of volumes from 400 to 7500 mL, which are as high as four times of the mean volume for any given gestational age.20,31–33 The huge range of the removed volume of fluid clearly demonstrated that the TTTS is patient-dependent, and consequently, the intervention should be tailored to the patient’s measurable physical characteristics.
The present results revealed that gradual removal of the excess amniotic fluid decreased the amniotic pressure with an exponential pattern that leveled-off with a plateau (Figure 2). The pre-AR amniotic pressures were higher than the reference range for normal pregnancies 25 (Figure 3), similar to previously published data.26,34–38 The mean difference between pre- and post-AR pressure was 9.1 ± 5.4 mmHg. Along with reduction of the amniotic pressure, the cervical length increased in more than half of the subjects.
The umbilical artery S/D ratio in uncomplicated twin pregnancy shows close agreement with the normal range for healthy singleton pregnancies. 39 Reduction of elevated amniotic fluid volume in polyhydramnios decreases the amniotic pressure which may lead to increased flow from the placenta to the fetus, if all other characteristics are unchanged. 40 In this study, the initial umbilical artery S/D ratio was above the 95th percentile of the normal range in 10 cases and within it in 8 cases (Figure 4). The most noticeable positive effect of the procedure on FVW was observed in three cases of severe TTTS with initial AEDF (i.e. patients 1, 4, and 8), but the post-AR S/D ratio did not reach the normal range for the given week of gestation. Nevertheless, this outcome is in agreement with previous reports on higher fetal mortality in cases of AEDF in either, the donor or the recipient.6,7,20,41 The post-AR S/D ratio changed to be within the normal range in 63.8% of cases, below it in four cases (11.2%) and above it in the remaining nine cases (25%). We did not observe a tendency in the S/D ratio changes due to the procedure. Nevertheless, the gradual drainage most likely allowed for better mechanical adaptation of the communicating vessels in the twins common placenta, and thereby, yielded improved regeneration of the balance between the bilateral blood circulations of the twins.
The prognosis of untreated severe TTTS is usually the loss of both twins. Accordingly, any intervention that leads to at least one alive twin is considered as a clinical success.1,2 Review of the published outcome of managed TTTS pregnancies revealed an overall success of live twins in the range of 53–78%5–9 post-AR29,36,42,43 and 50–88% post-FLA.8–10,13 This study with the controlled AR protocol demonstrated an impressive overall success with 86% survival of the twins beyond 6 months of age. The best way for classification and management of twin pregnancies is currently still debated with significant global variations.44,45 The present study is not a comparative analysis, and we believe that the TTTS intervention should be left for the patient’s choice subjected to providing all the known information.
The large variability of removed volumes of amniotic fluid in this study and in published reports indicates that draining of excess amniotic fluid is patient specific. Moreover, this significant inter-subject variability brings back to the frontline the unexplored questions such as how much of the amniotic fluid should be removed? or what may be a functional and objective criteria for termination of the fluid drainage? Currently, the recommended guidelines for termination of the fluid draining are based on imaging of the deepest vertical pocket which yields an estimation of the residual amniotic volume.2,4 In this study, we demonstrated that the amniotic pressure reaches a plateau during gradual fluid drainage and further draining of fluid is redundant. Thus, simultaneous continuous measurement of the amniotic pressure can also serve for objective termination of the AR procedure.
We also followed the long-term neurodevelopment of all the surviving twins by a semi-structured open interview. The analysis revealed that over 2/3 of the twins were neurologically optimal, about 1/4 demonstrated minor neurodevelopmental impairment and only one child (about 5%) was with a cerebral injury. The same parents responded to the same questionnaires twice, once for the donor twin and then for the recipient, and accordingly, we assume that the received data are consistent. However, this information is subjective and qualitative, and thus, we consider it to be similar to published outcomes.9,10,13,43,46
This study was based on manual measurements of the amniotic pressure and the removed volume of amniotic fluid with estimated errors of about 5%. Future studies should upgrade the methodology for computerized acquisition of the pressure and removed volume of the amniotic fluid with solid-state transducers similar to urodynamic devices. The relatively small group of this study is certainly another limitation. However, recruitment of subjects for long-term follow-up of years is obviously an obstacle as in similar publications. This study is not a comparative clinical study, and thus, the absence of a control group seems to be negligible. It is obvious that neither the healthcare provider nor the patient will take the risk of no management of severe TTTS. Another issue is the patient’s posture during the procedure, which may affect both the pressure and the fluid flow rate. In this study, removal of the excess amniotic volume was done by gravitational drainage. Some form of active removal of the fluid may be considered to shorten the intervention protocol.
In conclusion, we utilized controlled AR procedures for the management of TTTS pregnancies up to the 32nd week of gestation. The intervention protocol was based on gradual drainage of the amniotic fluid while the pressure and removed volume of the amniotic pressure were simultaneously measured. Analysis of the descending amniotic pressure as the removed amniotic fluid volume increases revealed exponential decay with a final plateau, and thus, further draining of amniotic fluid would have a negligible effect on the amniotic pressure. We used the final plateau of the amniotic pressure as an indicator for termination of the intervention, unless it was terminated due to the patient’s request. The overall outcome of this controlled AR management of TTTS revealed excellent survival of healthy twins with very little neurodevelopmental impairments.
Footnotes
Author contributions
AJJ conceived and supervised the study. The experiments were carried out by AJJ and ZG. AFV developed the questionnaire and analyzed the long-term neurodevelopmental data. All authors contributed to the data analysis and interpretation of the data. DE, AJJ, and ZG revised the manuscript together. All authors reviewed and approved the final submitted version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Compliance with ethical standards
Informed written consent was obtained from all participants under protocol approved by the Tel-Aviv Sourasky Medical Center IRB committee (No. 04-220; clinical trial identification No. NCT04148859).
