Abstract
Corrosion to metal is a great challenge to major industries. Anticorrosive coatings can effectively prevent metal corrosion. In this study, we propose a novel method to prepare silica nanoparticles-covered graphene oxide (SiO2-GO) nanohybrids and anticorrosion SiO2-GO/waterborne polyurethane acrylic (WPUA) coatings. Firstly, we obtained silane-functionalized graphene oxide (A-GO) via a simple covalent functionalization of graphene oxide (GO) with 3-aminopropyltriethoxysilane. Secondly, SiO2-GO was synthesized by a simple sol–gel method with tetraethoxysilane in water–alcohol solution. Finally, the obtained SiO2-GO nanofillers were added into WPUA to prepare SiO2-GO/WPUA coatings. GO, A-GO, and SiO2-GO nanohybrids could be confirmed by X-ray diffraction, Fourier transform infrared spectroscopy, Raman spectra, and transmission electron microscope. SiO2-GO nanohybrids showed small size compared with the unfunctionalized GO. Meanwhile, GO, A-GO, and SiO2-GO nanofillers were added into WPUA. The electrochemical impedance spectroscopy and field emission scanning electron microscope indicate that SiO2-GO nanohybrids can be homogeneously dispersed in the WPUA coatings at 0.4% loading level and the SiO2-GO/WPUA film exhibits excellent anticorrosion performance. SiO2-GO nanoparticles can effectively utilize in the area of anticorrosive nanofiller industry. This study provides a convenient method of anticorrosive coating production.
Keywords
Introduction
Corrosion to metallic structures is a great challenge to all industries. Organic coatings can be applied to protect metallic structures from corrosion. It is generally believed that organic coatings can form a physical barrier between metal substrate and corrosive environment. 1–3 With increasing health and environment awareness in recent years, waterborne polyurethane acrylic (WPUA) coatings have attracted more attention in environment-friendly coating industry. Compared to traditional coatings, WPUA coatings are favorably chosen due to low volatility, good physical and mechanical properties, durability, elasticity, and temperature-dependent hardness. 4,5 Nevertheless, WPUA coatings have poor resistance to water and weather. In addition, the poor adhesion between WPUA coatings and steel limits the practical application. 6 Extensive studies have been carried out to tackle the anticorrosion efficiency of WPUA coatings. 7,8
Graphene oxide (GO) is a two-dimensional nanomaterial of sp2-hybridized carbon atoms. 9 Graphene is well-known for its high stiffness and excellent thermal stability and electrical conductivity. 10 Recent studies indicate that GO can improve barrier performance and anticorrosion property of coatings. 11 –14 However, small particle size and large specific surface area of GO can lead to aggregation, which will eventually damage the barrier performance of WPUA coatings. Therefore, it is important to disperse graphene in polymer coatings homogeneously. 14 Fortunately, abundant functional groups, such as hydroxyls, epoxides, diols, ketones, and carboxyl groups, can be potentially modified. One of the solutions is grafting GO with nanoparticles preventing the GO aggregation. 15 An incomplete list of effective nanoparticles for GO surface modification may include platinum, silica, titanium oxide, aluminum oxide, zinc oxide, and silver. These nanoparticles can form space among GO sheets and hence prevent agglomeration effectively. 16 Consequently, nanoparticles grafting to GO are widely reported in recent studies. Pourhashem et al. 17 grafted 3-aminopropyltriethoxysilane (APTES) and 3-glysidyloxypropyl trimethoxysilane onto GO sheets to investigate the effect of silane type on the properties of nanocomposite coatings. They found that APTES-grafted GO sheets could endow composite coatings with better anticorrosion property on mild steel substrate. Tian et al. 1 prepared functionalized graphene nanoplates, which were modified by silane-coupling agent with a sol–gel method. Then, graphene nanoplates were incorporated in the nanocomposite coatings, which show excellent anticorrosion properties. Ramezanzadeh et al. 18 decorated GO with silica nanoparticles by a two-step in-situ sol–gel process, and silica nanoparticles-covered graphene oxide (SiO2-GO) nanohybrids showed better dispersion in the epoxy coating. Yu et al. 19 synthesized titanium dioxide (TiO2)-GO sheet hybrids with APTES, and TiO2-GO hybrids were incorporated in the epoxy coatings, which show better anticorrosion properties.
In this study, we utilized APTES to obtain silane-functionalized graphene oxide (A-GO), which are characterized by Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), Raman spectra (Raman), and transmission electron microscopy (TEM). As the next step, SiO2-GO nanohybrids are synthesized with tetraethyl orthosilicate (TEOS) in water–alcohol solution and characterized by FTIR, XRD, Raman, and TEM techniques. Finally, GO, A-GO, and SiO2-GO nanohybrids were added into WPUA to investigate the corrosion protection efficiency of WPUA coatings by electrochemical impedance spectroscopy (EIS) analysis.
Experimental
Materials
GO powder (approximately 1–5 µm) was purchased from Suzhou Tan feng Graphene Technology Co., Ltd (Suzhou, Jiangsu, China). Two-component WPUA resin coatings were supplied by Jinhua Yitu New Materials Co., Ltd. (Jinhua, China) APTES, absolute ethanol, acetic acid, and TEOS were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China) 316 stainless steel (6 cm × 6 cm × 60 µm).
Synthesis of A-GO
APTES (3 g) was added into 80 g alcohol–water solution (1:1 wt%/wt%) and the pH of mixture was adjusted at 4–5 by acetic acid. The obtained mixture was stirred at 38°C for 0.5 h. Subsequently, GO (0.1 g) was added into the obtained mixture and stirred at 60°C for 6 h. Finally, the mixture was filtrated with anhydrous ethanol and deionized water before dried at 60°C for 24 h.
Synthesis of SiO2-GO nanohybrids
TEOS (0.5 mL) and the ammonia water (120 µL) were added into 60 g alcohol–water solution (5:1 wt%/wt%), and then, the mixture was stirred for 0.5 h at room temperature. Subsequently, A-GO (0.05 g) was added into the above mixture and stirred at room temperature for 12 h. Finally, the mixture was filtrated with anhydrous ethanol and deionized water before dried at 60°C for 24 h. Figure 1 shows the schematic synthesis route of SiO2-GO nanohybrids.

The synthesis route of SiO2-GO. SiO2-GO: silica nanoparticles-covered graphene oxide.
Preparation of nanocomposite coatings
Nanofillers were dispersed in 6 g A component of WPUA coatings stirred for 12 h at room temperature. Then, 1 g B component of WPUA was added into the above mixture and stirred for 0.5 h at room temperature. Subsequently, the prepared coatings were coated on 316 stainless steel, which had already been cleaned by sanding and immersed in absolute ethanol for 20 min before use. And then, coated substrates were dried at 120°C, with coatings thickness of 60 ± 5 µm.
Characterizations
GO, A-GO, and SiO2-GO nanofillers were characterized by XRD technique (3KW D/MAX2200 V PC, copper K α), FTIR spectroscopy (NicoletiS10), and confocal microscopic Raman spectrometer (Raman, inVIA). The morphology of GO, A-GO, and SiO2-GO nanohybrids was observed by TEM (200CX). The contact angle of deionized water droplet (2 µL) on the coated substrate was measured at 25°C. Field emission scanning electron microscope (FE-SEM) was employed to observe dispersion of nanofillers in the matrix.
Corrosion test
EIS measurements were carried out by Parstat-2273 electrochemical workstation. Three electrode cells were used for electrochemical measurements. The coated substrate served as a working electrode with 1 cm2 working area. Platinum rod was used as a counter electrode and Ag/AgCl electrode as the reference electrode. The corrosive test was carried out in 3.5 wt% sodium chloride (NaCl) solution. EIS test was performed using AC amplitude in the frequency range of 100 kHz–10 MHz after different soaking times of 1, 14, and 28 days, respectively. Then, ZSimpWin software was employed to evaluate obtained the impedance data.
Result and discussion
Characterization of GO, A-GO, and SiO2-GO nanohybrids
XRD was employed to characterize the interlayer spacing of GO, A-GO, and SiO2-GO. The XRD patterns of GO, A-GO, and SiO2-GO are shown in Figure 2. The sharp peak at 2θ around 11.02° corresponds to (001) diffraction peak of GO. The sharp peak of GO sheets reveals that there are highly ordered structures in GO. The characteristic diffraction peak of 11.02° vanishes after modified by silane-coupling agent. A-GO presents a diffraction peak nearly 6.78°, indicating that the interlayer space of A-GO increases to 1.39 nm. Another peak of A-GO locates around 22.18°, which indicates that the interlayer space of (002) direction is 0.34 nm. The hump in SiO2-GO at 2θ about 21.84° is attributed to the amorphous structure and disordered surface of SiO2 after the successful combination between GO and TEOS. 15

XRD spectra of GO, A-GO, and SiO2. XRD: X-ray diffraction; SiO2: silica; A-GO: silane-functionalized graphene oxide; GO: graphene oxide.
Raman spectroscopy was employed to distinguish between ordered and disordered crystal structures of carbon. The D band reveals defects and disorder carbon. Raman patterns of GO, A-GO, and SiO2-GO are shown in Figure 3. The D and G characteristic peaks of GO can be observed in Raman spectra. Significantly, the G band of GO is 1581 cm−1 and shifts to 1587 cm−1 after chemically grafted with silane molecules. Furthermore, a high I D/I G ratio indicates defect concentration and disorder. I D/I G ratios for GO, A-GO, and SiO2-GO are 1.03, 1.07, and 1.10, respectively, which proves that the degree of disorder and defect concentration increase after GO combined with TEOS. 20 The full width at half maximum of GO, A-GO, and SiO2-GO is shown in Figure 3. The full width at half maximum of A-GO and SiO2-GO get significantly wider than that of GO, indicating that the degree of disorder and defect concentration increase after modification.
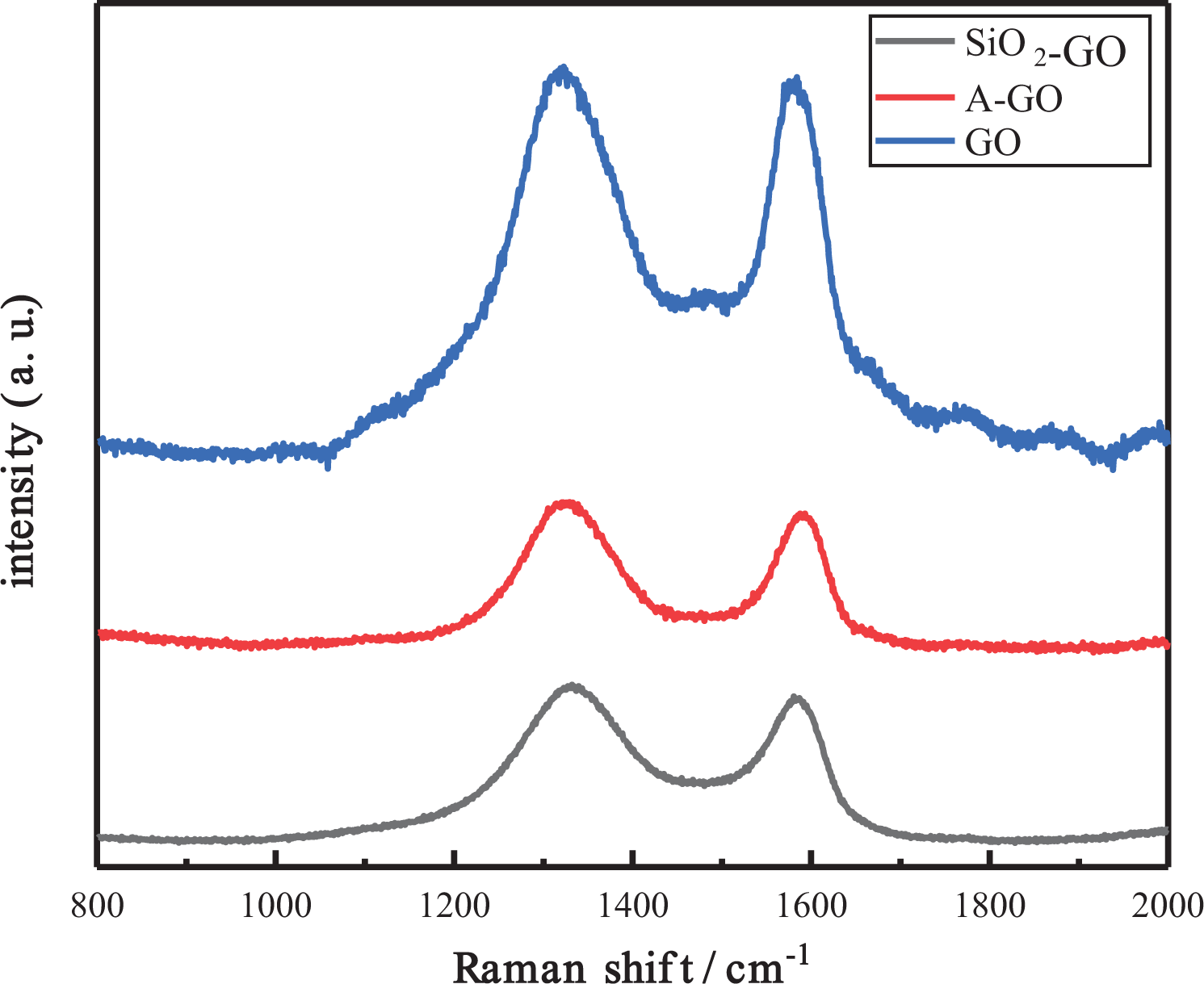
Raman spectra of GO, A-GO, and SiO2-GO. SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide; GO: graphene oxide.
The FTIR spectra of GO, A-GO, and SiO2-GO are shown in Figure 4. GO sample presents the following characteristic absorption peaks at 3415 cm−1 (OH), 2918 cm−1 (CH2), 1719 cm−1 (C=O), 1617 cm−1 (C=C), 1225 cm−1 (COO), and 1057 cm−1 (C–O–C). After GO was modified with APTES, the new characteristic absorption peaks appear at 3277 cm−1 (NH2), 1113 cm−1 (Si–O–Si), and 702 cm−1 (Si–O–C). Besides, the characteristic absorption peaks locate at 2926 and 2889 cm−1 corresponding to symmetric and asymmetric vibrations of CH2 group of A-GO. For SiO2-GO, new peaks appear at 3405 cm−1 (NH2), 1116 cm−1 (Si–O–Si), 1036 cm−1 (Si–O–C), and 916 cm−1 (Si–OH). 21 Moreover, there is also a new peak at 462 cm−1 (Si–O–Si). A-GO combination with TEOS leads to the symmetric and asymmetric vibrations of CH2 groups at 2925 and 2851 cm− 1. In addition, Si–O–Si bond shows the strongest peak, suggesting the successful combination between SiO2 and A-GO.

FTIR spectra of GO, A-GO, and SiO2-GO. FTIR: Fourier transform infrared; SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide; GO: graphene oxide.
Figure 5 shows the morphology of GO, A-GO, and SiO2-GO, respectively. The difference between GO and A-GO can be inferred in Figure 5(a) and (b). The size of GO gets smaller and the wrinkle of GO has decreased after the APTES grafted onto GO. It proves that the flake morphology of GO sheets has changed after the reaction with silane-coupling agents. The wrinkle of SiO2-GO shown in Figure 5(c) indicates the layered structure and morphology of GO. According to the previous studies, 15,21,22 the modification of GO surface with nanoparticles leads to superb dispersion and exfoliation of GO sheets.

TEM images of (a) GO, (b) A-GO, and (c) SiO2-GO. SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide; GO: graphene oxide; TEM: transmission electron microscopy.
Dispersion of SiO2-GO hybrids in WPUA coatings
To trace the surface and dispersion of GO, A-GO, and SiO2-GO hybrids in WPUA coatings, FE-SEM is employed. Figure 6 shows the surface of pure coatings, 0.4 wt% GO/WPUA, 0.4 wt% A-GO/WPUA, and 0.4 wt% SiO2-GO/WPUA. Figure 6(a) shows a glossy and smooth surface of the pure WPUA coatings. However, nonuniform density distributions and unsmooth surface can observe on coatings with GO and A-GO (Figure 6(b) and (c)), respectively. There is an abundant agglomeration in 0.4 wt% GO/WPUA coatings. Less agglomeration was observed in 0.4 wt% A-GO/WPUA coatings. It confirms that APTES could assist the dispersion of GO in the coatings. According to Figure 6(d), the surface of 0.4 wt% SiO2-GO/WPUA coatings is glossy and smooth, showing well-dispersed SiO2-GO hybrids. These results show that SiO2-GO hybrids perform better dispersion and exfoliation in the WPUA coatings. And better dispersion may help coatings get better anticorrosion performance. 21,23

SEM patterns of (a) pure coatings, (b) GO/WPUA, (c) A-GO/WPUA, and (d) SiO2-GO/WPUA. SEM: scanning electron microscopy; GO: graphene oxide; SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide; WPUA: waterborne polyurethane acrylic.
Corrosion resistance of coatings
EIS reveals the failure process of the studied coatings, which could be divided into three stages. At the initial immersion stage, coatings prevent water from reaching the metal. Corrosive substances permeate into coatings and enhance the capacitance and resistance of coatings. At the middle immersion stage, the corrosive substances reach the coating/metal boundary and form a microcorrosive battery. At this stage, the corrosion process of the metal substrate takes place and the adhesion of coatings declines, leading to coating blister and failure of adhesion to metal. However, no macroscopic pore can be observed at this stage. At last, the formation of rust spots and macroscopic pores at the final immersion stage indicates the failure of protection. Figure 7 shows the Nyquist, Bode, and phase angle spectra of pure coatings, 0.4 wt% GO, 0.4 wt% A-GO, and 0.4 wt% SiO2-GO coatings, respectively, after exposing to 3.5 wt% NaCl for 1, 14, and 28 days. Figure 8(a) to (c) show electrochemical impedance modeling of the initial immersion stage, the middle immersion stage, and the final immersion stage, respectively. While C c, R c, R s, C dl, R t, and R po represent coating capacitance, coating resistance, solution resistance, double-layer capacitance, charge transfer resistance, and pore resistance, respectively, 24 Figure 7(a), (b), (b1), and (c) clearly reflect the corrosion process. Figure 7(b) and (b1) shows one time constant, and Figure 7(c) shows one peak, which indicates the corrosion process at the initial immersion stage. It reveals that all the coatings reach the initial process after soaking for 1 day. Table 1 presents |Z|0.01 Hz (Ω·cm2) of all the coatings, which reflect corrosion resistance of coatings at low frequency, and the high value represents better anticorrosion performance. The |Z|0.01 Hz of 0.4 wt%SiO2-GO/WPUA coatings is 1.39 × 1010 Ω·cm2, showing better anticorrosion performance. Figure 7(e) and (e1) of pure coatings show two times constant and Figure 7(f) of pure coatings show two peaks, which indicate the corrosion process at the middle immersion stage. The |Z|0.01 Hz of pure coatings is 7.96 × 107 Ω·cm2 after 14 days of immersion. It indicates that pure coatings reach the middle immersion stage and other coatings reach the initial immersion stage after 14 days of immersion. It can be inferred that corrosive substances permeate into coatings leading to the failure of coating. Figure 7(h) and (h1) of 0.4 wt% SiO2-GO/WPUA coatings shows one time constant and Figure 7(i) of 0.4 wt% SiO2-GO/WPUA coatings shows one peak, which indicates corrosion process at the initial immersion stage after 28 days of immersion. The |Z|0.01 Hz of 0.4 wt% SiO2-GO/WPUA coatings is 9.46 × 108 Ω·cm2 after 28 days of immersion, showing better anticorrosion performance. The |Z|0.01 Hz of pure coatings declines from 108 Ω·cm2 to 105 Ω·cm2 after soaking in 3.5 wt% NaCl for 28 days. The |Z|0.01 Hz of other coatings decline substantially expecting 0.4 wt% SiO2-GO/WPUA coatings. These changes might be related to new emerging defects caused by a corrosive attack. EIS results show that 0.4 wt% SiO2-GO/WPUA coatings exhibit an excellent anticorrosion property.
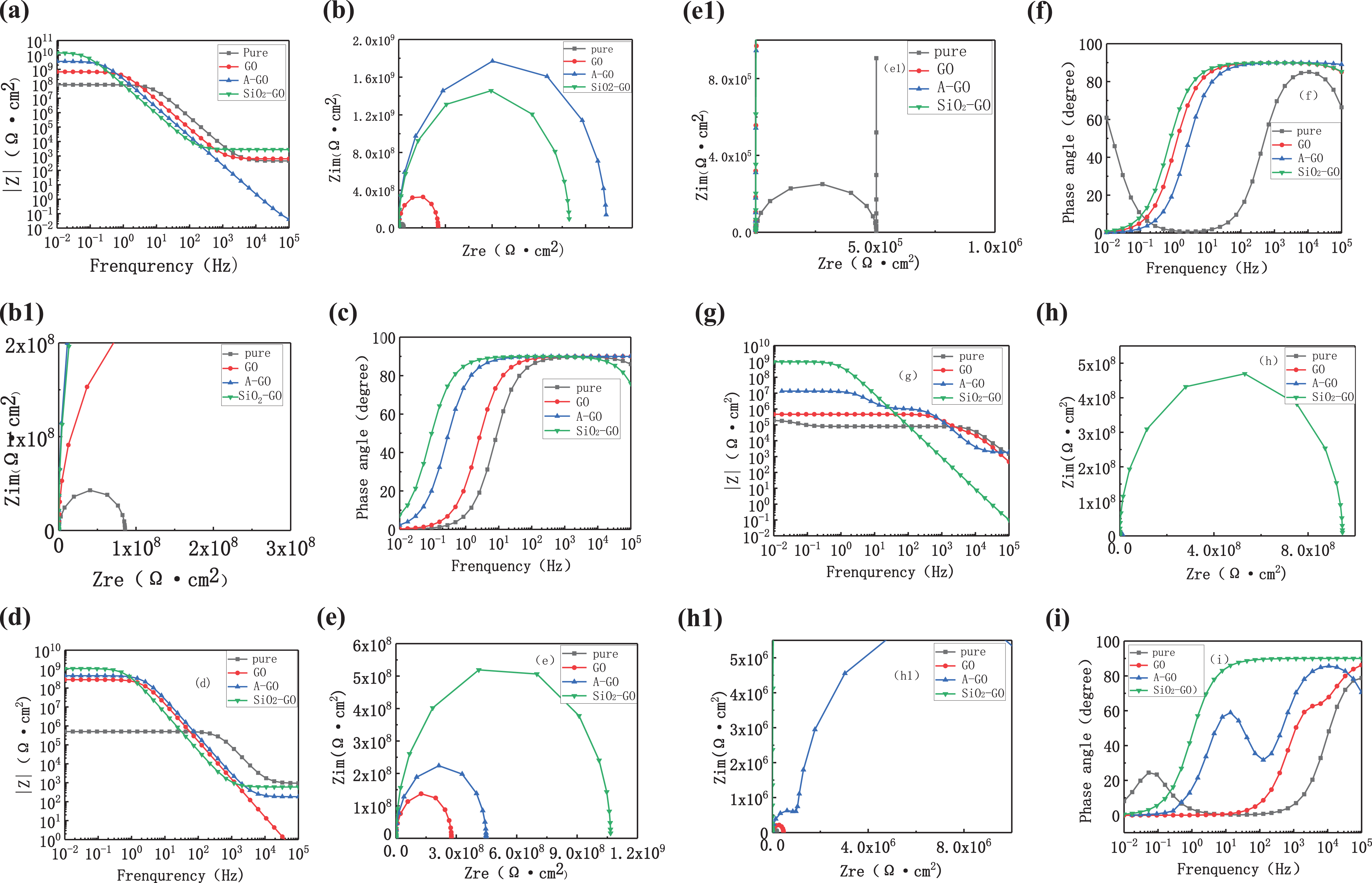
EIS measurements of the pure coatings, 0.4 wt% GO, 0.4 wt% A-GO, and 0.4 wt% A-GO-SiO2 coatings at different soaking time (a)–(c) soaking for 1 day, (d) –(f) soaking for 14 days, and (g)–(i) soaking for 28 days. (b1), (e1), and (h1) are larger images of (b), (e), and (h), respectively. GO: graphene oxide; SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide; EIS: electrochemical impedance spectroscopy.

Electrochemical impedance modeling: (a) electrochemical equivalent circuit for noncorrosive arising on metal substrates and (b, c) electrochemical equivalent circuit for corrosion arising on metal substrates.
Bode 0.01 Hz |Z| (Ω·cm2).

GO: graphene oxide; SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide.
Figure 9 shows the water contact angle of pure coatings, 0.4 wt% GO, 0.4 wt% A-GO, and 0.4 wt% SiO2-GO/WPUA coatings, respectively. The water contact angles of pure coatings, 0.4 wt% GO, 0.4 wt% A-GO, and 0.4 wt% SiO2-GO/WPUA coatings are 73.1° ± 1°, 82.9 ± 1°, 87.9 ± 1°, and 95.5° ± 1°, respectively. The contact angle represents the hydrophily of the surface. Water contact angle of 0.4 wt% SiO2-GO/WPUA coatings is higher than the others, suggesting that corrosive substances immersed into 0.4 wt% SiO2-GO/WPUA coatings are postponed.

Water contact angle of (a) original coatings, (b) containing 0.4 wt% GO, (c) 0.4 wt% A-GO, and (d) 0.4 wt% SiO2-GO hybrids nanocomposite coatings. GO: graphene oxide; SiO2-GO: silica nanoparticles-covered graphene oxide; A-GO: silane functionalized graphene oxide.
Figure 10 shows the SiO2-GO hybrids structure and Figure 11 shows the schematic of anticorrosive property of 0.4 wt% SiO2-GO/WPUA coatings. Organic coatings act as an effective barrier to prevent the solution from reaching the metal. The micropores on the coating surface assist the diffusion of corrosive substances, leading to metal corrosion. Adding SiO2-GO nanohybrids into WPUA coatings can dramatically reduce the amount of micropores. As the channel of invasion is hindered, the coating with SiO2-GO exhibits excellent anticorrosion property. Compared to GO and A-GO nanohybrids, SiO2-GO nanohybrids have a better dispersion in WPUA coatings, which significantly improve the corrosion resistance. Therefore, the aggregation of SiO2-GO nanohybrids is significantly declined. It has been reported in previous studies that silane-coupling agent in coatings can lead to excellent adhesions and anticorrosive performances. 25,26 Therefore, SiO2-GO nanofillers not only perform good dispersion but also improve the adhesion between metal and coatings. These evidences prove that 0.4 wt% SiO2-GO nanofillers can substantially enhance anticorrosive performance than others.

Synthetic SiO2-GO hybrids structure. SiO2-GO: silica nanoparticles-covered graphene oxide.

Schematic of anticorrosive property of SiO2–GO coatings. SiO2-GO: silica nanoparticles-covered graphene oxide.
Conclusions
In conclusion, we provide a sol–gel method to prepare SiO2-GO and anticorrosion SiO2-GO/WPUA coatings. XRD, Raman, FTIR, and TEM suggest the successful combination between SiO2 and A-GO. SEM spectra show well-dispersed SiO2-GO hybrids in WPUA coatings. SiO2-GO nanofillers exhibited excellent dispersion in coatings. Water contact angle of 0.4 wt% SiO2-GO/WPUA coatings is 95.5° ± 1°. The |Z|0.01 Hz of 0.4 wt% SiO2-GO/WPUA coatings is 9.46 × 108 Ω·cm2 after 28 days of immersion, showing better anticorrosion performance. SiO2-GO nanoparticles can effectively utilize in the area of anticorrosive nanofiller industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of China [No. 61403245].
