Abstract
Fe52Cr15Mo26C3B1Y3 amorphous particle-reinforced pure aluminum (Al) matrix composite was prepared by powder metallurgy. The ferrum (Fe)-based amorphous particles prepared by atomization method have good amorphous structure, and the circular reinforcement particles are evenly distributed in the Al matrix. The composite has high strength, hardness, and excellent corrosion resistance. The hardness of the composite increases gradually with the increase in the content of the reinforcement, from 46 Vickers hardness (HV) of pure Al to 220.5 HV, with remarkable effect. The tensile strength of the composite increases first and then decreases with the increase in the content of the reinforcement. When the content of reinforcement is 15%, the maximum tensile strength is 234 MPa, which is 154% higher than that of pure Al. The fracture mode of the composite is the mixture of plastic fracture and brittle fracture. The corrosion resistance of pure Al is significantly improved by the addition of reinforcements, which shows that the composite has a smaller corrosion current density and a more positive corrosion potential than that of pure Al.
Keywords
Introduction
Particle-reinforced metal matrix composites (MMCs) have been widely used in many fields. Aluminum (Al) matrix composites have become one of the engineering materials in great demand due to their lightweight, strong ductility, excellent mechanical properties and wear resistance, good thermal conductivity, and conductivity. They have been widely used in automotive and aerospace industries and other fields and have attracted wide attention. 1 –5
In traditional particle-reinforced MMCs, ceramic 6,7 (SiC, Al2O3), short carbon fiber, 8 and new nanomaterials 9,10 (carbon nanotubes, graphene) are widely used. However, due to the inherent characteristics between the reinforced materials and the metal materials, there are a series of problems in the preparation process, which limit the future research and application of this kind of composite materials. Ceramic-reinforced materials are easily agglomerated, which results in voids. The interface with the metal matrix is usually poor, and porosity reduces the mechanical properties of the composite material and the corrosion performance. 11,12 In addition, carbon and Al tend to react at high temperatures to form brittle compounds, leading to preferential initiation and propagation of cracks under load. 13,14
Metallic glass as a reinforcement for Al-based composites is an attractive alternative to ceramics and carbonaceous materials. At present, there are many studies on metallic glass-reinforced composites, but the previous research on the strength and plasticity of this new type of composites is mostly under compressive load. 15 –20 Metallic glass has extremely high strength, hardness, and elastic strain limit, as well as good corrosion resistance and wear resistance. 21,22 It has excellent properties as a reinforcing material in composites. Metal glass reinforcements are more compatible with metal substrates and have better interface bonding than traditional ceramic reinforcements. 23 If the sintering process can be carried out in the supercooled liquid region (ΔTx = Tx − T g), the metallic glass can act as a soft binder to eliminate the pores.
He et al. 24 prepared Fe43.2Co28.8B19.2Si4.8Nb4-reinforced 2024 Al alloy by hot-pressing sintering. The mechanical and tribological properties of composite materials were studied. The results show that the mechanical properties and friction properties of the composites are significantly improved by ferrum (Fe)-based amorphous particles. Ultimate tensile strength and ductility decrease with increasing particle size. Smaller particle size leads to lower wear resistance.
Zheng et al. 25 prepared Fe73Nb5Ge2P10C6B4-reinforced 2024 Al alloy by induction sintering. The research shows that compared with the matrix alloy, the mechanical properties of the composite have been significantly improved, but the density has only increased slightly. The yield strength and fracture strength of the composite are 403 MPa and 660 MPa, respectively. At the same time, a considerable fracture deformation of about 12% is retained, thereby achieving a significant combination of high strength and good plasticity. At present, most of them are prepared by hot-pressing sintering, but the cost is high, the cycle is long, and the material size and shape are more restricted. Pressureless sintering is an effective method for preparing Al-based amorphous composites. Its equipment is simple, sintering temperature is easy to control, and it is easy to industrialize production. However, there are few reports on the preparation of amorphous composites by pressureless sintering. Therefore, the composite materials are prepared by pressureless sintering.
At present, FeCrMoCBY series amorphous alloys are mainly used in coating research. 26,27 For this reason, a novel Al-based composite material is prepared after adjusting the composition of the amorphous powder. To explore the influence of amorphous alloys on the microstructure, mechanical properties and corrosion resistance of the composite under tensile loading, the related mechanism of amorphous particle-reinforced pure Al-based composites was analyzed.
Experimental details
Materials
In the experiment, Fe52Cr15Mo26C3B1Y3 amorphous powder was prepared by atomization method, with the particle size of 48 µm. The Fe-based amorphous powder was fully mixed with pure Al powder at 5%, 10%, 15%, and 20% (mass fraction) using QM-3SP4 (Nanjing NanDa Instrument Plant) planetary ball mill and dried in a vacuum drying oven for 2 h. A four-column hydraulic press was used to pre-compact the powder put into the steel mold into a billet at a pressure of 60 MPa, the sample size is ϕ36 mm, and then sintered in a common atmosphere sintering furnace. The temperature was controlled around the crystallization temperature (Tx ) 860 K of the Fe-based amorphous powder. The final sintering temperature was 823 K, and the temperature was maintained for 2 h. The sintered sample was placed in an induction heating furnace and heated to 793 K and immediately subjected to hot-pressing treatment at a pressure of 40 MPa.
Methods
X-Ray diffractometer (D8-FOCUS, D8-FOCUS:PANalytical B.V., scanning speed of 2° min−1, scanning angle of 10–90°, copper K α) was used for phase analysis of amorphous alloy powder and composite block. Differential scanning calorimetry (DSC) was used for thermal analysis of amorphous powder. Hitachi SU8010 (Hitachi Corporation of Japan, Japan) scanning electron microscope (SEM) was used to observe the microstructure of the sample, and the composition of the phases was analyzed in combination with an Energy Dispersive Spectrometer (EDS) spectrometer. The static tensile test of the composite was carried out on CTM9200 (Xie Qiang Instrument Manufacturing(Shanghai)Co.,Ltd) universal material testing machine. The CS350 (Shanghai Chenhua Instrument Co., Ltd) electrochemical workstation was used to test the Tafel polarization curve of the composite in 3.5% sodium chloride (NaCl) solution. Before the test, the sample was immersed in a 3.5% NaCl solution for 0.5 h. The measurement was started after the open-circuit potential was stable.
Results and discussion
X-Ray, DSC, and EDS analyses
To ensure that the reinforced body in the sintered Al-based composite material still maintains the amorphous characteristics and properties, in addition to controlling the influence of the sintering temperature, it is necessary to ensure that the original powder has a better amorphous structure. Figure 1 shows the micromorphology and X-ray diffraction (XRD) pattern of Fe-based amorphous powder. It can be seen from Figure 1 that the Fe-based amorphous powder prepared by atomization method is basically spherical and evenly distributed. And from the XRD pattern in the upper left corner of Figure 1, it can be seen that a single diffuse diffraction peak appears at 40° < 2θ < 45°, and no other crystal peaks appear. It shows that the amorphous powder is composed of a single amorphous phase structure and has an excellent amorphous structure.

Morphology and XRD pattern of Fe-based amorphous powder. XRD: X-ray diffraction.
Figure 2 is the DSC thermal analysis diagram of Fe52Cr15Mo26C3B1Y3 amorphous powder. During the heating process of Fe-based amorphous powder, two exothermic peaks can be seen obviously; it shows that the crystallization process of amorphous alloy is performed in two steps, which further proves that the Fe-based amorphous powder prepared by atomization method has complete amorphous phase. The glass transition temperature T g and crystallization onset temperature Tx of Fe-based amorphous powder are 670 K and 860 K, respectively, resulting in a subcooled liquid region of about 190 K (▵Tx = Tx − T g). It shows that it has good thermal stability. Therefore, the selected sintering temperature is 823 K, which ensures that the crystallization of the amorphous-reinforced material can be avoided during the heating process.

DSC trace of Fe52Cr15Mo26C3B1Y3 amorphous powder. DSC: differential scanning calorimetry.
Figure 3(a) is the XRD pattern of pure Al-based composites reinforced with Fe-based amorphous powders with different contents. It can be seen from XRD that new phases are formed in the sintering process of the composite, forming Fe9.7Mo0.3 compound and (Fe, Cr) solid solution. This is due to the solid-phase sintering between the Al matrix interface and the reinforcing phase during the sintering process. Fe- and Mo-rich elements will precipitate from the Fe-based amorphous powder and react. With the increase of content, the phenomenon of lattice distortion is obvious, which promotes the formation of a solid solution. Figure 3(b) is a partially enlarged view of the rectangular area in Figure 3(a). It can be seen from the figure that with the increase in the content of amorphous particles, the diffraction peak gradually changes from sharp diffraction peak to diffuse diffraction peak, and the diffuse diffraction peak becomes wider and wider. Secondly, with the increase in the content of amorphous particles, the diffraction peak also shifted to a larger 2θ angle. According to Bragg equation 2d sin θ = nλ, the lattice constant of the composite decreases with the increase in the content of the reinforcing phase, which indicates that the lattice distortion is enhanced. Therefore, the increased microstress will hinder the movement of the dislocations, thereby achieving the effect of improving the strength and hardness properties of the composite material. Figure 3(c) is a sintered sample of 15% reinforced phase composite material, which has a clear metallic luster, indicating that this method can be used to prepare a composite material that maintains Fe-based amorphous characteristics.

(a) XRD patterns of the Al-based composites reinforced with different volume fractions of Fe52Cr15Mo26C3B1Y3 glass particles, (b) enlargement plot of the XRD patterns at 44° < 2θ < 45°, and (c) sample composite reinforced by 15 vol.% Fe52Cr15Mo26C3B1Y3. XRD: X-ray diffraction.
Microstructure analysis
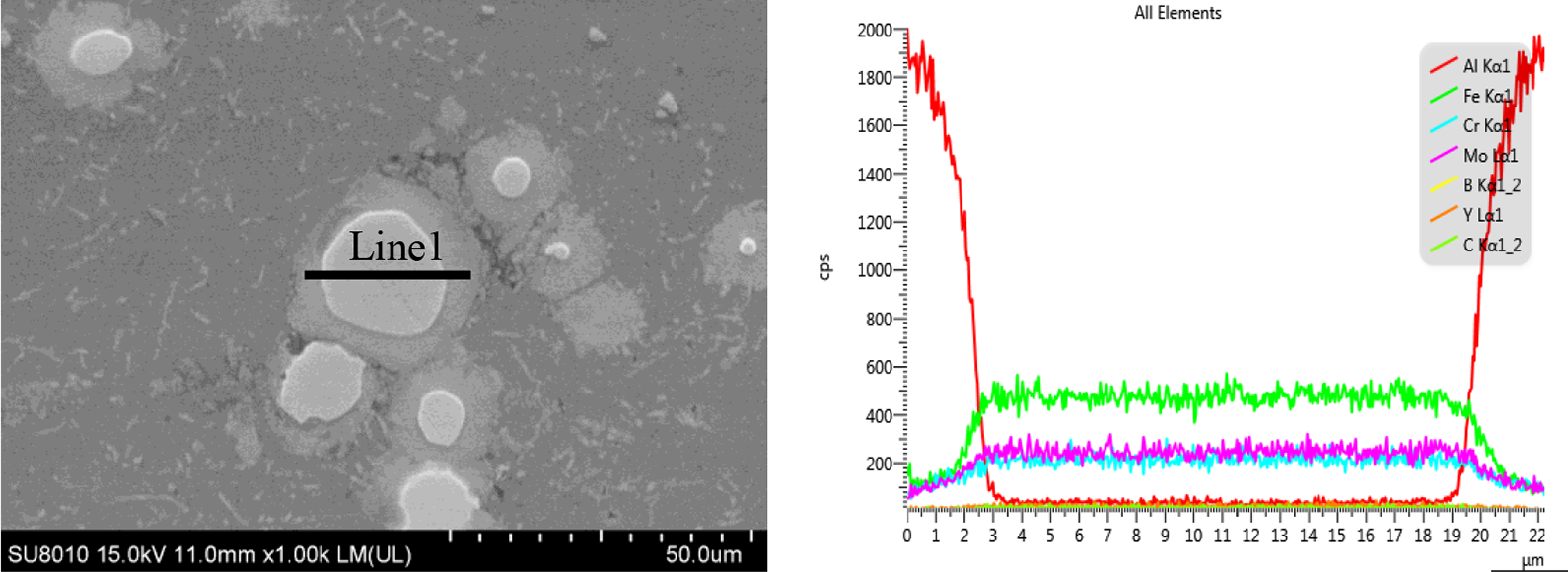
Figure 4 shows SEM images of Al-based composite materials reinforced with different contents of Fe52Cr15Mo26C3B1Y3. It can be seen from the figure that the particles of the reinforcement are evenly distributed in the matrix, the particles of the reinforcement phase–matrix are well combined, no obvious defects are generated, and the density is good. According to Figure 4(b), core–shell structure appears in the composite structure, 28 it can be seen from EDS line scanning (Figure 5) that the appearance in the middle of the structure is mainly Fe-based amorphous particles, and the continuous distribution of the dark phase is mainly Al. Core–shell structural particles are the main reinforcing material in composites, although thin strips of intermetallic compounds appear around their periphery. With the increase in the content of the reinforcement, agglomeration occurs between some reinforcement particles. Because of the poor binding property between the agglomerates, the properties of composite materials are affected. When the content of reinforcement reaches 20%, it can be seen from Figure 4(h), the composite material has small cracks, holes, and other defects. In the sintering-hot-pressing process, large particles are formed due to agglomeration, and the local residual stress near the large particles is relatively large, resulting in the phenomenon of detachment between the large particles and the matrix.

SEM images of Fe52Cr15Mo26C3B1Y3 reinforcements in Al-based composites with 5 vol.% ((a) and (b)), 10 vol.% ((c) and (d)), 15 vol.% ((e) and (f)), and 20 vol.% ((g) and (h)). SEM: scanning electron microscope.

((a) and (b)) EDS line scanning of the Al-based composites reinforced with 15 vol.% fractions of Fe52Cr15Mo26C3B1Y3 glass particles.
Figure 6 shows elemental mappings of 15 vol.% Fe-based/Al mixing powders and the corresponding micrograph. According to elemental mappings, each element of Fe-based amorphous particles is distributed in the reinforcement particles, which indicates that it is stable during sintering without obvious chemical changes.
Elemental mappings of 15 vol.% Fe-based/Al mixing powders and the corresponding micrograph: (a) EDS layered image 1, (b) electron image 1, (c) Al Kα1, (d) Fe Kα1, (e) Mo Lα1, (f) Cr Kα1, (g) C Kα1, (h) Y Lα1, (j) B Kα1.
Mechanical property analysis
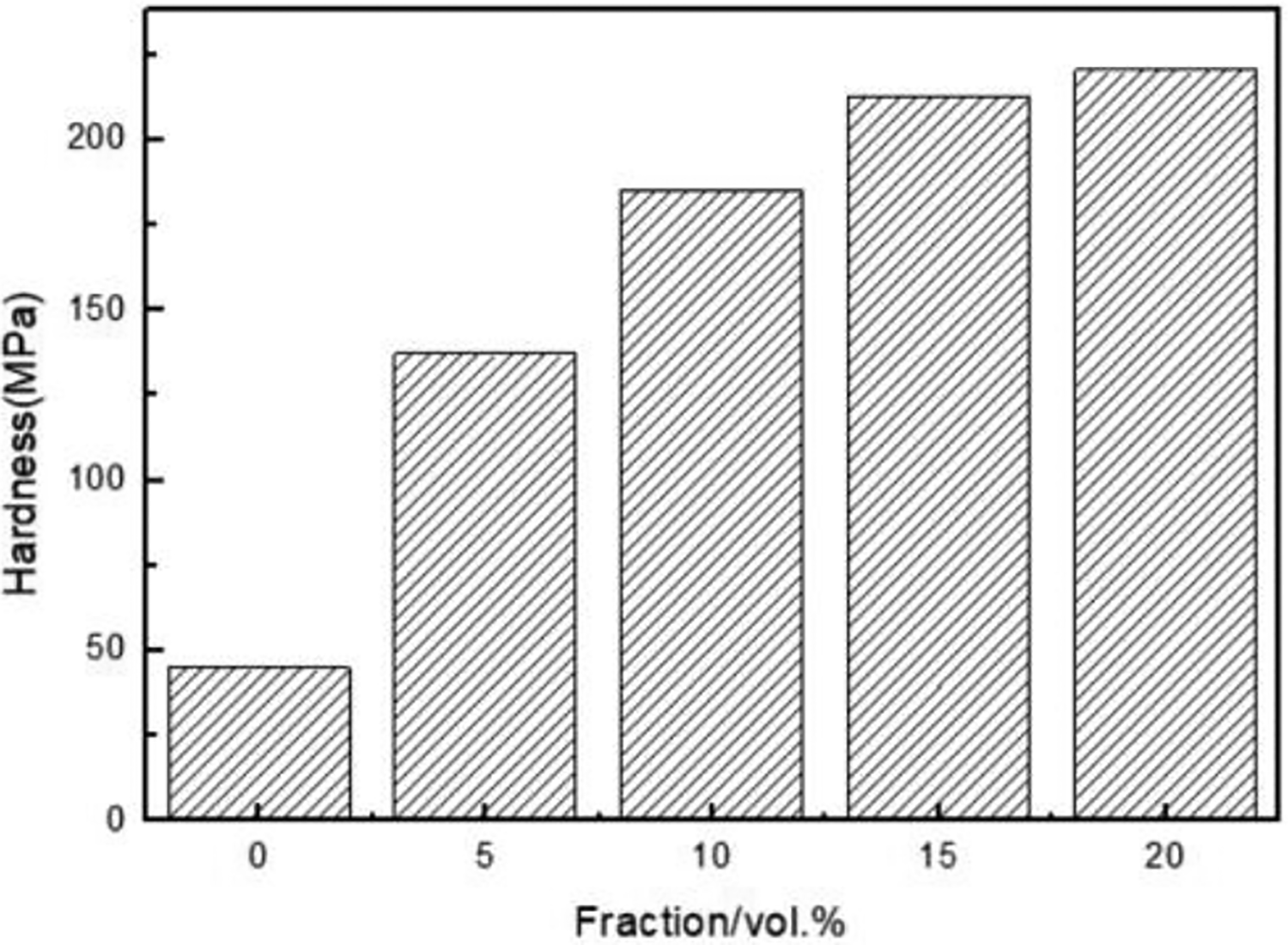
Figure 7 shows the Vickers microhardness (TEST-TECH(Shanghai)Co.,Ltd) of Al and composites reinforced with the Fe52Cr15Mo26C3B1Y3 glass particles. The Vickers microhardness was conducted according to the ASTM E-92 standard testing procedure. The Vickers hardness of the composite material increases with the increase in the content of the reinforcement and reaches the maximum when the content is 20%, which is 220.5 HV. The Vickers hardness of pure Al is 45 HV; therefore, the hardness of the composite material is significantly improved.
Microhardness of the Al-based composites reinforced with different volume fractions of Fe52Cr15Mo26C3B1Y3 glass particles.
Figure 8(a) shows the stress–strain curve of Al matrix composite under uniaxial tensile load at room temperature. The tensile strength was conducted according to the ASTM E-8 standard testing procedure. The tensile specimen is shown in Figure 8(b). With the addition of reinforcement particles, the tensile strength of the composite increases first and then decreases, which is much higher than that of pure Al. When the reinforcement content is 15%, the tensile strength of the composite reaches the maximum, which is 234 MPa. When the content of reinforcement is 20%, the tensile strength of the composite decreases to 192 MPa. During the sintering process, the addition of Fe-based amorphous powder as the second-phase particles has poor wettability with Al, and the thermal expansion coefficient is quite different from that of Al. As a result, when there is more reinforcement, the pure Al matrix cannot meet the better inclusion and combination with all the reinforcement particles, which makes the overall binding ability between the matrix and the reinforcement poor, so that under the effect of external load, the reinforcement particles come out of the matrix, break, and lead to the reduction of the strength of the composite. There are three reasons for the improvement of composite strength in this experiment. First, Fe-based amorphous powders are added as reinforcing phase particles. Hard particles can increase the ability to hinder the movement of dislocations, and round Fe-based reinforcing phase particles are more uniformly distributed in the matrix, which can avoid the degradation of the overall performance of the composite due to some defects. Secondly, the amorphous particles have high strength and hardness, which can inhibit the deformation of the matrix under an external load, that is, the force can be transferred to the reinforcing particles. Finally, the sintered composite has a good density and metallurgical bonding between the reinforcement and the matrix. Therefore, the strengthening mechanism of composite materials is mainly particle-bearing strengthening, fine-grain strengthening, and uniform distribution of amorphous particles.

(a) Room temperature compression stress–strain curves of the Al and Al-based composites reinforced with different volume fractions of Fe52Cr15Mo26C3B1Y3 glass particles. (b) Dimensions of the tensile sample (in mm).
Fracture morphology analysis
Figure 9 shows fracture surfaces of Fe52Cr15Mo26C3B1Y3/Al composites with different volume fractions of particles. It can be seen from Figure 9(a) that when the content of reinforcement is 5%, there are more dimples in the fracture, and the amorphous particles are well bonded to the matrix, indicating that the mechanism of fracture is ductile fracture. Figure 9(b) shows Al matrix can be found on the surface of Fe52Cr15Mo26C3B1Y3 particles, which means that due to the compatibility between the particle composition and the matrix, a solid metallurgical bond is established at the particle matrix boundary. When the content of reinforcement is 10%, as shown in Figure 8(d), some particles break and come out of the interface during stretching, and the tearing edge appears (as shown in the circle in the figure). With the increase of reinforcement content, dimple becomes shallow and tear edge becomes wider with the formation of cracks (Figure 9(e) and (g)). The decrease of the ductility of the composite indicates that the fracture mode of the composite gradually changes from ductile fracture to quasi-cleavage fracture. This is because with the increase of amorphous content, fine amorphous particles tend to agglomerate and cause the formation of large-sized particles; when the dislocation is pushed to the nondeformable particles under the action of strain, the voids will nucleate at the matrix

Fracture surfaces of Fe52Cr15Mo26C3B1Y3/Al composites with different volume fractions of particles: ((a) and (b)) 5%, ((c) and (d)) 10%, ((e) and (f)) 15%, and ((g) and (h)) 20%.
Electrochemical analysis
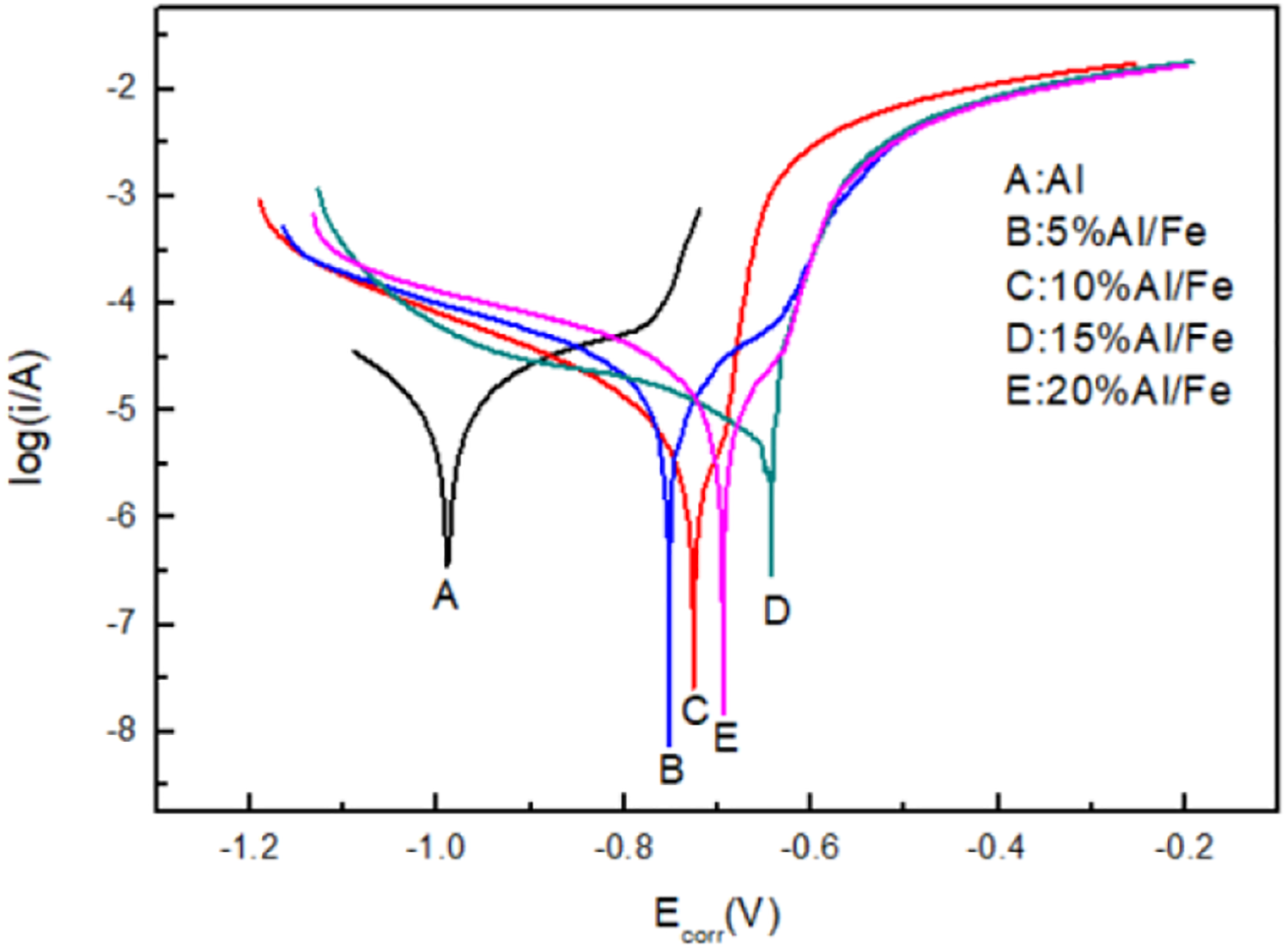
Figure 10 shows the Tafel polarization curves of Fe52Cr15Mo26C3B1Y3/Al amorphous composites with different contents in 3.5% NaCl solution. The corresponding electrochemical parameters E corr and J corr are presented in Table 1. The sample with 15% Fe52Cr15Mo26C3B1Y3/Al showed the most positive corrosion potential, which was −0.642 V; the sample with 5% Fe52Cr15Mo26C3B1Y3/Al showed the most negative corrosion potential, which was −0.752 V, and had a larger negative deviation compared with 15% Fe52Cr15Mo26C3B1Y3/Al. It can be seen from Table 1 that compared with sample Al, the J corr value of the composite with the reinforcement phase is reduced by at least one order of magnitude. When the content of amorphous exceeds 15%, the J corr value is reduced by two orders of magnitude. The minimum corrosion current density is 2.125 × 10−7 A cm−2 and the maximum corrosion current density is 1.694 × 10−6 A cm−2.
Potentiodynamic polarization curves for the Al and Al-based composites reinforced with different volume fractions of Fe52Cr15Mo26C3B1Y3 glass particles.
Summary of results of polarization for samples in 3.5% NaCl solution.

NaCl: sodium chloride; Al: aluminum; Fe: ferrum.
From the perspective of corrosion thermodynamics, corrosion potential represents the difficulty of corrosion. The more positive the value, the better the corrosion resistance of the material. 29 From the perspective of corrosion dynamics, corrosion current density represents the corrosion rate of materials. The smaller the corrosion current density, the better the corrosion resistance. 30 Therefore, as shown in Figure 10 and Table 1, when the amorphous content is 15%, the corrosion potential of the composite is the most positive and the corrosion current density is the least, showing good corrosion resistance.
Compared with pure Al, the composite material has a wider passivation range, indicating that the amorphous alloy has a better improvement in the corrosion resistance of the composite material. This is due to the elements Cr, Mo, and Y in Fe-based amorphous alloys, which have higher activity and can promote the rapid formation and expansion of passivation films. Metal elements promote the enrichment of ions in the membrane and thus passivation, which reduces the corrosion rate of the composite material and improves the corrosion resistance of the composite material. From the electrochemical reaction, the self-corrosion current is reduced and the corrosion potential is increased. Therefore, with the increase of amorphous content, the corrosion resistance of composite materials is getting better and better. When the content reaches 20%, the defects such as holes and cracks appear in the composite structure, increasing the internal corrosion area, thus accelerating the process of corrosion, leading to the decline of corrosion resistance, and forming more corrosive primary batteries, so the corrosion performance of the composite is slightly reduced.
Conclusions
In this article, Al-based composites reinforced by Fe52Cr15Mo26C3B1Y3 metallic glass particles have been successfully fabricated by powder metallurgy technique. The following conclusions have been drawn: The reinforcement particles are distributed in the matrix evenly, the reinforcement phase particles/matrix are well combined, there are no obvious defects, and the density is good. When the volume fraction reaches 20%, there are a few cracks, pores, and other defects in the composite. The maximum tensile strength of the composite is 234 MPa, which is 154% higher than that of pure Al. The microhardness of the composite increased from 46 HV to 220.5 HV. The fracture mechanism of composite materials is mainly a mixture of ductile fracture and brittle fracture. With the increase in the particle content of the reinforcement, the plasticity of the composite decreases gradually and there is a strong metallurgical bond at the boundary of the particle matrix. The corrosion resistance of the Al matrix composite was improved by the addition of amorphous particles. The corrosion potential increased from −0.99 V to −0.642 V, and the corrosion current decreased by two orders of magnitude.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
