Abstract
High-performance electrodes can solve problems of high voltage and large electricity consumption existing in chlor-alkali industry. A Ti/Al laminate composite (named as Ti/Al-LC) with three-layered structure (Ti/Al3Ti/Ti) is prepared as a new type of anode electrode for chlor-alkali electrolysis. Scanning electron microscope observation shows that the Ti/Al-LC is composited of a thicker inner layer with thickness about 700 µm and two thinner outer layers with thickness about 300 µm. From the X-ray diffraction pattern, it is known that the outer layers consisted of α-Ti and β-Ti phases, while the inner layer consisted of Al3Ti intermetallic phase. A saturated sodium chloride (NaCl) solution at 70°C is purposely chosen as the corrosion electrolyte to analyze the corrosion behavior of Ti/Al-LC as anode electrode for chlor-alkali electrolysis. Electrochemical tests, including potentiodynamic polarization and electrochemical impedance spectroscopy measurements, on a three-electrode system indicate that the Ti/Al-LC has a low corrosion rate with corrosion current density of 1.94 µA cm−2 and stable surface passive film in saturated NaCl solution at 70°C.
Keywords
Introduction
The chlor-alkali industry with products of chlorine (Cl2), hydrogen (H2) gases, and caustic soda forms a very important component of basic chemical building blocks that are essential for making thousands of downstream chemical products. 1,2 With the rapid development of chlorine alkali industry, it becomes an important research subject on how to reduce energy consumption, raise economic benefits, and improve the technology level. One of the ways to solve problems of high voltage and large electricity consumption existing in common electrolyzers is to select high-performance anode and cathode electrodes. Graphite is a traditional and widely used anode electrode in chlorine alkali industry due to its abundance in resources and low price. 3,4 However, the overpotential of Cl− ions is high, which increases the bath voltage during the electrolytic process. Besides, anode corrosion consumption of graphite not only reduces its useful life span but also blocks the membrane. Dimensional stable anode (DSA) with high selectivity for anodic reactions, good corrosion resistance, and long useful life span has attracted much attention in chlor-alkali industry. 5,6 However, the high cost of DSA, which is composited of a titanium substrate coated by a platinum-group metal (such as ruthenium) oxide and valve metal (such as titanium) oxide mixed coating, is the main disadvantage. In this work, a relatively low-price Ti/Al laminate composite (named as Ti/Al-LC) with three-layered structure (Ti/Al3Ti/Ti) is proposed as a new type of anode electrode for chlor-alkali electrolysis. Chlorine and chloride have serious causticity to the electrode, especially the anode electrode in the chlor-alkali industry. Therefore, the corrosion behavior of Ti/Al-LC was evaluated in the chlor-alkali electrolysis environment.
Experimental section
Preparation of the Ti/Al laminate composites
In this work, the Ti/Al laminate composites with three-layered structure (Ti/Al3Ti/Ti) were fabricated using Ti (TC4) and Al foils through vacuum hot-pressing sintering process in a ZRY-60-30 vacuum furnace. 7 –9 Before sintering, two Ti foil samples (thickness 0.5 mm) and one Al foil (thickness 0.7 mm) were pickled to remove the surface oxides, rinsed with deionized water, dried at room temperature, and overlapping placed (Ti/Al/Ti) in the furnace. During the sintering process, firstly, the sintering chamber was vacuumized to 0.01 Pa, then the samples were heated at 950 K, and then loaded a 2 MPa pressure for 10 min. After that, the pressure of the sample was unloaded to 0.5 MPa and the temperature was elevated to 960 K for 5 min to remove the residual liquid Al. Lastly, the loading pressure was increased to 4 MPa at 960 K for 1 h to get the Ti/Al laminate composites.
The crystalline structure of the Ti/Al laminate composites was verified by X-ray diffraction (XRD; D8 Advance, Germany). To analyze the phase composition of the inner layer, XRD test was applied on the inner layer by removing the outer layer of the Ti/Al laminate composites. The cross-sectional morphology as well as the thickness of the Ti/Al laminate composites was analyzed by a scanning electron microscope (SEM; JSM-6490LV, Japan).
Electrochemical tests
In the chlor-alkali electrolysis process, an aqueous saturated sodium chloride (NaCl) solution is fed to the anode side of the electrolyzer. The operation temperature is usually about 60–90°C. Therefore, in this research, a saturated NaCl solution at 70°C was purposely used as the corrosion electrolyte. Before electrochemical test, the samples were welded to a copper wire and the backside edges of the specimen were covered with high temperature silica gel, which guaranteed only the Ti surface exposing to the corrosion electrolyte. A three-electrode system comprising of a saturated calomel electrode (SCE) electrode (as the reference electrode), a platinum sheet (as the auxiliary electrode), and the samples (as the working electrode) was applied for the electrochemical tests. 10 –12 The corrosion behavior of the samples was characterized by potentiodynamic polarization on a CHI660E electrochemical workstation. Before each potentiodynamic test, the samples were first stabilized at open-circuit potential (OCP) for 5 min, then started potential sweep from −0.2 V (vs. OCP) to 2.0 V (vs. SCE) with a scanning rate of 10 mV s−1. Electrochemical impedance spectroscopy (EIS) measurements were carried out at OCP in a frequency range from 100,000 Hz to 0.01 Hz with the perturbation amplitude of 5 mV. The EIS data were analyzed by ZSimpWin 3.10 software.
Results and discussion
Characterization of the polylaminate coating
Figure 1(a) shows the cross-sectional SEM morphology of Ti/Al-LC. As observed in the figure, the Ti/Al-LC is composited of three layers: a thicker inner layer with thickness about 700 µm is covered by two thinner outer layers with thickness about 300 µm. Also, the outer layers display well metallurgical adhesion to the inner layer, which is free of defects such as pinhole, micropore, and microcracks. To verify the crystal structure of the outer and inner layers, XRD tests were carried out on the surface and inner layer (removing the outer by grinding) of Ti/Al-LC, respectively. Figure 1(b) depicts the XRD patterns of the surface and inner layer of Ti/Al-LC. The surface layer texture of the Ti/Al-LC consisted of α-Ti with (002) (2θ = 38.78°), (101) (2θ = 40.51°), and (102) (2θ = 53.31°) reflections and β-Ti phase with (211) (2θ = 71.42°) and (220) (2θ = 82.75°) reflections, which is consisted with the fact that TC4 is a two-phase (α/β) alloy. 13 –16 The thickness of the outer layer is about 300 nm, X-rays are able to penetrate the outer layer. Therefore, the appearance of reflection peak at 2θ = 80.66° on XRD pattern of surface Ti/Al-LC is the Al3Ti phase. The XRD pattern of the inner layer shows that the main phase is Al3Ti intermetallic alloys. It is also observed that no reflection peaks for Al phase have been observed, which indicates a complete reaction of Al foil with Ti. Through the above analysis, the Ti/Al-LC is consisted of two outer TC4 layers and an inner Al3Ti intermetallic layer.
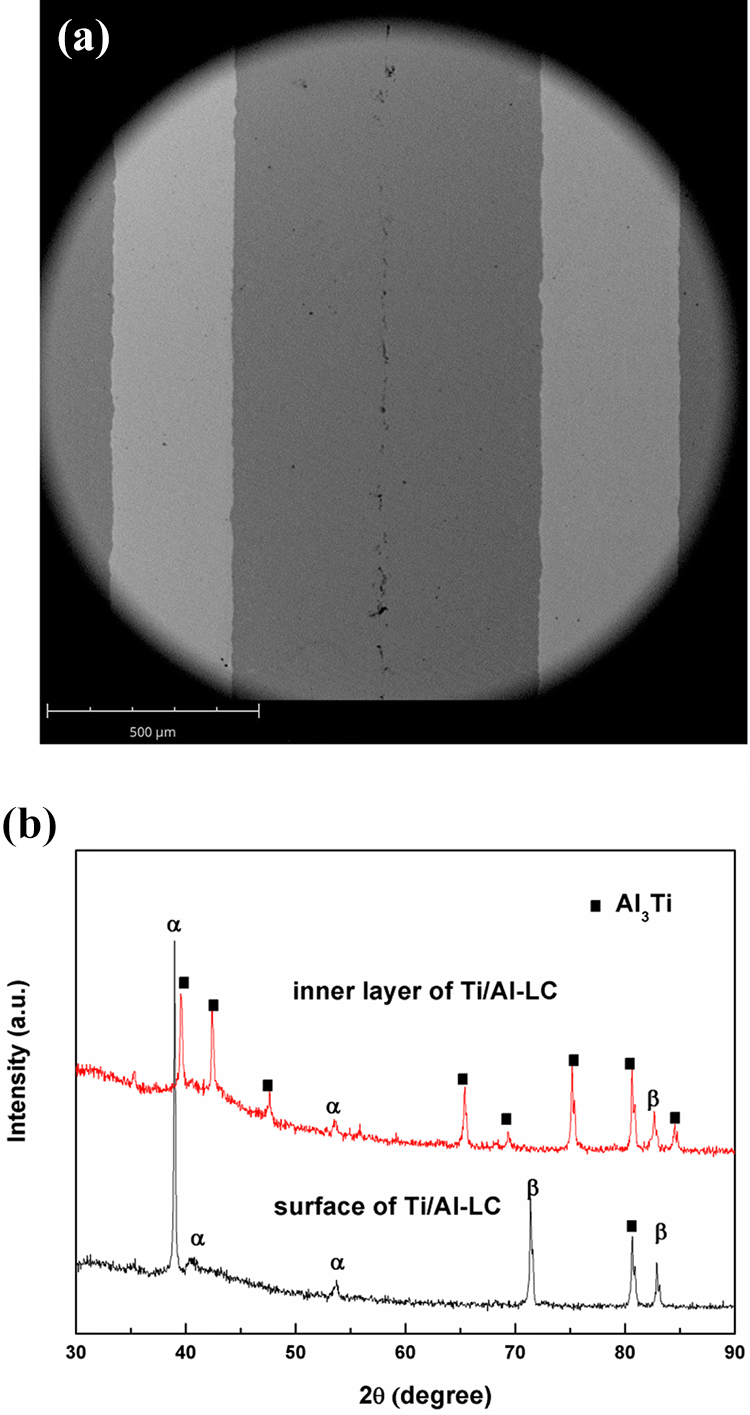
(a) Cross-sectional SEM morphology of Ti/Al-LC and (b) XRD patterns of the surface and inner layer of Ti/Al-LC.
Corrosion resistance and stability
Potentiodynamic polarization tests were used to estimate the general corrosion resistance of Ti/Al-LC in saturated NaCl solution at 70°C, as shown in Figure 2(a). It is observed from the potentiodynamic polarization curves that the Ti/Al-LC shows a typical passivation behavior. 17,18 The polarization curves can be divided into four regions: in the cathode polarization region (curve ab), the current density decreases with increasing potential, and H2 evolution is the main electrochemical process; in the anode polarization region (curve bc), the current density increases with increasing potential and the dissolving of the tested sample is the main electrochemical process. It is different from other passivation metals like stainless steel 19 that the Ti/Al-LC shows no obvious active–passive transition. The Ti/Al-LC is passivated directly in the anode polarization region indicating the easier self-passivating behavior. During the passivation process, a passive film begins to form on the tested sample surface; in the passive region (curve cd), when the potential exceeds point c, the current density stabilizes, the passive film covers the entire surface of the tested sample and the passivation current density is as low as 1.86 µA cm−2; in the transpassive region (curve de), the current density increases rapidly with increasing potential once the potential surpasses point d (about at 1.5 V), and the passive film dissolves. The corrosion current density I corr was also obtained by linear polarization method. Results show that the I corr of Ti/Al-LC is only 1.94 µA cm−2 in the saturated NaCl solution at 70°C. Such low-corrosion current density can guarantee a long and stable life of Ti/Al-LC in the chlor-alkali industry.

(a) Potentiodynamic polarization curve, (b) Nyquist plots, (c) Bode plots, and (d) equivalent circuit of Ti/Al-LC tested in saturated NaCl solution at 70°C.
EIS is a quasi-steady-state measurement, which is able to test the electrochemical behavior of electrode without exerting great impact on the electrochemical system due to the low-amplitude perturbation signal. 20 –22 The Nyquist plot for Ti/Al-LC obtained in saturated NaCl solution at 70°C is shown in Figure 2(b). It is observed that Nyquist plot displays one depressed semicircle within the test frequency range (from 100,000 Hz to 0.01 Hz). Figure 2(c) displays Bode plots of Ti/Al-LC. Two peaks are observed on the phase angle versus frequency plot indicating two time constants. Typically, when titanium or its alloy is exposed to the acid corrosive electrolyte, passive films will be formed due to the passivating element Ti. However, under more aggressive corrosion circumstances, the passive films can be destroyed leading to penetration of the electrolyte into the passive film/Ti interface. 23 The electrolyte can pass through the passive film and contact with the passive film/Ti interface directly, and therefore, two time constants appear on the Bode plots. According to the above analysis, an equivalent circuit (Figure 2(d)) is established to fit the impedance spectra of Ti/Al-LC tested in saturated NaCl solution at 70°C, where R s, R c, C dl, R f, and C f are denoted as the solution resistance (between the electrode reference and working electrode), charge transfer resistance, double-layer capacitance, passive film resistance, and passive film capacitance, respectively. Typically, due to the nonideal capacitive response of the double layer and passive film, constant phase elements (CPEs) are used to replace the C f and C dl to calculate the spectra. The CPE impedance can be presented as
where Q, ω, and n are the admittance magnitude of CPE, the angular frequency, and the CPE power, respectively. 24 The calculated results are listed in Table 1. The charge transfer resistance of Ti/Al-LC is 2.38 × 10−7 Ω cm2, which indicates less electrochemical reactions and stable surface passive film in chlor-alkali electrolysis operating condition.
Calculated results of EIS spectra for Ti/Al-LC.

EIS: electrochemical impedance spectroscopy; Ti/Al-LC: Ti/Al laminate composite; R s: solution resistance (between the electrode reference and working electrode); R f: passive film resistance.
Conclusions
A new type of anode electrode Ti/Al-LC with relatively low price for chlor-alkali electrolysis was prepared in this research. The Ti/Al-LC was consisted of two outer TC4 titanium alloy layers with thickness of 300 µm for each layer and an inner Al3Ti intermetallic layer with thickness of 700 µm. Furthermore, the outer layers displayed well metallurgical adhesion to the inner layer, which was free of defects such as pinhole, micropore, and microcracks. Chlorine and chloride have serious causticity to the electrode, especially the anode electrode in the chlor-alkali industry. A saturated NaCl solution at 70°C was used as the corrosion electrolyte to analyze the corrosion behavior of Ti/Al-LC as anode electrode for chlor-alkali electrolysis. Corrosion current density derived from the potentiodynamic polarization plots was as low as 1.94 µA cm−2, while the charge transfer resistance calculated from the EIS data was as high as 2.38×10−7 Ω cm2, which can guarantee a long and stable life of Ti/Al-LC in the chlor-alkali industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by The National Natural Science Foundation of China (nos 21506198 and 11602230), Young Talent Promotion Project of Henan Province (no. 2019HYTP002), Key Scientific Projects of University in Henan Province (no. 20B430021), and Mass Innovation Space Incubation Project (nos CLY20180158 and CLY20190140).

