Abstract
The stainless steel 304L (commercially supplied) and Monel K-500 that was prepared through cold pressing of metal matrix composite materials powder mixtures were joined by diffusion welding. Welding was performed under uniaxial compression using the 50 µm-thick nickel (Ni) and aluminum (Al) 2024 interlayers under 3 MPa at 950–980°C for 60 min in an argon gas atmosphere. After welding, the diffusion and intermediate zones of the samples were characterized using X-ray diffraction, optical microscopy, scanning electron microscopy, and energy-dispersive spectrometry analysis. As evaluating the diffusion zone with interlayer, it was determined that different interlayers expanded due to increasing temperature of the diffusion area. Interlayers expanded more on the K-500 side and relatively less on the 304L side. On the other hand, when samples with Ni interlayers were examined, high amount of Ni contained by both base material (304L) and interlayer led to the formation of rich Ni phases in the diffusion side. The brittle FeNi, Fe3Ni2, CuNi, Cu9Si, Cu0,81Ni0,19, Fe0,64Ni0,36, CrNi, Cr2Ni3, FeNi3, FeCu4, and Al0.71Cr0.3Fe17.65, which were identified to form irregularity in the diffusion zone. These intermetallic phases increased the hardness and significantly decreased the ductility of different material couples joined by diffusion welding. As a result, the microhardness and lap shear tests were applied to specimens to characterize the mechanical properties of the joint zones. The maximum hardness and maximum lap shear values were obtained at joint that made with the Al interlayer at 980°C as 432.8 HV and 165 MPa.
Introduction
The welding of dissimilar metal combinations has become a critical technology in many areas, given that it addresses the needs of advanced industry. 1 –3
The method of diffusion welding is a proper bonding method for electronics, nuclear, and aviation industries. 4 –6 Both similar and dissimilar materials are joined by using the diffusion welding. 7 Among the joining methods, diffusion welding is an intriguing technique that attempts to minimize the amount of the harmful effects on the material as much as possible (e.g. cracking, deterioration, separation, etc.) in order to form a bimetal. 8 –11 In recent years, great interest has been shown toward the production of diffusion-welded materials, given that diffusion welding solves many problems experienced between dissimilar materials during the joining process. 12,13
Diffusion welding depends on various parameters such as welding temperature, waiting period, and joining pressure. 14 The precise selection of these parameters, in combination with the thickness of the interlayer, results in the formation of an appropriate joining. Better bonding quality in the diffusion can be obtained by placing an interlayer between the joined materials. 15 This is done to minimize the thermal expansion, reduce the bonding temperature and pressure, and prevent the formation of brittle intermetallic phases by inhibiting the diffusion of unwanted elements. 16,17 Atabaki et al. 18 joined 304 stainless steel with copper interlayer by using the transient liquid phase (TLP) technique. On the other hand, He et al. 19 joined 304 stainless steel with nickel (Ni) interlayer by using the TLP technique.
When stainless steel materials are directly welded by diffusion welding, brittle intermetallic formations, which deteriorate the mechanical properties on the interface, occur. 20 The different thermal expansion of different materials further causes more stress due to the density difference. 21,22 Accordingly, a number of researchers are now using Ni, 23,24 silver (Ag), 22,25 and copper (Cu)-based 7,26,27 interlayers to alleviate the stress concentration.
In this joining method, the presence of improper intermetallic phases within the joining zone is inevitable. 28,29 Characteristically diffusion welding is an alternative method that can be used for joining the advanced metals. Advantages of the TLP processing comparison with conventional welding methods is the high capability of obtaining a bond zone carrying properties similar to those of the base metals. 30
Monel K-500 is an Ni-based, hardenable alloy having the face centered cubic surface (FCC) and containing a significant amount of Cu along with a small amount of iron, manganese, aluminum (Al), and titanium (Ti), 31,32 and this alloy has an excellent corrosion resistance. 33 This alloy is typically used in a broad range of applications, including pump shafts and impellers, oil well drilling tools, electronic parts, gyroscopes, fasteners, springs and surgical blades, marine services, chemical processing, and oil and gas production, as well as the electronic components industry. 34 These Ni-based superalloys are used in gas turbine engines to meet the requirements of those components being bonded via hot gas. These alloys are exposed to extreme working conditions whereby they require high-temperature creep, tensile strength, ductility, and oxidation resistance to withstand the applied load. 35,36 Rincon et al. 37 investigated the metallurgical impact of hydrogen embrittlement in heat exchange-based hydrogen–metal interactions on Monel K-500. Chen examined the corrosion behavior of both Monel K-500 and 316 stainless steel in artificial sea water and discussed how the specially created passive film contributed to damage recovery. This alloy showed an outstanding corrosion resistance both in the working area and during lab testing. 33,38 The Monel K-500 alloy is a Ni-based precipitation hardenable alloy containing a significant amount of Cu alongside small amounts of iron, manganese, Al, and Ti. While Cu is added to obtain a solid solution, Al and Ti precipitated into gamma-prime matrix using specific heat treatments while forming an intermetallic phase. The age hardening of this alloy occurs with the formation of consistent (Ni3 (Al, Ti)) precipitates. 31
This article sets out to explore and identify the mechanical properties and their microstructure compounds of the diffusion-welded materials produced with various methods. Products that combine in diffusion welding were revealed by using the optical microscopy, X-ray Diffraction (XRD), scanning electron microscopy/energy-dispersive X-ray spectroscopy (SEM/EDS) techniques. This information will help to establish a foundation for the properties of advanced welding process, indicating that TLP processes would potentially make great contributions to this welding category.
Materials and methods
In this study, metal matrix composites (MMCs) Monel K-500, manufactured using powder metallurgy method, as well as commercially supplied 304L stainless steel rods were used. Fifty-micrometer-thick pure Ni and Al 2024 foils were used as interlayers. The chemical compositions of the composite base materials are presented in Table 1.
Chemical composition of the composite base materials (wt%).

Ni: nickel; Cu: copper; Cr: chromium; Fe: iron; C: carbon; Mn: manganese; Si: silicon; S: sulfur; Al: aluminum; Ti: titanium; P: phosphorus; N: nitrogen.
The productions of cylindrical samples Monel K-500 were performed in two stages. In the first stage, metallic powders of 50–100 µm sizes with high purity (99.50–99.90%) were obtained from Alpha Aesar Company (Germany). Then, those powders were weighted by high precision (10−4 digits) digital scale (AND GR-200) for mixture them in weight percentage. To homogenize this mixture, a Ø100 mm conical mechanical blender with three wings that were mounted to the steel shaft was used for mixture operation. Mixture operation was performed for 15 min at room temperature condition. After that, this mixture was cold compressed to the 900 MPa by TecQuipment Universal Testing Machine (compressing capacity 1000 MPa and located in Dicle University) by mounting an apparatus. After pressing operations, the dimensions of testing samples were determined as Ø10 × 10 mm2. In the second stage, to strengthen the samples, sintering operation was performed. For sintering operation, argon gas was applied to the specimens for 45 min at 950°C in atmosphere-controlled furnace, followed by cooling at room temperature.
The joint surfaces (Figure 1) of the samples were smoothed out with parallel opposite surfaces using consecutively (i.e. 100-, 360-, 400-, 600-, 800-, and 1000-grit) sandpapers to reduce the surface roughness.

A schematic view of the samples.
Then the samples were cleaned ultrasonically with alcohol. The samples were welded by diffusion in argon gas controlled furnace, under 3 MPa of static pressure during period of 60 min at 950–980°C at the heating rate of 10°C min−1. The joined samples were left to cool down to an ambient temperature under argon gas atmosphere in the furnace.
The samples were cut perpendicular to the joining direction for metallographic examination. They were polished with diamond paste (3–6 µm) upon their surfaces being corrected with 1000- to 2000-grit sandpaper. Then, 37% hydrochloric acid (HCl) and 7 g of copper (II) sulphate (CuSO4) were applied on 304L material, which was made ready by means of a second sanding for the etching process by means of a second sanding, for 10 sec (Marble’s Reagent), and the etched samples were rinsed with water.
The Monel K-500 (MMCs) material was etched by applying mixture of 10 ml nitric acid (HNO3), 20 ml of hydrochloric acid (HCl), and 30 ml of glycerin (C3H3O3) for 30 sec. The etched samples were then cleaned with alcohol and dried with dry air. The polished surfaces of welded material pairs were examined in a backscattered mode (SEM-Zeiss Supra 50 VP) under a scanning electron microscope to obtain finer structural details in the diffusion zone. To characterize both the structure and the composition of element concentrations throughout the diffusion interfaces, values were obtained at an operating voltage of 15.00 kV. To understand the formation of intermetallics phases and to find the distribution of the phases, the XRD (Rigaku-Geigerflex X-ray diffractometer) analysis was performed to the region.
Lap shear tests of the samples combined with diffusion welding were performed on an automatic loading (Instron BS 8801) hydraulic tester machine that located in Dicle University. These tests were performed by applying a load at 1 mm min−1 with the apparatus shown in Figure 2. Shear strength values were determined by applying the cutting direction parallel to the intermediate layer direction and the cutting force on the intermediate layer.
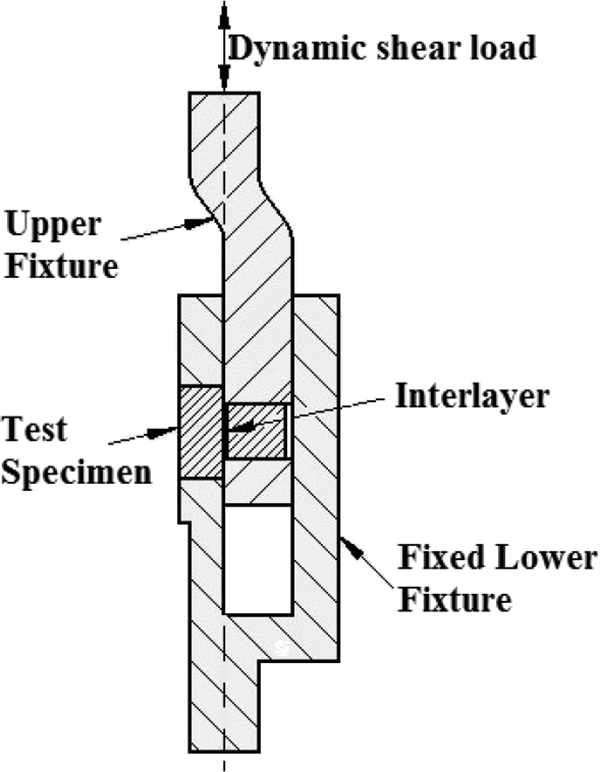
Schematic diagram of lap shear tests of 304L/Monel K-500 joints.
The microhardness measurements of the samples with Ni and Al 2024 interlayers which were diffusion welded at different temperatures were performed in the AOB-Vickers Hardness Testing Machine using a 10-g load for 20 s. To ensure that the results were accurate, four samples from the base materials, the diffusion transition zones shown in Figure 3, and the interlayers were tested by taking the values from 11 points in each operating parameter (temperature and interlayer). Figure 4 shows the hardness values of base metals and the interlayers.

The microhardness measurement scheme of different regions.

Vickers hardness of the base metal and the interlayers.
Results and assessment
Microstructure appearance in EDS and XRD results
Figures 5 and 6 show the optical images of the interlayered (Ni, Al 2024) samples welded at 950 and 980°C (Nikon Epiphot 200, Japan).

Optical images of the samples with Ni interlayer: (a) 950°C and (b) 980°C. Ni: nickel.

Optical images of the Al 2024 interlayered samples: (a) 950°C and (b) 980°C. Al: aluminum.
When SEM and EDS analyses of the Ni-interlayered samples were examined, it was remarkable that the spreading of the elements in the interface regions was the key parameter controlling the performance of the joining.
Temperature, pressure, and time parameters are the main parameters for atom diffusion. Also, among these parameters, temperature has the most effect in atom diffusion rate and diffusion velocity. Fick’s second law (equation (1)) shows that the diffusion rate of different atoms in the interface depends on time and distance, and further time is an advantage for atom diffusion. 39,40

An infinite time necessary for homogeneous structures in diffusion can be deduced. Diffusion coefficient that is a function of a temperature is an important parameter. 40,41

The temperature has the greatest effect on the coefficient diffusion rate and diffusion velocity. As a basic, equation (2) shows that the coefficient of diffusion depends on temperature (T) and activation energy (Q). The size of diffusion coefficient (D) is the indicator for diffused atom velocity. As the temperature increases, the quantities of atoms that make the diffusion action also increase. 41
EDS as well as SEM micrography were used to identify how the elements had spread to characterize the joining site of the interlayered samples. In terms of the thermal stimulation of the atoms in the interface at bonding temperatures of less than 950°C was not sufficient to join completely the material pairs. Increasing welding temperatures up to 950°C and 980°C was sufficient for joining of the matched surfaces in order to accelerate the diffusion in the interface. To understand microstructure better, the section (a) in Figure 7 was magnified by 2.00 KX, and Figure 8 shows the results of the linear EDS scan.

An SEM micrograph appearence of Ni-foiled bonded joints (980°C, 60 min). SEM: scanning electron microscopy.

The element content of the Ni-interlayer surface of the diffusion- bonded sample at various points at 980°C for the points subjected to EDS analysis. Ni: nickel; EDS: energy-dispersive X-ray spectroscopy.
The SEM micrographs of samples with Ni interlayer were obtained from both the Monel K-500 and 304L. What is more, the surfaces of the samples were analyzed using EDS, where upon the results are summarized in Table 2. Points 1, 2, 3, and 4 showed that this region was rich in Ni, Cu, and Al. In these regions, both the Ni-rich metals alongside the Ni/Cu/Al metals were consistent with the element compositions as presented in Table 2. The formations of Ni-rich in the diffusion zone (zones 5 or 6) also significantly increase the hardness value. While a Ni rich-region was determined in zones 7, 8, and 9 in the boundary of Ni interlayer of 304L material, an iron-rich region containing the base metal values was appeared when moving away from the boundary of the interlayer.
EDS analysis (wt) of the different regions marked in Figure 8.

EDS: energy-dispersive X-ray spectroscopy; Ni: nickel; Cu: copper; Al: aluminum; Mn: manganese; Fe: iron; Cr: chromium; Ti: titanium; Si: silicon.
When examining the linear ESD scan and SEM and EDS analyses of the Al-interlayered samples in Figure 9, it was determined that intermediate zone formed a reaction area, and there was the presence of a diffusion expansion in the side of composite Monel K-500 of the joint by 40 µm and the side of 304L by 50 µm.

The SEM micrographs of the samples welded with Al folio (980°C, 60 min). SEM: scanning electron microscopy; Al: aluminum.
As presented in Table 3 and Figure 10, the Ni (85.54) and Cu (13.34) element in reaction zone 1, Ni (82.01) and Cu (13.52) in phase zone 2, Ni (44.65), Cu (28.86), Al (26.64) element in zone 3, and Ni (25.75), Cu (42.61), Al (13.86), element in zone 4 were analyzed. When zone 5 was examined, it was determined that the presence of Cu (62.10), Ni (12.71), Fe (10.90), Al (9.43), and Cr (4.53) elements (in wt%) in the interface appeared in its microstructure. The main factor in this zone was aluminum interlayer and the copper content was lower than the base metal. The richness of Cu is caused by the diffusion of the base metal Monel K-500 close to the reaction zone. In zones 6, 7, 8, and 9, the elements near to the base metal values were obtained. The area shown in Figure 9 was enlarged to provide a better understanding of the microstructure of the samples which were joined with the Al foil for 60 min at 980°C.
EDS analysis (wt%) of the different regions marked in Figure 10.

EDS: energy-dispersive X-ray spectroscopy; Ni: nickel; Cu: copper; Al: aluminum; Mn: manganese; Fe: iron; Cr: chromium; Ti: titanium; Si: silicon.

The element contents of the Al-interlayered surface of the sample bonded by the diffusion at 980°C for the points subjected to EDS analysis. Al: aluminum; EDS: energy-dispersive X-ray spectroscopy.
Since the structure of the samples is polycrystalline, peaks of different crystallographic orientations are observed. The XRD patterns measured between 4.269° and 74.639° in the 2θ range of the Al 2024-bonded interfaces of stainless steel and Monel materials are shown in Figure 11. For all examples, the diffraction diagrams clearly show the presence of the phases FeNi, Fe3Ni2, CuNi, Cu0,81Ni0,19, Fe0,64Ni0,36, CrNi, Cr2Ni3, FeNi3 and Al0.71Cr0.3Fe17.65 (Figure 12).

XRD pattern of the diffusion bound Al intermediate layer sample. XRD: X-ray diffractometer; Al: aluminum.

The microhardness values of the diffusion welded samples: (a) Ni interlayer and (b) Al interlayer. Ni: nickel; Al: aluminum.
FeNi, Fe3Ni2, Cu9Si, FeCu4, Fe0.64Ni0.36, and CrNi phases were observed in the samples obtained by using Ni intermediate layer.
Microhardness results
Figure 13 shows the microhardness profiles of the 50-µm-thick Ni and Al 2024 interlayered joints. The welded samples were examined in five different sections: (1) 304L stainless steel, (2) its diffusion zone, (3) the interlayer (Ni, Al 2024), (4) diffusion zone, and (5) the Monel K-500 zone. When the zone 1 of the samples with the Ni interlayer was examined, it was determined that the hardness value as initiated by the stainless steel was 295 HV. As it approached the diffusion zone 2, the hardness values began to decrease, whereupon the lowest hardness value was found to be 153 HV at 950°C and 189 HV at 980°C in the interlayer area (3). In the side of Monel K-500, the hardness at the boundary between the interlayer and the diffusion zone (4) had increased abruptly at both temperatures and reached a peak value within the base metal (5), at the end of the diffusion zone. The hardness values obtained from the diffusion zone were 398 HV at 950°C and 412 HV at 980°C. In diffusion-welded samples, this result shows directly the presence of FeNi, Fe3Ni2, FeNi3, FeCu4, CuNi, Cu0.81Ni0.19, Cu9Si, CrNi, and Cr2Ni3, intermetallic compounds compared to the base metals (Figures 3 and 13(a)).

XRD pattern of the diffusion bound Ni intermediate layer sample. XRD: X-ray diffractometer; Ni: nickel.
Information has been provided about the microhardness values taken from the joint interfaces of the different materials, the composition scope of the welding, and dimension and diffusion character of the dispersion. When the Al interlayered samples were examined (Figure 13(b)), (1) the hardness value began to increase when approaching the diffusion zone (2) starting from 304L stainless steel and reached the peak in the interlayer zone (3) (356.7 HV at 950°C, 401.2 HV at 980°C). Furthermore, in the side of Monel K-500, the hardness level declined suddenly at the boundary of the interlayer and diffusion zone (284.1 HV at 950°C and 294.3 HV at 980°C), and peaked at the diffusion zone (432,8 HV). The reason for the change observed in the sample hardness at the interlayer boundary was most likely related to both microstructural changes and the precipitation of the Cu-rich phase. Another reason for this high hardness value is the Al2O3 phase which cannot be detected by XRD. 42,43 The properties of the phases forming during welding incate the mechanical properties of the material. The NiCu-, CrNi-, and FeNi-rich zones reached the hardnes values higher than 300 HV. The formation of NiCu intermetallic compounds that composed of Ni and Cu detected by XRD and these increase the hardness in that interlayer regions and then reached the values of base metals. It is compatible with the microhardness test results related to the diffusion zone width obtained from SEM images.
Lap shear test results
Lap shear tests were performed at room temperature to investigate the weld strength of the materials joined by diffusion welding method. In the determination of the shear strength, the interfaces of all the combined samples were deformed. Bonds formed by the intermediate layers can show high bond strength in bonding different materials. The shear strength of 304L/Monel K-500 materials joined at 950°C and 980°C affects the shear strength of the intermediate region, which significantly determines the hardness value and thickness of the reaction zones layer formed at different thicknesses. 21 In terms of microstructure, the thickness of the Ni interlayer samples was approximately 65 µm at 950°C and 95 µm at 980°C, depending on the temperature. It was found that the temperature of the intermediate layer was completely diffused at both temperatures and the diffusion width was approximately 950°CC and 100 µm and 980°C and 150 µm. Although Al hardness samples had high hardness, it was found that not all of the brittle phases that increase this hardness are within this interface region. These high temperature joints allow the Al intermediate to spread to both sides of the materials, especially to the Monel K-500, to a high degree of hardness to almost base metal.
The diffusion of the intermediate layer appears to be increased by increasing the bonding temperature. 44 There is a correlation between shear strength results (Figure 14) and intermediate layer thickness of Al and Ni intercalated samples combined at high temperature. Thus, it is concluded that a wide thickness of the diffusion layer (Figures 5 and 6) leads to maximum shear strength. Based on the test results, it can be said that the joint strength of all welded samples is sufficient (Figure 14).

Lap shear test results.
Conclusions
In this work, commercially supplied 304L stainless steel and Monel K-500 (MMCs) material that produced by using the powder metallurgy technique were successfully joined with diffusion welding using both pure Ni and Al2024 interlayers separately. Welding process were performed under the protected contitions of argon atmosphere during time period of 60 min at 950°C and 980°C. From this investigation, the following important conclusions are derived.
The key parameters that control the application of the joint area of diffusion of the elements of Ni and Al-foiled regions in the interface were noteworthy. The presence of Ni, which acted as a transition layer, prevented the Cu, Al, Fe, and Cr elements from infiltrating into the interlayer. On the other hand, the Al layer, had a 150-µm wide region containing Cu, Ni, Fe, and Cr elements at the ratios higher than the base metal. Under the same bond condition, the presence of some regions not bonded within the joint zone may be based on a relatively low duration. The highest lap shear value was obtained as 165 MPa in Al interlayer samples which were combined at 980°C where the intermediate diffusion was the largest. The FeNi, Fe3Ni2, FeNi3, FeCu4, CuNi, Cu0.81Ni0.19, Cu9Si, CrNi, and Cr2Ni3 phases detected in the XRD analyses caused hardness increase in base metals. The maximum hardness value was obtained in the Al foiled samples welded at 980°C.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
