Abstract
In the automotive and aviation fields, the complex use environment is critical to the performance requirements of materials, the corrosion resistance of materials severely limits the service life of materials, and improving the corrosion resistance of materials is of great significance in industrial production. In this article, in situ synthesis of powder metallurgy was used to prepare Al-Zr-Ti composites, and the effects of the addition of different amounts of Ti elements on the microstructure and corrosion resistance of Al-Zr-Ti composites were studied. The results show that Ti atoms form Al3(Zr, Ti) compounds by substituting Zr atoms in Al3Zr in composite materials to form substituted solid solutions to improve the properties of composite materials. With the addition of Ti, the corrosion resistance of the composites is improved compared with Al-Zr binary; when the Ti content is 10% (wt), the corrosion resistance of the composites reaches the best value, the corrosion potential is −0.67211 V, and the corrosion current is 1.4804 × 10−7 A cm−2.
Introduction
Aluminum matrix composites have become one of the most demanding engineering materials in metal matrix composites; because of their lightweight, excellent mechanical properties, and wear resistance, they have been widely used in automobile and aerospace industries.1–4 At present, researchers at home and abroad generally use in situ synthesis method to form reinforced phase particles in the matrix, so as to avoid the problem of uneven distribution caused by the addition of reinforced phase particles and improve the properties of aluminum matrix composites.5–8
In addition to the use of ceramic reinforcing materials, the use of intermetallic compounds (Al3Zr, Al3Ti, Al3Ni) to reinforce aluminum-based composite materials has also been extensively studied. Al3Zr has been paid more and more attention to Al-Zr binary compounds because of its excellent characteristics such as low density (4.11 g cm−3), high melting point (1580°C), high hardness, high elastic modulus (205 GPa), and good corrosion resistance. 9–15 Although Al3Zr is often used as a low density, the mechanical property is superior, and the high-temperature structural material is added into the Al alloy to improve the performance of the alloy, the reinforced effect of the single Al3Zr-reinforced relative aluminum-based composite material is not obvious, so the research personnel put forward the idea of hybrid-reinforced composite material. By adding two or more reinforcing phases at the same time, the material with excellent comprehensive performance can be obtained while maintaining the advantage of each component material. Hybrid composites can significantly improve or improve some properties of original single reinforcements due to different properties of various reinforcements, especially due to mixed effects. 16–25
In the Al-Zr-Ti ternary system, most of the researchers focus on the formation mechanism and microstructure of Al-Zr-Ti ternary system compounds. For example, Yang and Lu et al. 26,27 used electron probe microanalysis (EPMA) and X-ray diffraction (XRD) methods to study the ternary phase diagram of Al-Zr-Ti ternary alloys at 1273 K and 1073 K; the results show that in the Al-Zr-Ti ternary system, only elemental and binary phases are formed, no ternary phase is formed, and since the similarity of Ti and Zr elements is high, the Zr atom in the Al-Zr binary intermetallic compound is almost completely substituted by Ti atoms, so most of the Zr atoms in the Al-Zr intermetallic compound in the phase diagram are replaced by Ti atoms to form a replacement solid solution, thereby enhancing the properties of the composite. F. Yang et al. proposed theoretically that in the ternary system of Al-Ti-Zr, Ti element can replace Zr element in Al3Zr to form Al3 (Zr, Ti) compound. However, relevant researchers have little research on the influence of Al3 (Zr, Ti) compound formed by displacement reaction on the structure and corrosion resistance of the composite. In this article, Al-5Zr-xTi (x = 0, 5, 10, 15) composites were prepared by powder metallurgy in situ synthesis method to study the effect of Ti content on the microstructure and corrosion resistance of the composites.
Experimental procedure
Preparation of Al-Zr-Ti composites
Figure 1 shows the surface morphology of the original powder for the preparation of Al-Zr-Ti composites, in which the standard particle size of pure Al (99%) powder is 74–150 µm, the standard particle size of pure Zr (99.99%) powder is 48 µm, and the standard particle size of pure Ti (99.99%) powder is 48 µm.

SEM image of surface morphology of metal powder: (a) Al, (b) Zr, and (c) Ti. SEM: scanning electron microscope.
The corresponding Al, Zr, Ti powder, and magnesium stearate were weighed on the electronic balance, wherein magnesium stearate as a lubricant can reduce the friction of the mold wall to make the powder the compact is easy to demold. The weighing samples were mixed evenly by mechanical stirring, and the uniform mixed powder was cold-pressed on the press, and, finally, pressed pressure blocks were put into a vacuum atmosphere sintering furnace and heated to 800°C for 30 min under argon gas protection atmosphere and then cooled in the furnace. The sintered samples were heated to 400°C at a heating rate of 10°C min−1 and then hot deformation was carried out at the pressure of 40 MPa.
Test methods
Microstructure
Metallographic samples were first ground from 400 to 2000 grit emery papers and then polished with 0.5 µm diamond polish, and, finally, alcohol washed and blown dry. The microstructure was observed under an optical microscope without any corrosion, and the composition was analyzed with scanning electron microscope (SEM) and energy-dispersive X-ray spectroscopy (EDS).
Electrochemical test
The characterization methods of electrochemical corrosion resistance of the composites mainly include potentiodynamic polarization curve, an electrochemical impedance spectroscopy. According to corrosion potential, corrosion current density, impedance spectroscopy, and resistance value, the corrosion resistance and corrosion rate of the composite are studied.
The sample was working electrode, and platinum gauze was counter electrode, and saturated calcareous mercury was preference electrode, forming three-electrode workstation. The electrochemical properties of the sample were tested by a CHI660E electrochemical workstation (scanning rate was 0.001 V s−1). The effective working area of the sample was 1 cm2 and the electrolyte was 3.5% NaCl solution.
Each test was repeated at least three times to ensure the reliability of the data.
Results and discussion
Phase analysis of composite materials
Figure 2 shows the XRD pattern of Al-5Zr-xTi composite at 800°C sintering temperature. It can be seen from Figure 2 that the Al5Zr composite material contains an Al3Zr diffraction peak, indicating that a reaction occurs at a sintering temperature of 800°C to form Al3Zr. The XRD spectra of Al5Zr5Ti–Al5Zr15Ti composites in Figure 2 show that there are obvious Al3Zr diffraction peaks in Al5Zr5Ti–Al5Zr15Ti composites, but compared with the diffraction peaks of Al3Zr in Al5Zr composites, the position of Al3Zr diffraction peaks in Al5Zr5Ti–Al5Zr15Ti composites shifts slightly to the right. According to the research results of Yang et al., it can be known that the Al3(Zr, Ti) compounds are formed by adding Ti elements into Al5Zr5Ti–Al5Zr15Ti composites, in which Ti elements replace Zr elements in Al3Zr to form replacement solid solutions. In the displacement type solid solution, due to the dissolution of solute atoms, lattice distortion occurs, the lattice constant changes, and the partial crystal plane corresponds to a slight shift of the peak position of the diffraction peak, so that the diffraction peak shifts to the right in Figure 2.

XRD pattern of Al-5Zr-xTi composite. XRD: X-ray diffraction.
Microstructure of composite materials
Figure 3 shows the backscattering electron image of Al-5Zr-xTi composites. It can be seen from the figure that the phase morphology of the composite changes with the increase of Ti content. In Figure 3(a), a small amount of strip-like structure is formed inside the Al5Zr sample, and the rest are all aluminum matrix, the strip-like structure is unevenly distributed inside the matrix, and there are holes and crack defects on the matrix. From the microstructure of Figure 3(b) to (d) pilot sample, it can be seen that with the increase of Ti content, the internal strip structure of Al5Zr5Ti–Al5Zr15Ti sample has a process of growth compared with Al5Zr sample, and the strip structure is more uniform and dense on the matrix, which can play a good role in promoting the properties of the composites. However, the Ti content is not as good as possible; it can be seen from the observation that as the Ti content increases, the internal distribution of the composite material is more uniform and dense, but at the same time, the strip structure formed inside the composite material is also coarser, which will result in the formation of holes and cracks on the substrate adversely affects the performance of the composite.

Backscattered image of Al-5Zr-xTi composite: (a) Al5Zr, (b) Al5Zr5Ti, (c) Al5Zr10Ti, and (d) Al5Zr15Ti.
In samples of Al5Zr–Al5Zr15Ti shown in Figure 3, a strip-like structure was formed, a SEM was used to perform a point-scan analysis on the strip-shaped tissues of the Al5Zr and Al5Zr10Ticomposite, and the results of the scanning were shown in Figure 4. According to the EDS spectrum analysis of the composite material point scanning in Figure 4(b), the strip structure in the Al5Zr sample contains two atoms of Al and Zr, and the atomic percentage thereof is 3:1, so the internal strip of the Al5Zr sample can be known, and the tissue is an Al3Zr compound. Al5Zr5Ti–Al5Zr15Ti composite material also has strip-like structure inside, the EDS spectrum analysis of the composite material in Figure 4(d) shows that the inner strip structure of Al5Zr10Ti composite material contains Al, Ti, and Zr, and the element has an atomic percentage close to 18:5:1. According to the research of Yang et al., in the Al-Ti-Zr ternary system, Ti atoms can form Al3(Zr, Ti) compounds by replacing Zr atoms in Al3Zr compounds. According to EDS analysis results in Figure 4(d), it can be known that the structure in the Al5Zr10Ti sample may be an Al3(Zr, Ti) compound.
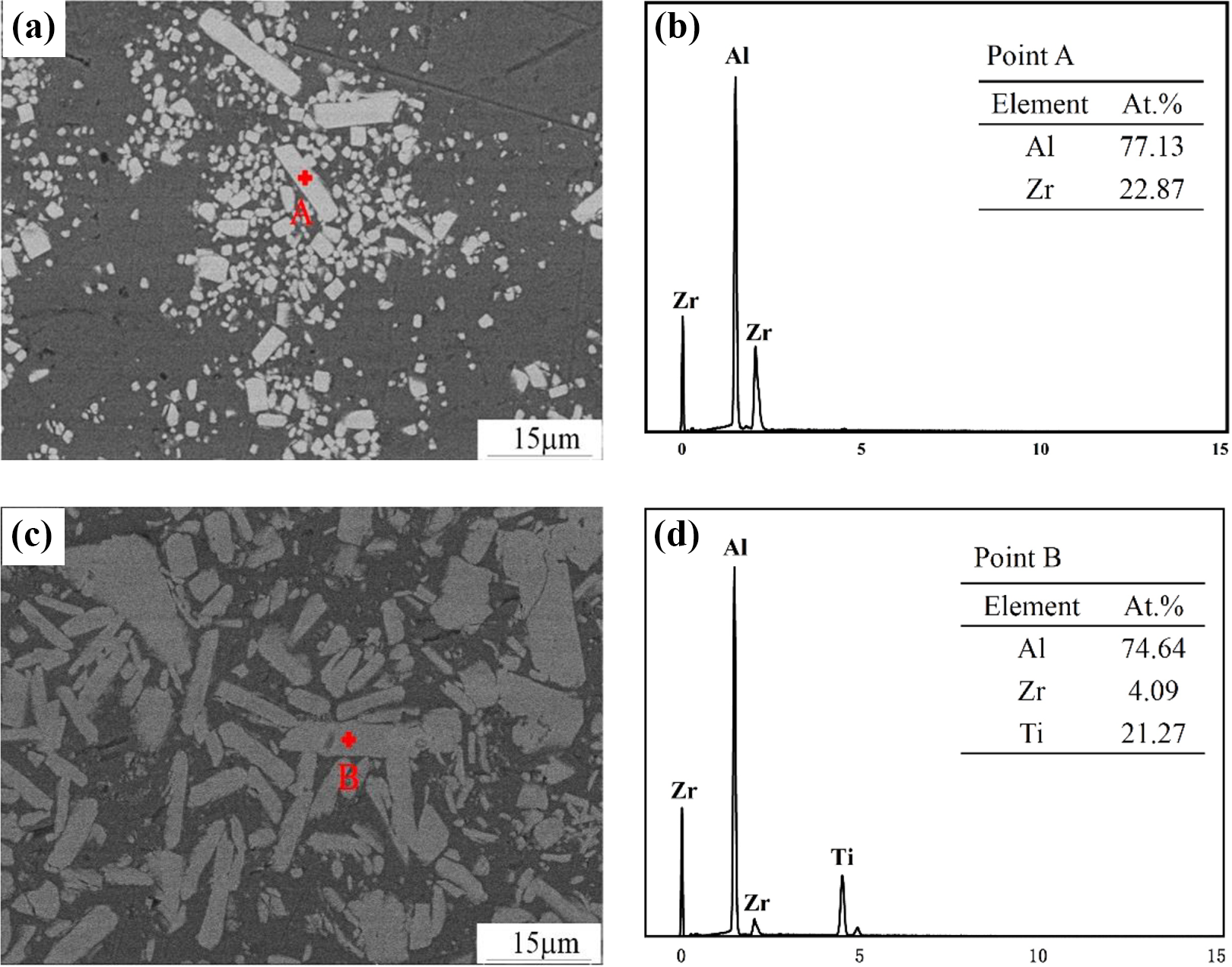
EDS spectrum of Al-5Zr-xTi composite material point scanning: (a) Al5Zr sample, (b) Al5Zr sample EDS spectrum, (c) Al5Zr10Ti sample, and (d) Al5Zr10Ti sample EDS spectrum. EDS: energy-dispersive X-ray spectroscopy.
The surface of the Al5Zr10Ti sample was scanned by SEM. The results of the scan are shown in Figure 5. Through the analysis of the surface scanning results of the Al5Zr10Ti composite material in Figure 5, it can be known that the Ti element and the Zr element are basically overlapped in the strip structure distribution. According to relevant research results at home and abroad, in the Al-Ti-Zr ternary system, since the similarity of the Ti and Zr elements is high, the Zr atom in the Al-Zr binary intermetallic compound can be almost completely substituted for the Ti atom to form a substitutional solid solution. Combined with the point of scanning results of Figure 4 and the surface scan results of Figure 5, it can be found that the strip structure is a Al3(Zr, Ti) compound, in which Ti atom forms Al3(Zr, Ti) compound by replacing Zr atom in Al3Zr compound.

Al-5Zr-xTi composite surface scan: (a) Al5Zr10Ti sample, (b) Al Ka1, (c) Ti Ka1, and (d) Zr La1.
Corrosion performance analysis of composite materials
Tafel polarization curve
Figure 6 shows the Tafer polarization curve of Al-5Zr-xTi composites measured in 3.5% NaCl solution, and the corresponding electrochemical parameters such as corrosion potential Ecorr and corrosion current density Icorr are presented in Table 1. It can be seen from Figure 6 that the order of corrosion potential of the four samples is as follows: Al5Zr10Ti > Al5Zr15Ti > Al5Zr5Ti > Al5Zr, wherein the Al5Zr10Ti sample exhibits the most positive corrosion potential, which is −0.67211 V; Al5Zr sample exhibits the most negative corrosion potential, which is −0.84615 V, which has a large negative offset compared with the Al5Zr10Ti sample. Combined with Table 1, the order of corrosion current density of the four samples is as follows: Al5Zr < Al5Zr5Ti < Al5Zr15Ti < Al5Zr10Ti, wherein the corrosion current density of the Al5Zr sample is the largest, 3.5907 × 10−6 A cm−2, and the corrosion current density of the Al5Zr10Ti sample is the smallest, 1.4804 × 10−7 A cm−2; compared with the Al5Zr, Al5Zr5Ti, and Al5Zr15Ti samples, the corrosion current density of the Al5Zr10Ti sample is reduced by an order of magnitude. From the point of view of corrosion thermodynamics, the corrosion potential represents the degree of corrosion, and the more positive the value, the better the corrosion resistance of the material. 28 From the perspective of corrosion kinetics, the corrosion current density represents the corrosion rate of the material, and the smaller the corrosion current density, the better the corrosion resistance. 29,30 It can be seen from Figure 6 and Table 1 that the corrosion potential of Al5Zr10Ti sample is the most positive, the corrosion current density is the smallest, and the corrosion resistance is better.

Polarization curve of Al-5Zr-xTi composite.
Corrosion potential and corrosion current density of Al-5Zr-xTi composites.

For the Al5Zr sample, the total reaction equation for electrochemical corrosion is as follows:
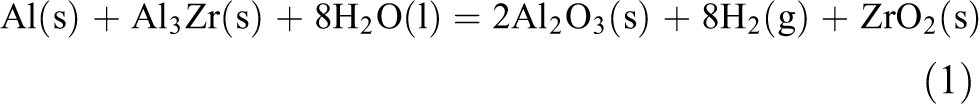
The deposited film of the corrosion product formed by the reaction can prevent the corrosion process from proceeding to a certain extent, the corrosion product of Al and Al3Zr is the precipitation phase of Al2O3, and the uniform and dense distribution of the Al2O3 film on the surface of the sample can effectively prevent the corrosion from proceeding. It can be known from formula (1) that ZrO2 is also generated during the corrosion reaction, and the precipitation phase of the corrosion product ZrO2 can further prevent the corrosion process from proceeding.
For the Al5Zr5Ti–Al5Zr15Ti sample, the total reaction equation for electrochemical corrosion is as follows:

It can be seen from the formula (2) that the corrosion product of Al and Al3(Zr, Ti) is the precipitation phase of Al2O3, and the Al2O3 film can effectively prevent the corrosion from proceeding. In addition, ZrO2 and TiO2 were also formed during the corrosion reaction, and the corrosion products ZrO2 and TiO2 precipitated phases can further prevent the corrosion process from proceeding. In addition, the TiO2 film formed by the reaction is firmly bonded to the surface of the sample, stable, and densely distributed on the surface of the sample, which can effectively prevent the corrosion. Therefore, with the increase of Ti content, the polarization curve of Al5Zr5Ti–Al5Zr15Ti composites gradually shifted to the right, and the corrosion resistance of Al5Zr5Ti–Al5Zr15Ti composites was improved compared with Al5Zr composites.
When the content of Ti is 10% (wt), the corrosion potential of the composite reaches the maximum, and the corrosion potential of the composite is −0.67211 V. When the content of Ti is more than 10%, the corrosion potential of the composite decreases gradually. The main reason for the above phenomena is that with the increase of Ti content, Ti replaces the Zr in Al3Zr to form Al3(Zr, Ti) compound, and the replaced Zr is difficult to distribute evenly on the surface of the matrix, so it will lead to the formation of pores. At the same time, with the increase of Ti content, the microstructure of Al5Zr5Ti–Al5Zr15Ti composite in Figure 3 can be observed, the microstructure of Al5Zr5Ti–Al5Zr15Ti composite is gradually increasing, and the coarsening of grains, the imbalance between the grain and the grain boundary, is increased, which increases the formation trend of the galvanic cell and leads to an increase in the electrochemical corrosion tendency. Therefore, with the increase of Ti content, the corrosion potential of Al5Zr5Ti–Al5Zr15Ti composites increases at first and then decreases.
Analysis of corrosion surface morphology
Figure 7 shows the surface morphology of the Al-5Zr-xTi composite after corrosion. Observing Figure 7(a) to (d) corrosion surface morphology of the composite material, it can be seen that pitting occurred on the surface of the Al-5Zr-xTi composite material, and the passivation film on the material surface was damaged corrosive media continue to enter the interior of the composite, leaving black holes on the surface of the composite. The black holes in the surface of the Al5Zr composite after corrosion are larger than the surface holes of the composite after adding Ti, indicating that the corrosion resistance of the Al5Zr composite is inferior to that of the Al5Zr5Ti–Al5Zr15Ti composite. After adding Ti, the black holes on the corroded surface of the composite gradually decreased compared with Al5Zr, indicating that the addition of Ti can improve the corrosion resistance of the composite. The corrosion surface morphology of the composite is the best when the Ti content is 10%, indicating that the Al5Zr10Ti composite has better corrosion resistance, consistent with the results in Figure 6.

Surface morphology of Al-5Zr-xTi composite after corrosion: (a) Al5Zr, (b) Al5Zr5Ti, (c) Al5Zr10Ti, and (d) Al5Zr15Ti.
Electrochemical impedance spectroscopy
The equivalent circuits that are simulated based on the impedance spectroscopy data of the composites with the Z-view software are shown in Figure 8. In the equivalent circuit, there are mainly resistance, Warburg impedance, and constant phase angle element (CPE). CPE can be written as

Equivalent circuit diagram of Al-5Zr-xTi composite.

In formula (3), ZW represents Warburg impedance.
Figure 9 shows the Nyquist plot of the Al-5Zr-xTi composite. It can be seen from the figure that there is a Warburg impedance32, 33 in all four samples of Al5Zr–Al5Zr15Ti, that is to say, behind the tolerant arc in the high-frequency region, there is a straight line with a tangent of 45°. The presence of Warburg impedance shows a significant difference in the concentration of the reactants and the concentration of the solution near the surface of the electrode, so the diffusion phenomenon occurs. 34

Nyquist maps of Al-5Zr-xTi composite.
In Figure 9, we can see that the radius of the capacitive arcs are in order from large to small: Al5Zr10Ti>Al5Zr15Ti>Al5Zr5Ti>Al5Zr, the Al5Zr10Ti sample is the biggest, and Al5Zr is the smallest, and there is a big difference in size. The results show that the larger the radius of the impedance arc in the Nyquist diagram, the larger the impedance value of the material and the more difficult the material is to corrode. 35,36 Therefore, in the Al-5Zr-xTi composite, the Al5Zr10Ti sample has the largest impedance arc radius, so the impedance value is the largest and the corrosion resistance is the best, which is consistent with the polarization result.
Conclusion
The Al-5Zr-xTi composites were prepared by powder metallurgy at 800°C; the effects of Ti content on the microstructure and properties of the composites were studied, the following conclusions can be drawn: After adding Ti elements, the internal products of the composites are more uniform and compact Ti elements replace the Zr elements in the Al3Zr to form Al3(Zr, Ti) compounds, thus forming a displacement solid solution to improve the properties of the composites. Compared with the Al-Zr binary, the corrosion resistance of the composites is improved after the addition of Ti. The corrosion resistance of the composites increases first and then decreases with the increase of Ti content. When the Ti content is 10% (wt), the corrosion resistance of the composite material is better because it has the most positive corrosion potential (−0.67211 V), the minimum corrosion current density (1.4804 × 10−7Acm−2), and the maximum impedance arc radius.
Footnotes
Acknowledgment
The authors thank the Hubei Provincial Key Laboratory of Green Materials for Light Industry, Hubei University of Technology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
