Abstract
The objective of this study was to prepare composite materials based on mesoporous silica/carbon, functionalized with acid groups, and also to analyze its application in the adsorption of dyes. These materials were prepared in a simplified unconventional route. The carbon phase was generated by direct carbonization of organic surface-active templates, which are used in the synthesis of meso-ordered silicas. The resulting composite materials were characterized in terms of their structure, texture, chemical composition, and surface chemical reactivity. Data obtained demonstrate uniform and ordered porous structures of approximately 70–200 Å, with a developed surface area of approximately 400–500 m2 g−1 and a large total pore volume of approximately 1.0–1.5 cm3 g−1. The potential of synthesized composite materials functionalized by oxygenated groups as adsorbents of cationic dyes was analyzed, the values of the maximum adsorption capacity between 317 mg g−1 and 245 mg g−1 were obtained. The adsorbing properties of silica/carbon composite materials obtained in an unconventional method using an organic template as the only carbon source were compared with the properties of silica/carbon composite materials manufactured by the conventional method using external precursors of the carbon phase. It has been proved that composite materials synthesized using an unconventional procedure, despite the fact that they have a lower carbon phase content, exhibit adsorption properties that are comparable or superior to materials elaborated in the traditional way, removing up to 85–90% of methylene blue from water contaminated with dye.
Introduction
The design of new composite nanomaterials (composites) that combine and/or improve the properties of individual components is an area of research of growing interest. In this way, mesoporous silica/carbon composites have been developed for use in the design of materials with special properties for specific applications. It has been reported that in many processes, silica/carbon composites exhibit superior properties to their components separately, as result of a synergy between the properties of meso-ordered silica (elevated surface area and ordering in a wide range of porous structure) and the properties of active carbons (electrical conductivity, thermal stability, and easy modification of its surface chemistry by functionalization). 1 –7
The most common technique to produce a layer of carbon on the surface of a meso-ordered silica is to anchor the organic compound on the surface of previously prepared pure silica and subsequently carbonize it by oxidation with sulfuric acid (H2SO4).2 The second important method is chemical vapor deposition, which, compared to the first method, requires more complex and expensive equipment. 2 Silica/carbon composites have also been prepared using a molding organic template and additional organic matter as carbon source. Thus, Yu et al. 8 synthesized mesoporous carbon material from hybrid silica MCM-48/surfactant and divinylbenzene as an additional carbon precursor, while Lu et al. 9 synthesized composite silica/carbon materials using a combination of 1,4-bis(triethoxysilyl)benzene and Pluronic P123 as precursors to the carbon phase.
As an alternative to the above methodologies used for the synthesis of mixed meso-ordered composite materials based on silica/carbon, in this work, these types of materials were prepared directly from hybrid materials based on silica and surfactant agents used as precursors of mesostructured silicas Santa Barbara Amorphous No. 15 (SBA-15), Korean Institute of Technology No. 6 (KIT-6) and meso-structured cellular foam (MCF). Organic template was employed both directly as a structure-directing agent and as the sole source for the formation of a carbon layer on the surface of the pure meso-structured silica. Direct carbonization of the structuring template for generating the carbon phase is a novel synthesis strategy that simplifies the production of silica/carbon composite materials, since it eliminates both the use of additional substances as carbon sources and some additional transformation stages used in conventional synthesis procedures. 10,11 Based on these considerations, the general aim of this work was to develop simple, clean, and cost-effective procedures for the production of meso-ordered silica/carbon composites, suitable for use as a platform in the elaboration of materials with specific adsorption properties.
Experimental section
Preparation of silica/surfactant hybrids
The SBA-15/P123 hybrid material was prepared according to the procedures reported by Zhao et al. and Newalkar et al., 12,13 using tetraethyl orthosilicate (TEOS) as a source of silicon and Pluronic P123 triblock copolymer as a template for structuring porosity, in a reaction mixture with a molar composition: TEOS:Pluronic 123:HCl (2 M):H2O = 1:0.17:5.7:193. The mixture was kept under agitation at 35°C for 20 h and in static hydrothermal conditions at 93°C another 2 h; the resulting solid material was recovered by filtered under vacuum, washed with ethanol, and dried at room temperature.
The hybrid material KIT-6/P123 was synthesized according to the procedure given by Kleitz et al. 14,15 from the reaction mixture with a molar composition: TEOS:Pluronic 123:HCl (2 M):H2O:nButOH = 1:0.17:1.83:195:1.31. The mixture was kept under stirring at 35°C for 24 h and under static hydrothermal conditions at 130°C for another 24 h. The resulting solid was recovered by filtration under vacuum, washed with ethanol, and dried at room temperature; n-butanol was included as a cosolvent and as an aid to a molding agent to expand pore size.
The hybrid precursor material of the mesocellular silica was synthesized according to the report of Schmidt-Winkel et al. 16 from the reaction mixture of the following molar composition: TEOS:Pluronic 123:HCl (2 M):H2O:ammonium fluoride (NH4F):1,3,5-trimethylbenzene (TMB) = 1:0.02:5.71:171.95:0.03:0.81. The mixture was kept under stirring for 20 h at 40°C and 24 h at 100°C in static hydrothermal conditions. The formed solid was recovered by vacuum filtration, washed several times with ethanol, and dried at room temperature; TMB was used as a micellar expander, and NH4F was used to improve the ordering of the structure of silicon oxide. 17
Direct preparation of silica/carbon composite materials
Composite materials based on silicon dioxide and carbon were obtained from synthesized hybrid materials by direct carbonization of the space-filling template. 10,11 The thermal decomposition of Pluronic P123 copolymer template was carried out by a two-stage process:
Partial oxidation of the organic matter of the molding template with a solution of 0.2 M H2SO4 (mass/volume ratio = 1:1). The mixture was heated under static condition for 5 h at 100°C and for another 12 h at 150°C; the resulting aqueous dispersion was filtered under vacuum to separate the solid material in powder form.
Full carbonization of partially decomposed template was completed by calcination of the filtered solid material at 800°C for 1 h under nitrogen flow.
Strict control of contact time with acid solutions, as well as carbonization times and temperatures, is necessary to achieve good reproducibility of silica/carbon composites. The synthesized silica/carbon composites were labeled as S/TC, K/TC, and M/TC to indicate that the inorganic phase is one of the meso-ordered silicas (S, K, M) and that the carbon phase was produced by direct carbonization of the molding template (TC); the digit 8 has been added to these codes to indicate the final carbonization temperature.
Functionalization of silica/carbon composite materials obtained by direct technique
The chemistry of the carbon layers deposited on the surface of the silica/carbon composites, obtained by the direct method described above, was modified by generating oxygenated acid groups. 17,18 For the partial oxidation of the carbon layer, 1 g of the composite material was treated with 10 mL of solution of (NH4)2S2O8 in H2SO4 (mass to mass ratio = 1:1.77). The dispersion was kept at room temperature under agitation for 60 h, then the solid was separated by vacuum filtration and washed with distilled water until neutral pH. The letter O was added to the codes of the synthesized silica/carbon composites to denote its surface functionalization with oxygen-containing groups.
Preparation of silica/carbon composite materials by conventional method
First, the SBA-15 (SMS), KIT-6 (SMK), and MCF (SMM) meso-structured pure silicas were synthesized by calcining corresponding silica/surfactant hybrids in an extra dry air flow (70 mL/min) at 500°C during 4 h to completely eliminate the mold template by combustion (“hard method”). 18 The same meso-structured silicas were also prepared by extraction of the molding organic template under reflux conditions at room temperature, using a mixture of 100 mL of ethanol and 2 mL of HCl per 0.5 g of hybrid (“soft method”). 17 At the next stage, the resulting pure silicas were impregnated with 2,3-dihydroxynaphthalene (DN), dissolved in acetone, for 12 h at room temperature, to be used as an external precursor of the carbon phase (EC). Finally, the materials were dried and calcined under nitrogen flow (60 mL/min) at 300°C for 1 h and then at 800°C for 2 h to achieve complete carbonization of the precursor of carbon layer. The composites prepared by this procedure were identified by the S/EC, K/EC, and M/EC codes, including the digit 8 to indicate carbonization temperature.
Characterization of synthesized materials
Textural properties were measured by nitrogen physisorption technique (Praxair, grade 5) at boiling temperature of liquid nitrogen (−196°C), using an AS1-C-MS Quantachrome physisorption analyzer. Previous degassing of the solid samples was carried out in vacuum conditions (approximately10−3 torr) at 150°C for 24 h. The properties of acidity and superficial acid strength of functionalized composite materials were determined by NH3 desorption at programmed temperature technique, using the same equipment configured for chemisorption analysis. The arrangement printed by the molding template of surfactant P123 in the porous structure of the materials and their phase composition was confirmed by X-ray diffraction analysis at low angle (SAXS) and at high angle (WAXS), respectively. A Bruker Model D8 Discover diffractometer with copper K α as the radiation source (λ = 1.54 Å) was used; linear detector was operated at 40 kV and 40 mA, and the scans were performed at intervals of 0.02° in the range 0.5° < 2θ < 5° and 10° < 2θ < 70°. The morphology and superficial texture of the particles of the synthesized materials were analyzed using a JEOL Model JSM 6610LV (Japan) scanning electron microscope, with Everhard Thormley secondary electron detector and 15 keV electronic beam acceleration voltage. This same microscope with Oxford Model X-Max accessory equipped with a lithium-doped silicon detector was used to obtain the X-ray energy dispersion spectra (XEDS) and identify the characteristic energy lines of the chemical elements present in the materials studied. The acceleration voltage of the primary beam was 20 keV. The presence of oxygenated groups in the functionalized materials was confirmed by detecting their characteristic modes of vibration, using a Digilab spectrophotometer of SCIMITAR series. Finally, the surface activity of the functionalized materials was assessed in the adsorption of methylene blue (MB) from standard aqueous solutions. The remaining concentration of the dye in the aqueous solution of MB after certain contact times was measured by ultraviolet–visible (UV-Vis) spectroscopy at λ = 663 nm, using an Agilent UV-Vis spectrophotometer model Cary 100 Scan.
Results and discussion
Structural characteristics of synthesized materials
The diffractograms obtained at low angle (SAXS) for pure silicas, as well as for functionalized and nonfunctionalized silica/carbon composite materials, confirmed the presence of meso-scale periodicity in the porous structures of the solids. The diffractograms obtained for silica SMS and composite materials S/C (Figure 1(a)) show signals at 2θ = 0.8–1.0° (intense), 2θ = 1.3–1.6°, and 2θ = 1.7–2.0° (weak), corresponding to characteristic d 100, d 210, and d 200 diffractions for meso-structured silica SBA-15 with hexagonal spatial symmetry p6mm 2D. The diffractograms for silica SMK and composite materials K/C (Figure 1(b)) show signals at 2θ = 0.8–1.0° (intense) and 2θ = 1.5–1.8° (low intensity) corresponding to d 211 and d 220 diffractions, characteristic for meso-structured silica KIT-6 with porous system of bicontinuous cubic spatial symmetry Ia3d. 4,19,20 The structural analysis confirmed the preservation of ordering of the porous structure during the carbonization of external agent or the molding template as well as during functionalization of the formed carbon phase.

Diffractograms at low angle for (a) SMS silica and its material derivatives; (b) SMK silica and its material derivatives.
Surface texture and elemental composition of synthesized materials
A high-resolution scanning electron microscopy (HRSEM) study also confirmed that carbonization and functionalization processes did not lead to significant changes in the textural characteristics printed by the molding template on silica matrices. Micrographs (Figure 2(a) and (b)) for the S/TC composite material show a cylindrical shape characteristic of particles of this type of siliceous materials as well as a system of longitudinally viewed cylindrical pores. The porous structure of the particles can also be seen in their cross section (Figure 2(c)), noting slightly deformed hexagonal arrays of cylindrical pores with an average size of 10 nm (Figure 2(d)).

(a to d) HRSEM micrographs present morphology and pore system for the particles of the S/TC material. HRSEM: high-resolution scanning electron microscopy.
HRSEM micrographs for the M/TC composite material showed well-developed porosity with the appearance of foam (Figure 3(a) and (b)), a large cellular structure characteristic of mesocellular silica and relative uniformity of the shape and size of the porous structure (Figure 3(c) and (d)). Pores ranging in size from 30 nm to 40 nm and wall thickness from 7 nm to 9 nm were detected. A study on the chemical composition of the silica/carbon compounds and their functionalized derivative materials allows us to determine that, in general, composite materials obtained from 2,3-dihydroxynaphthalene (DN) as an external precursor of carbon contain a greater percentage of this element than in silica/carbon materials obtained by direct carbonization of the molding template (Table 1). These differences in carbon content, depending on the precursor used, are the result of how carbon was deposited on the surface of the silica. In the case of impregnation with DN, a carbon layer is formed on the outer surface of the silica particles and on the inner surface of its pores, whereas during direct carbonization of the template a carbon layer is formed only on the inner surface of the pores.

(a to d) HRSEM micrographs showing morphology and system of pores for particles of material M/TC. HRSEM: high-resolution scanning electron microscopy.
Elemental composition obtained by XEDS microanalysis.

These results confirm the lower carbon content in the silica/carbon composites obtained by direct carbonization of the molding template as well as a correlation between the carbon content and the C/Si ratio, with the expected pore size and structure for each material.
Surface functional groups of functionalized composite materials
Fourier transform infrared results confirmed that the oxygenated groups responsible for adsorption and the reactive properties of synthesized materials were generated on the carbon surfaces of composite materials after chemical activation. 10,11,21,22 Since all silica/carbon composites derived from SMS, SMK, and SMM silicas exhibited very similar IR spectra, only spectra for composite materials K/TC8, K/TC8O, and K/TC8 S are presented in Figure 4, as representative of the complete set of materials studied (K/TC8S is included to emphasize the oxidation effect of the carbon phase).

IR spectra of silica/carbon K/TC materials, functionalized and nonfunctionalized. IR: infrared.
The XRD analysis showed that the K/TC8 composite material largely retained the characteristics of the porous system of the meso-structured silica SMK, while the result of the IR analysis presented in Figure 4 indicates that K/TC8 also has structural characteristics similar to those of the silica xerogel SMK. 23 This result indicates that the carbonization of the molding template formed low-dense carbon layers, whose characteristic IR bands did not overshadow those of pure silica, on which they overlapped by appearing in similar frequency ranges 10,11 ; XEDS analysis showed that materials obtained by direct method contain on average up to 40% less carbon, than those obtained by conventional method.
The carbon phase present in the K/TC8 material was oxidized or sulfonated to produce the K/TC8O and K/TC8S composites with acidic sites on their surfaces; the case of the K/TC8S material is considered only to highlight the effect of oxidation on carbon surfaces. In general, the IR spectra of these materials also showed the same band pattern as K/TC8, however, some variations in the intensity and widening of certain bands are noticeable, as well as the appearance of new ones of low intensity. Since the functionalized composites K/TC8O and K/TC8S were prepared from K/TC8, we can infer that the increase in the intensity of the IR bands by 464, 803, 1085, and 3436 cm−1 is evidence of tension vibrations of new OH and carboxyl groups, produced by the oxidation of the carbon phase in material K/TC8; among the oxygenated groups produced by oxidation of carbon surfaces with H2SO4, those that determine the acidity of these materials are the carboxyl and phenolic groups. 24,25
In the case of material K/TC8S, whose characterization and application are not reported in this work, the treatment of the carbon phase with H2SO4 also produced sulfonic groups (–SO3H), which were recognized by their IR signals in the range 1189–1040 cm−1 and in 631 cm−1, produced by tension vibrations of the C–S bond. 26,27 The IR spectrum of K/TC8O also shows three small signals in the range 1664–1773 cm−1 that indicate the presence of carboxyl, ketone, and lactone functional groups, which are characteristic of active carbons. Acid coals are characterized by high concentrations of this type of oxygenated groups superficial. Oxidation of the carbon layers with ammonium persulfate in H2SO4 preferably generates carboxylic and phenolic groups, while sulfonation generates carboxylic and sulfonic groups. The ionization of these groups in aqueous media results in a negative charge density on the surface of the carbon layers, which can be used for the retention of cationic species in solution, as is the case with the MB dye that is reported. 24,25
Textural characteristics of synthesized materials
Data on the adsorption–desorption of nitrogen at the boiling temperature allow an accurate evaluation of the structural properties of porous solids. In the textural analysis of the synthesized composite materials, the isotherms of the pure silica, silica/carbon compounds manufactured by conventional method using external carbon precursors, and the composites obtained by the nonconventional method through direct carbonization of the organic template were compared. The textural properties of materials, determined by quantitative processing of sorption data using standard Brunauer-Emmett-Teller (BET) and Brunauer-Joyner-Halenda (BJH) methods, are summarized in Table 2.
Texture properties of the synthesized materials.
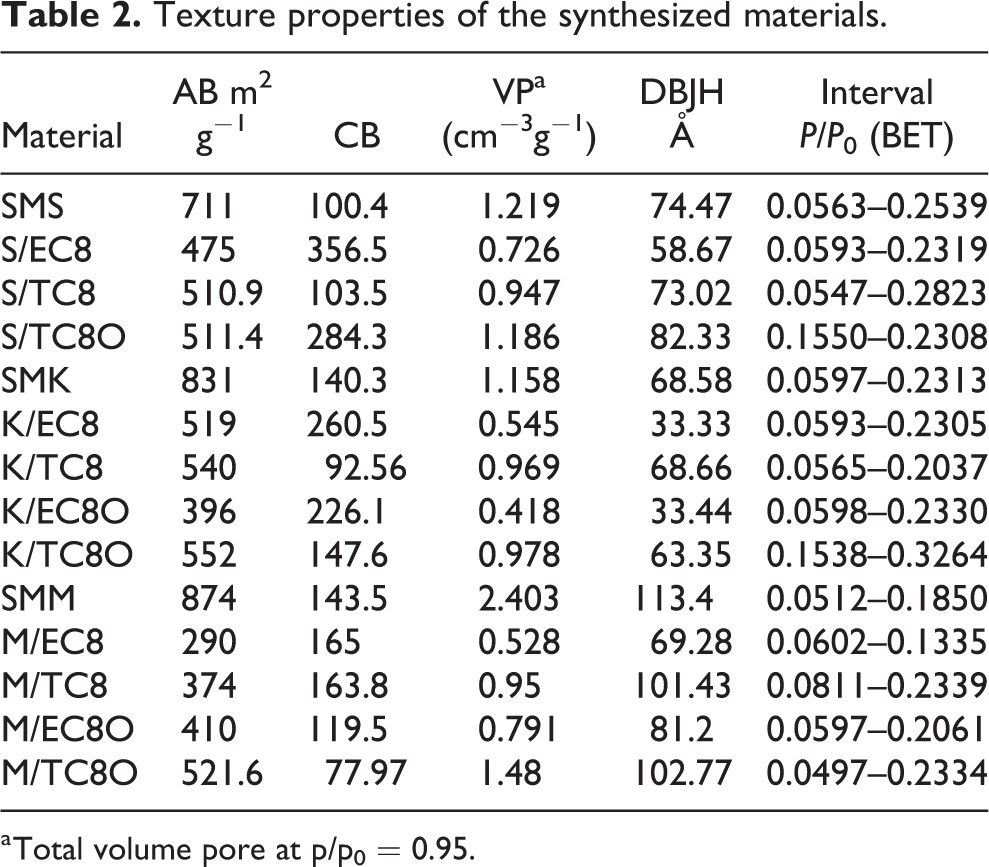
a Total volume pore at p/p0 = 0.95.
All silica/carbon materials derived from SMS silica exhibited Type IV adsorption isotherms (IUPAC) presented in Figure 5, with a typical capillary condensation step of materials with mesopores greater than 40 Å (P/P 0 > 0.6) and with H1 hysteresis cycle that reveals relatively uniform porous systems in shape and size 28 –30 ; transmission electron microscopy analysis revealed hexagonal arrangements of long cylindrical pores, similar to those of SBA-15 silica.

(a) Sorption of N2 on SMS meso-structured silica and silica/carbon derived composites: obtained from external (S/TC8) and internal (S/TC8) precursor and by functionalization (S/TC8O); (b) pore size distributions of pure SMS silica and derived silica/carbon materials.
The comparison of the isotherms for the S/TC and S/EC materials with those of pure SMS silica shows that the carbon precursor carbonization process produces a decrease in the specific surface area and the total pore volume. However, textural properties are better preserved in S/TC material due to the formation of a thinner carbon layer. The pore size distributions (PSDs) for S/C type materials, presented in Figure 5(b), were unimodal and narrow in the range of 50–90 Å. In general, the comparison between the adsorption isotherms of pure silicas and those for materials of the S/TC and S/EC classes shows that the carbonization process of any carbon precursor (external or internal) causes a decrease in specific surface area and volume total pore (Table 2). However, textural properties are best preserved in S/TC type materials, due to the formation of less dense carbon layers.
On the other hand, according to the adsorption–desorption results shown in Figure 6, materials K/TC8 and K/TC8O retained the uniformity of the pore system as in SMK, but the hysteresis H1 of the isotherms for materials K/EC8 and K/EC8O reflect a notable reduction in specific surface area, pore size, and pore total volume (Table 2). Both SMK silica and derived silica/carbon composites have Type IV isotherms and H1 hysteresis cycles, but with notable differences. 28 –30 As a general rule, composite materials of class K/C retain the three-dimensional meso-ordered structure of pure SMK silica but exhibit varying degrees of deformation. Adsorption isotherms and smaller hysteresis cycles for conventional K/EC materials indicate a greater loss of textural characteristics after the formation of the carbon layer and functionalization processes.

(a) Sorption of N2 on SMK mesostructured silica and silica/carbon derived composites: obtained from external (K/TC8) and internal (K/TC8) precursor and by functionalization (K/TC8O); (b) pore size distributions of pure SMK silica and derived silica/carbon materials.
PSDs presented in Figure 6(b) for this group of materials confirm the presence of relatively uniform mesopores and reflect the expanding effect of n-butanol between the self-assembled hydrophobic arrangements of P123, which results in mesopores larger than those in materials derived from SMS silica as well as in a slight loss in pore size uniformity.
Both pure SMM silica and silica/carbon derived composite materials exhibited the Type IV adsorption isotherms presented in Figure 7(a). Sorption in SMM, M/TC8, and M/TC8O exhibits a large cycle of H1 hysteresis, displaced at high relative pressures, corresponding to materials with pores much larger than those present in SMS and SMK silicas. The shape of the adsorption isotherm and the hysteresis cycle for SMM are consistent with the “ink bottle” mesopores, consisting of wide and uniform bodies, and interconnected by smaller necks. 31,32

(a) Sorption of N2 on SMM meso-structured silica and silica/carbon-derived composites: obtained from external (M/TC8) and internal (M/TC8) precursor and by functionalization (M/TC8O); (b) pore size distributions of pure SMM silica and derived silica/carbon materials.
Similarly, while adsorption isotherms and H1 hysteresis cycles for SMM and M/TC8O materials indicate large and uniform spherical mesopores (P/P 0 > 0.75), for M/TC8 they suggest a large blockage of the connections between the spherical cavities of the mesocellular structure of the material and a specific surface reduction compared to SMM (Table 2). This negative effect was greater in the case of conventional materials M/EC8 and M/EC8O and was remarkably reversed in the case of unconventional material M/TC8O during treatment with acid solution for its functionalization. For composite materials M/EC and M/EC8O, the great deformation of the hysteresis cycles H1 suggests changes in pore geometry and relative losses in the uniformity of the porous structure; this can be the result of accumulation of carbon within the large cavities of the mesocellular silica, which also affects the specific surface and total pore volume of the materials.
The pore size distributions for the M/C materials presented in Figure 7(b) proved to be quite wide compared to those for the S/C and K/C composites. The large mesopore size is a result of the dissolution of the TMB auxiliary in the nucleus of the supramolecular template of Pluronic P123, which causes an increase in the pore size of the inorganic phase due to a swelling effect. The TMB/P123 ratio used in the synthesis of these materials induces a phase transition from a hexagonally arranged cylindrical mesoporous structure as in SBA-15 silica to an MCF structure with a continuous three-dimensional system of large spherical pores uniformly interconnected by windows. 33 M/C composites have heterogeneous PSD in the range of 30–200 Å. This difference with respect to the PSD for pure SMM silica is probably due to the irregular thickness of the carbon layer that is generated and the different effects on the porous structure produced by the carbonization and functionalization processes (blockages, unblocks, or structural contractions).
The PSDs presented in Figures 5(b), 6(b), and 7(b), for all the materials studied, reflect the more or less negative effects on the textural properties of the carbonization and functionalization processes. Obviously, carbonization processes can lead to partial blockage of the porous system. Thus, in conventional silica/EC materials, the blocking effect is more pronounced than in silica/TC composite materials. Conventional impregnation of the porous structure with an external precursor (DN) to obtain the carbon phase leads to the generation of a larger amount of carbon both on the outer surface of the solid particles and on the walls of their pores (16–22%), in contrast to a smaller amount (8–15%) that produces by the carbonization of the surfactant template (P123) only on the walls of pores; this consideration was confirmed by the study of the elemental composition by the method of XEDS. A large amount of carbon can reduce the diameter and change the morphology of the pores. A total blockage of a fraction of the porous system may also occur, which leads to a decrease in the specific surface area of the solid and its total pore volume. In contrast, the synthesis of silica/carbon composite materials from direct carbonization of the molding template, using the low H2SO4:silica/surfactant hybrid ratio, generates a lower proportion of carbon phase. Thus, as can be seen from the results presented in Table 1 for the silica/carbon composites obtained by the direct process, the shape and dimensions of the pores of the parent material are better preserved and the specific surface reduction is also minor.
Adsorption activity studies
In the removal of contaminating dyes from industrial wastewater, physicochemical methods such as adsorption are most widely used. The silica/carbon composite materials functionalized with oxygenated groups and designated as S/TC8O, K/TC8O, and M/TC8O were tested as MB adsorbents (Figure 8) in an aqueous medium.

Methylene blue molecule.
In this part of the work, standard aqueous solutions of MB with concentrations of 20, 50, 100, 200, and 300 ppm were used. In a typical assay, 10 mg of the functionalized material is dispersed in 40 mL of the MB solution under ambient temperature conditions and constant agitation; in all cases, the maximum contact time was 2 h. The residual concentration of the MB solutions in contact with the functionalized material was assessed using UV spectroscopy by measuring the maximum absorption at 663 nm, which is the wavelength at which the dye typically absorbs. Functionalized composite materials S/TC8O, K/TC8O, and M/TC8O removed 85% of the dye from an aqueous solution with 20 ppm after 2 h of treatment (Figure 9); the characteristic absorption band of the MB almost disappears.

Efficiency of S/TC8O, K/TC8O, and M/TC8O materials in the adsorption of aqueous MB at pH ∼6.5, C I = 20 ppm.
The amounts of adsorbed MB in equilibrium over materials synthesized by conventional method and composites obtained by carbonization of the template are presented in Table 3.
Comparison of the MB quantities adsorbed in equilibrium from standard aqueous solutions of different initial concentration by Silica/EC8O and Silica/TC8O composites.

The adsorption equilibrium data for the composite-MB system were quantitatively treated according to the adsorption models of Langmuir and Freundlich.
34
Figure 10(a) shows the adsorption isotherms of the composite materials of the silica/CP8O family. The Langmuir model assumes a selective chemisorption mechanism on energetically equal adsorption sites, leading to the formation of an adsorbate monolayer that saturates the surface exhibited by the solid at the adsorption–desorption equilibrium. This model is formulated mathematically by the equation: (a) MB adsorption isotherms for S/TC8O, K/TC8O, and M/TC8O; Y-axis displacement: 0, 0.1, 0.2; (b) quantitative analysis of the adsorption data using the Langmuir model for the composite materials S/TC8O, K/TC8O, and M/TC8O; Y-axis displacement: 0, 5, 10; (c) quantitative analysis of the adsorption data using the Freundlich model for the composite materials S/TC8O, K/TC8O, and M/TC8O; Y-axis displacement: 0.3, 0, −0.3.

or, more conveniently, by its linear form

Quantitative analysis according to Langmuir and Freundlich models for the adsorption of aqueous MB on Silica/TC8O type composite materials.


where K f and n are the Freundlich constants related to the adsorption capacity and the factors that favor the adsorption process, respectively. The linear form of the model is expressed as

A plot of log q e against log C e should produce a straight line with slope equal to 1/n and intercept equal to log K f, from which the constants n and K f can be calculated (Table 4). Figure 10(c) represents the graphs of the Freundlich isotherms for the three functionalized composite materials studied.
Table 4 summarizes the n max monolayer capacity values and the corresponding adsorption–desorption equilibrium constants. Of the two adsorption models tested, the one that best fits the experimental adsorption data is chosen from the value of the regression coefficient (R 2). The comparable value of R 2 observed for the three MB-adsorbent systems studied suggests that the Langmuir or Freundlich models can satisfactorily describe the adsorption of MB on the composite materials S/TC8O, K/TC8O, and M/TC8O. The relatively large values of the equilibrium constants K L suggest that the adsorption of MB on the surface of the materials is favored under the conditions (temperature, pH) at which the physical contact between the adsorbate and the adsorbent occurs.
The functionalized composites obtained by direct carbonization of molding template as a carbon source, exhibited a great capacity of adsorption, similar or superior to that reported for other adsorbent materials by different authors, which operate under restrictive conditions in same applications type (Table 5).
Comparison of adsorption capacities and operating conditions for various adsorbing materials in the removal of methylene blue in aqueous medium.

a0.25 g L−1.
b0.50 g L−1.
c0.20 g L−1.
Comparison of the results obtained for Silica/TC8O and Silica/EC8O composite materials (Table 3) reveals that both types of materials adsorbed the similar amounts of MB from aqueous solutions with initial dye concentrations less than 100 ppm. But for initial concentrations in the 200–300 ppm range, the retention capacity exhibited by the Silica/TC8O materials was lower (approximately 40%) than that of the Silica/EC8O compounds (Table 3), probably due to the absence of a layer of functionalized carbon on the outside of the particles in the first materials and, therefore, smaller populations of acidic sites. Despite this result, the adsorption capacity of composite materials of the Silica/TC8O family is potentially competitive with respect to other adsorbing materials tested in the retention of cationic dyes in an aqueous medium (Table 5). In addition, the Silica/TC8O materials did not undergo any reactivation treatment prior to its application, and adsorption tests were developed under conditions similar to those of wastewater treatment without adjusting the pH (pH 6–6.5), avoiding the need to add any other substances that must be neutralized or extracted later.
Conclusions
As an alternative to the procedure commonly used to produce mesoporous silica/carbon composites, this work took into account the underutilized idea of the direct carbonization of the structuring organic template contained in the precursor hybrid materials of mesostructured silicas of type SBA-15, KIT-6, and MCF. The implementation of this idea allowed to develop a short, cost-effective, and reproducible procedure to prepare silica/carbon composites containing mesoporous of controlled sizes and shapes. In certain aspects, it should be noted that the characteristics of this procedure is consistent with postulates of green chemistry.
The structural and textural characterization of materials labeled S/TC8, K/TC8, M/TC8, S/TC8O, K/TC8O, and M/TC8O showed that the carbonization and functionalization processes do not significantly modify the structural qualities planned for porous systems (Table 1, Figures 5 to 7). This is a great advantage over conventional procedure to obtain the same type of silica/carbon composite material, since in this case, the impregnated precursor covers the entire surface and fills any cavity in the pure silica matrix. Its subsequent carbonization may partially or completely overlap the mouth of the pores and cause a decrease in specific surface area and total pore volume (Figure 2 to 7, Table 2). The carbon layer of the silica/carbon composite materials obtained by the direct method was subsequently oxidized to generate oxygenated groups anchored on its surface (–COOH, –C=O, –C–OH, –C–O–) (Figure 4). When these functionalized materials are used to treat wastewater contaminated with cationic dyes, these oxygenated groups are responsible for retention of the dye simply by electrostatic attractions. Due to the nature of the interaction between the adsorbate (MB) and the functionalized silica/carbon materials, adsorption can be satisfactorily interpreted in terms of the Langmuir and Freundlich models. Monitoring the concentration of the dye in solution using UV-Vis spectroscopy only recorded a decrease in the absorption band characteristic of MB at 663 nm, excluding the possible degradation of the dye on the surface of functionalized silica/carbon composites.
In addition, the silica/carbon composite materials prepared by direct method have structural and textural properties, which make them ideal material platforms for the design of advanced materials with applications in adsorption and catalysis (Table 2). The carbonization processes and the subsequent incorporation of oxygenated functional groups into the carbon layer did not significantly affect the porous structure of the silicon oxide matrices taken as base ingredients to produce materials with porous systems of appropriate sizes and shapes to allow mass flow of gaseous or soluble chemical species in applications such as adsorbent materials, as well as reagents and products in possible catalytic applications. In general, materials functionalized with oxygenated groups, whose silica/carbon matrices were obtained by direct carbonization of the structuring template of the porous system, exhibited adsorption capacities that allow their direct use in the remediation of wastewater contaminated with cationic dyes such as blue methyl or with cations heavy metals such as Pb, Cr, or As, without further conditioning requirements for the adsorbent or for the water to be purified.
Footnotes
Acknowledgments
The authors sincerely appreciate the financial support of the National Council of Science and Technology (CONACYT, Mexico) through the scholarship No. 423957. The authors also acknowledge the technical support provided by Faculty of Chemical Sciences of the Autonomous University of Puebla (BUAP), Institute of Physics of Autonomous University of Puebla (IFUAP), Waste Revaluation Laboratory, CINVESTAV—Unidad Saltillo, University Center for Linking and Transfer of Technology, BUAP (CUVyTT-BUAP), and Body of Research in Zeolites, BUAP-CA-95.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
