Abstract
Intraspecies aggression is commonly focused on securing reproductive resources such as food, territory, and mates, and it is often males who do the fighting. In humans, individual acts of overt physical aggression seem maladaptive and probably represent dysregulation of the pathways underlying aggression. Such acts are often associated with ethanol consumption. The Drosophila melanogaster model system, which has long been used to study how ethanol affects the nervous system and behavior, has also been used to study the molecular origins of aggression. In addition, ethanol-induced aggression has been demonstrated in flies. Recent publications show that ethanol stimulates Drosophila aggression in 2 ways: the odor of ethanol and the consumption of ethanol both make males more aggressive. These ethanol effects occur at concentrations that flies likely experience in the wild. A picture emerges of males arriving on their preferred reproductive site—fermenting plant matter—and being stimulated by ethanol to fight harder to secure the site for their own use. Fly fighting assays appear to be a suitable bioassay for studying how low doses of ethanol reshape neural signaling.
The Purpose of Aggression
Intraspecies aggression is commonly focused on securing reproductive resources such as food, territory, and mates, and it is most often males who fight each other for control of reproductive resources. This is usually considered separately from interspecies aggression, which—while it can involve the control of resources—often involves a predator-prey relationship and is clearly subject to different evolutionary pressures. However, both types of aggressive actions appear to be evolutionarily appropriate in that dominance in the competition helps secure reproductive success. Evolutionary inappropriate or pathological aggression is probably maladaptive, having no positive effect on reproduction, and may arise from dysregulation of the signaling pathways and circuits that underlie aggressive behaviors.1,2 With regard to individual acts of overt physical aggression, humans seem biased toward maladaptive forms of aggression. In humans, ethanol consumption increases the probability of pathological physical violence between individuals. Government statistics and forensic studies show a positive correlation between habitual alcohol consumption and violent behavior of all types including homicide (summarized in Alcohol and Violence, 3 and Attwood and Munafò 4 ).Furthermore, after adjusting for past aggression and a number of other covariates, a recent epidemiological meta-analysis of 4 cohort studies, totaling 6706 persons, demonstrated that heavy episodic drinking of alcohol is predictive of future aggression. 5
The molecular origins of aggression have been studied in a wide variety of animals and appear to reveal distinctly different underlying mechanisms. However, some of these apparent differences may be artifacts of the techniques available for use in each animal model system. For instance, vertebrates are large enough that surgeries, electrode placement, and the measurement of neurotransmitter, neuropeptide, or hormone levels were quickly practical and their use led to a description of the seeds of aggression in terms of changes in the activity of brain regions and changes in the balance of these molecules.1,6 However, in vertebrates genetic screens to identify the master regulatory switches of aggression have not been feasible. In the Drosophila invertebrate model system, the opposite is true, and genetics has been a practical way to identify master gene switches and neural circuits that by themselves are sufficient and/or necessary for producing aggression. The respective advantages and disadvantages of each model system have led to different stories on the origins of aggression. Despite this, the 2 fundamentally differing approaches have generated a picture of aggression that is starting to overlap (eg, Asahina 7 ).
As in mammals, in D. melanogaster male-male aggression for control of reproductive resources is the norm. While females do fight, they seem less aggressive than males, because even after a fight the winner will usually share the reproductive site with the loser (ie, lasting dominance hierarchies do not emerge as a product of the fighting8,9). Fighting between male flies has many of the properties that one might expect: larger males tend to win their fights; defending a food patch is more often associated with success than is displacement of a resident male; fights usually end before damage occurs; and fighting appears stressful in that, even in the absence of obvious physical damage, repeated fighting compromises the health of the animal. In flies, as in mammals, aggression is also modulated by prior experience—males alter their fighting strategy depending on their past performance and also show a clear loser effect and fighting between males establishes stable hierarchical relationships. Male flies even remember individuals they have previously defeated or lost to and behave differently in accordance with this information. Losing a fight seems to undermine a male’s confidence, making him more likely to lose any subsequent bout. Territorial male behavior has been seen in the wild but only on small food surfaces, suggesting that the resource must be limiting to make fighting worthwhile.10-12
Early studies on the interaction of D. melanogaster with ethanol focused on the fact that decaying plant matter provides a reproductive niche (Figure 1) for many Drosophila species whose occupancy correlated with the degree of microbial decay. 13 Studies of this kind often took advantage of the controlled introduction of fermenting fruits into a landscape in the form of wineries (these research sites no doubt also had nonscientific advantages for the investigators). McKenzie and McKechnie 14 compared the distribution of D. melanogaster and D. simulans surrounding a winery and reported that while larvae and pupae of both species were observed in post-fermentation residues, essentially only D. melanogaster larvae and pupae were present during the fermentation stage. The unusual success of D. melanogaster in exploiting fermenting foods as a protected niche was well illustrated in the observations of Marks et al 15 who showed that D. melanogaster is often the only Drosophilid within a winery even if it is a minority member of a guild of 7 to 8 Drosophila species living in the area around the winery. The reason for this appears to be that D. melanogaster adults, larvae, and eggs tolerate ethanol concentrations that are poisonous to some other Drosophilid species.16,17 Thus, while the conversion of sugar in the food to ethanol reduces the food's net caloric value, ethanol helps make the food a partially protected niche for D. melanogaster by reducing the number of competitors. Furthermore, ethanol’s ability to inhibit the growth of parasites and perhaps other pathogens makes the food safer for consumption 18 —a benefit that at one time also promoted the consumption of ethanol-rich beverages and food in humans. D. melanogaster larvae further exploit the medicinal value of ethanol when they cure an endoparasitoid wasp (Leptopilina) infection by increasing their consumption of ethanol-rich food. 19 The importance of consuming ethanol-rich food to cure a parasitic wasp infection is a nontrivial consideration given that Leptopilina parasitism of D. melanogaster larvae is typically 5 to 40% (with 90% parasitism rates not unheard 20 ). Ethanol clearly plays an outsized role in the life of D. melanogaster, and it would be surprising if this role were limited to the aspects above.

Male and female Drosophila melanogaster associating on fermenting grapes. The scent of the ethanol and the consumption of the ethanol-rich food causes males to become more aggressive.
In the context of alcohol-use disorder (AUD), D. melanogaster have long been used as a model system for studying the behavioral effects of ethanol and identifying genes and signaling pathways important for the adaptation of the nervous system to ethanol. 21 This is largely because the genetics of Drosophila greatly outpaced the capacity to genetically manipulate mammalian model systems, albeit this advantage may be slipping away. There are many similarities in the behavioral and physiological responses of flies and humans to ethanol. Both voluntarily consume ethanol and use it for medicinal purposes. Both also show similar dose-dependent responses to ethanol, becoming behaviorally hyperactive at low doses, incoordinated at intermediate doses, and sedated at high doses. Flies can also acquire ethanol tolerance (ethanol-induced ethanol resistance) that is dependent on the adaptation of the nervous system to ethanol. Like humans, D. melanogaster can be made functionally dependent on ethanol and show signs of ethanol withdrawal.22,23 Flies also perceive ethanol as positively rewarding. 24 The genes and pathways that underlie these and other responses are also shared between flies and mammals and include neurotransmitter receptors, pathways involved in learning, the neuroimmune system, and circadian rhythms (reviewed in Park et al, 21 Petruccelli and Kaun 25 ). In my laboratory, we asked whether D. melanogaster was a good model for studying ethanol-induced aggression. As in humans, in flies ethanol exposure correlated with a change in aggressive behaviors. Unexpectedly, both the odor of ethanol and elevated blood ethanol concentration (BEC; also synonymously called blood alcohol concentration and also abbreviated BAC) enhanced aggression.
Just the Scent of Ethanol Makes Flies Aggressive
Because in antennae the same protein that helps recognize the pheromone 11-cis-vaccenyl acetate (cVA) also binds ethanol, it seems sensible to suspect that the odor of ethanol might influence aggression. cVA is a male fly pheromone that can trigger different behavioral responses depending on concentration and duration of exposure. The behavioral effects of cVA include aggregation of flies, female aphrodisia, the inhibition of male courtship (after transfer to the female), and aggression between males upon their first encounter with another's cVA. 26 The cVA pheromone is bound by the LUSH odorant binding protein in the antenna, and the cVA:LUSH complex then interacts with the OR67d/ORCO cation channel, thereby activating the olfactory neurons to signal detection of cVA (reviewed in Ziegler et al 27 ). The LUSH protein was shown to be required for the detection of toxic levels of ethanol and was the first non-enzymatic protein shown to physically bind ethanol. A LUSH:ethanol crystal structure was solved to better understand the structure of high-affinity ethanol binding.28,29
Monitoring Drosophila fighting is a simple exercise. Traditionally, one only needs 2 males, a small plastic chamber, a platform of food with or without a decapitated female in the center, and a video camera. The purpose of the food and the female are primarily to localize the 2 males into a predicable location so that they are in the plane of focus of a camera. However, the presence of food and the female can increase male aggression.30,31 Normal male aggression requires the visual, olfactory, and gustatory systems. Males in a fighting chamber see each other and sense the olfactory pheromone cVA. They approach and begin fencing. When males fence, they reach out and touch each other, and the gustatory receptors on their legs taste the other animal’s pheromones. 26 The detection of a second male pheromone, (z)-7-tricosene, drives more overt acts of aggression. 32 One then commonly sees lunging (rising on the back 4 legs to strike down on the opponent fly with their 2 front legs) and wing threats (flashing a V-shaped wing position), and, less commonly, shoving, tussling, and boxing.
In Park et al 33 the response of flies to the odor of ethanol was investigated, and it was shown that the aggressiveness of males was increased by the presence of ethanol in the food of the fighting platform. The peak aggressiveness occurred at 5% ethanol for time spent fighting, number of fights, and number of lunges and at 10% ethanol for latency to lunge and proportion of flies lunging. These concentrations did not cause an increase in blood ethanol, probably because males rarely fed on the food and because the duration (30 min) in the chamber was too short to produce a systemic change in blood ethanol. An ethologically irrelevant 20% ethanol concentration, which elevated the BEC, produced a general reduction in aggressiveness. The mechanism underlying the ethanol odor-induced aggression was accounted for by the response of the sensory neuron. Recording from the cVA-sensing T1 antennal neurons showed that ethanol vapor potentiated the response of the neuron to the cVA pheromone.
Potentiation of a behavioral response to an odor may be a fundamental attribute of ethanol in a fly's world. Demonstration that the scent of ethanol enhances the attractiveness of a food odor is an old discovery. One of the first D. melanogaster publications, by Barrows, 34 showed that flies use their sense of smell to find food and that food odorants (including ethanol) synergize to be much more attractive than either odorant alone. Since then, this experiment has been repeated and improved upon many times by many laboratories (eg, Zhu et al 35 ). Park et al 33 reported that ethanol vapor potentiated the behavioral response to farnesol—an attractive odorant common to citrus fruits. An olfactory trap assay was used to determine the behavioral preference of flies for various concentrations of farnesol and ethanol. Farnesol plus ethanol was much more attractive than either ethanol or farnesol alone. The ai2 sensilla that senses farnesol is highly specific for this odorant and does not respond to ethanol alone. 36 Nonetheless, Park et al 33 showed that ethanol potentiated the response of these sensory neurons to farnesol. One would like to generalize the mechanism of synergy proposed for cVA-sensing neurons, but we cannot because other odorant binding proteins have not been tested for ethanol binding and recognizing an ethanol-binding pocket by sequence analysis is impractical. 37
Increased Blood Ethanol Level Also Promotes Aggression
The olfactory responses to ethanol described in Park et al 33 did not involve a detectable change in blood ethanol concentration (BEC). The question of how elevating blood ethanol level affected aggression was addressed in a paper the following year (Park et al 33 ). Three different BECs were tested by treating flies in an ethanol-vapor chamber. They were 1) a standard rapidly sedating dose of ethanol (~170 mM BEC), 2) a very low dose of ethanol (~1 mM BEC) and 3) an extremely low dose of ethanol (~0.3 mM BEC).38,39 For the corresponding standard %BEC units used for humans in the United States, these are approximately 0.78, 0.0046, and 0.0015 %BEC, respectively. The highest-dose animals were behaviorally assayed 24 h after recovery from treatment, an interval needed to let the animals fully recover from sedation. Animals experiencing the intermediate dose were assayed immediately after treatment because the animals appeared ostensibly normal (did not show incoordination or ethanol-induced hyperactivity). The lowest dose was produced by allowing the intermediate dosed animal to metabolize ethanol for 1 h prior to testing. This last paradigm was not ideal, however, it was a tractable way to reproducibly produce such a low blood ethanol level. The top 2 doses of ethanol caused the males to be unusually docile while the lowest dose of ethanol caused the males to be much more aggressive (Figure 2). This aggression-producing ethanol treatment was referred to as the PEA (post-ethanol aggression) treatment. The mechanism underlying this increase in aggression appears distinct from olfactory-induced aggression because no potentiation of cVA signaling was observed in recordings from the cVA-sensing T1 antennal neurons.
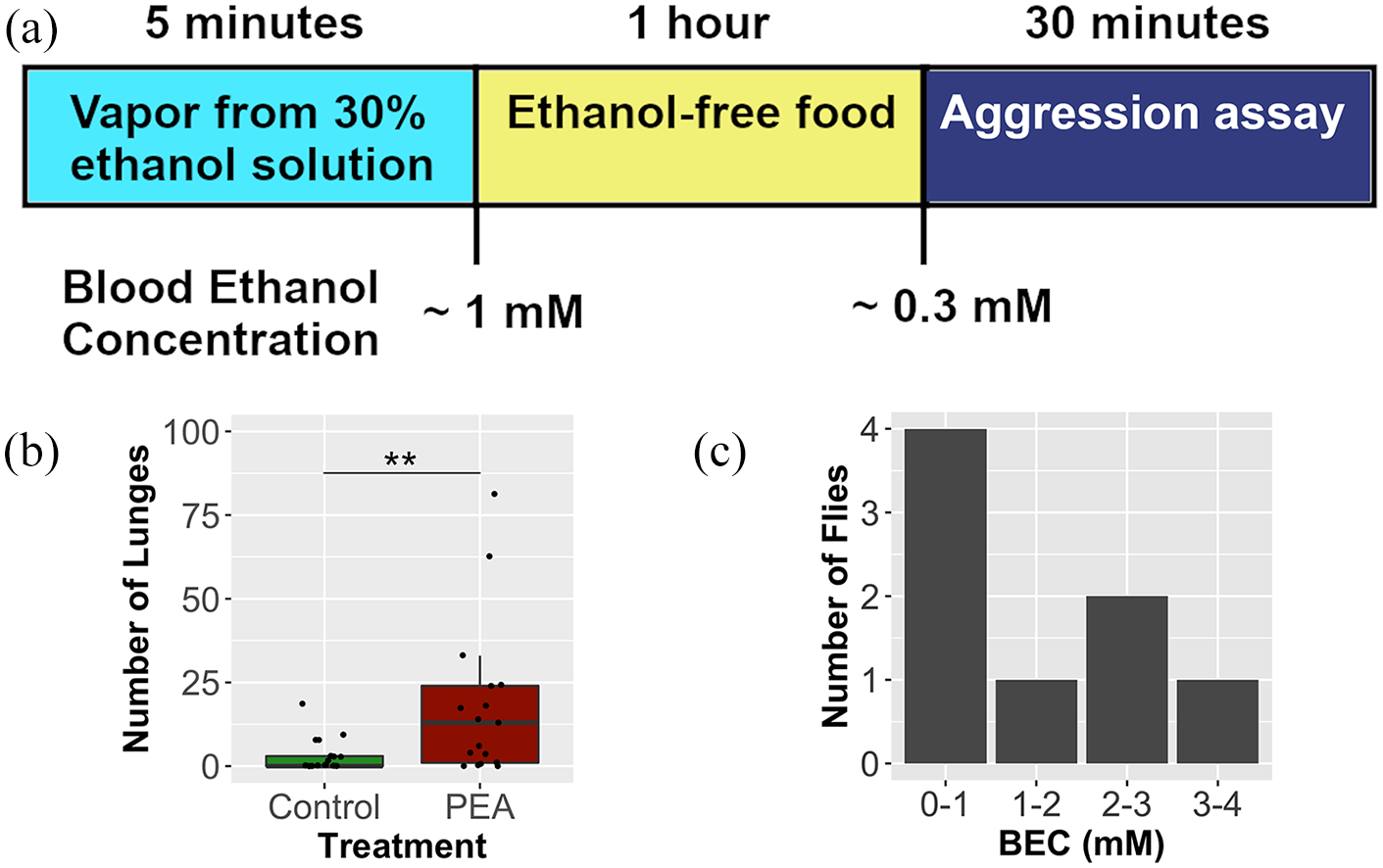
Aggression-inducing BEC can be induced by consumption of ethanol concentrations that occur in the wild: (a) the PEA (Post-Ethanol-Aggression) treatment that induces male aggression produces an initial 1 mM BEC. After 1 h this has dropped to 0.3 mM BEC and the males show increased aggression, (b) replot of data from Park et al 39 shows that PEA treatment increases aggression, and (c) data from Pohl et al 43 shows the BEC of 8 flies immediately after they voluntarily take a single sip of 10% ethanol food. BEC ranges up to 4 mM were observed, indicating that aggression-producing BEC can be produced by feeding.
Park et al 39 showed that the PEA treatment significantly reduced the latency to the first lunge and elevated the incidence of lunging and shoving. In this paper, the level of aggression was correlated with the expression of the FruM transcription factor. FruM is a male-specific transcription factor largely responsible for the exhibition of male-specific behaviors.40,41 The ethanol treatment that increased aggression also increased FruM protein abundance and ethanol treatments that reduced aggression reduced FruM protein abundance. Suppressing FruM induction also blocked ethanol-induced aggression. This correlation was inferred to mean ethanol modulation of FruM abundance could be responsible for PEA-induced aggressivity. Since FruM is a master regulator that generates male behavioral patterns, it is enticing to think that an ethanol-induced increase in FruM increases aggression because it increases maleness while a higher dose of ethanol, that decreases FruM, decreases aggression because it reduces maleness. Changes in FruM expression have been shown to affect male neurophysiology in ways predicted to alter behavior. 42
Are the 2 modes of ethanol-induction of aggression (olfactory and systemic, Figure 3) truly distinct? This is perhaps an open question. It is true that the olfactory study in Park et al 33 showed that systemic BEC was not increased by the olfactory treatment, and the systemic ethanol study in Park et al 39 showed that the PEA treatment increased BEC but did not potentiate the response of the cVA-sensitive antennal sensory neurons. However, remaining to be tested is whether or not the PEA treatment potentiates leg gustatory receptors used to detect the pheromone (z)-7-tricosene. This pheromone, tasted during fencing, is required for cVA-induced aggression. 32 This would be consistent with ethanol's role as a potentiator of sensory responses.
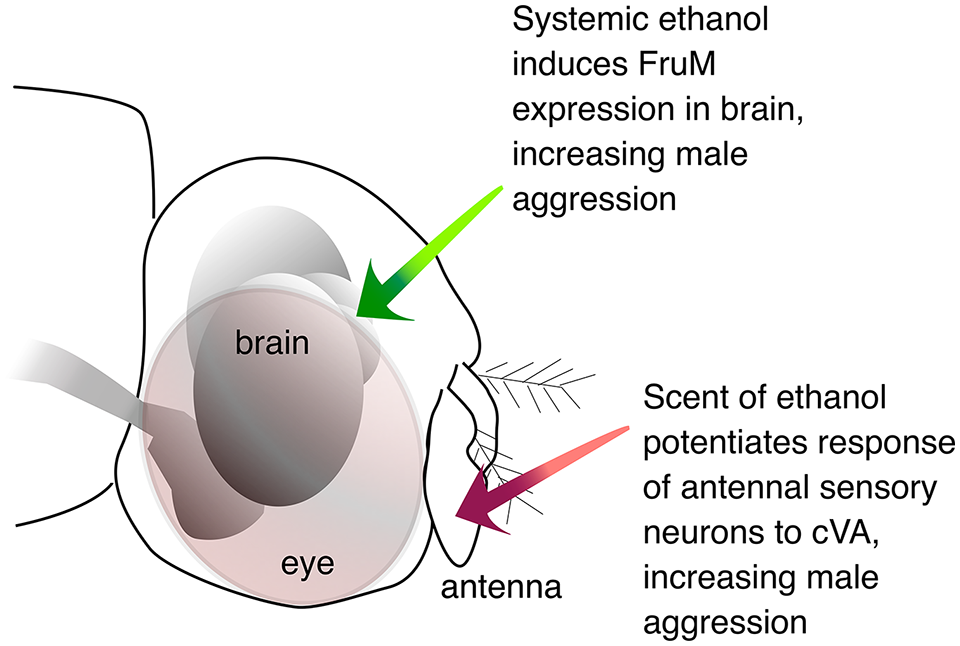
Summary. Ethanol acts at 2 positions to promote male aggression.
Relevance to the Natural History of Drosophila melanogaster
Are the effects of ethanol on aggression relevant to the behavior of D. melanogaster under natural conditions? Aggression has not been monitored in the wild in any detail. However, it appears likely that natural levels of ethanol will enhance male aggression. We propose that upon arrival on a fermenting substrate, the odor of ethanol will make flies more aggressive and that if the male consumes the food, ethanol will act systemically to promote aggression. This proposal is based on the following facts. Natural fermentation of fruits easily hit the 5% to 10% ethanol range and, in the lab, the odor of food supplemented with 5% ethanol increased male fighting. 33 What about feeding? Can a fly really consume enough ethanol-rich food to raise blood ethanol to the needed level? The answer is clearly yes. Pohl et al 43 showed that a single sip of 10% ethanol food can produce an internal BEC of 1 to 3.08 mM ethanol, indicating that even a brief feeding on fermented food could easily achieve systemic BEC levels needed to potentiate male aggression (Figure 2c). Therefore, not only is a food scented with ethanol more attractive to flies, such food would make males more aggressive. In this scenario, ethanol signals to the male that he has arrived on prime reproductive real estate and that he should fight harder for control of it and for the females who share it.
Do the Low Level Effects of Ethanol in Flies Have Relevance Toward Humans?
Ethanol has long been proposed to promote reproductive success of D. melanogaster. Ethanol is a positive component that demarcates preferred reproductive sites and provides the impetus for flies to hang onto them. Because 2 independent mechanisms have evolved that make males value ethanol-rich sites, I posit that the importance of ethanol is much greater than originally envisioned. The integration of ethanol into the natural history of D. melanogaster means that the fly's relationship with ethanol is of a different type than our relationship to ethanol. The role of ethanol in a fly's life is nonpathological. Does this mean that flies are a poor model for studying the pathological behavioral effects of ethanol? No. Each behavioral response to ethanol is a tool that the Drosophila behavioral geneticist can exploit to describe the underlying signal pathways. Thus, the extensive natural relationship of flies to ethanol in all likelihood represents opportunity for investigators.
In Park et al, 39 1 h after a treatment that produced a blood ethanol level of 1 mM, the authors observed increased aggression. By this time, the blood alcohol level had dropped to ~0.3 mM (so-called PEA treatment). What is not clear is whether the increase in aggression is a direct consequence of ~0.3 mM ethanol or whether it is a time-delayed consequence of the 1 mM ethanol BEC. For instance, aggression might be caused by the acetate produced as metabolism lowers the BEC from 1 to 0.3 mM. Acetate has recently been implicated as an important contributor responsible for some physiological changes previously ascribed to ethanol. 44 Flies may prove an ideal system for studying the role of acetate in ethanol responses.
Some may be disturbed by the idea that such a very low dose of ethanol (⩽1 mM) has any behavioral consequence at all. Usually, investigators focus on clearly intoxicating levels of ethanol (~ 17 mM range). However, a number of mammalian neuronal receptors and channels (eg, GABAA receptors, glycine receptors, and BK channels) are modulated by ethanol in the 1 mM range. 45 And, most impressively, mammalian neuronal acetylcholine receptor channels—which contribute to a great many ethanol responses 46 —are modulated by micromolar ethanol (EC50 [half maximal effective concentration] = 0.0885 mM). 47 Low doses of ethanol may subtly alter the nervous system and behavior to lay the groundwork for future more problematic effects of ethanol. The Drosophila aggression response may be a excellent model for studying how very low doses of ethanol, popularly perceived as benign, reshape brain function.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institute on Alcohol Abuse and Alcoholism Grant 2R01AA018037-06A1 to NSA. We thank Jane Kirschman for copyediting and feedback on the manuscript.
Declaration of conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NSA researched the article, wrote the article, and designed and drew the figures.
