Abstract
Background:
Outcome-based agreements (OBAs) may facilitate earlier patient access to promising therapies, particularly when evidence is limited. The authors of this paper investigated how to operationalize an OBA using real-world data (RWD) from the Canadian Neuromuscular Disease Registry (CNDR).
Objective:
The first objective of this research was to determine which spinal muscular atrophy (SMA) health outcomes in the CNDR are suitable for an OBA. The second objective was to evaluate the current process of data collection in the CNDR and explore how to operationalize data processes to support an OBA, including identifying gaps and proposing solutions.
Design:
This qualitative expert-led assessment was conducted through a series of focus group discussions with selected experts.
Methods:
A selected group of experts participated in 17 focus group meetings. The ability of the CNDR to generate real-world evidence (RWE) for an OBA was evaluated for eight SMA health outcomes. The criteria used were data readiness, interpretability, and timeframe within the CNDR. Next, the processes involved were evaluated, specifically to determine how data tracking within the CNDR could be operationalized for an OBA, including identifying gaps and possible solutions. Based on the findings, the group proposed a future state for an OBA process using the CNDR. The group followed up with stakeholder feedback interviews to validate the findings of this research and to gather insights.
Results:
Five SMA outcomes within the CNDR were identified as potentially suitable outcomes for OBA. Three process gaps were identified in the current state of the CNDR, and corresponding solutions were proposed. A proposed future-state process flow for CNDR was developed to support RWE generation for OBA.
Conclusions:
Expert consultations suggest that operationalizing an OBA using CNDR RWD is feasible.
Plain language summary
Outcomes-based agreements (OBA) are one way to give patients faster access to new therapies, especially when long-term clinical data is limited. These agreements link how well a drug works in the real world to how much is paid for it. For rare diseases like spinal muscular atrophy (SMA), real-world data (RWD) from patient registries can play a key role in supporting these types of agreements. This assessment looked at whether the Canadian Neuromuscular Disease Registry (CNDR) could be used to help operationalize an OBA for SMA treatments in Canada.
A group of experts took part in 17 focus group meetings to examine two main questions:
1. Which SMA outcomes currently tracked in the CNDR are most suitable for use in an OBA?
2. What are the current data collection strengths and gaps in the CNDR, and how could the registry be used to support OBA?
The group also proposed a step-by-step process for how the CNDR could be used to generate real-world evidence (RWE) for OBA and gathered feedback from healthcare stakeholders to validate the findings.
The group identified five SMA outcomes in the CNDR as suitable for inclusion in an OBA, based on criteria such as data readiness, clarity, and timeliness. They also found three key gaps in how data is currently collected and tracked. For each gap, practical solutions were proposed. A “future state” process was developed to show how CNDR data could be used to generate RWE to support of an OBA.
This assessment suggests it is feasible to use CNDR data to support OBA for SMA treatments in Canada. With some improvements to data processes, the CNDR could help enable more flexible, evidence-based and timely access to SMA therapies. Further research should also be conducted to empirically test the CNDR data quality.
Keywords
Introduction
Access to therapies for rare diseases
In Canada, rare diseases are those that affect fewer than 5 in 10,000 individuals. 1 Although each rare disease affects a relatively small number of people, the large number of distinct conditions means that approximately 1 million Canadians are living with a rare disease. 2 Despite the promise of novel therapies, Canada lacks a fully established national strategy for funding rare disease treatments. 3 The small patient population, low number of potential participants in clinical trials, and heterogeneous presentation of disease make it challenging to generate data to support healthcare decision-making. 3 There are a number of novel therapies coming to market for unmet needs in rare diseases that, although promising, have immature data and clinical uncertainty. This, along with long reimbursement timelines, restricts or delays the availability of these treatments via public plans in Canada, often by over 1.5 years. 4 While not commonplace in Canada, innovative funding solutions such as outcome-based agreements (OBAs) have sometimes been employed to enable patient access to such novel therapies. 4 Leveraging real-world evidence (RWE) within such agreements can enhance decision-making and accelerate access to treatments for patients with rare diseases. 5
Outcome-based agreements and real-world evidence
The growing number of promising therapies with imperfect data, coupled with long reimbursement timelines—particularly in rare disease and precision oncology—has made timely access to new therapies increasingly challenging. 6 Addressing such market challenges calls for novel funding policies, with OBA a notable emerging option. An OBA consists of an agreement between a manufacturer and payer in which the manufacturer will issue a refund or rebate to the payer based on a therapy’s performance in a real-world patient population, measured against an agreed-upon, pre-defined set of benchmarks. 7 OBAs enable patients to access promising new therapies while allowing additional RWE to be generated to resolve uncertainty and mitigating the risk of non-performance for payers. 8 OBAs are most suitable for high-cost therapies for which clinical trials may not be possible and treatment effectiveness is uncertain. Although these therapies pose significant financial risk to payers, they are often the only option for patients with few or no alternatives. 6 Challenges to the implementation of OBAs in Canada include a lack of access to quality real-world data (RWD), resources, infrastructure, and relevant knowledge, among others. 4
Prospective randomized controlled trials (RCTs) are the standard for determining the efficacy and safety of drug treatments. When the patient population is small and RCTs are not possible, patient registries can be an innovative way to generate evidence to inform decision-making. 9 An emerging option in Canada, patient registries collect valuable RWD data from people currently living with a disease. Canada’s Drug Agency (CDA-AMC) supports the investment in rare disease registries that collect RWD to address evidentiary uncertainties and strengthen medical decision-making. 10 The Canadian Neuromuscular Disease Registry (CNDR) is well-positioned to support this objective and contribute to the broader adoption of OBA in Canada. 11
Canadian neuromuscular disease registry
The CNDR is a Canada-wide registry of people diagnosed with neuromuscular diseases including spinal muscular atrophy (SMA, 5.3% of the registry), Duchenne muscular dystrophy (DMD, 13.3% of the registry), and amyotrophic lateral sclerosis (ALS; 36% of the registry), among others. Launched in 2010, this clinic-based registry is led by researchers at the University of Calgary, and governed and operated by a national investigator network. Research ethics board approval is obtained at the lead site, and at participating clinic site. The registry has been funded through a mix of government support, charitable organizations, and industry sponsors.12,13 Initially, the registry’s primary goal was to increase efficient patient access to cutting-edge research and clinical trials. Secondary objectives were to increase the understanding of the natural history and epidemiology of neuromuscular disease across Canada and facilitate a more collaborative research community. Over the past decade, the CNDR has expanded its scope to include improving health outcomes and supporting pharmacoeconomic analyses. More recently, it has begun collecting post-approval RWE for novel therapies. 11 Additionally, the CNDR participates in a coordinated multinational effort to recruit patients for high-impact research and supplies data to global registries. 12 The CNDR benefits patients and physicians by enabling long-term patient follow-up, access to comparators from across Canada, possible clinical trial recruitment, and notifications of new drugs.11,12
The SMA dataset within the CNDR was first launched in 2012 as a longitudinal, prospective, observational study to evaluate the safety and effectiveness of novel therapies for SMA and provide practical information unattainable in clinical trials. 14 This dataset was expanded in 2019 and now includes a number of recorded health outcomes such as motor function, pulmonary function, patient-reported outcomes, number of hospitalizations, and types of therapies used. Currently, 27 clinic sites across Canada provide SMA data to the CNDR, from both pediatric and adult patients. The only data available in the CNDR come from patients who have provided informed consent and registered. All data are collected from the patient’s chart, manually extracted, and entered into the CNDR.
Spinal muscular atrophy: Treatments, coverage gaps, and patient impact
SMA is a recessive genetic neuromuscular disorder that affects 1–2 per 100,000 people. The irreversible loss of motor neurons that characterizes SMA leads to progressive muscle weakness and atrophy, making early detection and treatment essential for maintaining muscle function.15,16 There are currently three therapies for SMA available in Canada: nusinersen (Spinraza), onasemnogene abeparvovec (Zolgensma), and risdiplam (Evrysdi). 16 Eligibility for coverage varies by province,16,17 which has led to reimbursement gaps and inequities in patient access.18,19 For symptomatic SMA patients, therapy aims primarily to maintain function or slow the progression of disease, or occasionally to improve symptoms.
As described by the focus group’s SMA patient expert, without access to therapy, patients experience a decline in their physical abilities as the disease progresses. Along with the decline of gross motor skills, patients continue to experience a progressive loss of life skills such as dressing themselves, feeding themselves, and transferring. They lose the strength in their voice, making communication difficult. Patients without treatment lose their stamina which impacts their ability to maintain employment or attend school regularly. They experience an increase in hospitalizations and need for supportive equipment. Their lives continue to become more isolated due to health risks and complications. As untreated patients lose the ability to maintain physical function, they require alterations to their homes for accessibility which has a considerable financial impact. Patients slowly lose their independence as they lose function and, with that physical loss, and the need for mental health support increases—not only for the patient but for the patient’s extended family as well. As the patient experiences the loss of function, the greater the burden of care; increased financial, emotional, and psychological burden is placed on the family unit.
Innovative funding mechanisms for SMA treatments
Managed entry agreements (MEAs) are tools to help manage the uncertainties inherent in financing novel therapies with high evidence uncertainties. MEAs have been used in international jurisdictions to enable timely access to SMA therapies.8,20 There are currently 44 publicly available MEAs for the above-mentioned SMA treatments. 20 In some jurisdictions, access to SMA therapies has been enabled by other vehicles such as OBAs. A review of these agreements shows that, although complex to implement, they can play an important role in facilitating timely drug reimbursement and access. 8 Canada currently has fixed discounts or rebates and restricted coverage for all three SMA treatments 20 and, at the review of the latest publicly available information, has not leveraged innovative mechanisms or agreements to support drug access.4,7
Exploring the operationalization of an OBA in Canada through RWE generation by the CNDR
A notable rare disease category is neuromuscular disease, which includes SMA. The CNDR collects RWD for SMA and other neuromuscular diseases and may be a feasible option for generating RWE to support OBAs in Canada. This assessment presents the findings from a series of expert-led focus group discussions (FGDs) and stakeholder interviews to determine whether an OBA can be operationalized using RWE from the CNDR. The first objective of the assessment was to determine which SMA health outcomes in the CNDR are suitable for an OBA. The second objective was to evaluate the processes involved, specifically to determine how data tracking within the CNDR could be operationalized for an OBA, to identify gaps, and to explore corresponding solutions. Based on these discussions, the group proposed a future-state process for implementing OBAs using CNDR data.
Methods
Focus group discussions
This assessment used the qualitative research method and data collection technique called a FGD, which conforms to the Consolidated Criteria for Reporting Qualitative Studies (COREQ) statement
21
(Supplemental Material 1). A selected group of experts discussed the research topic in-depth, facilitated by the project’s lead author and investigator,
The above-noted research team, moderated by Arif Mitha, held 17 1-h virtual focus group meetings on Microsoft Teams between February and November 2024, with each meeting centered on a different discussion topic. Topics included the CNDR, SMA and its health outcomes, data collection processes, current available SMA therapies, international OBA, payer processes for current drugs, manufacturer patient support programs, process gaps to enable an OBA, solutions, and a future-state OBA process. These semi-structured discussion groups were designed to explore each topic until reaching saturation. The process to determine saturation was having the group review the summary of the discussion from the previous meeting. This was done iteratively until the review no longer provided new insights or outcomes, and it was deemed that saturation was reached. External sources used as input to the discussions included documents identified in literature reviews and interviews with subject matter experts. The meetings were recorded and the moderator collected field notes. An iterative process was used to summarize the data collected and develop findings. The group reviewed the results to ensure that they were sufficient and to achieve consensus. The results were then reviewed for validation and additional insights by 14 external key stakeholders including SMA-treating physicians, public payers (including provincial jurisdictions and a representative of the pan-Canadian Pharmaceutical Alliance (pCPA)), private insurers, pharmaceutical manufacturers, and health technology assessors. The feedback from the external stakeholders was then shared with the members of the focus group, who made adjustments to the results based on consensus.
Evaluation of SMA health outcomes for an OBA using data from the CNDR
In order to evaluate the suitability of SMA health outcomes for an OBA using data from the CNDR, the focus group experts first identified a list of eight health outcomes (Table 1). External inputs used in the discussion included clinical trials, HTA records, Health Canada product monographs, academic journals, and international MEA. Next, the suitability of each health outcome was determined based on three criteria: data readiness, data interpretation, and data timeframe. Data readiness was defined as completeness, accuracy, and accessibility for an OBA. Data interpretation was based on whether the outcome has a clearly defined interpretation such as a binary yes/no or can be scored on a simple scale. Data timeframe was defined as the time needed for the results of each health outcome to appear in the CNDR. The next step, defining the current state process, involved analyzing publicly available information for current renewal criteria and processes for SMA therapies in British Columbia, Saskatchewan and Ontario. All three provinces use their exceptional access programs (EAPs)—which evaluate patients on a case-by-case basis to provide access to therapies that are not listed for automatic coverage under public drug benefit plans 22 —to assess and grant access to SMA therapies. Private insurer prior authorization (PA) forms—which have the same function as the EAP process in public payer systems—were also reviewed. Finally, gaps in the proposed OBA process using the CNDR were identified and solutions to each gap were proposed.
SMA health outcome data readiness, interpretation, and timeframes in the Canadian Neuromuscular Disease Registry.

Note on interpreting this table: For all health outcomes listed, patient data are only available in the CNDR if the patient has registered in the CNDR. All data are collected from the patient chart.
CNDR, Canadian Neuromuscular Disease Registry; HTA, health technology assessment; HRQoL, health-related quality of life; OBA, outcome-based agreement; PROMs, patient reported outcome measures; PEDS QL, Pediatric Quality of Life Inventory; QALY, quality adjusted life year; SMA, spinal muscular atrophy; SMAFRS, Spinal Muscular Atrophy Functional Rating Scale; SMAIS, Spinal Muscular Atrophy Independence Scale.
Assessment findings
SMA health outcomes data within the CNDR
The focus group identified eight SMA health outcomes to evaluate for their suitability for an OBA (Table 1), based on an evaluation of data readiness, interpretation, and timeframe within the CNDR. For all eight health outcomes, the source of the data was the patient chart. As previously noted, data are manually extracted from the patient chart and entered into the CNDR.
The first health outcome evaluated was motor function scales, which were found to be suitable for an OBA. Age-appropriate motor function scales include the Hammersmith Infant Neurological Examination Section 2, 23 Hammersmith Functional Motor Scale—Expanded, 24 Revised Upper Limb Module, 25 Children’s Hospital of Philadelphia Adult Test of Neuromuscular Disorders, 26 and the 6-Minute Walk Test. 27 For data readiness, motor function scales are currently collected for patients when required as part of a payer’s EAP (for public payers) or PA (for private insurers) processes, at the frequency required by the payer. For other patients, motor function scales are conducted at the physician’s discretion, as is their timing. For data interpretation, motor function scales were the primary outcome in the clinical trials for all three drugs, and both public and private payers are currently using motor scales in their EAP and PA processes to decide if patients can continue on treatment at each renewal period (Figure 1). The focus group also agreed that for motor function scales to be suitable for an OBA, a baseline measurement would be required and a retry period would be required to accommodate patients for exceptions (e.g., patient has the flu, broke an ankle, etc.). Physicians who participated in the project described success on current SMA therapies as maintenance or even improvement of a patient’s disease state and specified that “meaningful change” in scale results would need to incorporate the age-related natural disease progression (decline) of SMA.

Current state of CNDR process flow.
Health outcomes 2–5 in Table 1—including overall survival (OS), permanent ventilation, use of a feeding tube, and duration of therapy—were found to be suitable for an OBA in combination with motor scales. All of these outcomes are captured in the CNDR, and data readiness was found to be suitable. OS is easily interpreted (binary yes/no) and is mainly relevant for SMA type 1 patients whose survival is limited to childhood; other SMA types have a longer life expectancy. Ventilation status and episodes are recorded in the patient chart, and payers are currently using this outcome as a criterion to start (“exclusion”) and stop (“discontinuation”) treatment (Table 2). A standard definition for permanent ventilation within the OBA (e.g., use of respiratory assistance for 16 h or more per day for more than 21 consecutive days) would be required. 28 The downside of using permanent ventilation as an outcome is that loss of respiratory function varies across the patient population. This heterogeneity compromises the suitability of this outcome, though it is appropriate in combination with other outcomes. Feeding tube status can be exclusive or supplemental to eating food. This information is collected as part of the CNDR data. Again, a standard definition for feeding tube within the OBA (e.g., absence of exclusive nasogastric or gastrostomy feeding) would be required. 29 Duration of therapy refers to whether a patient continues to refill their prescription and therefore remains on the therapy for the agreed-upon timeframe. Reasons for discontinuing treatment typically include progression or worsening of disease (e.g., permanent ventilation), adverse events, and patient desire to stop (often due to lack of improvement or poor tolerability). Duration of therapy is clear to interpret health outcome, as long as a standard definition for discontinuation is used (e.g., patient stops treatment for 3 months).
Health outcome 6 in Table 1—patient reported outcome measures (PROMs)—was found to be unsuitable for an OBA, but could be considered in the future. The CNDR captures PROMs through standardized questionnaires such as the Pediatric Quality of Life Inventory (PEDS QL), 30 Spinal Muscular Atrophy Functional Rating Scale (SMAFRS), 31 and Spinal Muscular Atrophy Independence Scale (SMAIS). 32 While these instruments are part of patient charts, they are not administered consistently to all patients, and there were no instances of PROMs being used as the primary outcome for healthcare decision-making. Additionally, payers noted in their feedback that current HTA recommendations do not include PROMs, so they would be difficult to accept. Of note, physicians described PROMs as more granular than motor function scales, with the ability to capture minute functions like brushing teeth and combing hair that reflect important improvements in quality of life (QOL). To assess patients, physicians reported using a combination of clinical outcomes, PROMs, and QOL criteria, including asking patients how they are doing. Physicians confirmed that PROMs are administered by trained physicians or occupational therapists, rather than captured by patient self-reporting.
Health outcomes 7 and 8 in Table 1—hospitalizations and scoliosis surgery—were found to be unsuitable for an OBA based on their lack of data readiness and interpretability within the CNDR. The number of hospitalizations was currently unsuitable as the data quality was not validated. Additionally, a baseline measurement would be required, and the reason for each hospitalization would also need to be analyzed in correlation to the effectiveness of a therapy. Scoliosis surgery was also deemed to be unsuitable for an OBA because, although this outcome is easy to collect as it is in the patient chart, it is not applicable to all SMA patients and the timing can vary based on alternative treatments used such as braces.
In terms of data timeframe: for health outcomes 1–6, the data could be made available for an OBA in the CNDR within 3 months of being populated in the patient chart. This was deemed suitable for an OBA by the focus group.
Current state processes for SMA drug access and renewal; gaps and solutions; proposed future-state OBA process using the CNDR
The focus group mapped out the current-state process for accessing the SMA drug therapies nusinersen and risdiplam (Table 2, Figure 1). The one-time gene therapy onasemnogene abeparvovec was intentionally excluded from this research, as the assessment focused solely on therapies that require ongoing administration and periodic reimbursement renewals. Both nusinersen and risdiplam are managed within public and private payer EAP and PA processes. Physicians complete the necessary forms with the required health outcome data to show the effectiveness of the treatment and submit it to the payers, most commonly annually, to obtain approval for the following reimbursement year. Physicians or their delegated research staff obtain patient consent to participate in the CNDR. CNDR staff manually extract data from registered patients’ clinic charts and enter them into the CNDR.
Three process gaps were identified in the current-state process and possible solutions to each gap were discussed. The first gap is that only approximately 30% of patients with SMA are currently registered in the CNDR, as only CNDR-registered patients can have their data transferred from the patient chart to the CNDR. An individual-level OBA would require all patients reimbursed by a specific payer be part of the CNDR. For a population-level OBA, the patient numbers for each payer would be small in Canada, given the >10 public payers and >200 private insurers in the country, 33 and would need to be further stratified by SMA type. A solution to this gap would be for payers to require patients who get reimbursed via an OBA to register in the CNDR to gain access to treatment. This is analogous to the current requirement for patients to provide their health outcomes data to enable continuation on therapy as part of the EAP and PA processes. The FGD found this solution plausible as patients are already agreeing to provide their data to public and private drug plans to qualify for initial coverage and for renewal. The second gap is the issue of privacy, specifically consent for data sharing. The current privacy consent agreed to by CNDR participants does not allow external sharing of individuals’ data. The CNDR is currently revising their consent statement to allow external data sharing and reconsenting existing patients. This new consent will have to be evaluated to determine if it is sufficient for OBA. If insufficient, additional OBA-specific consent would need to be created, and patients partaking in the OBA would have agree to it as part of the OBA eligibility requirements. The third gap is the data-reporting timeline. To generate a report, the CNDR currently requires a 3-month workback schedule to carry out data quality processes, including CNDR analysts running the report and then contacting clinic sites to correct any data quality issues. This would constrain the timeline to produce OBA reports, but may be sufficient.
Figure 2 presents a proposed future state for an OBA process using the CNDR, based on the current state and solutions to the three gaps identified. Other than mandated registration in the CNDR, the process would not require administration for physicians or patients above the current state. The frequency of testing for health outcomes would be every 12 months, which physicians deemed appropriate in interviews and literature. 15 To enable data sharing at an individual level, a new CNDR consent statement would have to be implemented and existing patients partaking in the OBA would need to be reconsented. As previously mentioned, the lead time for CNDR reports is 3 months. Payers would be able to validate the CNDR reports with their own data collected via the OBA form (currently the EAP and PA forms). Based on the SMA literature review, FGD, and feedback from stakeholder interviews, an appropriate length of the OBA term would be 3 years, with an optional three-year extension. Physicians reported that achieving meaningful results in adults would take a minimum of 3 years and up to 5 years. Public payers agreed that 3 years was reasonable but did not consider a 5-year timeframe appropriate, as being locked into contracts for that length of time would prevent patients from using any other therapies that emerged during the period of the agreement and would be detrimental to payers’ budgeting practices. Private payers echoed this feedback and expressed a preference for shorter OBA contracts as patients could potentially switch insurance plans before the end of the assessment period.
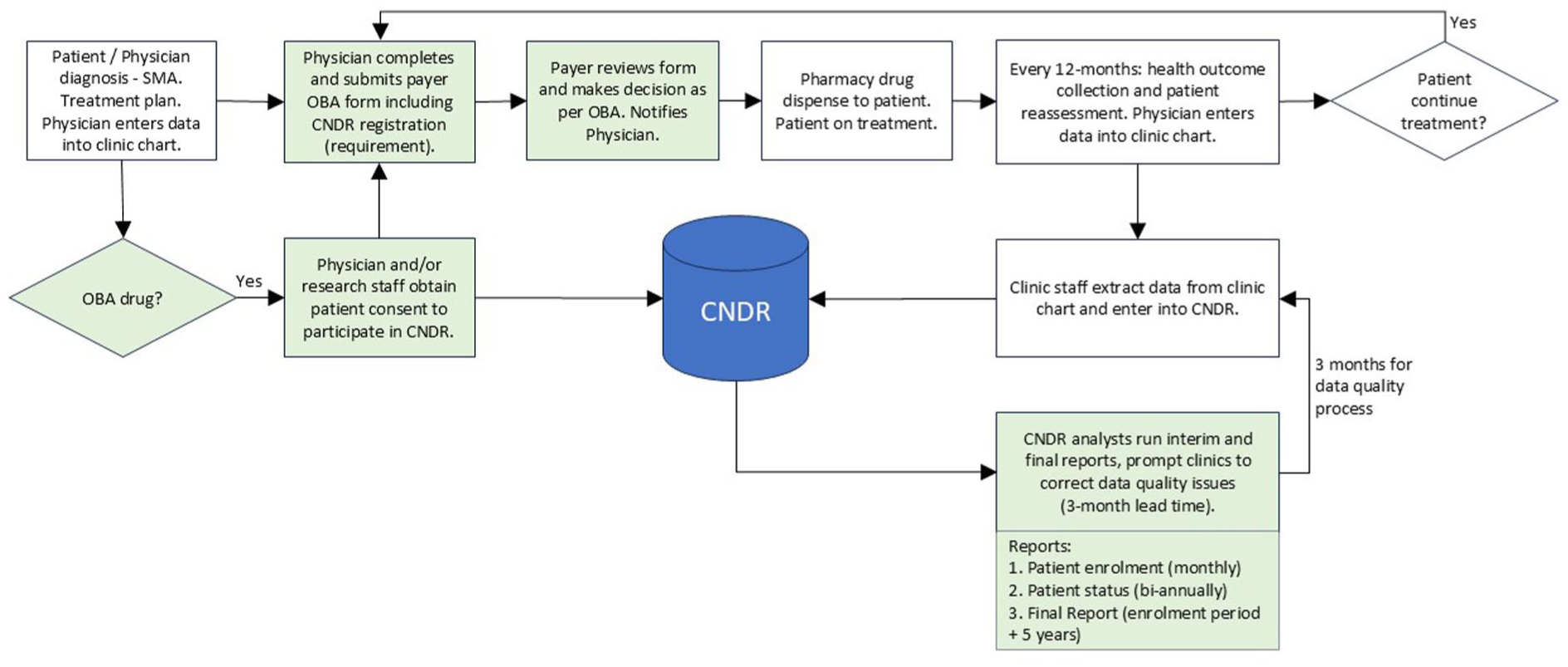
Future state of CNDR process flow.
Discussion
Although this assessment was conducted in SMA, it is important to note that this assessment approach and its findings are applicable beyond SMA—to other CNDR-tracked conditions such as ALS and DMD, and to other patient registries.
A key finding of this assessment is that both public and private payers are already collecting health outcomes data as part of their EAP and PA processes for SMA drugs, and that they encompass several key features required for the implementation of an OBA, including: (1) a process control for patient eligibility to take part in the OBA—physicians and patients provide payers eligibility criteria, such as gene tests confirming disease, baseline health outcomes, age, etc.; (2) a data validation method—payers can validate the CNDR’s data with their own data collected through the EAP and PA processes; and (3) a method to ensure that health outcomes are being collected and reported at the desired frequency—which is often not the case in the real world. In the case of SMA, motor scales are used as per the payers’ EAP/PA frequency requirements. This finding is applicable to all therapies for rare diseases for payers to leverage their EAP/PA processes to align with an OBA: payers are already creating the eligibility and renewal criteria, doing the data collection work, and making funding decisions, and physicians and patients are already providing the data. A limitation noted in the research acknowledged by both public and private payers is that currently data collected through EAP/PA processes would have to be analyzed manually as they are not stored electronically.
Public payers noted that they would only use health outcomes to support listing decisions recommended in HTAs provided by the CDA-AMC. This feedback highlights the opportunity for the CDA-AMC to recommend when an OBA could be appropriate and help public payers understand the health outcomes needed to support such agreements. In addition to the outcomes reviewed in this research, private payers were also interested in health outcomes such as short- and long- term disability; however, these were not in scope.
This assessment brought an important question to the forefront, namely whether it should be mandatory to report newborn screening data for SMA into the CNDR to improve incidence and prevalence information. Further research would be required to evaluate if this is feasible.
This research led to additional learnings regarding the infrastructure required to support patients on SMA therapies and to successfully support an OBA, which can also be applied to other diseases. When a drug first launches, this infrastructure may not be in place and can take years to establish. For example, when the first SMA therapy was approved by Health Canada in 2017, missing infrastructure elements included treatment guidelines for adult SMA patients, the ability to collect PROMs, an advanced patient registry that captures clinical data, resources for physicians to conduct motor function scale assessment, and newborn screening, among others. These elements took time and resources to establish and are now in place.
A strength of this assessment was the inclusion of a diverse set of experts who could speak to each aspect of the research.
Limitations
Limitations of this assessment include the fact that the CNDR data were not accessed as part of the assessment and thus were not validated for quality due to confidentiality concerns. A follow-up to this assessment should include a feasibility study and empirical validation of the CNDR data quality for an OBA. Notably, the study should validate that the data readiness, interpretation, and timeframe are sufficient for current patients enrolled in the CNDR who are receiving therapy via a payer EAP or PA process that requires providing health outcomes such as motor scales. Additional limitations include a limited number of participants in the FGDs, and limited stakeholder interviews.
Conclusion
In summary, this expert-led assessment demonstrates the potential feasibility of operationalizing OBA using CNDR-derived RWD, provided that the identified gaps are addressed and an empirical validation of CNDR data quality for an OBA is conducted. These findings aim to deepen Canadian stakeholders’ understanding of how patient registries can generate RWE to support OBA and other innovative funding mechanisms, ultimately improving timely and equitable access to rare disease therapies. Further opportunities for discussion and engagement between payers, data holders, and manufacturers are needed to establish how to implement such data strategies for OBA at a pan-Canadian level.
Supplemental Material
sj-pdf-1-trd-10.1177_26330040261433035 – Supplemental material for Could an outcome-based agreement be operationalized using real-world data from the Canadian Neuromuscular Disease Registry? Perspectives from an expert-led assessment in spinal muscular atrophy
Supplemental material, sj-pdf-1-trd-10.1177_26330040261433035 for Could an outcome-based agreement be operationalized using real-world data from the Canadian Neuromuscular Disease Registry? Perspectives from an expert-led assessment in spinal muscular atrophy by Arif Mitha, Victoria Hodgkinson, Susi Vander Wyk, Chris Cameron and Allison Wills in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors would like to acknowledge the SMA patient community, the CNDR investigator network, and the stakeholders who participated in interviews with the focus group for this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

