Abstract
Background:
Approximately 25% of cancer patients are diagnosed with rare cancers and face unique challenges. Decentralized patient-partnered research efforts, like Count Me In provide an avenue for patients to participate in research that overcomes key barriers to address disparities in rare cancer research to accelerate discovery.
Objectives:
Projects in metastatic breast cancer (The Metastatic Breast Cancer Project; MBCproject) and angiosarcoma (The Angiosarcoma Project; ASCproject) highlight disparities that exist for all cancer patients and underscore those that are compounded for rare cancer patients.
Design:
Through Count Me In’s research platform, patients visit a website to enroll in the study and complete surveys, which allows us to access their medical records and biospecimens. Clinically annotated sequencing data are de-identified and released on research platforms.
Methods:
MBCproject and ASCproject data were analyzed to identify differences between patients with a more common and rare cancer, respectively. The analysis included outreach strategies, patient-reported themes, and distance traveled for care.
Results:
As of September 28, 2023, 3742 patients have enrolled in MBCproject and 491 patients have enrolled in the ASCproject from across the United States and Canada. Outreach strategies were tailored to resource availability. Using survey information, it was observed that patients with a rare cancer (angiosarcoma) traveled longer distances to receive care than those with a more common cancer (metastatic breast cancer) for three major cancer centers. Patients with rare and common cancers highlighted different themes when asked about their disease experience. Themes like misdiagnosis and discontent with resource availability came up more often for rare cancer patients. Data sharing and collaboration in angiosarcoma research enabled rapid discoveries with clinical impact.
Conclusion:
Count Me In’s platform has led to unprecedented data generation and findings in rare cancer through partnering with patients. Directly engaging with patients to generate and share data while emphasizing collaboration sets the foundation for a more equitable future.
Plain language summary
About 25% of all patients diagnosed with cancer are diagnosed with a cancer that’s considered rare. These patients face many challenges, since less is known by researchers and physicians about how to treat cancers that aren’t as common. There is also limited information about their disease available to these patients and their families. One way to help better understand rare cancers is to make it easier for patients to participate in research so they can share information that can help researchers study and better treat these diseases.
An initiative called Count Me In has developed a way for patients from anywhere in the United States or Canada to participate in research by registering online, sharing their experience through surveys, and allowing researchers to access their medical history and samples. Studies in angiosarcoma (The Angiosarcoma Project), a very rare cancer, and metastatic breast cancer (The Metastatic Breast Cancer Project), a more common cancer, have highlighted the challenges that cancer patients face, particularly those faced by rare cancer patients.
Keywords
Introduction
Twenty-five percent of cancers are considered rare, and patients within these populations face unique challenges, including lack of standard of care for treatment, lack of clinical expertise, and lack of clinical trial availability. 1 Furthermore, lack of funding to support translational research across the spectrum of rare cancers coupled with insufficient numbers of patients in any given subtype creates additional barriers to participate in research that might impact the future of people with their disease. Count Me In (CMI), an initiative aimed at democratizing the ability to participate in cancer research, enables cancer patients from across the United States and Canada to share their clinical information, tumor samples, and biological samples with researchers and has enabled rare cancer patients to participate in research alongside people with equally as devastating, but more common cancers.
CMI launched in 2015, with The Metastatic Breast Cancer Project (MBCproject), a cancer that leads to over 43,000 deaths in the United States per year. 2 The goal was to engage directly with cancer patients through remote participation to generate clinically annotated genomics data sets and then share the data widely to accelerate discoveries in cancer research. By leveraging social media and patient networks online, CMI was built to overcome geographic dispersion and enrolled over 6500 patients with MBC from across the United States and Canada, and now represents one of the most comprehensive studies to date in this disease. 3
The MBCproject was developed alongside patients, patient advocates, and caregivers, and provided insight into best practices for patient engagement specific to the metastatic breast cancer patient community. All aspects of the online study were built with iterative feedback from metastatic breast cancer patients and advocates to ensure resonance within the MBC community. Patients in the United States or Canada could join via a web portal, MBCproject.org, to register and sign an electronic consent form, fill out surveys, and provide information to facilitate sample and medical record collection. Online enrollment eliminated geographic barriers to research participation. This flagship program provided insights and infrastructure to launch an additional 10 patient-centered research projects across the cancer compendium, three of which focused on rare cancers, including angiosarcoma, the initiative’s first endeavor to study an exceedingly rare cancer.
Angiosarcoma is an extremely rare endothelial sarcoma that occurs in one in 1 million people. 4 It is a highly aggressive malignant tumor with approximately 350 people diagnosed every year in the United States. The median survival is 16 months, with a 5-year survival of approximately 30%–40%. There has been little advancement in treatment options and currently, if diagnosed, therapies are not rooted in precision medicine and often include surgery, radiation therapy, and chemotherapy. With its rarity and poor prognosis, the development of large-scale research initiatives has been non-existent. CMI, however, was able to leverage the infrastructure built with the metastatic breast cancer community to rapidly generate data at scale with this exceedingly rare patient population, leading to discoveries that supported changes in clinical practice. In addition, these efforts generated insights regarding unique challenges faced by patients with rare cancers. 4
CMI launched the ASCproject in March 2017, and within the first day, over 60 patients living with this disease joined the project. By 2019, over 250 patients had consented and data from 36 patients (48 samples) was in the public domain. Sarcoma clinicians leveraged the data to support the development of clinical trials as well as add additional cohorts to existing trials. 5 CMI published original findings from these data within a year of the first clinical trial publishing clinical outcomes that leveraged the same data, showcasing the ability to rapidly impact clinical care through patient-partnered research.4,5
The ability to conduct decentralized research with patient communities creates a powerful path forward toward discoveries for both common and rare cancers. Having conducted studies in both, CMI is uniquely poised to describe disparities that exist throughout the cancer landscape, but which are exacerbated within rare cancer communities. Resource disparities mean that the outreach landscape for common and rare cancers is different and must be tailored as such. Patients from both rare and common cancer communities are willing to generously donate their data and share information, but the themes that they want to inform researchers of appear to be different. For example, when comparing patients with angiosarcoma to patients with metastatic breast cancer, angiosarcoma patients face an increased travel burden for their care. Understanding and addressing the unique challenges faced by rare cancer patients is essential for progress.
Rare cancers
According to the National Cancer Institute, a rare cancer is defined as a cancer that occurs in fewer than 15 people out of 100,000 every year. 6 Due to the small number of cases of these cancers, research funding lags far behind that for common cancers. In 2020, the NCI funded 6.4 billion U.S. dollars for cancer research, but only a small portion was allocated for rare cancers. 7 The number of research dollars allocated to different types of cancer ultimately impacts the number of available treatments for these cancers. As such, as listed by the National Cancer Institute, there are 84 FDA-approved treatments for breast cancer, and only 17 FDA-approved treatments for soft-tissue sarcomas, none of which are specific to angiosarcoma, highlighting the unmet need and the unique challenge faced for patients diagnosed with angiosarcoma (see Figure 1). 8

FDA-approved drugs for common and rare cancers. According to the National Cancer Institute’s list of approved drugs, as of 2023, there are 84 FDA-approved therapeutics available to treat breast cancer. By contrast, there are 17 FDA-approved therapeutics available to treat all known soft tissue sarcomas, none of which are specific to angiosarcoma (National Cancer Institute). 8
There are systemic reasons for this difference. Translational research consists of several steps and often begins with sample collection to further understand the pathophysiology and genetics of the underlying disease. The timeline for new drugs to enter the public market specifically focused on rare cancer solutions can take well beyond 20 years, which could only ensue after research produced discoveries that could be translated into the clinic. 9 The CMI model was developed to overcome these systemic challenges that hinder progress in translational research for patients with devastating cancer diagnoses.
Methods
Data from both the MBCproject and the ASCproject (N = 3742 patients for the MBCproject (from October 2015 to September 2023) N = 491 patients for the ASCproject (from January 2017 to September 28, 2023)) were analyzed to identify key differences between participating patients in a more common cancer population and a rare cancer, respectively. The study design, collection of patient information and clinical samples, and the distance traveled for care analysis are described herein. The RECORD guidelines were consulted when revising the manuscript. 10
Patient engagement
Outreach and engagement strategies were built in alignment with each cancer community to facilitate scalable outreach to patients regardless of where they lived or were treated. Partnerships were explored and established with disease-specific advocacy organizations prior to the study launch. These partnerships helped to establish trust in the community and ensured the study team was aware of existing efforts in the space. Partnerships were bespoke to each organization, with an overarching goal to spread awareness of CMI through newsletters, social media, webinars, and conferences. Partnerships were approached with the aim of respectful and sustainable amplification of messaging to spread awareness.
Study design
This research is an observational study designed to collect data from cancer patients at any point in their cancer. Study workflows were developed alongside patients and advocates prior to launching the MBCproject and have been modified over time in response to patient and community feedback to improve operations, outreach, and engagement. To facilitate sample and medical record collection by the study team, participants provide information about the institutions where they received care for their cancer. Surveys collect information about cancer experience and demographics, including the timeline for diagnosis, subtype, treatment, and race/ethnicity. Patients receive sample kit(s) in the mail, which enables them to provide a saliva sample and/or blood sample, at no cost to them. In addition to sending out and receiving sample kits, the team requests the patient’s medical records based on the information provided on the patient’s medical release forms. Key pieces of clinical information are abstracted, including detailed procedure histories, so that tumor samples can be collected for patients who opt-in to share their archived specimens. Tumor, saliva, and blood samples are sequenced (via whole exome sequencing and/or transcriptome capture); medical record and survey data are aggregated and de-identified; and all data are deposited to publicly accessible databases such as cBioPortal11,12 and the Genomic Data Commons (Genomic Data Commons Data Portal) that are available to researchers around the world to increase the potential for discovery.
The workflow for both patients and the study staff and points of attrition for the ASCproject are outlined in Figure 2(a) and (b), respectively. Processes inherent to the study workflow prohibit the ability to collect all data and material(s) for every patient that provides consent. For example, medical record departments may require a pen-to-paper signature for record collection, and patients are not always able to provide this. Some pathology departments dispose of archived tissue samples after 10 years, so the acquisition of tumor samples for patients who are long-term survivors may not be possible. Before data are released, sequencing quality is assessed via computational pipelines, and some samples may drop out of the dataset due to low sample purity.

CMI study workflow. (a) CMI study process. Steps outlined in orange are patient steps and steps outlined in green are study team steps. Patients visit study websites, fill out basic information, and have the option to complete an electronic consent form to share survey information, copies of medical records, and a saliva sample, and can opt-in to share portions of archived tumor tissue. Patients complete a medical release form to indicate where they have received treatment. Pts fill out survey(s) about their cancer experience and/or demographic information. Pts receive a saliva and/or blood kit in the mail to return via a self-addressed envelope. The study team sends kits to consented participants and requests copies of medical records and archived tissue samples. All data from medical records are clinically abstracted, patient-reported data are cleaned and standardized, and samples are sequenced. All de-identified linked data are shared on publicly accessible platforms. (b) Key points in workflow and points of attrition for the ASCproject. A total of 738 pts registered for the study. Of those pts, 491 completed the consent form. All consented pts were sent a saliva kit. A total of 317 pts returned their saliva sample kit. Medical records for all pts who returned saliva kit were requested. Medical records were successfully obtained for 275 pts. A total of 179 pts had tissue that was able to be requested. A total of 226 tissue samples were received for 151 pts. A total of 136 samples were successfully processed and sequenced.
Patient-reported themes analysis
Patients were asked the following question on the intake survey for each project: “Tell us anything else you would like us to know about yourself and your cancer.” A total of 100 respondents from each project were selected from each dataset and were analyzed to determine a list of themes. A set of responses to this question was cleaned to remove potentially identifiable data and included in Figure 3.
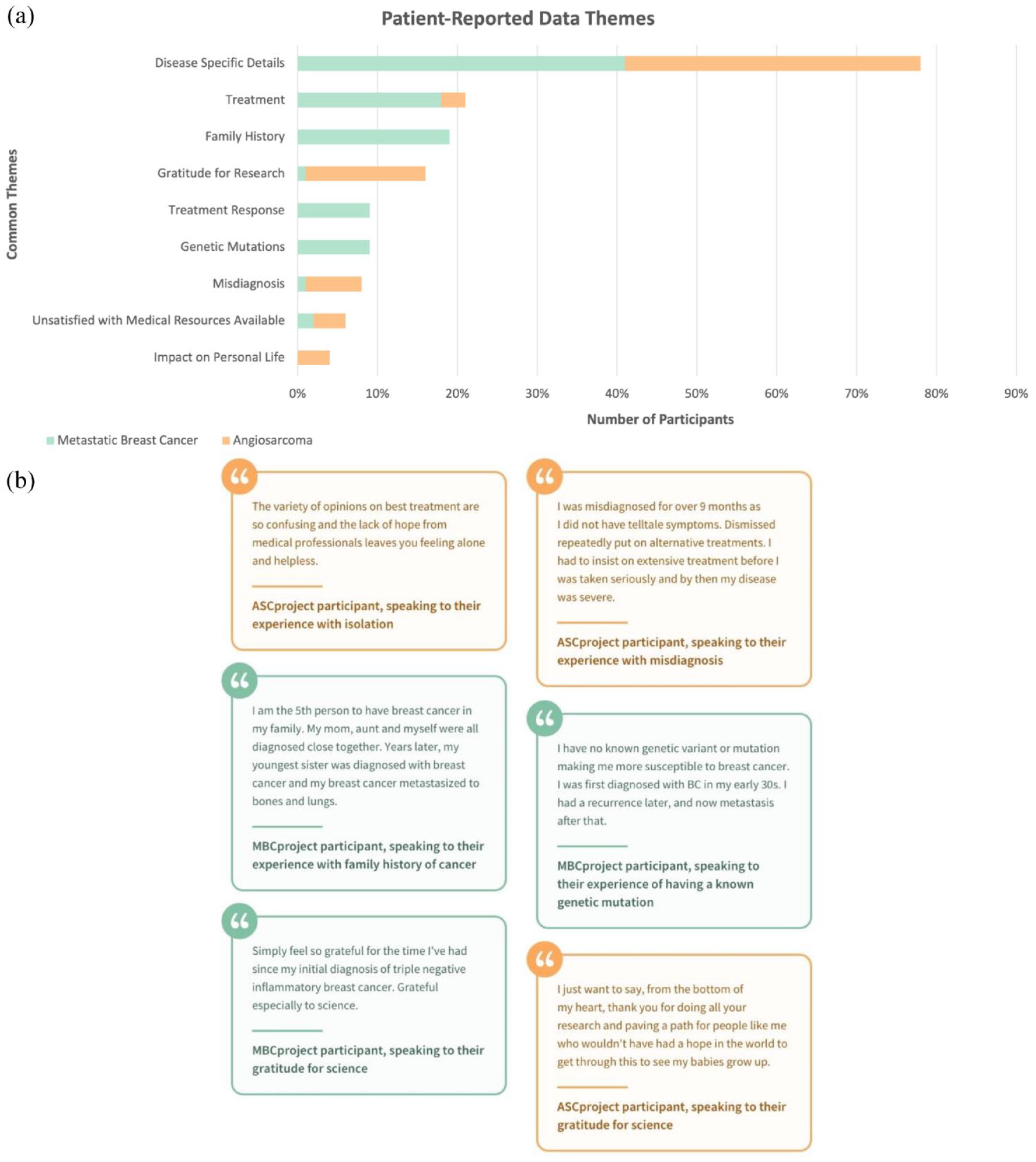
Patient-reported themes from the MBCproject and ASCproject. (a) Patient-reported data collected from intake surveys for the MBCproject and ASCproject. A total of 100 responses to the question “Tell us anything else you would like us to know about yourself and your cancer” from each project were categorized into themes. (b) Select responses to the survey question “Tell us anything else you would like us to know about yourself and your cancer.” All potentially identifying information was removed from responses.
Distance traveled for care analysis
Patient-reported data from surveys completed by MBCproject and ASCproject participants were used to identify those who received medical care at the selected medical institutions by using keywords that identify these institutions, followed by manual curation. The selected medical institutions were MD Anderson Cancer Center (MD Anderson), Memorial Sloan Kettering Cancer Center (MSKCC), and Dana-Farber Cancer Institute (DFCI).
The distance between the home and medical institution for participants was calculated using the home zip code from the survey and the institution zip code. The geographical distance between zip codes was calculated using the latitude and longitude of the zip codes using the distm function from the geosphere R package. The latitude and longitude of zip codes were obtained from the zipcodeR R package (USA, using the five-digit zip code) and the Service Objects database (Canada).
A total of N = 463 ASCproject and N = 3307 MBCproject participants completed these surveys and provided home/institution information. A subset of participants had valid home zip codes and reported receiving medical at MD Anderson (ASCproject, N = 71 patients; MBCproject, N = 105 patients), MSKCC (ASCproject, N = 30 patients; MBCproject, N = 93patients), and DFCI (ASCproject, N = 33patients; MBCproject, N = 88 patients), and was included in Figure 4.

Enrolled participants across MBCprojects and ASCprojects. Geographic distribution of consented participants in the ASCproject (491 pts; orange dots) and MBCproject (3742 pts; green dots).
Statistical analysis
Data from MBCproject and ASCproject were used retrospectively for all analyses and sample sizes were not pre-planned. For the analysis of patient-reported themes, the sample size of 100 was determined based on the feasibility of the manual process involved in determining common themes. For the analysis of distance traveled, sample sizes were determined by the number of patients at the data cutoff with data available for each project. A two-sided Wilcoxon rank sum test was used to compare the distributions of distance traveled. A p value of <0.05 was considered to be statistically significant. All statistical analysis was performed using R (version 4.3.2).
Results
Since the launch of both studies, patients from across the United States and Canada have consented to join the study (3742 for the MBCproject and 491 for the ASCproject as of September 28, 2023). The distribution of participants follows a similar pattern to the population density (Figure 4).
Outreach
The MBCproject had 36 breast cancer organizations as advocacy partners, and the ASCproject had 6 sarcoma organizations as partners, only one of which is specific to angiosarcoma (see Figure 5). This is not an exhaustive list of all organizations that exist in the space and instead represents the organizations that CMI was connected to through the studies. The difference in the number of advocacy partner organizations for each project highlights two key aspects that speak to the differences in rare versus more common cancers. One area highlighted is the clear disparity in resource availability. A single partner organization is focused solely on angiosarcoma. Although metastatic breast cancer patients face challenges in the availability of resources and organizations specific to metastatic breast cancer, several partners focus on raising awareness, education, or research funding for metastatic breast cancer.
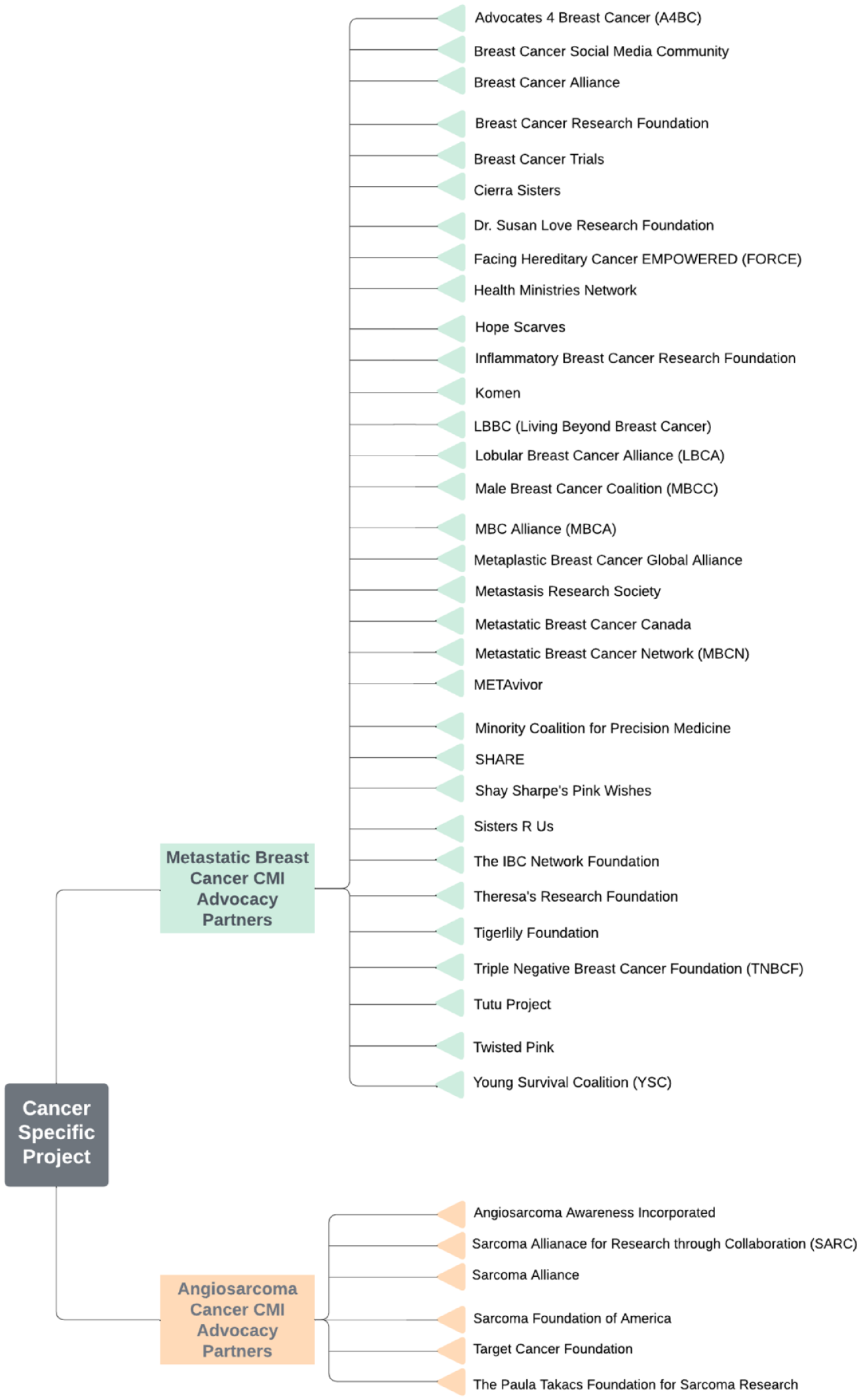
Count Me advocacy partner organizations: the metastatic breast cancer and angiosarcoma projects. The MBCproject has 38 advocacy partner organizations, while the ASCproject has 6 advocacy partner organizations, one of which is specific to angiosarcoma. All organizations help to inform project outreach strategies.
The difference in resource availability also impacts outreach. For the MBCproject, the CMI study staff connected with many organizations for awareness. The number of organizations and initiatives meant that numerous connections were forged to ensure messaging about the project would reach patients. For the ASCproject, the partnership was limited to the only known online support group specifically focused on angiosarcoma. In both studies, there was a tremendous initial response and sustained engagement, despite differences in accessibility and resources. Once trust was established, patients and participants in both projects were willing to post about the project in their closed Facebook group regularly. Because there was only one group representing the angiosarcoma community, messaging about the project was more bespoke and tailored. Posts were informational and explained the project in a personal context, helping potential participants see themselves in the message, which aided in their consideration for joining the project themselves.
Working with advocacy partner organizations led to valuable lessons about the importance of tailoring outreach approaches based on cancer type, which includes considerations about how messages are amplified and the landscape of resource availability. Regardless of cancer type, the key lesson is to enter new spaces to listen, learn, and adjust approaches to resonate with patients.
Travel burden—Distance traveled for care
Participants in both the MBC and ASCprojects provided information about where they live and where they receive care for their cancer. For patients in each project who received care at three major NCI comprehensive cancer centers (MD Anderson Cancer Center, Memorial Sloan Kettering Cancer Center, and Dana-Farber Cancer Institute), it was observed that ASCproject patients faced a greater travel burden, traveling a longer home-to-institution distance in each of these three institutions based on the quartiles of the distance distribution (Figure 6). For example, the median distances in ASCproject versus MBCproject were 1314 versus 555 km for MD Anderson Cancer Center, 134 versus 48 km for Memorial Sloan Kettering Cancer Center, and 89 versus 63 km for Dana-Farber Cancer Institute. The difference in travel distance was statistically significant for MD Anderson Cancer Center
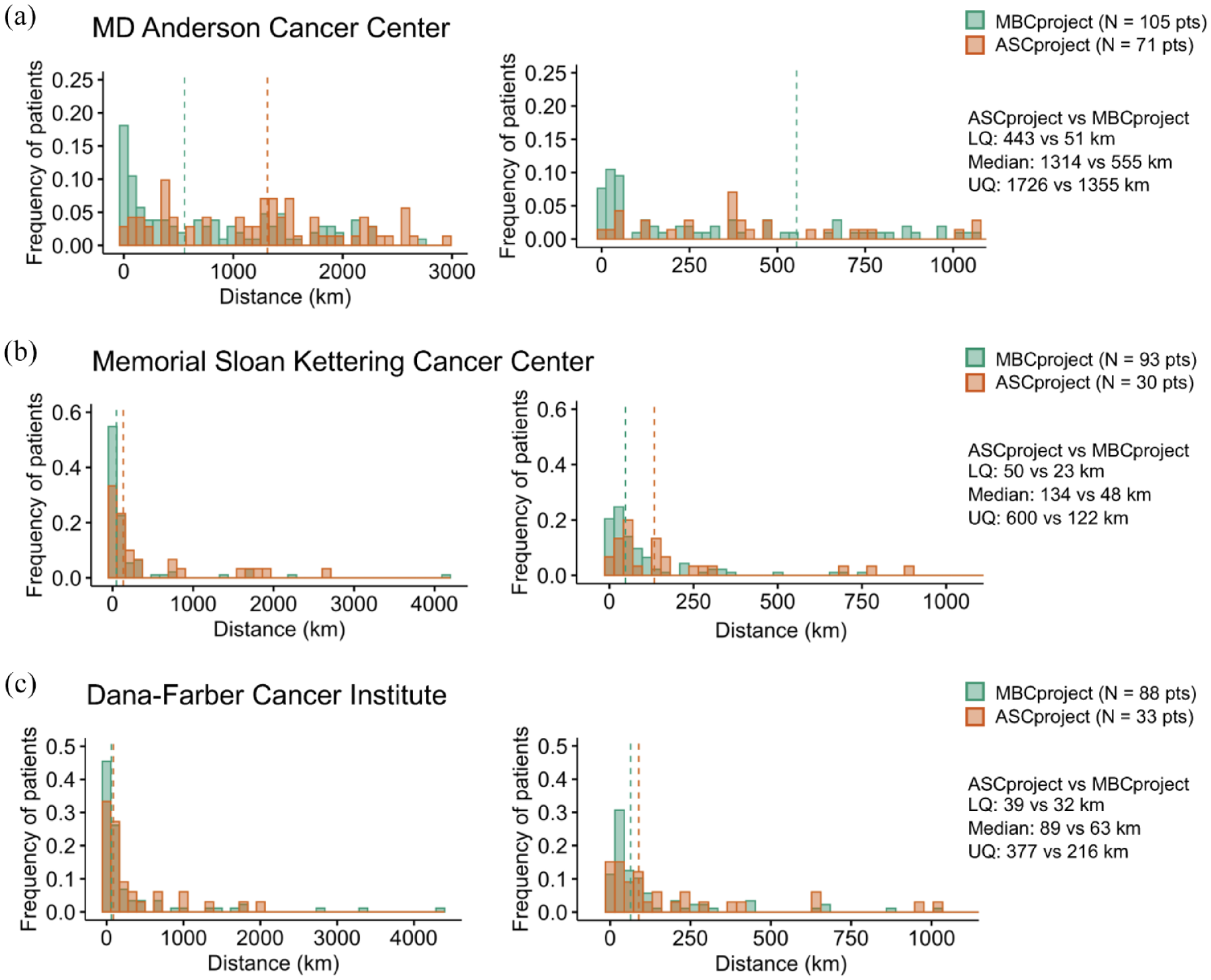
Distribution of distance between home and treatment institution in the ASCproject and MBCproject for three major NCI comprehensive cancer centers. The treatment institutions were MD Anderson Cancer Center (a) (ASCproject, N = 71 pts; MBCproject, N = 105 pts), Memorial Sloan Kettering Cancer Center (b) (ASCproject, N = 30 pts; MBCproject, N = 93 pts), and Dana-Farber Cancer Institute (c) (ASCproject, N = 33 pts; MBCproject, N = 88 pts). Patients included reported receiving care at said medical institutions. Distances were calculated from the home and medical institution zip codes. Quantiles for the distance distribution of each project are shown on the right side of each panel. Based on the quantiles of the distributions, the home-to-institution distance for ASCproject patients was longer than that for MBCproject patients for each of the treatment institutions.
Another observation was that a larger fraction of patients was treated at these three major cancer centers in ASCproject compared to MBCproject (71/463 = 15.3% vs 105/3307 = 3.2% for MD Anderson Cancer Center; 30/463 = 6.5% vs 93/3307 = 2.8% for Memorial Sloan Kettering Cancer Center, and 33/463 = 7.1% vs 88/3307 = 2.7% for Dana-Farber Cancer Institute). The larger fraction of patients in ASCproject is consistent with the observed greater travel burden: patients with a rare cancer like angiosarcoma are more likely to have to travel far from their home and local medical institutions to receive care, and thus, are more likely to receive care at a major cancer center.
Overall, using survey data from CMI studies, we found that patients with rare cancer (angiosarcoma) travel larger distances to receive care than those with a more common cancer (metastatic breast cancer) for three major cancer centers, which highlights the insights on patient’s medical care and experiences that can be gained from patient-reported data, as some of our previous work has also shown (see Figure 6).3,13
Patient-reported themes
One of the many benefits of partnering directly with patients and collecting their information is the ability to develop and deploy surveys to ask participants to provide their direct input. In addition to structured survey questions about cancer diagnosis, treatment, and demographics, space is provided in each survey for patients to tell us more. When prompted with the question, “Tell us anything else you would like us to know about yourself and your cancer,” participants provided a variety of responses that covered an array of themes. By examining responses from metastatic breast cancer patients and comparing them to those from angiosarcoma patients, unique differences between the populations of patients were highlighted while other themes were common regardless of cancer type.
As seen in Figure 3(a), the most prevalent theme to emerge across both cohorts was a focus on describing what was categorized as “disease-specific details.” Responses in this group included information about sites of disease (current or past), subtype or histological details, and information about symptoms at disease onset. Many patients used the question as a space to expand upon the structured questions that were asked earlier on in the survey, and this was true for patients with both types of cancer.
The next theme that emerged was information about treatment, which came up more often for metastatic breast cancer patients (approximately 20% of all patients) and included information about specific drugs and/or radiation treatment. A theme that came up only for metastatic breast cancer patients is family history. Since there are known hereditary components to breast cancer, the existence (or lack thereof) of a familial component was mentioned by MBC patients when asked an open-ended question about the patient’s experiences that they thought the study staff should know. Both “treatment response” and “genetic mutations” were themes that came up for many metastatic breast cancer patients. Responses categorized as “treatment response” were those that spoke to the effectiveness of particular therapies, while those categorized as “genetic mutations” were those that mentioned specific tumor mutations that were present. In summary, many of the ways that MBCproject participants chose to answer this question were related to their clinical experience with the disease.
Many angiosarcoma patients focused on “gratitude for research.” Participants spoke of the lack of available resources and treatments for angiosarcoma, and how they were grateful for the ability to contribute to research that could change the future for angiosarcoma patients. This may speak to the scarcity of research projects available to this community of patients.
Misdiagnosis was another commonly discussed theme within the responses from angiosarcoma patients. Responses in this category included patients who described they were told that they had something other than angiosarcoma or metastatic breast cancer prior to receiving their diagnosis. Many patients who mentioned misdiagnosis expressed concern as to whether their prognosis was poorer given the delay in definitive diagnosis. Relatedly 7% of patients expressed dissatisfaction with the lack of medical resources available. Lastly, “impact on personal life” came up for angiosarcoma patients as well. Participants spoke about the impact that their diagnosis had on their ability to be present for family, friends, or career.
The data provide insight into the themes that patients want researchers to be aware of and shows potential differences in how rare cancer patients choose to highlight their experience versus patients with MBC. Obtaining this information is only possible by asking patients directly, which is a benefit of the CMI model. To build upon these observations, the next step is to examine responses from all patients consented to the studies and determine whether response themes may be correlated to other factors, like geography, socioeconomic status, or time from initial diagnosis to study registration.
In addition to broad themes, the quotes from patients in Figure 3(b) underscore the importance of the patient’s voice. All responses shown in the figure have been scrubbed of any potentially identifying information but have otherwise been kept in the form that they were input to provide insight into areas that are important for patients to highlight to the research community. Survey questions like those deployed through CMI underscore the ability to collect nuanced data directly from patients and could provide foundational knowledge for additional efforts to increase what is attainable through efforts that engage patients directly in research. In the case of angiosarcoma patients enrolled in this study, themes of isolation, lack of resources, and misdiagnosis are prevalent and distinct relative to MBC.
The power of data sharing
In the context of rare cancers, a significant impediment to scientific advancement stems from the scarcity of accessible data conducive to discovery. Data from ASCproject were released several years in advance of the project’s first publication (see Figure 7). 14 The dataset included data on the first 227 consented patients, and whole exome sequencing (WES) analysis on 47 samples from 36 patients. The pre-published data were leveraged by several sarcoma groups, including Wagner et al, 5 who published findings from a multicenter phase II trial study of ipilimumab and nivolumab, which showed an objective response rate of 25% in angiosarcoma patients who presented a high tumor mutational burden.5,15 The manuscripts have been cited over 200 times combined in over 100 journals, underscoring the impact that collaboration, data availability, and data sharing can have in the bid to change the future for rare cancers.

Rapid data sharing in ASCproject. Timeline of data generation, release, and discovery through collaboration and rapid data sharing in CMI’s ASCproject.
Discussion
The ability to conduct decentralized research enables patients to participate regardless of where they live. Eliminating this barrier democratizes access to research for patients with both common and rare cancers. In the case of rare cancers, a decentralized approach may provide the only opportunity for patients to participate in a study focused on their subtype of cancer. Leveraging a patient-partnered approach enables studies to be built bespoke to each community, which, in turn, facilitates enrollment and dissemination of findings directly to the contributing patients.
CMI releases data uncoupled to publication, which spurred a rapid translation of discoveries from ASCproject. Because this cancer is exceedingly rare, and there was a dearth of data previously available to the sarcoma community of clinicians, utilization of the clinically annotated data was swift and impactful, leading to changes in clinical practice for this population of patients.
As a result of the discoveries made and the impact of studying angiosarcoma, the initiative has been able to expand into additional rare cancers, osteosarcoma (OS), and leiomyosarcoma (LMS). OS is a sarcoma that occurs in the bone and affects mostly younger adults and teenagers. The 5-year relative survival rate for OS patients with localized disease is approximately 76%. That number decreases for patients whose disease spreads to regional lymph nodes (64%) or distant sites (24%). 14 LMS is a cancer that grows in the smooth muscles, with a 5-year survival rate of 35%. 16 These patient communities are enthusiastic about the opportunity for research participation, as patients diagnosed with these rare cancers would benefit greatly if targeted treatment strategies were discovered. There are limitations to this type of research study. Power analysis for sample size calculation was not done as the goal was to enroll as many patients as possible with this rare cancer. Over the next several years, it is essential to consider how to continue to address the disparities that exist for rare cancer patients. Continued and increased focus in this area, both at the organizational and systemic levels, is what will drive progress toward a more equitable future. CMI has made some noteworthy contributions in the rare cancer space. By involving participants from different backgrounds, the research process increases accuracy and efficiency in producing widely applicable therapies that can address underfunded cancers in underserved spaces. The role of the patient in this process is indispensable.
As CMI continues to evolve, an area of opportunity is to develop additional surveys to better understand the insights and experiences of rare cancer patients to provide researchers with more comprehensive knowledge on the impact of their medical conditions as well as possible interventions. Patients can directly provide robust data to researchers, so additional, tailored questions should be developed and deployed to better understand the rare cancer patient experience. How long did it take the patient to receive a definitive diagnosis? How many times were patients misdiagnosed? Did the patient and their family have to relocate to better facilitate expert care? How far do patients travel regularly for treatment? A better understanding of the rare cancer experience can enable the research community to better address disparities for these patients.
Furthermore, increasing equity in the healthcare system by focusing efforts on involving underrepresented populations that are historically marginalized and underserved, including racial and ethnic minorities is critical to providing equitable opportunity and generating representative data. To date, enrollment in CMI has been low, with approximately 12% of participants enrolled in CMI projects identifying as being part of racial and/or ethnic minorities. However, through community engagement initiatives, CMI is committed to building inroads into communities that experience disparities. An initial initiative was to make the CMI projects available in Spanish, with feedback from the community, to increase accessibility to the Spanish-speaking Hispanic and Latino/Latina/Latinx/Latine people with cancer (e.g., www.MBCprojectenEspanol.org). A recent initiative is the “Amplifying Black Voices” campaign (www.BlackCancerVoices.org), which highlights the experiences of Black cancer patients, caregivers, and loved ones affected by cancer through written stories and videos. Ongoing efforts are underway with key advocacy groups and pioneers within the Black and African American cancer community to develop culturally relevant materials for participation into CMI research studies. As CMI continues to gain momentum, consistent involvement of marginalized populations in the research process with transparency and compassion will be imperative.
Conclusion
Patients diagnosed with rare cancers face significant disparities in their experience with cancer, ranging from lack of clinical trials to greater difficulty finding clinical expertise for their treatment. In addition to these disparities, rare cancer patients have limited options for research participation that could be leveraged to address such disparities. CMI’s ability to reach patients across the United States and Canada has led to unprecedented data generation by partnering with patients. CMI’s research projects in metastatic breast cancer and angiosarcoma have enabled the ability to observe challenges that exist for all cancer patients, but that are particularly compounded for individuals with a rare cancer diagnosis. Outreach to rare cancer patients must consider the lack of resources and organizations available to this community. Patients have important themes that they want to share with researchers about their cancer experience, with rare cancer patients highlighting challenges with their experience like misdiagnosis or dissatisfaction with medical resources. In comparing the two communities, rare cancer patients also appear to face a unique travel burden for their cancer care. Working directly with patients to release and generate data and prioritizing collaboration in the field lays the groundwork for a more equitable future.
