Abstract
Despite the unmet needs of patients living with cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD) and the challenges facing a rare population with small patient numbers, now is a time of unprecedented opportunities to turn scientific breakthroughs into safe and effective treatments for families of CDD patients. New data collected for over a decade and an evolution in genetics technologies have resulted in transformational new treatments currently in development for CDD. This progress is in great part due to the patient advocacy efforts early on to drive development of stakeholder research tools necessary to de-risk industry entry into the CDD space, family participation in longitudinal natural history studies, and a robust caregiver-reported database. Cumulatively, these efforts offered new insights into CDD, specifically patterns in disease progression, helped identify the most burdensome symptoms to patients and caregivers, improved clinical trial design, and reduced financial barriers for therapeutic development for potential industry partners. This paper documents the growth of a small patient community through relationship building and collaboration. The International Foundation for CDKL5 Research is mindful of ongoing challenges namely the long research timelines, high development and production costs, and inequitable access to approved therapies. Therefore, sustaining strong early resources while recognizing opportunities that engagement, advocacy, and funding can accelerate progress remains at the heart of the agile foundation strategy.
Plain language summary
Effectively operating a rare disease patient advocacy foundation presents obstacles that are difficult to anticipate, yet there is silver lining in learning from opportunities lost to regroup and refocus on our mission. The IFCR initially formed to drive research forward and support families while fostering new scientists to study the disorder. This remains the core objective and the organization has successfully contributed to the development of a robust CDD community and important research assets to facilitated research progress. On the heels of the first approved treatment for CDD, we find ourselves contemplating our future in a much different light, asking propelling questions and making tough decisions. Given the constraints that most rare disease communities face, how can we best use limited resources? How can we partner with others to realize timely progress? What gaps exist that require CDD family thought leadership and engagement along the continuum of drug development? Reflecting on past years, it is remarkable how fast science is moving. Genetic therapies are under early development for CDKL5 Deficiency Disorder. We must prepare for any future we are afforded to trial disease-modifying treatments. Multistakeholder engagement is required pre-clinically, during clinical trials and post approval.
Keywords
Introduction
International Foundation for CDKL5 Research
The International Foundation for CDKL5 Research (IFCR) is the largest global patient advocacy organization serving cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD). The organization began when a group of parents received this diagnosis for their children and found nowhere to turn for answers or support. They soon recognized that building a global patient community and a knowledge base to ignite therapeutic development was essential. They formalized these efforts in 2009 and incorporated the IFCR as a nonprofit organization.
The IFCR’s effort to fund the creation of needed biological models and other groundbreaking research and establish CDKL5 Centers of Excellence (COEs) has laid the foundation for a thriving research, clinician, and patient network. Collaboration has strengthened the organization’s ability to accelerate its mission. By leveraging partnerships where there were shared interests, the organization has been able to both maintain research momentum and provide critical professional and family education. A focus on key partnerships continues in recent work targeting gaps in clinical trial readiness for CDD. At the same time, family support and education have been a priority of the organization, ensuring that those diagnosed can live well with CDD.
What is CDD?
Pathological mutations in CDKL5 result in CDD. Separate case studies linked various mutations in the gene to a neurodevelopmental disorder around 2004 similar to a phenotype that overlapped with Rett Syndrome (RTT).1,2 CDKL5 was previously known as serine/threonine protein kinase 9 (STK9). 3 While previously considered the “Hanefeld Rett variant,” this is a unique disorder, but with overlapping features of many disorders.
Through efforts previously funded by the IFCR, we know that CDD is associated with severe, medically refractory epilepsy, motor, cognitive, visual, and autonomic disturbances. The majority of CDD cases are identified through diagnostic investigations of early-onset seizures. Children are now being diagnosed when younger because of greater use of genetic testing, but parent journeys are variable because this is a rare disorder. Epilepsy typically begins during the first few weeks or months of life; few children can walk or speak words, and gastrointestinal and sleep dysfunction is frequent. 4 CDD is considered a Developmental and Epileptic Encephalopathy (DEE) and has significant impacts on both the child and their family. Recent studies suggest CDD has an incidence of around 1 in 40,000–60,000 and is likely one of the three most common genetic causes of DEE. 5
The early years—de-risking
The IFCR’s initial focus was supporting (and learning from) families while raising funds to establish a basic understanding of the gene function and a clinical research tool kit to foster drug development. The organization understood the need for a global CDKL5 patient advocacy organization to advance clinical research and care in such a rare disease.
Early work with an Australia-based team built the foundation for a robust patient-reported natural history study known as the International CDKL5 Disorder Database (ICDD) (Figure 1). Seed funding a post-doc to develop early questionnaires and the initial governance framework led to what is now a comprehensive database contributing to further understanding of the heterogeneity of symptoms within and across patients. This ongoing work gave evidence for CDD to diagnostically stand alone as a disorder independent of RTT. In January 2020, CDD was assigned an ICD-10 code. 5
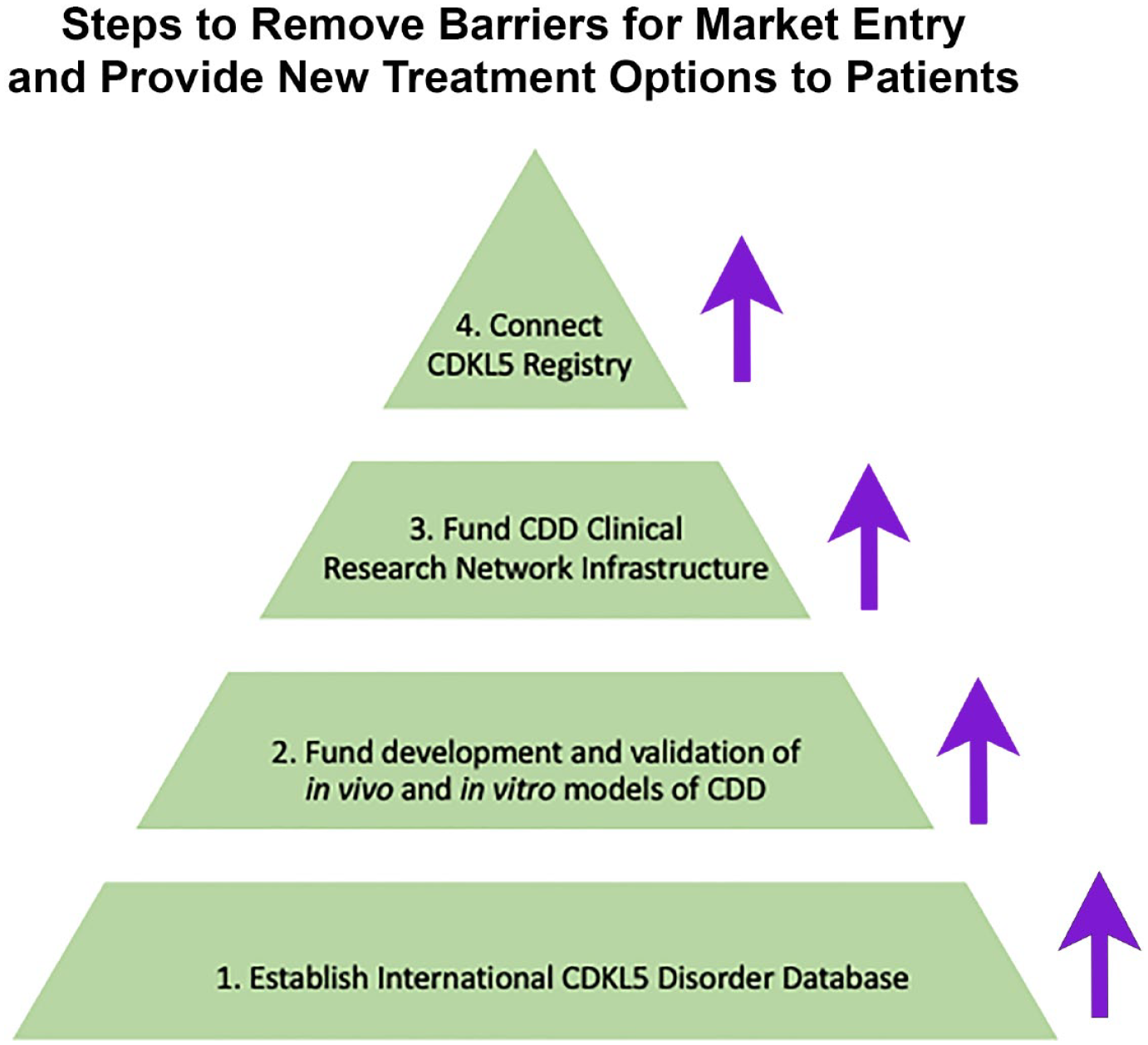
De-risking development of new therapeutics via an organized community with research-ready data and tools. Foundational assets developed by the CDD community include:(1) the ICDD which provides a deeper understanding of clinical complexities and burden of CDD including genotype-phenotype interactions and clinical management; (2) human cell models, organoids, and animal models of CDD that are required for pre-clinical therapy development and further studies aimed at understanding CDKL5 function; (3) shared infrastructure for the ICCRN which allows for sustained natural history collection and jumpstarts development of clinically meaningful CDD-specific Clinical Outcome Assessments (COAs); and (4) the Connect CDKL5 Registry which facilitates patient identification, is a channel for clinical trial recruitment, and a method for providing caregiver education and family support.
Simultaneously, work was underway to develop key partnerships to align ICDD with clinician-collected data to work toward discovering and validating hallmark symptoms. Establishing CDKL5 COEs was the next logical step. As a young foundation, the IFCR leveraged commonalities with RTT, and many of the multidisciplinary consult care centers overlap with RTT clinics. The first CDKL5 COE at Colorado Children’s Hospital is co-funded with a local foundation, Rocky Mountain Rett Association. Likewise, the IFCR’s inaugural Family and Science Symposium in 2012 was co-sponsored and organized with the International Rett Syndrome Foundation (IRSF). This partnership with IRSF continued for the subsequent two conferences. Since 2017, the science symposium has shifted to annual hosting by the Loulou Foundation. These collaborative efforts helped open the door to participation in the Rett and Rett-like Disorders U54 NIH Natural History Study (Table 1).
Pre-clinical and clinical research tools available to the research stakeholder community.

CDLK5, cyclin-dependent kinase-like 5.
The early years—research roadmap benchmarks
Models
The CDD research community has generated mouse and zebrafish lines and made them publicly available for further disease function and drug development research. In addition, a repository of iPSC cell lines from male and female CDD patients is housed with appropriate isogenic control lines at Coriell Institute to support the discovery of potential disease-modifying therapeutics. Work in CDD animal models has demonstrated the ability for symptom reversal; worldwide efforts are already underway to develop therapeutic strategies (including gene therapy) to treat and potentially cure CDD. With the potential of disease-modifying therapies, there is a critical need to develop clinical trial readiness for CDD. 6 Lastly, we collaborate with the Harvard Brain Tissue Resource Center to allow postmortem samples to be stored safely for future investigation (Table 1 and Figure 1).
International CDKL5 disorder database
Much of our knowledge of CDD comes from the unique ICDD, a rich natural history database collecting parent-reported and genetic data since 2012. The largest collection of cases worldwide (330 patients across 40 countries), the database continues to inform research on genotype-phenotype, quality of life, incidence, and prevalence. A catalog of CDKL5 sequence variations, including pathogenic mutations, nonpathogenic polymorphisms, and sequence variations of uncertain significance, can be found at the RettBASE website (Table 1 and Figure 1).
Centers of excellence
In 2013, the first CDKL5 COEs serving the complex needs of patients with CDD and their families were established by IFCR at Children’s Hospital Colorado, Boston Children’s Hospital, and Cleveland Clinic. In this consult care model, COEs provide multidisciplinary care including neurology, development, genetics, gastrointestinal (GI), rehabilitation, pulmonary, and therapy (physical, occupational, and speech) specialties. This care model has served as the template for future COEs in our network (currently 10 sites across the United States).
RTT and Rett-related disorders natural history study
Beginning in 2015, 67 CDD patients aged infant through 30 participated in longitudinal data collection of core clinical features—an NIH U54 grant funded this research, including RTT, methyl CpG binding protein 2 (MECP2) duplication, CDKL5, and Forkhead-box G1 (FOXG1) disorders. The clinical research team worked closely with patient advocacy representatives from each disorder to ensure study success. This work provided foundational data to guide outcome measure development in the respective disorders (Table 1). 7
In recent years—current momentum and challenges
The CDD research roadmap at large and ongoing international de-risking efforts for future therapeutic development have benefited from funding from additional country-based CDKL5 patient advocacy groups and was accelerated by the founding of the Loulou Foundation in 2015. Because of its full spectrum of symptoms, CDD is the ideal DEE for clinical tool development; thus, our efforts are translatable to other similar disorders. The IFCR continues to focus on building key partnerships and has since targeted gaps in clinical trial readiness in the CDD community, and the effort is ongoing to date.
The NIH-funded U54 natural history study clarified the need for a CDD-focused continuation of CDD-specific longitudinal data collection. It also laid the groundwork for the International CDKL5 Clinical Research Network (ICCRN). The IFCR invested substantially in a central data collection repository and Streamlined, Multisite, Accelerated Resources for Trials Institutional Review Board (SMART IRB) with data-sharing agreements to continue data collection and foster further NIH funding of this effort. It was a fruitful investment as a multi-year clinical trial readiness U01 grant was awarded to the ICCRN in 2020 entitled “Multi-Site Validation of Biomarkers and Core Clinical Outcome Measures for Clinical Trials Readiness in CDKL5 Deficiency Disorder (PI: Timothy Benke, U01-NS114312-01A1).”
Over 10 years of collected natural history has shown that, with age, the clinical manifestation of CDD changes and varies by patient, thus complicating treatment options. Caregivers and families of diagnosed patients need evidence-based resources to make informed decisions regarding age-dependent disease progression and first-line treatments for all comorbidities of CDD. As an organization, one of our many priorities is developing clinical standards of care that authentically incorporate patient needs. Doing this will entail convening topic-based working groups with our multidisciplinary clinical experts to address the best treatment approaches, early intervention strategies, and specialty care required while also considering the social determinants of health for those without access to COEs. Once established, these guidelines, co-produced with caregivers, will be published, and disseminated to the broader community to empower families and providers with a roadmap for effective clinical care options.
In recent years—research roadmap benchmarks
International CDKL5 Clinical Research Network
Combining patient and clinician-collected data efforts, the ICCRN was born. This effort merged the ICDD patient-reported dataset with the U.S. COE infrastructure. The IFCR-funded COEs (1) deliver multidisciplinary care to CDD patients, (2) participate in uniform, well-governed data collection via the ICCRN, and (3) openly share with other data repositories and stakeholders. The ICCRN is conducting clinical trial readiness studies to refine and validate a suite of clinical outcome measures necessary to assess disease modification in CDD. The ICCRN presents an excellent capacity to apply and scale new diagnostic testing or clinical outcomes approaches. Additionally, the sites serve as a clinical trial network for industry partners. However, diagnosing patients in rural and underserved areas is vital to accelerate caregiver-driven research.
Connect CDKL5
Connect CDKL5 is a global contact registry in its most basic form. It is a tool that collects basic patient information, demographics, and details about receiving the diagnosis. It is the only database that allows patients to provide these details without being asked to participate in further data collection, which is often overwhelming and burdensome to caregivers early in the diagnostic journey. Our Connect CDKL5 platform allows individuals living with CDD to learn about and connect with researchers in ways that inform the global community. It also alerts caregivers to opportunities to participate in focus groups, attend events, and advocate for their loved ones living with CDD. We currently offer data collection in Spanish and rely on browser translations for all other languages. We have around 500 participants from nearly 45 countries.
CDD clinical trials
The CDD community is fortunate to have both observational and interventional clinical trial experience. IFCR has gained much experience maintaining effective industry partnerships to serve the global community as disease-modifying treatments are approaching the pipeline.
An investigator-initiated pilot study of Ataluren started in 2016. It was ineffective in reducing seizure frequency or improving cognitive, motor, or behavioral function or quality of life in subjects with CDD with nonsense variants. 8
Ovid Therapeutics led a phase II open-label ARCADE study of OV935/TAK-935 (soticlestat) in pediatric patients with CDD or Duplication 15q (Dup15q) syndrome in 2018. In 2020, all patients who had completed the phase II ARCADE study to date opted to enroll in the ENDYMION open-label extension study, which is ongoing. 9
UCB’s GEMZ Study (NCT05064878) is an ongoing phase III, randomized, double-blind, placebo-controlled, fixed-dose, multicenter study to examine the efficacy and safety of ZX008 (fenfluramine hydrochloride) and seizure reduction. This study follows an investigator-initiated open-label study in six patients completed in 2022. 10
Marinus Pharmaceuticals conducted a double-blind phase of a randomized, placebo-controlled, phase III trial to study the safety and efficacy of ganaxolone. 11 In 2022, these results led to the first FDA-approved treatment for seizures in CDD, Ztalmy (ganaxolone). This milestone widened our advocacy lens and the need to expand our priorities to address unmet community needs for access to treatments today and the novel therapies the future promises.
Discussion
Looking toward the future
This paper is a testament to a small patient advocacy organization’s integral role in driving community growth through relationship building and collaboration as a trusted partner to all stakeholders. The success of drug development, particularly gene and cell therapies as alternative or adjunct therapies, will depend on appropriate and early engagement with the CDD community as a whole. To date, the IFCR has continued to meet the moment despite current capacity challenges. Forward momentum requires an expansion of capacity, both organizationally and in regard to regulators’, clinical researchers’, patients’, and caregivers’ capacity to participate (Figure 2).

The integral role of the patient community is to drive therapeutic development, impact policy, advise regulatory decisions, and improve care. IFCR intentionally built a diverse stakeholder and caregiver community to impact and support all aspects of life with CDD from diagnosis management and living with the disease to informing clinical trial design. IFCR serves a critical role in education on CDD for healthcare providers, researchers, and most importantly, caregivers. As the largest global patient advocacy organization, IFCR holds a central role in progressing research and in getting new products approved through regulatory agencies. In their role as partners with trialists, they have a positive impact on patient recruitment, patient retention, and developing patient-centric protocols with meaningful endpoints, which can potentially lead to more effective therapeutics. As advocates, IFCR understands their work does not end with approval of a therapeutic and works tirelessly at the state and federal level to ensure all families have access to new therapies.
As new patients continue to receive complex diagnoses like CDD and are desperate for a cure, providing education on the complexities of drug development is paramount. IFCR’s roadmap over the years has been mindful of the necessary data and biological models, regulatory requirements, and substantial financial investment needed to bring therapies to market. We know that success is not achieved alone but through a cohesive rare disease community rallying around timely treatments for all patients.
While this review specifically focuses on the important contributions of IFCR through the lens of the history of the organization, it is limited in its scope and a light should be placed on the resources developed by the country-based patient advocacy groups. Some of the pre-clinical resources mentioned in Table 1 were co-funded with other groups, and it should not be assumed that assets discussed here were independently funded by IFCR. The list may be incomplete in some cases if there are animal models or cell lines independently funded by individual patient advocacy groups dedicated to CDD research that are not yet publicly available. Each CDD patient advocacy is contributing to research and patient support to the extent they are able. Researchers and families are encouraged to reach out to local patient advocacy groups to learn about their funded research and programs.
Conclusion
A challenge for most patient advocacy groups is the need to support families, fund research, and develop future therapies with limited capacity. The CDD community that grew out of IFCR’s early efforts has no walls and branches to international foundations that are able to focus on the unmet needs of families under their umbrella, developing relationships with local clinicians, and the unique healthcare systems that exist within each of those countries. Likewise, the thriving Loulou Foundation fuels research and development toward new therapies. IFCR will continue its commitment to clinical research and care while supporting families. A unique aspect of the CDD community shines through as researchers develop their niche expertise in the CDD landscape, and in the same vein, the individual CDD foundations have developed missions that contribute to the community accelerating research and supporting families collectively with shared goals and objectives strengthening the community effort together.
Footnotes
Appendix
Acknowledgements
The authors would like to acknowledge the families that participate in the substantial natural history study, observational studies, and interventional clinical trials. Without their contributions, none of the progress discussed in this paper would be possible.
