Abstract
Background:
Tuberous sclerosis complex (TSC) is an autosomal dominant neurocutaneous disorder of non-malignant tumor growths throughout major organ systems and neurological, neuropsychiatric, renal, and pulmonary co-morbidities. Skin manifestations are readily visible, often develop early in life, and are major features that contribute to TSC diagnosis. Medical photographs of such manifestations are commonly shown as examples from White individuals creating a potential barrier to accurately identifying these features in darker skinned individuals.
Objectives:
The aim of this report is to raise awareness of dermatological manifestations associated with TSC, compare their appearance by race, and consider how recognition of these features could impact diagnosis and treatment of TSC.
Design and Methods:
We conducted a retrospective chart review at the TSC Center of Excellence (TSCOE) at the Kennedy Krieger Institute, which included all patients in the center from 2009 (inception) through the end of the calendar year 2015 and analyzed data from the TSC Alliance Natural History Database (NHD).
Results:
Among TSCOE patients, 50% of Black patients were diagnosed before the age of 1 year, compared with 70% of White patients. NHD data corroborated this trend showing a significant difference with only 38% of Blacks as compared with 50% of Whites were diagnosed at age ⩽1 year. A significant difference was observed where White participants had higher odds of having received genetic testing in both data sets. While no differences in the total number of TSC features was observed in either data set, shagreen patches and cephalic fibrous plaques were more frequently recorded in the NHD for Black individuals.
Conclusion:
We highlight a disparity in the representation of Black participants within the NHD, TSCOE, and TSC trials, in addition to differences in utilization of molecular testing and topical mechanistic target of rapamycin (mTOR) inhibitor therapy between Black and White individuals. We show a trend toward later diagnosis age in Black individuals. These differences between races warrant further study across additional clinical sites and other minority groups.
Plain Language Summary
Keywords
Introduction
Tuberous sclerosis complex (TSC) is a neurocutaneous, genetic condition diagnosed in approximately 1 in 6000 live births that results from an autosomal dominant or spontaneous mutation in either the TSC1 or TSC2 genes.1,2 Clinical diagnosis of TSC is based on the presence of a specific combination of major and minor features, of which four major and one minor are dermatological manifestations (Table 1). 3 Skin manifestations are readily visible and often develop early in life. 4 Hypomelanotic macules, also called ash-leaf spots, are the most common dermatologic feature of TSC in patients under the age of 1 year (Figure 1). 5 A complete skin examination utilizing a wood’s lamp is recommended in those with lighter skin complexions to visualize these findings. They are often present on the limbs, trunk, and buttocks, but can be identified throughout the body. Later in childhood, facial angiofibromas, also referred as adenoma sebaceum, are a hallmark feature of TSC (Figure 2). 6 Other common skin lesions in TSC include ungual fibromas, shagreen patches, and cephalic fibrous plaques. 4 Examples of hypomelanotic macules, facial angiofibromas, cephalic fibrous plaques, and shagreen patches in both Black and White individuals are shown in Figures 1–4, respectively. Dermatological features of TSC are typically non-life threatening and are less severe than other TSC features; however, early recognition in combination with other features can help expedite a clinical diagnosis to promote early intervention strategies that are shown to improve cognitive outcomes.3,7 In some cases, the dermatological examination alone is sufficient to establish a definite diagnosis of TSC. 8 Furthermore, cutaneous manifestations are often the only visual manifestation of TSC for individuals with mild to moderate phenotypes of TSC. These features can negatively impact one’s body image, self-acceptance, and quality of life. 9
Clinical diagnostic criteria for tuberous sclerosis complex [reprinted under the terms of the Creative Commons Attribution-NonCommercial-No Derivatives License (CC BY NC ND) from the study by Northrup et al.]. 3
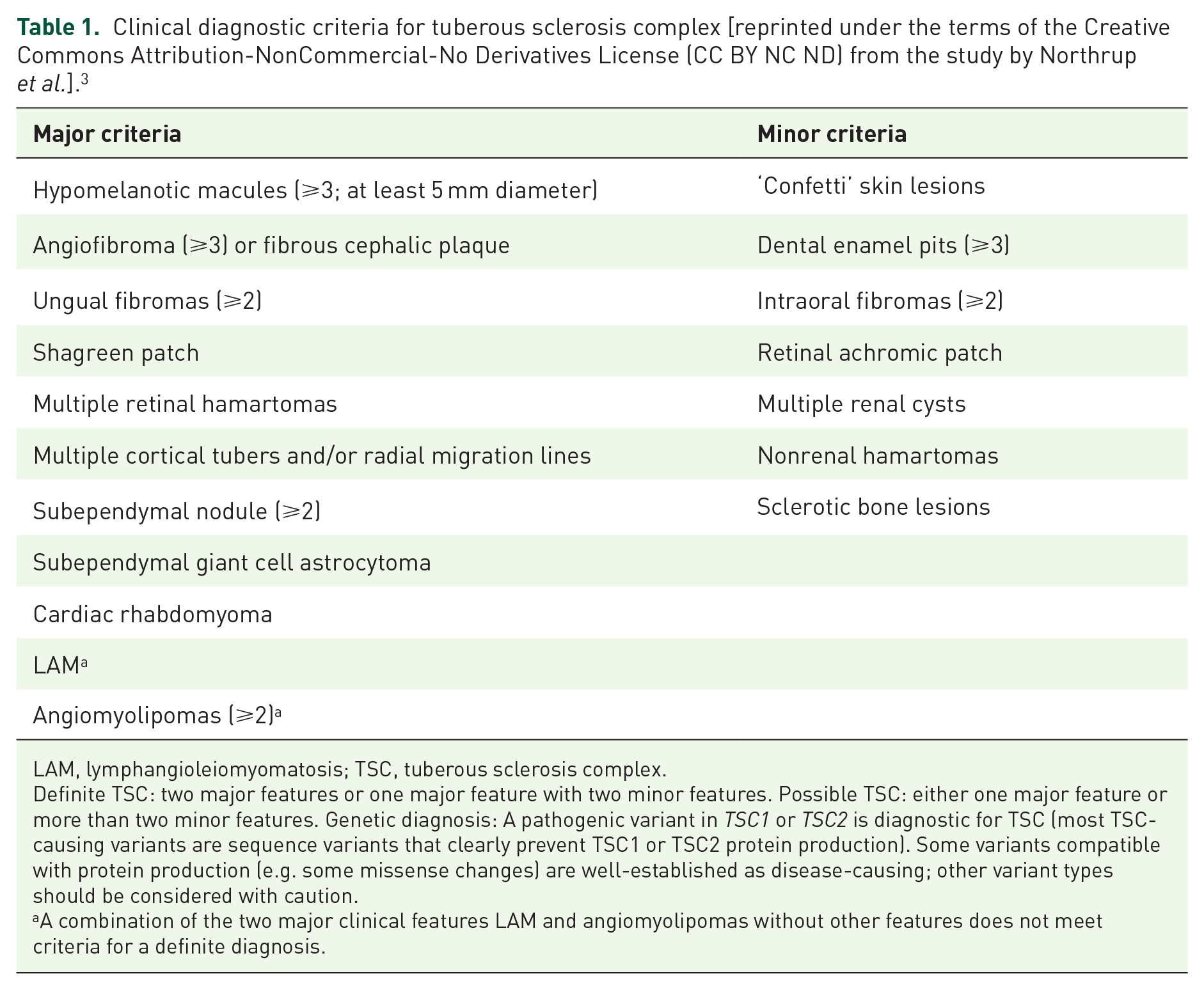
LAM, lymphangioleiomyomatosis; TSC, tuberous sclerosis complex.
Definite TSC: two major features or one major feature with two minor features. Possible TSC: either one major feature or more than two minor features. Genetic diagnosis: A pathogenic variant in TSC1 or TSC2 is diagnostic for TSC (most TSC-causing variants are sequence variants that clearly prevent TSC1 or TSC2 protein production). Some variants compatible with protein production (e.g. some missense changes) are well-established as disease-causing; other variant types should be considered with caution.
A combination of the two major clinical features LAM and angiomyolipomas without other features does not meet criteria for a definite diagnosis.

Hypomelanotic macules in Black and White individuals with TSC. (a–d) Black individuals with hypomelanotic macules. (a) Age 11 male, on forehead. (b) Age 11 male, on back. (c) Age 14 male, on cheek. (d) Age 14 male, on leg. (e–h) White individuals with hypomelanotic macules. (e) Age 8 male, on neck. (f) Age 7 female, on arm. (g) Age 4 female, on arm. (h) Age 15 male, on leg.
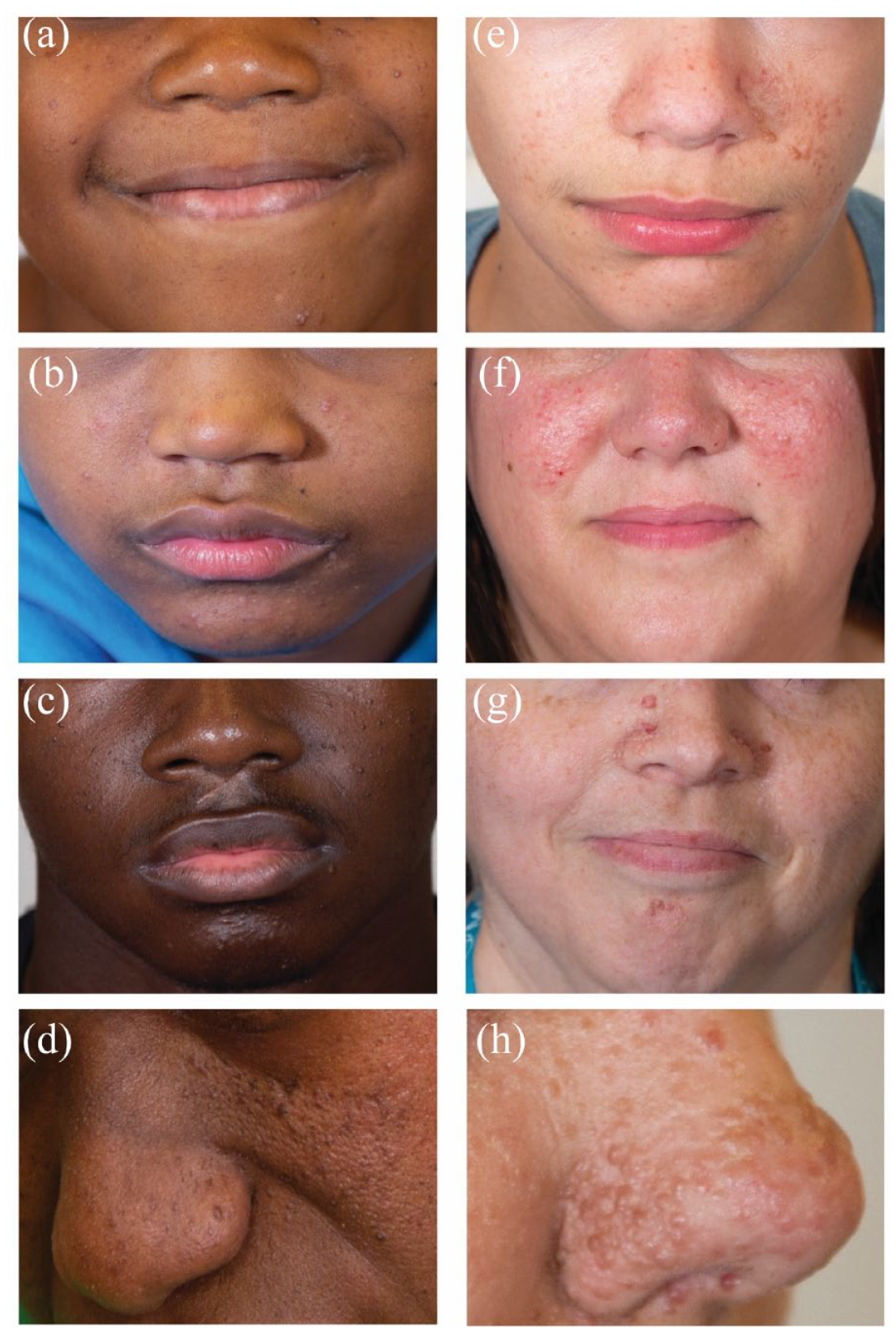
Facial angiofibromas in Black and White individuals with TSC. (a–d) Black individuals with facial angiofibromas, ages 11, 14, 16, and 28, respectively. (e–h) White individuals with facial angiofibromas, ages 13, 37, 39, and 28, respectively.
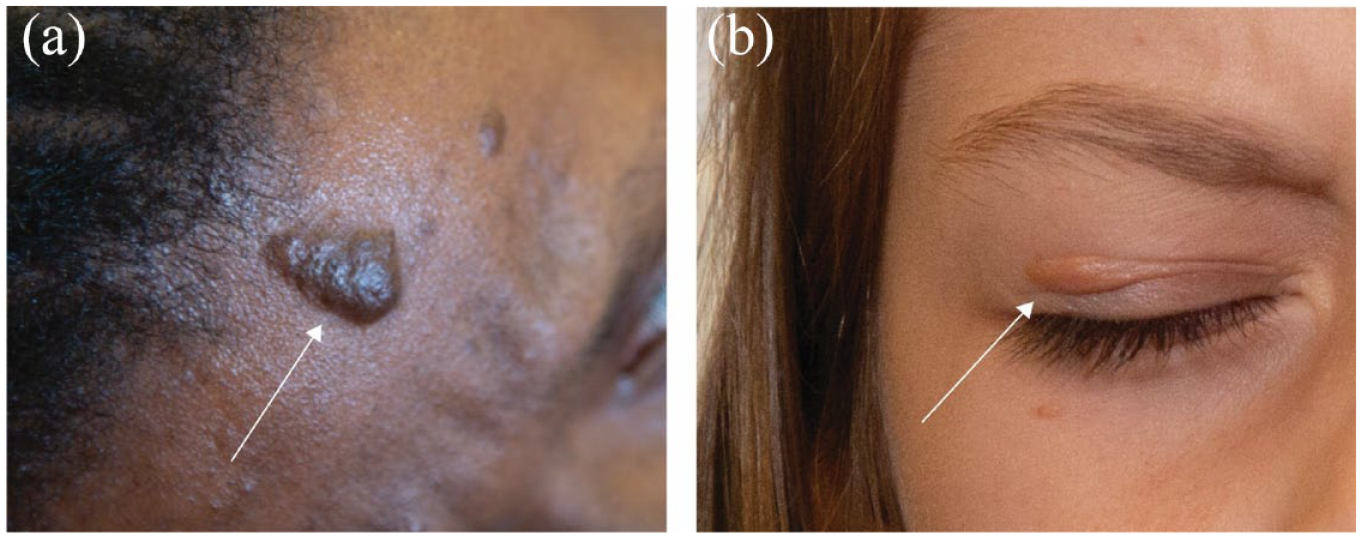
Cephalic fibrous plaques (CFPs) in Black and White individuals with TSC. (a) A 28-year-old Black female and (b) a 7-year-old White female with cephalic fibrous plaques (CFPs) on the forehead and eyelid, respectively. White arrows indicate location of CFPs.

Shagreen patches in a Black and White individual with TSC. (a) An 11-year-old Black male with a shagreen patch on the back and (b) a 37-year-old White female with a shagreen patch on the back.
Facial angiofibromas occur in up to 75% of patients and become more prominent with age. 3 Facial angiofibromas often appear erythematous, raised, and in a butterfly-shaped appearance across the nasolabial folds; however, in Black individuals, they are typically hyperpigmented (Figure 2). In Black individuals, facial angiofibromas may be confused for seborrheic keratoses or other benign epidermal growths or even other neurocutaneous disorders. 10 To date, medical photographs of facial angiofibromas and other TSC skin manifestations are commonly shown as examples from White or other individuals with lighter skin tones, creating a barrier for clinicians to accurately compare in Black or other individuals with darker skin tones. There is an evident lack of published resources for individuals with darker skin. Recent case studies of Black individuals emphasize the importance of early diagnosis, wherein a late diagnosis at the age of 37 in a Black patient led to complications including hemodialysis and a complete bilateral nephrectomy and complications involving the central nervous, renal, and respiratory systems in a 24-year-old, respectively.11,12
Recognizing that skin manifestations of TSC may have implications for diagnosis and treatment, this study seeks to provide examples of how these manifestations present by comparing Black and White individuals from a TSC Center of Excellence (TSCOE) and from participants in the TSC Alliance Natural History Database (NHD) on TSC.
Methods
Study design and ethics approval
We conducted a retrospective chart review of patients from the TSCOE at the Kennedy Krieger Institute to extract the age at diagnosis of TSC by a medical professional, the number and type of clinical features of TSC, and the individual’s race. The Johns Hopkins Institutional Review Board (IRB) approved protocol NA 00076475 Interdisciplinary Protocol for Patients with TSC allowing use of de-identified data from the Kennedy Krieger Institute clinical database for research purposes. Data on skin manifestations, race, genetic testing, and topical treatments from the TSC Alliance NHD were obtained with permission from the NHD and Biosample Repository Steering Committee. The TSC Alliance has IRB approval from Ethical and Independent Review Services for the NHD study (Protocol 15039-08) that permits use of de-identified clinical data for TSC research. STROBE guidelines were followed when preparing the article.
Settings
Data were obtained via retrospective chart review from the TSCOE and data exports from the TSC Alliance NHD. The Tuberous Sclerosis Clinic at Kennedy Krieger Institute is dedicated specifically to the care of individuals affected by TSC and their families. The TSC NHD was implemented in 2006 and captures clinical data to document the impact of the disease on a person’s health over his or her lifetime. More than 2000 people with TSC are enrolled in the project among 18 US-based clinical sites and the TSC Alliance site. The TSC Alliance provides funding to participating clinics to perform data entry, monitors the integrity of the database, and makes data available to investigators to answer specific research questions and identify potential participants for clinical trials and studies. The TSC Alliance utilizes Studytrax as their platform to store and monitor natural history data. Race was obtained from clinic sites via recorded race in the medical record, self-report by participant or participant’s family, or provider observation of skin color. Race obtained from participants consented via the TSC Alliance was obtained via self-report by participant or participant’s family or recorded race in the medical record. Patient or parental consent via a signed photo release was obtained to use images collected from individuals with TSC. All images were captured by the same photographer and camera at a TSC Alliance event.
Participants and data sources
Data from the TSCOE at the Kennedy Krieger Institute from 2009 to 2015 were utilized in these analyses. For the TSCOE cohort, patients with a confirmed clinical diagnosis of TSC and a recorded age at diagnosis were utilized for analysis. Diagnostic criteria can be found in Table 1. Out of 41 patients, 18 were Black, 21 were White, and 2 were classified as other for race (1 Asian, 1 Middle Eastern). Given the limited number of participants in the other category, we restricted our analyses to individuals who were recorded as Black or White. Data collected since 2006 were utilized in an export from the NHD on January 5, 2022. For all NHD data analyses, blank entries were excluded. For the NHD, data from 1966 participants with a confirmed clinical diagnosis of TSC and race recorded as either Black or White out of a total of 2375 participants were utilized in this analysis. Where relevant, the total number of participants used in each comparison is noted. In the NHD, 77% (1,816) of total participants were White and 6% (150) were Black. At the time of analysis, the NHD was underrepresented for Black participants and overrepresented for White individuals as compared with US Census calculations. 13 We selected Black and White participants with a recorded TSC diagnosis date in the NHD (n = 1487; 107 Black and 1380 White) for further analyses. Study size was not previously determined using power analysis but was limited by availability of data within the clinic cohort and the NHD.
Bias
Selection bias was addressed by incorporating analysis of the TSC Alliance NHD to our research question as we recognize that analysis of a single clinic cohort introduces potential bias for geographic location, socioeconomic status, or other variables. We did observe that the number of Black participants in the NHD was underrepresented as compared with the US census; however, due to the retrospective study design, we were limited to enrolled participants at the time of data export.
Statistical analyses
For TSCOE data, statistical analyses were conducted using GraphPad Prism 9 software and reviewed via consultation with the Biostatistics, Epidemiology, and Research Design Unit at The University of Tennessee Health Science Center. No power analyses were conducted, and eligibility included any TSC patient at the TSCOE from 2009 to 2015 with race identified as Black or White. A Fisher’s exact test for the hypotheses of independence between the variables of race and age at diagnosis, race and number of features identified, race and molecular testing, and race and identification of specific features (e.g. facial angiofibromas, hypopigmented macules, and cephalic plaques) was conducted. We further fitted a logistic regression model in which the outcome was being diagnosed at the age of ⩽1 year versus age of ⩾2 years, and the predictor variables were sex, race, and number of features identified.
For NHD data, statistical analyses were conducted using GraphPad Prism 9 software and reviewed via consultation with an independent statistical consultant. No power analyses were conducted, and the full NHD cohort was considered (outlined in Figure 5). Specific tests used are noted in corresponding figure legends. For all NHD data analyses, blank entries were excluded, and the number of records included in each analysis is noted in the figure legends. A Fisher’s exact test for the hypotheses of independence between the variables of race and presence of specific skin manifestations, molecular testing, treatment between races, participation in clinical trials, and age at diagnosis when comparing groups at age ⩽1 year with age ⩾2 years. Unpaired t-tests with Welch’s correction were utilized to compare average age at diagnosis and count of TSC features between races.

TSC participant selection from the NHD.
Results
Participants and descriptive data
The study population from the TSCOE included 41 individuals with a median age of 13 years at time of the study with 44% (18 out of 41) identified as Black and 56% were White (23 out of 44; Table 2). The distribution of age at diagnosis by race in the TSCOE cohort is provided in Table 3. The process of selecting NHD participant records for analysis is shown in Figure 5.
Patient demographics and characteristics at TSCOE.

IQR, interquartile range; TSC, tuberous sclerosis complex; TSCOE, TSC Center of Excellence.
Distribution of age at diagnosis in TSCOE by race.
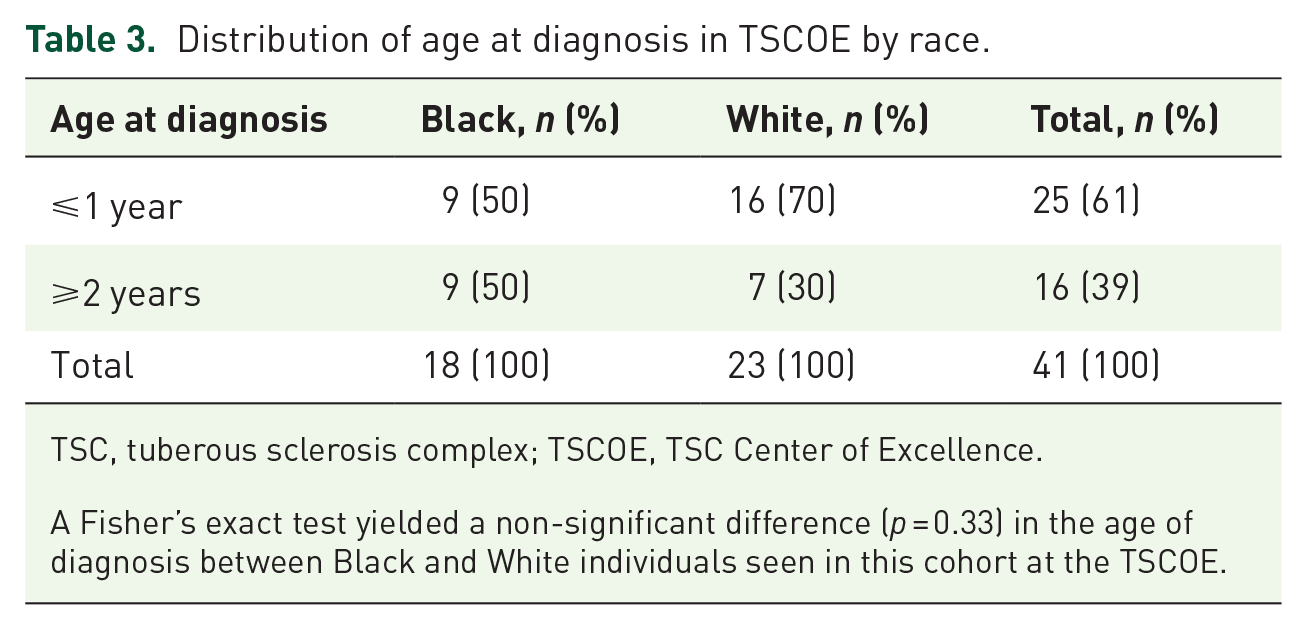
TSC, tuberous sclerosis complex; TSCOE, TSC Center of Excellence.
A Fisher’s exact test yielded a non-significant difference (p = 0.33) in the age of diagnosis between Black and White individuals seen in this cohort at the TSCOE.
Main results
A trend was observed showing that Black patients were less likely to be diagnosed at an earlier age, with 50% (9 out of 18) diagnosed at ⩽1 year compared with 70% (16 out of 23) of White patients in the TSCOE cohort, although this finding was not statistically significant (p = 0.33). We did not have recorded diagnosis dates specific to the dermatological manifestations to assess which features may have been present in the group diagnosed with TSC at age ⩽1 year versus those diagnosed ⩾2 years to support the early diagnosis. The distribution of the number of major TSC features by race at TSCOE is shown in Table 4. A Fisher’s exact test for independence found no evidence of an association between the number of major TSC features and race (p = 0.76). The tests for independence between race and the presence of each of three specific dermatological features – facial angiofibromas, hypopigmented macules, and cephalic fibrous plaques – indicated that race was independent of whether these features were identified (p > 0.05 for all; Supplementary Figure 1).
Distribution of number of major features by race in TSCOE cohort.

TSC, tuberous sclerosis complex; TSCOE, TSC Center of Excellence.
A Fisher’s exact test yielded a non-significant difference (p = 0.76) in the number of major TSC features between Black and White individuals seen in this cohort at the TSCOE.
A multiple logistic regression model in which the outcome was age at diagnosis (⩽1 year versus ⩾2 years) was fitted for the TSCOE cohort. For this model, the predictor variables were race, sex, and number of major features. The results from this model indicate that White individuals had higher odds of being diagnosed by age 1 year; however, this association was non-significant (p = 0.10; Table 5). In addition, we found that a higher number of major features was associated with a significantly increased probability of having been diagnosed earlier: with each increase of one feature, the probability of having been diagnosed at ⩽1 year was increased by a factor of ~1.5 (p = 0.045).
Multiple logistic regression model for being diagnosed before or at age 1 year versus after age 1 year in TSCOE cohort.

TSC, tuberous sclerosis complex; TSCOE, TSC Center of Excellence.
A multiple logistic regression model in which the outcome was age at diagnosis was fitted with the predictor variables of race, sex, and number of major TSC features. The association between sex was not significant. The association between race showed a non-significant trend that White individuals had higher odds of being diagnosed by age 1 year. The number of TSC features did show a significant association, wherein an increased number of features was associated with an increased likelihood of diagnosis at age at ⩽1 year.
denotes statistical significance
For participants that had a recorded TSC diagnosis date in the NHD (n = 107 Black, 1380 White), the average age of TSC diagnosis was similar between White (5.7 years) and Black (5.4 years) individuals (Figure 6). To further compare with the cohort data from the TSCOE, we investigated the proportion of Black and White individuals diagnosed at age ⩽1 year versus ⩾2 years in the NHD. We found a statistically significant difference (p = 0.03) with 38% of Black participants diagnosed at age ⩽1 year compared with 50% of White participants (Figure 7). Furthermore, broadly comparing all Black and White individuals in the NHD, we found no evidence that the number of major or minor TSC features differed between the groups, with both Black and White participants having on average five major and less than one minor features (Figure 8). Looking specifically at TSC dermatological manifestations, we assessed the number of participants with a record of having facial angiofibromas in the NHD. Similar proportions of Black (64%) and White (69%) individuals had facial angiofibromas, and a similar diagnosis age was observed with the average age at facial angiofibroma diagnosis as 11.3 years for Black and 10.7 years for White participants (Figure 9). Further analysis showed that Black participants had a significantly increased probability of having shagreen patches (p < 0.0001) and cephalic fibrous plaques (p = 0.014) as compared with White participants in the NHD (Figure 10). No significant differences were observed for the hypomelanotic macule, ungual fibroma, nor confetti lesion manifestations (Supplementary Table 1). We did not have data on the ungual fibroma nor confetti lesion manifestations from the TSCOE cohort to compare these features further. Overall, these findings highlight potential differences in the presentation of dermatological symptoms in TSC between Black and White individuals that warrants further exploration clinically.
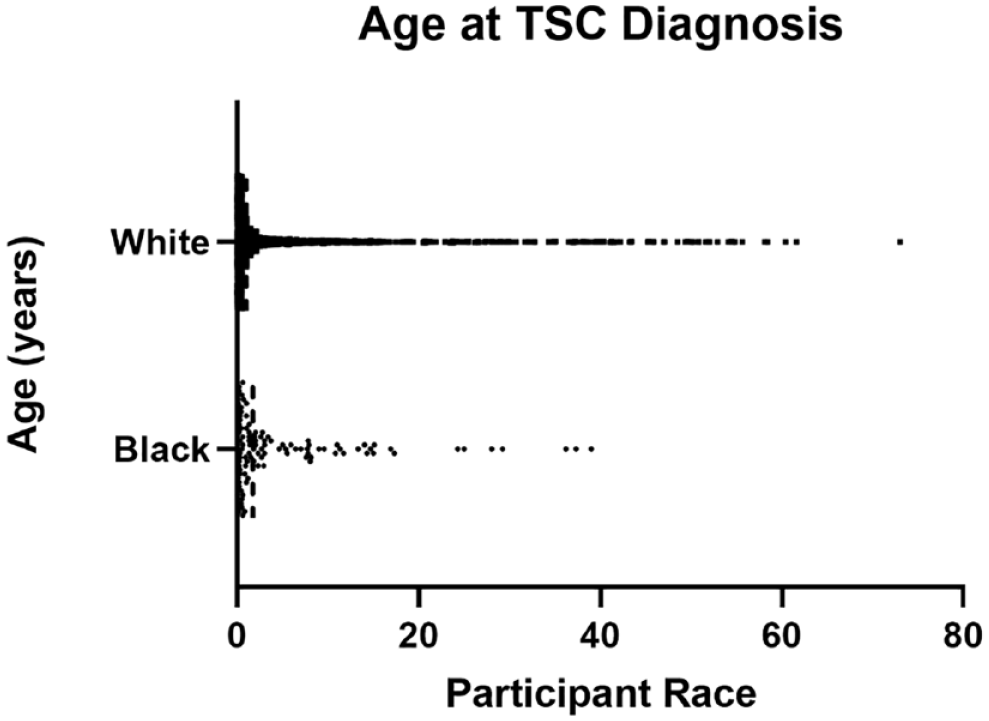
Age at TSC diagnosis for Black and White participants in the TSC natural history database.
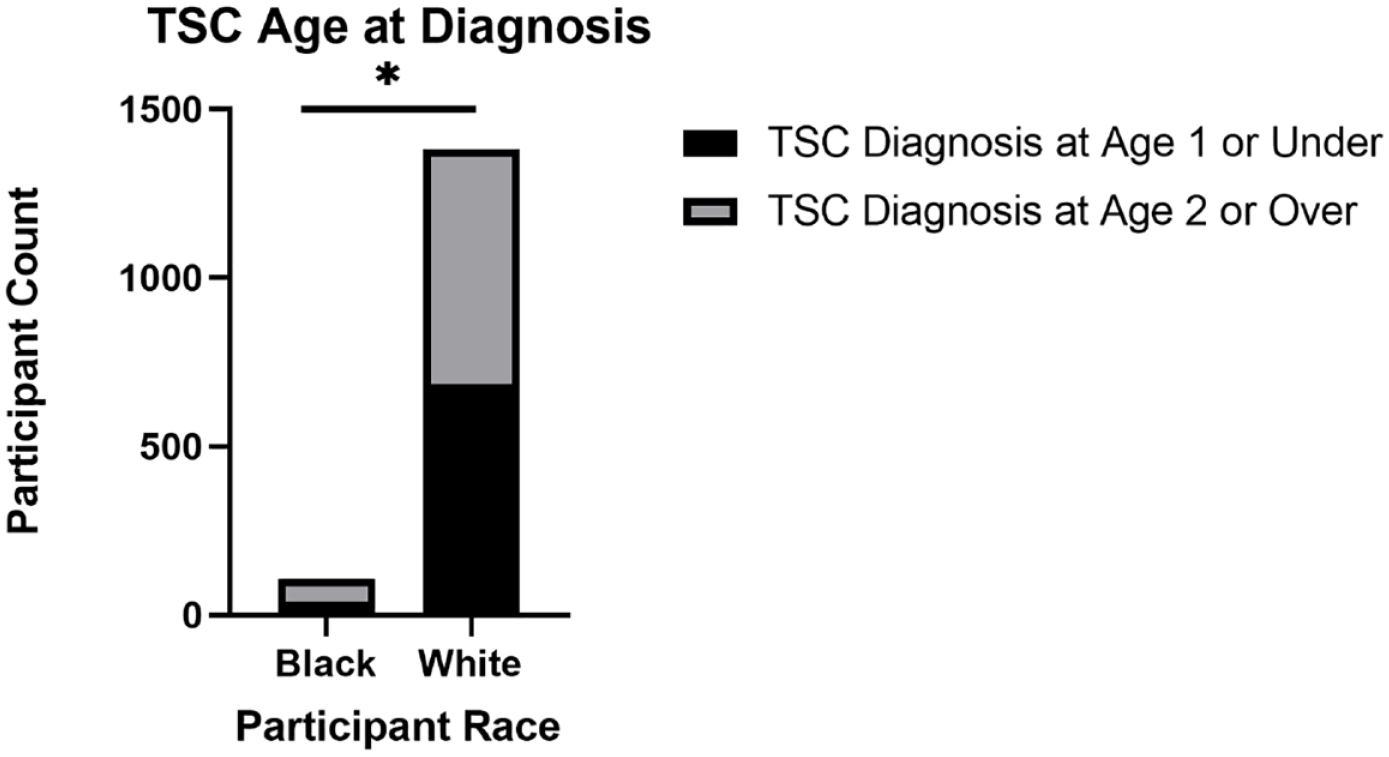
Comparison of age at TSC diagnosis for Black and White participants in the TSC natural history database.
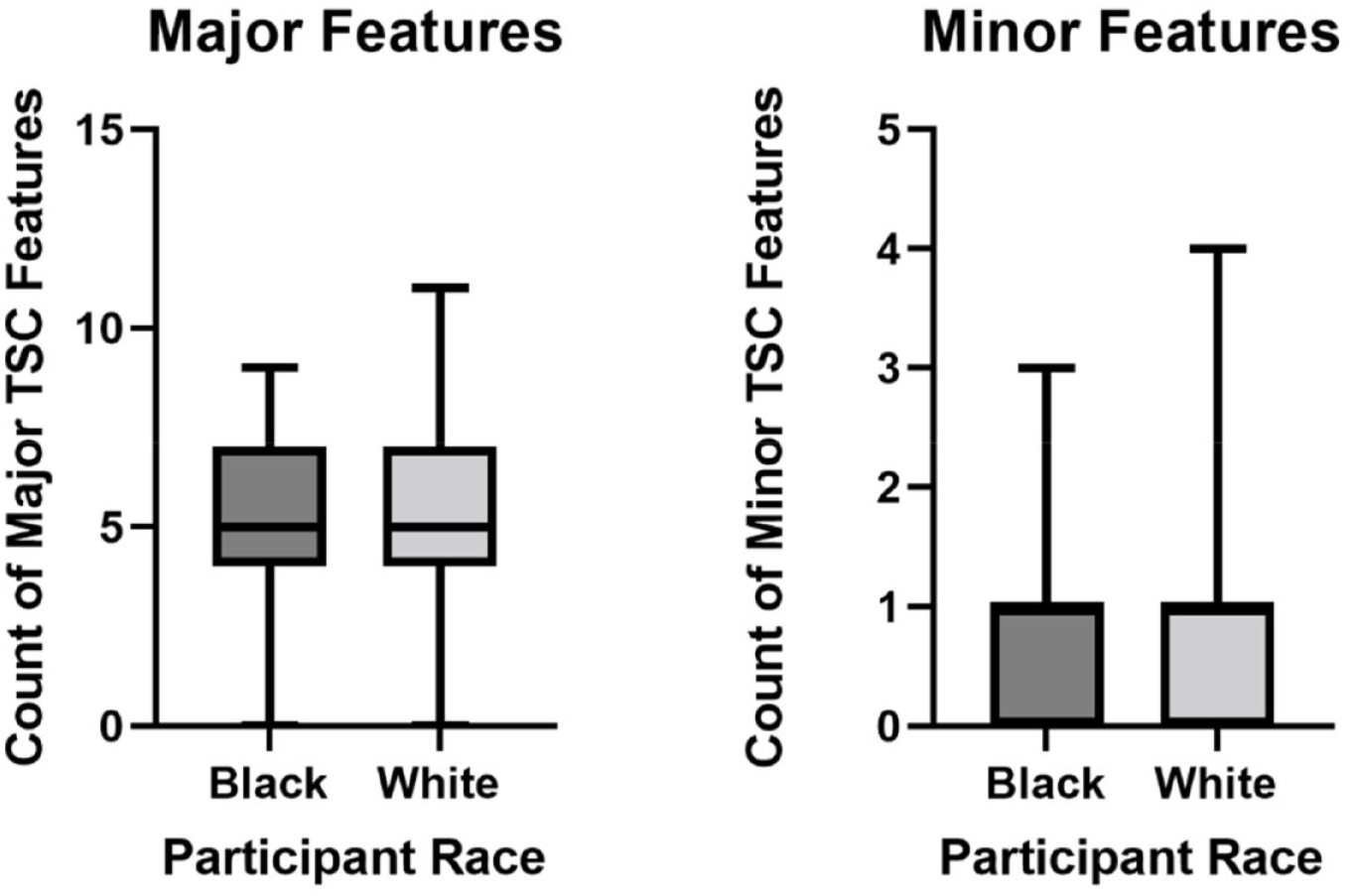
Count of major and minor TSC features in Black and White participants in the TSC natural history database.

Facial angiofibromas in Black and White participants in the TSC natural history database. (a) Count of Black and White individuals with or without facial angiofibromas. Out of 144 Black individuals with a recorded response, a total of 92 (64%) were reported to have facial angiofibromas. Out of a total of 1682 White individuals with a recorded response, a total of 1166 (66%) were reported to have facial angiofibromas. A Fisher’s exact text showed a non-significant difference (p = 0.189) in the percentages of Black and White individuals with facial angiofibroma. (b) A total of 67 Black participants and 791 White participants had a date recorded for the age at angiofibroma diagnosis. An unpaired t-test with Welch’s correction was conducted determining a non-significant difference in the age at facial angiofibroma diagnosis (p = 0.6198) between Black (average age 11.25 years) and White (average age 10.72 years) participants. Mean illustrated by red lines in (b).

Presence of dermatological features in Black and White Participants in the TSC Natural History Database. (a) A total of 141 Black participants and 1639 White participants had a recorded answer for the presence or absence of shagreen patch. A Fisher’s exact test showed a significant difference (p < 0.0001) in the presence of shagreen patch between Black and White Participants, with 55.3% of Black and 37.6% of White individuals noted to have a shagreen patch. Odds ratio of 2.051 (95% CI: 1.457–2.919) indicates that Black individuals have an increased probability of having a shagreen patch. (b) A total of 142 Black participants and 1626 White participants had a recorded answer for the presence or absence of cephalic fibrous plaque. A Fisher’s exact test showed a significant difference (p = 0.0139) in the presence of cephalic fibrous plaque between Black and White participants, with 29.6% of Black and 20.5% of White individuals noted to have a shagreen patch. Odds ratio of 1.625 (95% CI: 1.105–2.358) indicates that Black individuals have an increased probability of having a shagreen patch.
We further investigated the use of a topical mechanistic target of rapamycin inhibitor (mTORi) for facial angiofibromas in the NHD cohort. Analysis showed a greater percentage of White participants utilized a topical mTORi as compared with Black participants (26% versus 13%, respectively; Figure 11). A potential variable within this data set was the lack of FDA approval for the topical mTORi rapamycin to treat facial angiofibroma at the time of data entry in the NHD. Another potential contributing variable was participation in clinical trials for mTORi among NHD participants. To address this, we analyzed NHD participants enrolled in the ‘Topical Rapamycin to Erase Angiofibromas in TSC (Treatment)’ (NCT01526356) clinical trial and observed that 43 White individuals and only 2 Black individuals in the NHD were recorded as participants. Since this trial first published results in 2018, it is possible that there was reluctancy to utilize topical mTORi prior to the publication of trial results. 14 To further investigate, we analyzed use of topical mTORi between Black and White participants diagnosed with facial angiofibromas in year 2018 or later. Unfortunately, we only found 1 Black participant with a recorded facial angiofibroma diagnosis date ⩾ 2018 versus 27 White individuals. Of these, 41% (11 out of 27) of White participants and the single Black participant were recorded to be taking a topical mTORi. Further analysis is needed to assess prescription patterns of topical mTORi in Black and White individuals across multiple TSC clinic sites, especially given the recent FDA approval of Hyftor sirolimus topical gel in 2022. 15
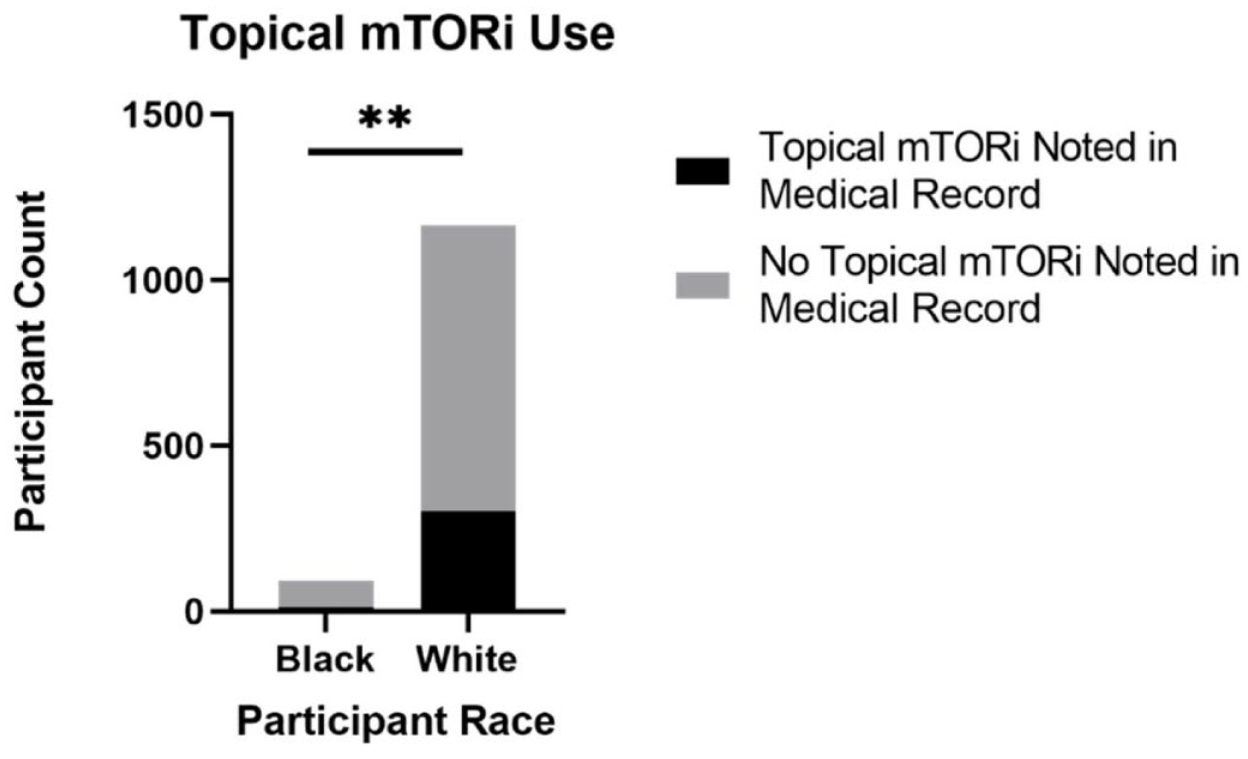
Use of topical rapamycin for facial angiofibroma in Black and White participants in the TSC natural history database.
Although systemic mTORi therapies are not currently approved for facial angiofibromas, reduction in the appearance of facial angiofibromas has been noted in patients taking them for other major features of TSC. 16 We assessed the use of systemic mTORi across Black and White participants with a facial angiofibroma diagnosis and found 8 Black participants (8 out of 92, 9%) and 86 White individuals (86 out of 1166, 7%) that were prescribed a systemic mTORi for SEGA (subependymal giant cell astrocytomas), and 9 Black participants (9 out of 92, 10%) and 107 White participants (107 out of 1166, 9%) treated with a systemic mTORi for renal angiomyolipoma, suggesting the disparity we observed in mTORi utilization was specific to topical mTORi use. Further analysis is needed to investigate this finding in a clinical setting. Given the small number of recorded Black participants in the topical rapamycin trial, we further investigated research participation recorded in the NHD. Participation of NHD participants is recorded for 17 distinct clinical trials (Supplementary Table 2). While 217 (217 out of 1816, 11.9%) White individuals had participated in at least one clinical trial recorded in the NHD, only 8 (8 out of 150, 5.3%) of Black individuals had, illustrating a significant difference in clinical trial participation (p = 0.01; Table 6). We observed a similar trend looking at participation in the TSC Alliance’s Biosample Repository project, wherein only 22 Black (22 out of 150, 14.7%) versus 516 White (516 out of 1816, 28.4%) individuals were participating at the time of analysis. This result could be due to lack of representation in the NHD as it is a research study itself or for other reasons as further highlighted in the discussion.
Participation of Black and White participants in the TSC natural history database in recorded TSC clinical trials.

TSC, tuberous sclerosis complex.
A total 217 (11.9%) White participants have participated in at least one clinical trial recorded in the TSC Natural History Database versus only 8 (5.3%) Black participants. Clinical trials reported in the NHD shown in Supplementary Table 2. A Fisher’s exact test shows a significant difference (p = 0.01) between Black and White participation in TSC clinical trials. Odds ratio 2.4 (95% CI: 1.187–5.028).
Since genetic testing is one route of obtaining a TSC diagnosis, we further investigated the frequency of molecular testing for the TSC1 or TSC2 genes and found a significant difference (p = 0.009) between Black and White participants. A total of 145 Black participants and 1692 White participants had a recorded answer for molecular testing, with 79 Black (79 out of 145, 54.5%) and 1108 White (1108 out of 1692, 65.5%) having confirmed molecular testing related to TSC recorded in the NHD (Figure 12). This finding was also observed in the TSCOE cohort with a significant difference noted (p = 0.049), wherein only 44% of Black versus 78% of White participants had molecular testing noted in their medical records (Figure 13). Further analysis is warranted to assess why this discrepancy exists specifically in the context of genetic testing for TSC.

Molecular testing in Black and White participants in the TSC natural history database.
Discussion
A late diagnosis of TSC can delay appropriate treatment and lead to unnecessary and sometimes devastating consequences. For example, subependymal giant cell astrocytomas carry the risk of growing large enough to obstruct ventricular flow in the brain, leading to hydrocephalus and death without emergent surgical intervention. 17 Renal angiomyolipomas, which contribute to renal disease are the most common cause of mortality in TSC 18 and can encroach on normal renal tissue, leading to end-stage renal disease. 19 Growing renal angiomyolipomas also carry the risk of causing aneurysms and life-threatening hemorrhages. 19 Lymphangioleiomyomatosis (LAM), which occurs primarily in women with TSC, can lead to progressive decline in lung function. 10 It is important to note that systemic and topical therapies in the form of mTORi are available and recommended by the 2021 TSC consensus guidelines to treat specific manifestations of TSC. 20 Early treatment of epilepsy in TSC also has implications for neurodevelopmental outcomes in TSC-associated neuropsychiatric disorders (TAND), including intellectual disability and autism spectrum disorder. Results from the TSC Autism Center of Excellence Network (TACERN) study determined that infants with TSC who develop seizures early in life are more likely to exhibit global developmental deficits by 24 months of age than infants with TSC that do not experience early seizures.21,22 Although dermatological features are not life threatening and can manifest at different points across the lifespan, the dermatological features that present in the early years of life are important indicators of a TSC clinical diagnosis. As a result, prompt diagnosis is critical to optimize neurocognitive outcomes, minimize life-threatening complications, and improve the quality of life for those with TSC. 23
Skin lesions are the most readily visible manifestations of TSC and provide an opportunity to identify the disease without invasive medical tests. However, we observed that facial angiofibromas, which are recognizable even without a complete skin exam, may appear differently (Figure 2). Other skin manifestations such as hypomelanotic macules, cephalic fibrous plaques, and shagreen patches may also appear differently across skin tones (Figures 1, 3, 4) and can present at different frequencies across races (Figure 10). While we recognize that our observed differences in diagnosis frequencies at age ≤1 were likely not due to the appearance of facial angiofibromas as they most often appear later in childhood, the other skin manifestations may play a role and warrant further exploration in other clinic data sets. We did not have sufficient data on diagnosis dates for these features to analyze at the time of data export.
Therefore, we hope this publication increases recognition of these features and leads to improved early identification of this disorder to provide proactive intervention and avoid irreversible and catastrophic outcomes. Regarding our finding from NHD data related to underutilization of topical mTORi in Blacks relative to Whites, other potential barriers exist that could contribute including economic status (e.g. not able to afford the medication and/or lack of insurance or insurance coverage), access issues (e.g. no compounding pharmacies nearby to make the topical cream, living far away from a clinical trial site), and potential mistrust by minority groups of the medical or research community for treatments that are not yet FDA approved.24,25 The latter point has been observed in many cancer clinical trials, with Black patients constituting less than 4% of all patients enrolled across multiple trials. 26 Not only does this perpetuate underrepresentation of minority groups, but also has the potential to expand outcome disparities due to the possibility of unique biological differences inherent to different racial groups. We noted a similar trend in our data showing that White individuals were twice as likely than Black individuals to participate in a clinical trial looking at 17 different TSC trials in the NHD. We observed a comparable trend when assessing participation in the TSC Alliance’s Biosample Repository project, wherein Black individuals were participating at a lower rate than White individuals. Similarly, genetic testing in data sets from both the TSCOE and NHD illustrated that Black individuals were less likely to receive genetic testing than White individuals (Figures 12 and 13). This result may be due to lower awareness of genetic testing as suggested by Canedo et al., 27 concerns about trust, privacy, and the benefits of genomic studies, 28 or other factors not considered here. While we understand some of the barriers to clinical trial participation 29 and medication use, further analysis of specific barriers to participation is warranted in the context of TSC. In summary, these data highlight that racial disparities are not limited to common diseases such as cancer, but that they also exist in the rare disease space. It may be difficult for diseases that are rarer than TSC to obtain sufficient numbers of patients to conduct similar analyses.30,31

Molecular testing in Black and White participants in the TSCOE.
Limitations
While we aim to bring awareness to racial disparities in the context of TSC, our analyses are limited. A limitation to the NHD data analysis is that we do not know if our data set is a representative sample size. Total numbers differ across figures due to several blank (i.e., unknown) responses regarding the feature being analyzed. For each feature, analysis was limited to records with a recorded value, further restricting our sample size for each comparison. Where applicable, the number of records is included in the text or figure legend. In addition, the NHD was underrepresented for Black individuals and overrepresented for White individuals as compared with the US census at the time of data export. Increased participation in research by minority groups is crucial to properly analyze disparities and potentially discover relevant differences to approach treatment based on race. Another major limitation was the cohort size at the TSCOE (n = 41). Replication across additional TSC clinics would be helpful to expand on these analyses.
Conclusion
There is limited understanding about how racial and ethnic disparities impact patient care outcomes in the TSC community. In this publication, we highlight a disparity between Black and White individuals with TSC specifically in the underrepresentation of Black patients in research studies and clinical trials, age at TSC diagnosis, differences in the utilization of topical mTORi therapy and molecular testing, and appearances of skin symptoms that are major features for diagnosis. Identifying these disparities and their potential as barriers to care is the first step to implementing evidence-based practices to optimize patient-centered clinical outcomes. These differences warrant further study across additional clinic sites and other races in the future.
Supplemental Material
sj-docx-1-trd-10.1177_26330040221140125 – Supplemental material for Racial differences in the dermatological manifestations of tuberous sclerosis complex and the potential effects on diagnosis and care
Supplemental material, sj-docx-1-trd-10.1177_26330040221140125 for Racial differences in the dermatological manifestations of tuberous sclerosis complex and the potential effects on diagnosis and care by Ashley J. Pounders, Gabrielle V. Rushing, Sonal Mahida, Bareng Aletta Sanny Nonyane, Emily A. Thomas, Rabiah Sundus Tameez and Tanjala T. Gipson in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors thank the participants of the TSC Alliance Natural History Database project for allowing their data to be used for research. They also thank the Biostatistics, Epidemiology, and Research Design Unit at The University of Tennessee Health Science Center and Rich Mushlin for consulting with them regarding the statistical tests utilized in these analyses. They acknowledge Katherine B. Puttgen, Anna L. Grossberg, and Bernard Cohen who provided the dermatological differential diagnosis at the TSCOE. This publication was made possible by the Johns Hopkins Institute for Clinical and Translational Research (ICTR) which is funded in part by Grant Number UL1 TR003098 from the National Center for Advancing Translational Sciences (NCATS) a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the Johns Hopkins ICTR, NCATS or NIH.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
