Abstract
Introduction:
Barth syndrome (BTHS) is a rare X-linked disorder characterized by cardiomyopathy, neutropenia, growth abnormalities, and skeletal myopathy. There have been few studies investigating health-related quality of life (HRQoL) in this population. This study investigated the impact of BTHS on HRQoL and select physiologic measures in affected boys and men.
Methods:
In this study, we characterize HRQoL in boys and men with BTHS through cross-sectional analysis of a variety of outcome measures including the Pediatric Quality of Life Inventory (PedsQLTM) Version 4.0 Generic Core Scales, PedsQLTM Multidimensional Fatigue Scale, Barth Syndrome Symptom Assessment, the PROMISTM Fatigue Short Form, the EuroQol Group EQ-5DTM, the Patient Global Impression of Symptoms (PGIS), and the Caregiver Global Impression of Symptoms (CaGIS). For a specific subset of participants, physiologic data were available in addition to HRQoL data.
Results:
For the PedsQLTM questionnaires, 18 unique child and parent reports were analyzed for children aged 5–18 years, and nine unique parent reports were analyzed for children aged 2–4 years. For the other HRQoL outcome measures and physiologic measurements, the data from 12 subjects (age range 12–35 years) were analyzed. Based on parent and child reports, HRQoL is significantly impaired in boys and men with BTHS, especially in school functioning and physical functioning. Parent and child reports of more severe fatigue are significantly correlated with more impaired HRQoL. When exploring the potential relationship between physiology and HRQoL, the CaGIS as a whole for pediatric subjects and individual questionnaire items from the PGIS and CaGIS for pediatric subjects assessing tiredness, muscle weakness, and muscle pain showed the strongest correlations.
Conclusion:
This study provides a unique characterization of the HRQoL in boys and men with BTHS using a variety of outcome measures, and it highlights the negative impact of fatigue and muscle weakness on HRQoL in BTHS.
Trial registry name::
A Trial to Evaluate Safety, Tolerability and Efficacy of Elamipretide in Subjects with Barth Syndrome (TAZPOWER). https://clinicaltrials.gov/ct2/show/NCT03098797.
Registration Number: NCT03098797
Plain Language Summary
Barth syndrome is a rare disorder characterized by heart issues, muscle weakness, tiredness, exercise intolerance, and growth delays. The study was done to determine the effect of Barth syndrome on health-related quality of life of the boys and men affected. We analyzed health-related quality of life questionnaires completed by subjects and/or their parents from the following:
• Interdisciplinary Barth Syndrome Clinic at Kennedy Krieger Institute. There were 24 subjects in total from this clinic.
• Baseline data from a clinical drug trial for Barth Syndrome that included both health-related quality of life data and physical function data. There were data from 12 subjects in total from the trial.
We discovered that health-related quality of life is significantly impaired in boys and men with Barth syndrome, especially in school and physical function. Parent and child reports of more severe tiredness are significantly linked with impaired health-related quality of life. There are strong relationships between some health-related quality of life reports and physical function measurements. Tiredness and muscle weakness negatively impact health-related quality of life. We are hopeful that the results of this study will be used in the treatment of boys and men with Barth syndrome to result in improved health-related quality of life.
Introduction
Barth syndrome (BTHS, MIM no. 302060) 1 is a rare X-linked disorder with an estimated prevalence of 1 in 1,000,000 men 2 characterized by cardiomyopathy, neutropenia, growth abnormalities, and skeletal myopathy. 3 It is associated with pathogenic variation of the TAZ gene (MIM * 300394), 4 which encodes a mitochondrial transacylase involved in cardiolipin metabolism. Boys and men with BTHS present with eating difficulties, pain, attention difficulties, memory problems, fatigue, and muscle weakness.5–7 The most frequently reported symptoms of boys and men with BTHS are fatigue/tiredness and muscle weakness. 7 The cardinal characteristics of BTHS have been thought to contribute to difficulties in school, work, and activities of daily living. 8 Boys and men with BTHS have a significantly increased risk of premature death. 9 Many children die during early childhood secondary to heart failure or infection. Those who survive into adulthood have a shortened life expectancy. 10
The World Health Organization defines health as ‘not only the absence of disease and infirmity, but the presence of physical, mental and social well-being’. 11 The Centers for Disease Control and Prevention defines health-related quality of life (HRQoL) as ‘an individual’s perceived physical and mental health over time’. 12 It is recommended that HRQoL assessments be routinely implemented in pediatric health care. 13 When assessing HRQoL in childhood-onset disorders, parent and child reports are recommended when possible to ensure an evaluation is as comprehensive as possible since children and parents base their judgments on HRQoL on different experiences. 14 Previous studies of HRQoL ratings in boys and men with BTHS using the Pediatric Quality of Life Inventory (PedsQLTM) Version 4.0 demonstrated HRQoL was lower in BTHS in comparison with healthy controls and a cohort with cardiac disease. 8 They have also demonstrated good agreement between parent and child report in BTHS using the PedsQLTM.8,15 The authors had the opportunity to assess HRQoL in patients with BTHS through multiple avenues including an interdisciplinary clinic that provides routine clinical care as well as during baseline assessment to consider participation in a clinical drug trial.
Aim and research objectives
The goal of this study is to provide a comprehensive overview of HRQoL in boys and men with BTHS based on the use of PedsQLTM questionnaires (Version 4.0 Generic Core Scales and Multidimensional Fatigue Scale) from our interdisciplinary clinic as well as a battery of symptom and HRQoL questionnaires from baseline data obtained for a clinical drug trial. For a specific subset of participants, physiologic data were available in addition to HRQoL data. Given the wealth of information provided by these measurements in research,7,8,13–16 we are hoping to inspire clinicians to incorporate a battery of HRQoL measures into routine care for patients with BTHS to help make treatment recommendations as targeted as possible.
Methods
The study was cross-sectional in nature. As this was an observational, non-interventional study, power analysis was not performed for this study. This study was approved by the Johns Hopkins University IRB protocol ‘NA_0008316 Clinical Studies in Barth Syndrome’ and ‘IRB00124162 A Phase 2 Randomized, Double-blind, placebo controlled crossover trial to evaluate the safety, tolerability, and efficacy of subcutaneous injections of Elamipretide (MTP-131) in subjects with genetically confirmed Barth Syndrome’.
All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki of 1975, as revised in 2013. Informed written consent was obtained from all participants in the studies. Proof that informed consent was obtained is available upon request.
Outcome measures
PedsQL
Patients were seen in Interdisciplinary BTHS Clinic at Kennedy Krieger Institute (KKI) from 4 June 2013 to 3 March 2021. Patients were eligible to participate if they had a diagnosis of BTHS, attended the Interdisciplinary clinic, and were between 2 and 18 years of age (as this is the age range for the PedsQL).
The PedsQLTM Version 4.0 Generic Core Scales (PedsQLTM) and PedsQLTM Multidimensional Fatigue Scale were used to measure the HRQoL of boys with BTHS, via both child and parent proxy report. The questionnaires were provided to subjects and families via email prior to arrival at Interdisciplinary BTHS Clinic at KKI in Baltimore, MD. The questionnaires were either returned via email or provided on the day of clinic. The questionnaires were scored by the physical therapist then entered into the medical record and RedCap database.
The PedsQLTM 4.0 Generic Core Scales assesses the following: physical functioning (eight items), emotional functioning (five items), social functioning (five items), and school functioning (five items). There are both child self-report and parent proxy report formats. The subjects are asked how much of a problem each item has been during the past 1 month. 17 The child self-report includes ages 5–7, 8–12, and 13–18 years. The parent proxy report includes ages 2–4 years in addition to the older age groups. The content is similar across forms and differs only in developmentally appropriate language and first- or third-person tense. A 5-point Likert-type scale is used across child self-report for ages 8–18 years and parent proxy report (0, never a problem; 1, almost never a problem; 2, sometimes a problem; 3, often a problem; and 4, almost always a problem). For the young child self-report (ages 5–7 years), the Likert-type scale is simplified to 3 points (0, not at all problem; 2, sometimes a problem; 4, a problem for you a lot). 18 Items are reverse-scored and linearly transformed to a scale from 0 to 100 points with higher scores indicating a better QoL. In addition to a total score, a 0 to 100 subscale score can also be generated for each of the four domains (physical functioning, emotional functioning, social functioning, and school functioning). 17 A psychosocial health summary score is calculated by computing the mean as the sum of the items divided by the number of the items in the emotional, social, and school functioning scales. 18
The reliability and validity of the PedsQLTM 4.0 Generic Core Scales have been demonstrated in children and parents in both the healthy and chronic disease population across the world.17–21 Clinically meaningful cut-off scores for identification of an ‘at-risk’ status or significantly impaired HRQoL has been determined approximating 1 SD below the mean in the general pediatric population sample, which includes both healthy children and children with chronic health conditions. For example, the child self-report PedsQL 4.0 Total Scale Score cut-off point score was 69.7 (parent proxy report score was 65.4). 17 Huang et al. 22 suggested clinical cut points for total score on the PedsQL for children less than 8 years old set at 77 for major chronic conditions and 70 for children greater than or equal to 8 years old.
The PedsQLTM Multidimensional Fatigue Scale was designed to measure fatigue across pediatric populations. It consists of three subscales: general fatigue (six items), sleep/rest fatigue (six items), and cognitive fatigue (six items). 23 The PedsQLTM Multidimensional Fatigue Scale has demonstrated reliability and validity in pediatric patients, aged 5–18 years, with cancer, rheumatologic conditions (e.g. fibromyalgia), type I diabetes, and in healthy children and adolescents.23–25
BTHS-SA, PFSF, EQ-5D, PGIS, CaGIS, physiologic data
Additional data on 12 subjects were collected as part of a clinical trial protocol between 19 July 2017 and 26 February 2018 at Johns Hopkins Hospital, which has been previously described 26 (Clinicaltrials.gov NCT03098797). Subjects were eligible to participate in the study if they were 12 years of age or older, medically stable, ambulatory, and able to complete the 6-min walk test. For additional information regarding inclusion and exclusion criteria, please see Reid Thompson et al. 26 The data analyzed in this study were from the baseline portion of the trial only (prior to participants beginning any medication as part of the trial). The average age was 19.5 years (range, 12–35 years).
A large battery of patient-, caregiver-, and physician-reported outcomes were chosen for the clinical trial given the small sample size and since the study team did not want to miss the opportunity to assess effects in all domains of HRQoL.
In brief, HRQoL data were collected via administration of age-appropriate versions of questionnaires including the BTHS-SA, the PROMISTM Fatigue Short Form (PFSF), the EuroQol Group EQ-5DTM (EQ-5D), the Patient Global Impression of Symptoms (PGIS), the Caregiver Global Impression of Symptoms (CaGIS), and the Clinician Global Impression of Symptoms (CGIS) at the time of the first study visit and prior to intervention. Physiologic data were collected using the AVIVO Mobile Patient Monitoring System (AVIVO-MPMS), which includes a non-invasive body-adherent PiiX device equipped with linear electrodes and an accelerometer.27,28 Subjects were monitored using the AVIVO-MPMS for an average of 7.5 days (range = 6–9 days) after the first study visit and prior to intervention. The physiologic data collected included: heart rate (HR), respiratory rate (RR), impedance (as a marker of fluid status), activity duration, activity intensity, and resting posture.
The Barth Syndrome Symptom Assessment (BTHS-SA) is a content-valid and psychometrically sound BTHS symptom-focused PRO questionnaire for use in clinical trials of investigational treatments. 7 It has adolescent and adult versions and has a 24-h recall.
The PFSF was developed using psychometric methods for the assessment of HRQoL measures as a part of the Patient-Reported Outcomes Measurement Information System (PROMIS) project funded by the National Institutes of Health. 29 The 10-item pediatric version was administered for subjects less than 18 years of age, and the 8-item adult version was administered for subjects 18 years of age or older. 30
The EQ-5D was developed to enable the standardized assessment of HRQoL measures using a five dimension descriptive system and a valuation scale. 31 The five health status dimensions assessed include Mobility, Self-Care, Usual Activities, Pain/Discomfort, and Anxiety/Depression. The valuation scale has a range of 0–100, with 0 representing the worst health imaginable and 100 representing the best health imaginable. The pediatric version was administered for subjects less than 16 years of age, and the adult version was administered for subjects 16 years of age or older.32,33
The Clinical Global Impression (CGI) scales were used for the investigator to provide an overall assessment of the subject’s symptoms related to their diagnosis of BTHS.
The CaGIS scales were used to assess the caregiver’s overall assessment of the subject’s symptoms related to their diagnosis of BTHS.
The PGIS, CaGIS, and CGIS assessed the severity of BTHS-related symptoms during the preceding week according to a 5-point scale that includes the following: no symptoms, mild symptoms, moderate symptoms, severe symptoms, and very severe symptoms. 34 All global impression scales assessed the severity of BTHS-related symptoms overall. The pediatric versions of the PGIS and CaGIS were administered for subjects less than 16 years of age, and they also assessed the severity of: tiredness at rest, tiredness during activities, muscle weakness at rest, muscle weakness during activities, muscle pain at rest, muscle pain due to activities, early fullness when eating, difficulty eating, and headache. The adult versions of the PGIS and the CaGIS were administered for subjects 16 years of age or older, and they also assessed the severity of: tiredness at rest, tiredness during activities, muscle weakness at rest, muscle weakness during activities, muscle pain at rest, muscle pain due to activities, dizziness/lightheadedness, and shortness of breath.
Statistical analyses
PedsQLTM
All data were normally distributed as assessed using the Shapiro-Wilk Test. For analysis, we made sure to consider the profile (various dimensions) as opposed to focusing on only the sum score as was recommended by Pinquart, 13 given that declines in HRQoL can vary across dimensions. The Pearson Correlation Coefficient (Pearson r) was used to test consistency in rank order relationships between child and parent reports. 15 Paired t tests were also performed to assess for differences between child and parent report. Pearson r was used to assess correlation between all survey HRQoL domains and age. Pearson r was also used to assess for correlations among the PedsQL Multidimensional Fatigue Scales and the PedsQL 4.0 Generic Core Scales.
BTHS-SA, PFSF, EQ-5D, PGIS, CaGIS, physiologic data
The data were not normally distributed as assessed using the Shapiro–Wilk Test. For analysis, Spearman’s Rank Correlation Coefficient (Spearman ρ) was used to assess correlation between questionnaire responses and physiologic measurements as well as age. It was also used to assess correlation between the responses of the clinician and the responses of the patients and families to their corresponding questionnaires.
Correlations were considered very strong if the magnitudes were greater than or equal to 0.7, strong if between 0.4 and 0.69, moderate if between 0.3 and 0.39, weak if between 0.2 and 0.29, and none/negligible if between 0.01 and 0.19.
Results
PedsQL
From 4 June 1013, to 3 March 2021, we had a total of 23 unique responses to the PedsQLTM (parent and/or pediatric version) 4.0 Generic Core Scales and PedsQLTM Multidimensional Fatigue Scale for children aged 5–18 years. From these 23 responses, one record was removed as it was an incomplete data set. We had a total of 18 unique participants (for the participants who had completed the age 5–18 questionnaire on multiple visits, the most recent response was utilized for analysis to reduce problems with dependent data). Both parent report and child report forms were analyzed. The average participant age was 10.42 years [standard deviation (SD) = 3.31 years, median = 9.6 years, minimum = 5.2 years, maximum = 17.2 years], and for additional participant characteristics, please see Table 1. There were no consistent medication trends that would allow for correlation outcomes. The means, SDs, Pearson r correlations and p values are summarized in Table 2 (average domain scores for child and parent questionnaires are displayed via bar graph in Supplemental Figure 1). A very strong correlation between parent proxy and patient report was observed for: school function (r = 0.81), psychosocial health (r = 0.71), and PedsQL total score (r = 0.72). A strong correlation between parent proxy and patient report was observed for physical functioning (r = 0.68), emotional functioning (r = 0.66), social functioning (r = 0.51), general fatigue (r = 0.60), sleep/rest fatigue (r = 0.60), cognitive fatigue (r = 0.65), and total fatigue (r = 0.66). A strong negative correlation was found between age and: child report general fatigue (r = –0.56) and child report sleep/rest fatigue (r = –0.60).
Study participant characteristics, Interdisciplinary BTHS Clinic.

BS, Barth syndrome.
When parent and child completed the questionnaire, parent answer was provided first.
Not answered.
Domains combined from PedsQL Quality of Life Inventory and Multidimensional Fatigue Scale for children and parent proxy reports for children aged 5–18 years (n = 18 for child and parent reports).

HRQoL, health-related quality of life; PedsQL, Pediatric Quality of Life Inventory.
Means and standard deviations provided for each domains as well as Pearson r for parent and child reports. p value provided from paired t test of child and parent reports.
At-risk status for impaired HRQoL relative to population sample. 17
Below cut-off scores for major chronic health conditions, 22 using total score: <8 years = 77, for children 8+ years = 70.
Using previously recommended clinical cut points for the PedsQLTM 4.0 Generic Core Scales 20 (Table 2), average of the child reports demonstrated significantly impaired HRQoL in the following domains: physical functioning, emotional functioning, and school function. The average of parent reports demonstrate significantly impaired HRQoL in the following domains: emotional functioning, social functioning, school function, and psychosocial health. The percentage of responses demonstrating impaired HRQoL using the PedsQLTM was also analyzed (Table 3) for the cohort of 18 children and parents who completed questionnaires for age 5–18 years as well as the nine parent reports for the cohort of children aged 2–4 years. In the child and parent reports for children aged 5–18 years, the largest percentage of respondents reported significantly impaired HRQoL in the physical health domain (66.7%, 72.2%). The parent report for children aged 2–4 years demonstrated the largest percentage of those reporting significantly impaired HRQoL in social function domain (44.4%).
Age 5–18 child and parent report and age 2–4 parent report – percentage of subjects demonstrating at-risk status for impaired HRQoL relative to population sample using PedsQL 4.0.

HRQoL, health-related quality of life; PedsQL, Pediatric Quality of Life Inventory.
In all domains, utilizing a paired t test, there was not a statistically significant difference between parent and child response (Table 2).
Pearson r correlations among the PedsQL Multidimensional Fatigue Scales and the PedsQL 4.0 Generic Core Scales are displayed in Table 4. More severe fatigue was correlated with more impaired HRQoL. On both the parent and child reports, there was a strong to very strong correlation between total fatigue and physical function, psychosocial health, social functioning, and school functioning. There were strong to very strong correlations across all fatigue levels (total fatigue, general fatigue, sleep/rest fatigue, and cognitive fatigue) and school functioning in both the parent and child report. The parent report revealed very strong correlations across all fatigue levels and emotional functioning while the child report only demonstrated moderate to strong correlations between all fatigue levels and emotional functioning.
Pearson r correlations among PedsQL Multidimensional Fatigue Scales and PedsQL 4.0 Generic Core Scales for child self-report and parent proxy report for ages 5–18 years (n = 18).

PedsQL, Pediatric Quality of Life Inventory.
There was a total of nine complete PedsQLTM Generic Core Scales and Multidimensional Fatigue Scale parent reports for children aged 2–4 years (there were three subjects whose parents completed questionnaires when their children were aged 2–4 years and returned to clinic when their children were older that completed questionnaires for children aged 5–18 years as well). The average patient age was 3.36 years (SD = 0.75 years, median = 3.3 years, minimum = 2.3 years, maximum = 4.7 years). Based on averages of these nine responses (Figure 1), the domain that is most impaired is social functioning (mean = 62.36 ± 22.05) and the domain that is least impaired is sleep/rest fatigue (mean = 83.32 ± 12.15).
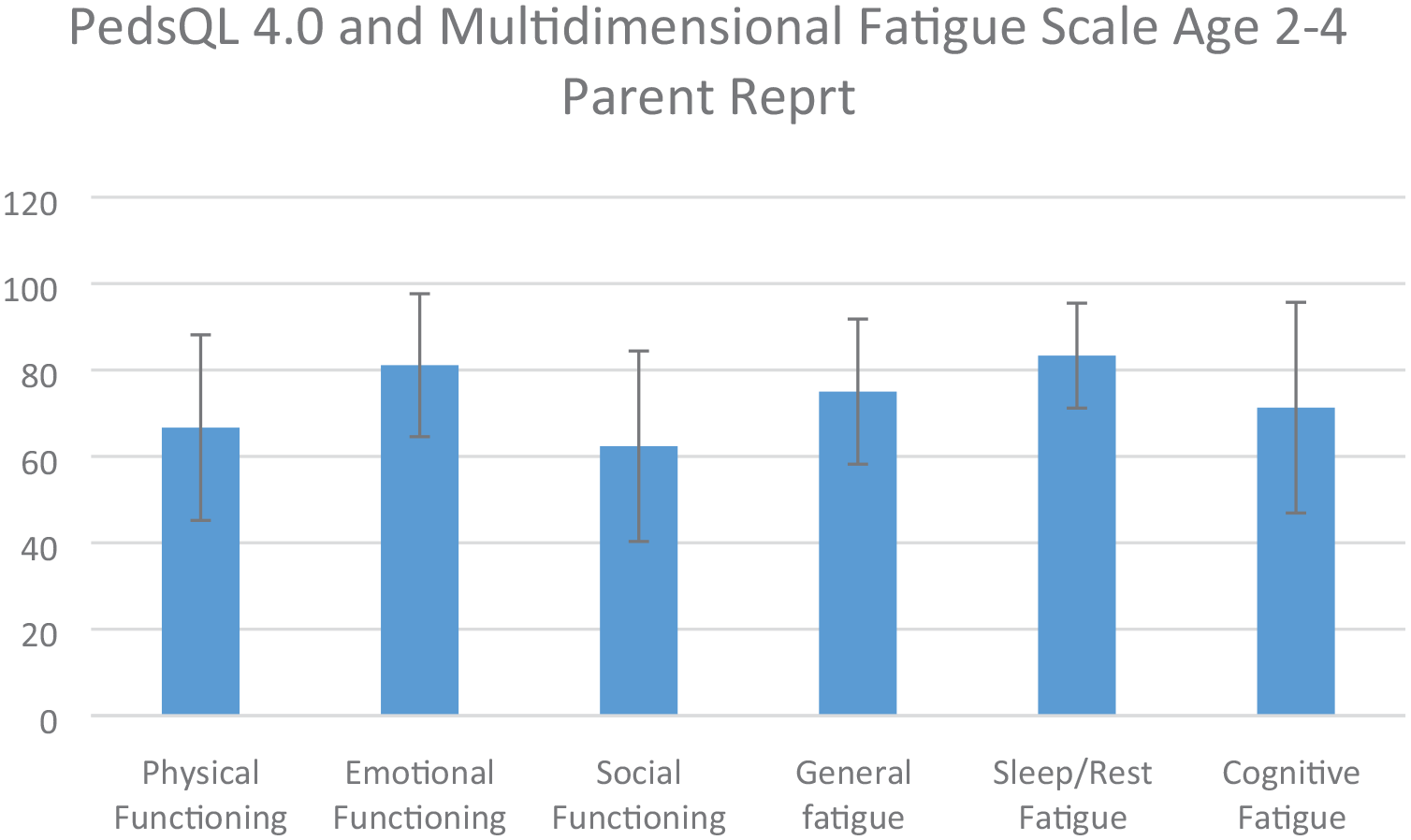
Average scores of individual domains of PedsQL 4.0 Generic Core Scales and Fatigue Module for parent proxy reports for ages 2–4 years for most recent response of each participant (n = 9).
Physiologic data, BTHS-SA, PFSF, EQ-5D, PGIS, CaGIS
Baseline physiologic data were collected from 12 subjects between 19 July 2017 and 26 February 2018 (four of these subjects completed the PedsQL questionnaires discussed previously in our multidisciplinary clinic 2–4 years prior to baseline data collection for the clinical trial). For additional information regarding participant characteristics for these subjects, see Table 5.
Study participant characteristics, baseline data of clinical drug trial for physiologic data and Clinician Global Impression scale.

BPM, beats per minute; CGI, Clinician Global Impression; HR, heart rate; RR, respiratory rate.
There were a total of 4 unique responses to the adolescent BTHS-SA, 8 to the adult BTHS-SA, eight to the pediatric PFSF, 4 to the adult PFSF, four to the pediatric EQ-5D, 8 to the adult EQ-5D, 4 to the pediatric PGIS, 8 to the adult PGIS, 4 to the pediatric CaGIS, 7 to the adult CaGIS, and 12 to the CGIS to establish the pre-intervention baseline. A baseline CaGIS was not available for one adult subject. The aggregated means, SDs, medians, minimums, and maximums for questionnaire responses are summarized in Supplemental Tables 1–11.
Since the number of days each subject was monitored differed, the average value across the monitoring period was calculated for each parameter to allow for additional analysis. The aggregated means and standard deviations for each parameter are summarized in Supplemental Table 12.
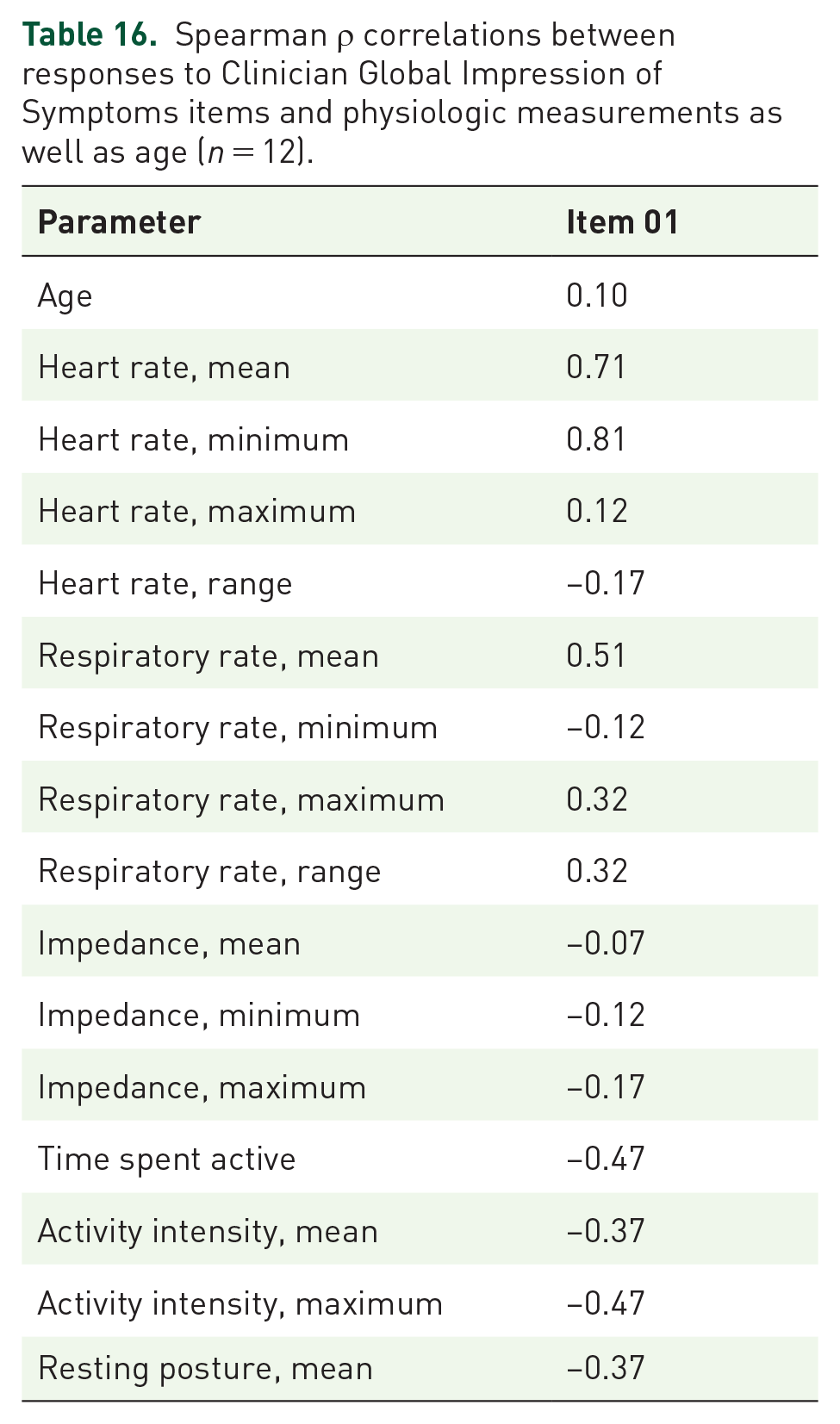
Spearman ρ correlations between questionnaire responses and physiologic measurements as well as age are summarized in Tables 6–16. The CaGIS as a whole had the largest fraction of very strong correlations at 65 out of 170 total. All were positive. Among these, maximum RR, RR range, and maximum activity intensity had the highest number of very strong positive correlations with responses to CaGIS items at 8. Responses to pediatric PGIS Item 3 (tiredness during activities) and pediatric CaGIS Items 5–7 (muscle weakness during activities, muscle pain at rest, muscle pain due to activities) had the highest number of very strong correlations at 11 each. Responses to these individual questionnaire items also had very strong positive correlations with the same physiologic measurements (i.e. mean HR, minimum HR, maximum HR, HR range, maximum RR, RR range, mean impedance, minimum impedance, time spent active, mean activity intensity, maximum activity intensity).
Spearman ρ correlations between responses to pediatric Barth Syndrome Symptom Assessment items and physiologic measurements as well as age for subjects less than 16 years of age (n = 4).

CGIS, Clinician Global Impression of Symptoms.
Spearman ρ correlations between responses to adult Barth Syndrome Symptom Assessment items and physiologic measurements as well as age for subjects 16 years of age or older (n = 8).

CGIS, Clinician Global Impression of Symptoms.
Spearman ρ correlations between responses to pediatric PROMISTM Fatigue Short Form items and physiologic measurements as well as age for subjects less than 18 years of age (n = 8).

CGIS, Clinician Global Impression of Symptoms; PROMIS, Patient-Reported Outcomes Measurement Information System.
Spearman ρ correlations between responses to adult PROMISTM Fatigue Short Form items and physiologic measurements as well as age for subjects 18 years of age or older (n = 4).

CGIS, Clinician Global Impression of Symptoms; PROMIS, Patient-Reported Outcomes Measurement Information System.
Spearman ρ correlations between responses to pediatric EQ-5DTM items and physiologic measurements as well as age for subjects less than 16 years of age (n = 4).
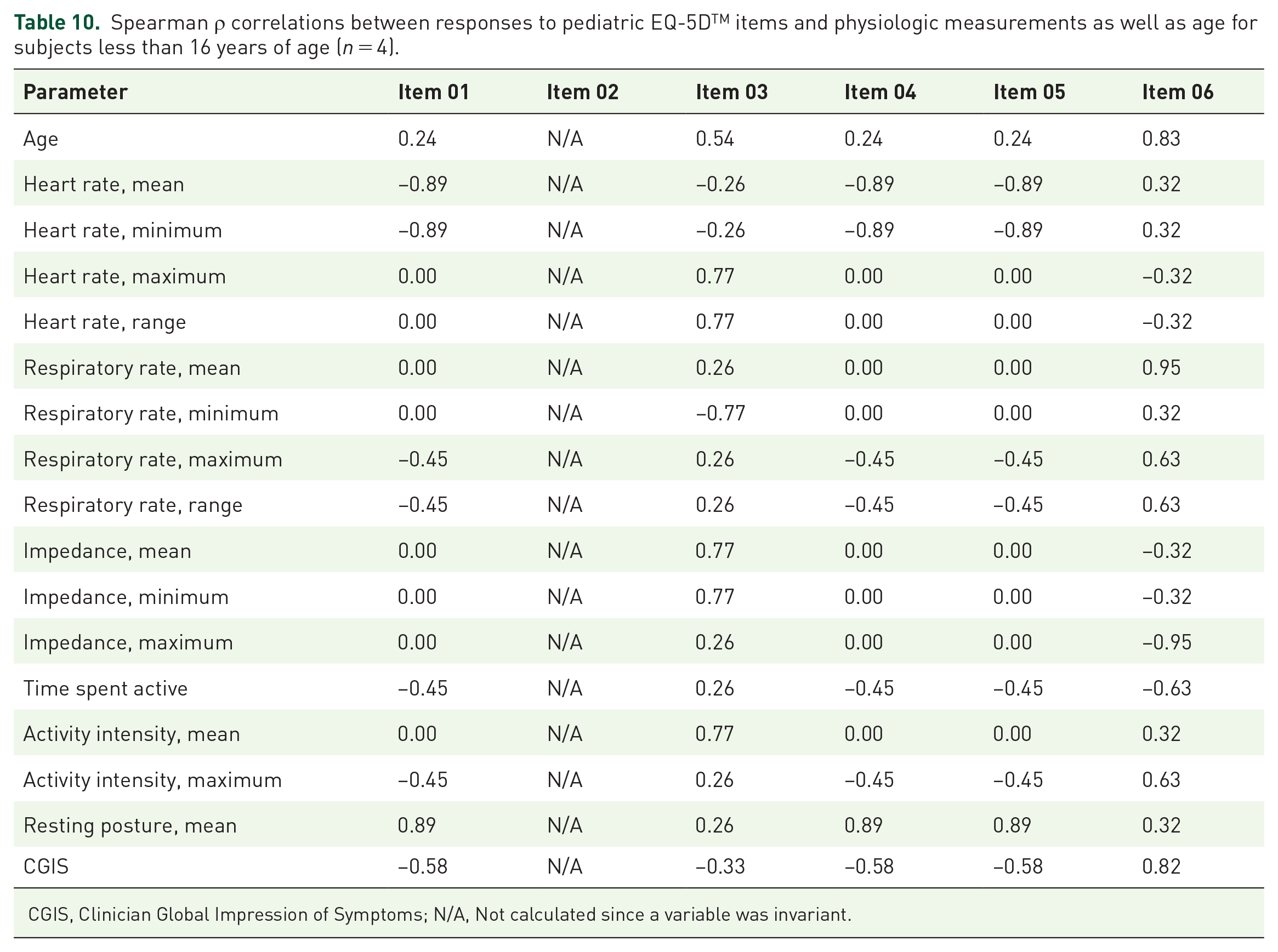
CGIS, Clinician Global Impression of Symptoms; N/A, Not calculated since a variable was invariant.
Spearman ρ correlations between responses to adult EQ-5DTM items and physiologic measurements as well as age for subjects 16 years of age or older (n = 8).

CGIS, Clinician Global Impression of Symptoms; EQ-5D: EuroQol Group.
Spearman ρ correlations between responses to pediatric Patient Global Impression of Symptoms items and physiologic measurements as well as age for subjects less than 16 years of age (n = 4).

CGIS, Clinician Global Impression of Symptoms.
Spearman ρ correlations between responses to adult Patient Global Impression of Symptoms items and physiologic measurements as well as age for subjects 16 years of age or older (n = 8).

CGIS, Clinician Global Impression of Symptoms.
Spearman ρ correlations between responses to pediatric Caregiver Global Impression of Symptoms items and physiologic measurements as well as age for subjects less than 16 years of age (n = 4).

CGIS, Clinician Global Impression of Symptoms; N/A, Not calculated since a variable was invariant.
Spearman ρ correlations between responses to adult Caregiver Global Impression of Symptoms items and physiologic measurements as well as age for subjects 16 years of age or older (n = 7).
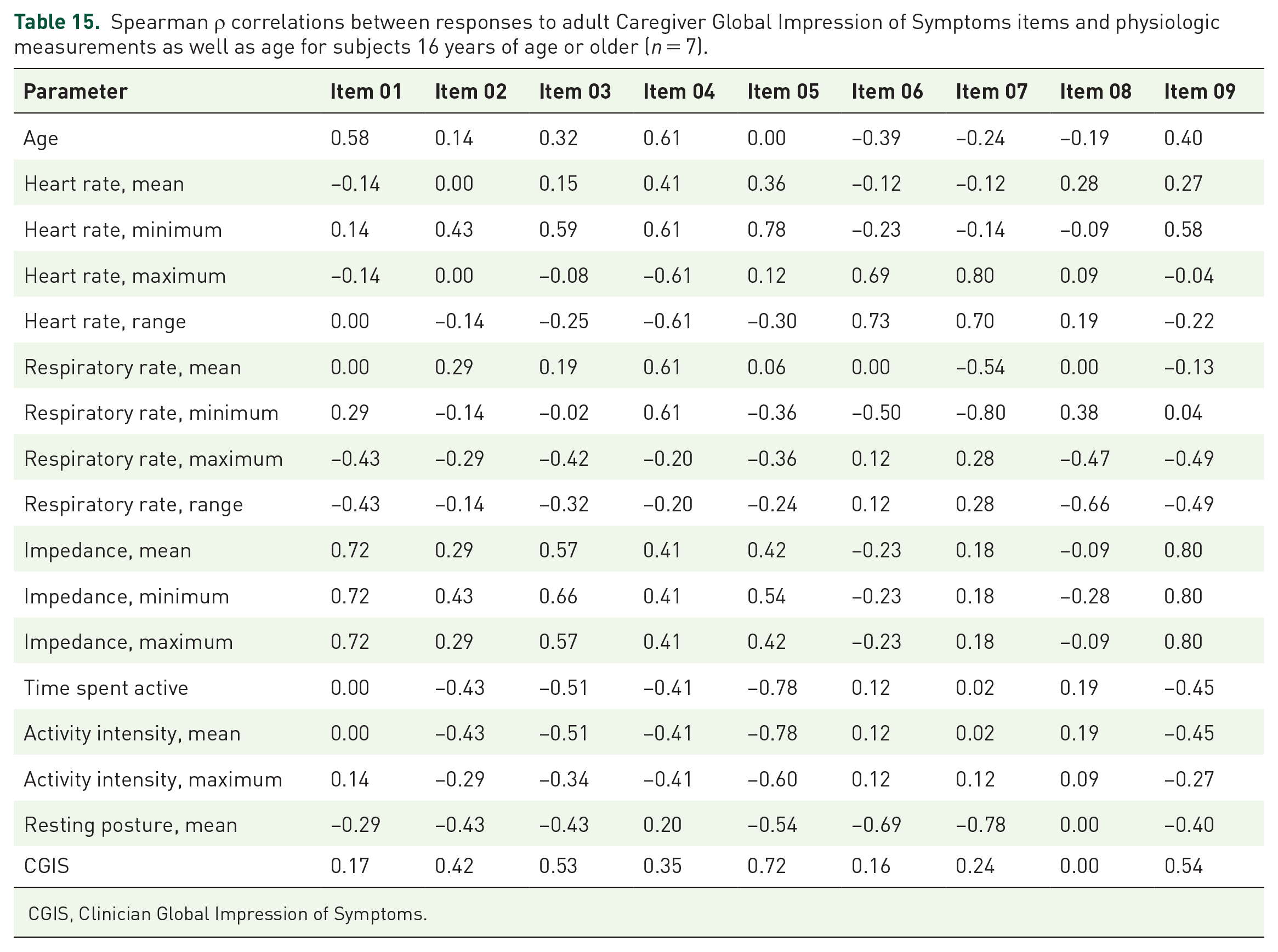
CGIS, Clinician Global Impression of Symptoms.
Spearman ρ correlations between responses to Clinician Global Impression of Symptoms items and physiologic measurements as well as age (n = 12).
Discussion
This study demonstrated that HRQoL is significantly impaired in boys and men with BTHS as perceived by both the subject themselves as well as their caregivers. In addition, this study highlights the impact of fatigue and muscle weakness on HRQoL in BTHS. This study also provides the first characterization of HRQoL in children 2–4 years old with BTHS via parent proxy report.
This study found strong agreement between child and parent proxy report of HRQoL (for children aged 5–18 years) using the PedsQLTM, specifically in the domains of physical functioning and school functioning, consistent with previous studies. 15 As in previous studies, 8 boys with BTHS rated their own psychosocial functioning in a similar manner to parents, which is not typical in families of children with chronic illness as parents typically report more observed problems.18,21,23,35,36 This may be attributable to increased communication between the parent and child given the severity of BTHS. 8 In the cohort of children with BTHS aged 5–18 years, 50% reported significantly impaired psychosocial QoL, significantly more than the 20% of children with cardiovascular disease in the study by Uzark et al. 37 This is most likely secondary to the fact that BTHS is a multisystem disorder and additional systems besides the cardiovascular system, such as the immune system, are largely affected. Boys and men with BTHS aged 5–18 years reported most impairment in HRQoL school functioning followed by physical functioning. This is similar to previous studies of 5- to 16-year-olds with chronic conditions who report the largest decline in school functioning, followed by physical functioning. 20
The results of this quantitative study are in agreement with a recent qualitative study 16 in that parents of boys and men with BTHS are aware of their child’s fatigue and how it impacts daily life. This study also demonstrated that ‘the self-regulation and coping in boys with BTHS was interpersonal and contingent on parental awareness’. 16 Combined, this information suggests that healthcare professionals should ensure that education is provided to both the parent/caregiver as well as the patient with BTHS so that all players are on the same page, for increased cohesiveness of the family unit, while navigating this complex disease course.
Parent report of children aged 2–4 years with BTHS demonstrated significantly impaired physical functioning in 33.3% of respondents and significantly impaired social functioning in 44.4% of respondents, and this is similar to previous studies of younger children with chronic diseases that found large differences between children and their healthy peers in physical function. 38
Our findings that more severe fatigue is significantly correlated with more impaired generic HRQoL are similar to findings of children with other chronic diseases. 23 This study also demonstrated strong to very strong correlations between age and self-reported fatigue on PedsQLTM 4.0 Multidimensional Fatigue Scale, PROMISTM and BTHS-SA. Fatigue/tiredness was the most frequently reported symptom during interviews of children and adults with Barth Syndrome.7,10 Fatigue is an understudied area in the BTHS and requires additional insight given its possible ability to negatively affect aspects of HRQoL.
When exploring the potential relationship between physiology and HRQoL, there were a few emerging trends. Caregiver report of more severe symptoms in pediatric CaGIS as a whole showed the most very strong correlations. The same was true for self-report of more severe symptoms in the pediatric PGIS and in the adult PFSF. Individual questionnaire items from the CaGIS and PGIS for pediatric subjects assessing tiredness, muscle weakness, and muscle pain showed the most very strong correlations. Further study is needed to determine whether these findings are reproducible, and if so to investigate the underlying reasons.
Limitations of the study include our small number of participants, the fact that the data is cross-sectional in nature, and the possible cognitive fatigue and memory issues some boys and men with Barth Syndrome face, which could limit the accuracy of data generated by self-reported instruments with long recall times (e.g. 1 month recall time with the PedsQL). In terms of the parent proxy measures used (such as PedsQL), we do not have information on which caregiver completed the questionnaire. While several studies have looked at the effects of race and socioeconomic status on QoL in chronic disease, 13 we were unable to do that in our study given our small sample size.
To provide additional insight into the causation of the impaired HRQoL noted in our participants, longitudinal HRQoL studies are ongoing in our group.
Conclusion
This study provides the most comprehensive characterization of HRQoL in boys and men with BTHS to date as well as the first analysis of HRQoL in boys and men with BTHS and physiologic data. The data demonstrate significant impairment in many aspects of HRQoL including physical function, emotional function, social function, and fatigue. The data highlight the negative impact of fatigue and muscle weakness on HRQoL in BTHS. It is recommended that HRQoL measures be incorporated into the routine care of patients with BTHS to promote identification and treatment of the impaired domains to improve the QoL of boys and men with BTHS as well as their families and assess the efficacy of potential new treatments.
Supplemental Material
sj-docx-1-trd-10.1177_26330040221093743 – Supplemental material for Quality of life in Barth syndrome
Supplemental material, sj-docx-1-trd-10.1177_26330040221093743 for Quality of life in Barth syndrome by Alexander Y. Kim, Hilary Vernon, Ryan Manuel, Mohammed Almuqbil and Brittany Hornby in Therapeutic Advances in Rare Disease
Supplemental Material
sj-pdf-1-trd-10.1177_26330040221093743 – Supplemental material for Quality of life in Barth syndrome
Supplemental material, sj-pdf-1-trd-10.1177_26330040221093743 for Quality of life in Barth syndrome by Alexander Y. Kim, Hilary Vernon, Ryan Manuel, Mohammed Almuqbil and Brittany Hornby in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors thank all the patients with Barth syndrome and their families who participated in this study. The authors gratefully acknowledge the statistical consultation with Andy Zabel.
Ethics approval and consent to participate
This study was approved by the Johns Hopkins University IRB protocol ‘NA_0008316 Clinical Studies in Barth Syndrome’ and ‘IRB00124162 A Phase 2 Randomized, Double-blind, placebo controlled crossover trial to evaluate the safety, tolerability, and efficacy of subcutaneous injections of Elamipretide (MTP-131) in subjects with genetically confirmed Barth Syndrome’. All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki of 1975, as revised in 2013. Informed written consent was obtained from all patients for being included in the study.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The clinical trial portion of this work was supported by Stealth BioTherapeutics. No funding was provided for the clinical studies in Barth Syndrome study. The authors confirm independence from sponsors; the content of the article has not been influenced by the sponsors.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hilary Vernon, Ryan Manuel, and Brittany Hornby received clinical trial funding from Stealth BioTherapeutics. Hilary Vernon also received research support from Stealth BioTherapeutics.
Data that support the findings of this study are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
