Abstract
Background:
Lysosomal acid lipase deficiency (LAL-D) is a very rare genetic abnormality caused by LIPA gene mutation. The disease has two distinct clinical variants in humans: Wolman disease in infants and cholesteryl ester storage disease in children and adults. Both conditions are characterized by elevated serum transaminases, dyslipidaemia, severe liver steatosis and accelerated fibrosis or cirrhosis, contributing to its high rate of early mortality. Recently sebelipase alfa (recombinant human LAL) was launched to address its underlying pathology. This systematic review evaluates the safety and efficacy of sebelipase alfa for LAL-D.
Methods:
This systematic review was performed following the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Clinical trial records were systematically searched in PubMed/Medline, ClinicalTrials.gov., Cochrane Library and Google Scholar up to September 2020. Records that have reported at least one of the included outcomes were included. Baseline and endpoint mean and standard deviation (SD) for all outcomes were recorded. For safety, frequency and overall distribution of different adverse events were included.
Results:
A total of seven records from five individual studies with 110 LAL-D patients were included into this study. The mean age ranged from 2.57 months in infants to 31.6 years among adults. Serum transaminases (alanine aminotransferase and aspartate aminotransferase), serum lipids (total cholesterol, triglycerides, high-density lipoprotein cholesterol and low-density lipoprotein cholesterol), gamma-glutamyl transferase and liver volume were included as efficacy outcomes. Final pooled results were synthesized as a change from baseline to end of the treatment. A significant effect on both serum transaminases and other serum lipid was achieved (p < 0.01), while non-significant differences were seen for GGT and liver volume as p = 0.35 and p = 0.08 was observed. Mostly the adverse events related to the infusions were infrequent and mild-to-moderate in severity.
Conclusion:
Sebelipase alfa as an enzyme replacement provides an effective, safe and well tolerated treatment in both variants of LAL-D.
Plain language summary
Lysosomal acid lipase deficiency (LAL-D) is a rare, progressive, genetic disorder caused by functional mutations in the LIPA gene, which encodes LAL enzyme. This enzyme maintains lipid homeostasis by hydrolysing the cholesterol esters and triglycerides. Patients with deficient LAL activity are seen with abnormal liver functions which keep them at a high risk of early mortality. Clinical diagnosis of this disease is very challenging due to both its low prevalence and low awareness among patients/clinicians and additionally due to its overlap with other liver/lipid disorders. Also, owing to lack of safe and effective treatment, dietary modifications and some lipid modifying drugs are usually used to control the LAL-D manifestations. Recently, recombinant human LAL named as sebelipase alfa (Kanuma™, Alexion Pharmaceuticals, Inc., New Haven, Connecticut, USA) was approved in 2015 for the European Union and subsequently in the United States as an enzyme replacement therapy for LAL deficiency. The initial clinical trial data indicate that sebelipase alfa produces a significant improvement in all of the wide range of LAL-D manifestations. However, the cumulative evidence is not reported regarding its safety and effective use. Therefore, a systematic literature review of all the clinical trial records by following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines was undertaken. From all of the available clinical trial records, 110 LAL-D patients treated with sebelipase alfa were included. Serum transaminases, serum lipids, gamma-glutamyl transferase (GGT) and liver volume were included as efficacy outcomes. Final pooled results were synthesized as a change from baseline to end of the treatment. A significant effect on both serum transaminases and other serum lipids was achieved (p < 0.01), while non-significant differences were observed for GGT and liver volume, with p = 0.35 and p = 0.08 respectively. Mostly the adverse events related to the infusions were infrequent and mild-to-moderate in severity. The enzyme replacement provides an effective, safe and well tolerated treatment in both variants of LAL-D.
Introduction
Lysosomal acid lipase deficiency (LAL-D) is a rare, progressive, genetic disorder caused by functional mutations in the LIPA gene, which encodes lysosomal acid lipase (LAL) enzyme. 1 The enzyme plays a pivotal role in lipid homeostasis, by hydrolysing the cholesterol esters and triglycerides, reported in both humans and animal models. 2 This autosomal recessive abnormality shows two distinct clinical variants in humans: Wolman disease (WD) in infants and cholesteryl ester storage disease (CESD) in children and adults. 3 WD shows a more serious profile and more rapid progression than CESD and usually the patient dies within 6 months of age. Both conditions are characterized by elevated serum transaminases, hepatic lipid accumulation, dyslipidaemia, a severe liver steatosis and accelerated fibrosis or cirrhosis, contributing to its high rate of early mortality. 4 The most commonly found mutation is seen with the E8SJM variant of LIPA-gene, reported in 50–70% of LAL-D cases. 5 Clinical diagnosis of this disease is very challenging owing to both its low prevalence and low awareness among patients/clinicians and additionally to its overlap with other liver/lipid disorders. 6 Measurement of LAL enzyme activity in dried blood spots, leukocytes or fibroblasts is used to confirm the diagnosis of this metabolic disease.7,8
Due to lack of safe and effective treatment which can directly address the underlying pathology, dietary modifications and some lipid modifying drugs are usually used to control the LAL-D manifestations. The agents, including statins alone or in combination with other hypolipidaemics, are used to improve the lipid profile by reducing serum cholesterol and triglyceride (TG) concentrations. However, they cannot prevent liver injury or disease progression. 9 At the advanced stages, the only option left is liver transplantation. The decision at this point relies upon a number of associated complications. 10 Among the different therapeutic approaches, enzyme replacement therapy (ERT) as a treatment option is recommended for several conditions where natural enzyme is either deficient or completely absent. For a number of lysosomal storage disorders, such as Gaucher disease, Fabry disease, Pompe disease and mucopolysaccharidoses, ERT has been developed. 11 On the same lines, recombinant human LAL named as sebelipase alfa (Kanuma™, Alexion Pharmaceuticals, Inc., New Haven, Connecticut, USA) was approved in 2015 for the European Union and subsequently in the United States as an ERT for LAL deficiency. 12 Sebelipase alfa is produced by utilizing a technique which allows a targeted expression of a gene sequence in hen oviduct cells. 13 Those expressed sequences then encode for the amino acid sequences which are the same as the native human LAL enzyme.
The individual clinical trial reports indicate that in both clinical classes, that is, WD and CESD, sebelipase alfa produces a significant improvement in all of the wide range of LAL-D manifestations. However, the cumulative evidence is not reported regarding its safety and effective use. Therefore, a systematic literature review of all of the clinical trial records to evaluate its safety and efficacy profile was undertaken. Owing to the disease rarity and lack of enough observational data, results from all clinical trials with their dose escalation or extension studies to provide a better understanding of available evidence have been included.
Materials and methods
The systematic review is registered at PROSPERO with registration number CRD42020218022 and was performed following the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Literature search strategy
Four electronic English databases (PubMed/Medline, ClinicalTrials.gov., Cochrane Library and Google Scholar) were searched to identify clinical trial records published up to the end of September 2020. Further, the potentially relevant back references from the searched studies were included. A search strategy using a combination of the following relevant medical subject heading terms and key words was applied: “Cholesterol Ester Storage Disease”[MeSH] OR “Wolman Disease”[MeSH] OR “Lysosomal acid lipase deficiency”[tiab] OR “Lysosomal Storage Diseases”[tiab] OR “LAL-D”[tiab] OR “CESD”[tiab] OR “Lysosomal Storage Diseases”[tiab] OR “LIPA deficiency”[tiab] OR “LAL Deficiency”[tiab] AND “Sebelipase alfa”[tiab] OR “Kanuma”[tiab] OR “recombinant LAL”[tiab] OR “Recombinant Proteins”[MeSH] OR “Sterol Esterase”[MeSH] OR “Enzyme Replacement Therapy”[tw]. The search results were updated before the final analysis.
Inclusion and exclusion criteria
Two reviewers (AB and PT) screened and identified studies according to predefined inclusion and exclusion criteria. The studies having patients with LAL-D of any age or gender were eligible. Clinical trial records that have reported at least one of the included outcomes were included. The studies that investigated safety and efficacy of sebelipase alfa as treatment of LAL-D at any dose were reviewed. The studies were excluded if they had included patients with clinically significant concurrent disease, abnormal values on laboratory screening tests other than liver function or lipid panel and had any other liver related complication. All observational studies, case reports, case series, conference abstracts, letters, historical articles, editorials, review articles or non-English language articles were excluded.
Study selection
Both authors (AB and PT) independently screened the titles and abstracts after removal of duplicate and irrelevant studies. Randomized controlled trials (RCTs), open label dose escalation (OLDE) studies of RCTs and open label extension (OLE) studies of RCTs were included. After checking eligibility and relevance against inclusion criteria, the reviewers evaluated the full text of eligible articles. In the case of multiple publications of the same study, all publications with dose escalation or study extension were considered owing to the very small prevalence of this orphan disease condition. A third independent reviewer (AD) was consulted to resolve any discrepancies between the reviewers. The literature selection process is as shown in Figure 1.

Flowchart of study selection process.
Outcomes
Change in liver function indicators, such as alanine aminotransferase (ALT) and aspartate aminotransferase levels (AST) normalization, was used as the primary outcome for efficacy assessment. Additionally levels of other biochemical markers of liver function, serum lipid levels [total cholesterol, triglyceride, low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol levels (HDL-C)], gamma-glutamyl transferase (GGT) and liver volume measurement were also considered. Baseline and endpoint mean and standard deviation (SD) for all outcomes were recorded. For safety, frequency and overall distribution of different adverse events were included.
Data extraction and quality assessment
Data extraction was independently performed by both reviewers with the help of an electronic extraction sheet using Microsoft Excel. Discrepancies were resolved by group discussion or by consulting the third reviewer. The following data were extracted: publication characteristics (author, year, country and journal), population characteristics (gender, age), methodological quality (sample size, study design), treatment dosage and duration, efficacy and safety outcomes. Supplementary attachments were searched when additional information was needed. Also, corresponding authors and trial sponsor were contacted when required. The included studies were assessed for risk of bias by using the Cochrane Risk of Bias tool, which address all the key aspects, such as selection bias, performance bias, detection bias, attrition bias and reporting bias. 14
Data synthesis and analysis
This systematic review presents a comprehensive qualitative synthesis of results from the included studies. During the data extraction, the effect size for all included outcomes from each study was presented as means and SDs. Mean differences with 95% confidence interval (MDs, 95% CI) as final results after analysing baseline and after the intervention results were reported. The MDs, 95% CI estimation and risk of bias assessment was performed by using Review Manager version 5.3.5 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014), while adverse event proportions presented through forest plots were synthesized by using R software.
Results
Study selection
The initial systematic search identified 664 records from all of the data sources. After removal of duplicate records, 514 records were screened for the titles and abstracts. Of these, 454 were not relevant. Among the remaining 60 records which were screened for eligibility in full-text version, 53 were excluded based on inclusion and exclusion criteria. The excluded records were review articles (n = 15), case series/reports (n = 13), abstracts (n = 24), editorials (n = 1). Seven records from five individual studies (RCTs=1, OLDEs=4, OLEs=2) qualified for systematic review and were included for qualitative synthesis.15–21 The PRISMA flow diagram of the detailed literature selection process is illustrated in Figure 1.
Study characterization
This systematic review comprises a total of seven prospective reports from five international multicentre trials. The data of 110 patients diagnosed with LAL-D, including patients from two extension results of a primary study by Balwani et al., 21 were included. Studies with both phenotypes of LAL-D, that is, WD and CESD patients, were considered. The mean age varied between 2.57 months in WD to 31.6 years among CESD. Only one study, by Burton et al., 19 has a double blind, placebo-control design while all others were with open label study design. In all of the included studies, patients received sebelipase alfa as treatment, starting with a dose of 0.35 mg/kg-q.w., to an escalation up to 7.0 mg/kg-q.w. The study characteristics of the included studies are presented in Table 1.
Table of basic characteristics of included studies.
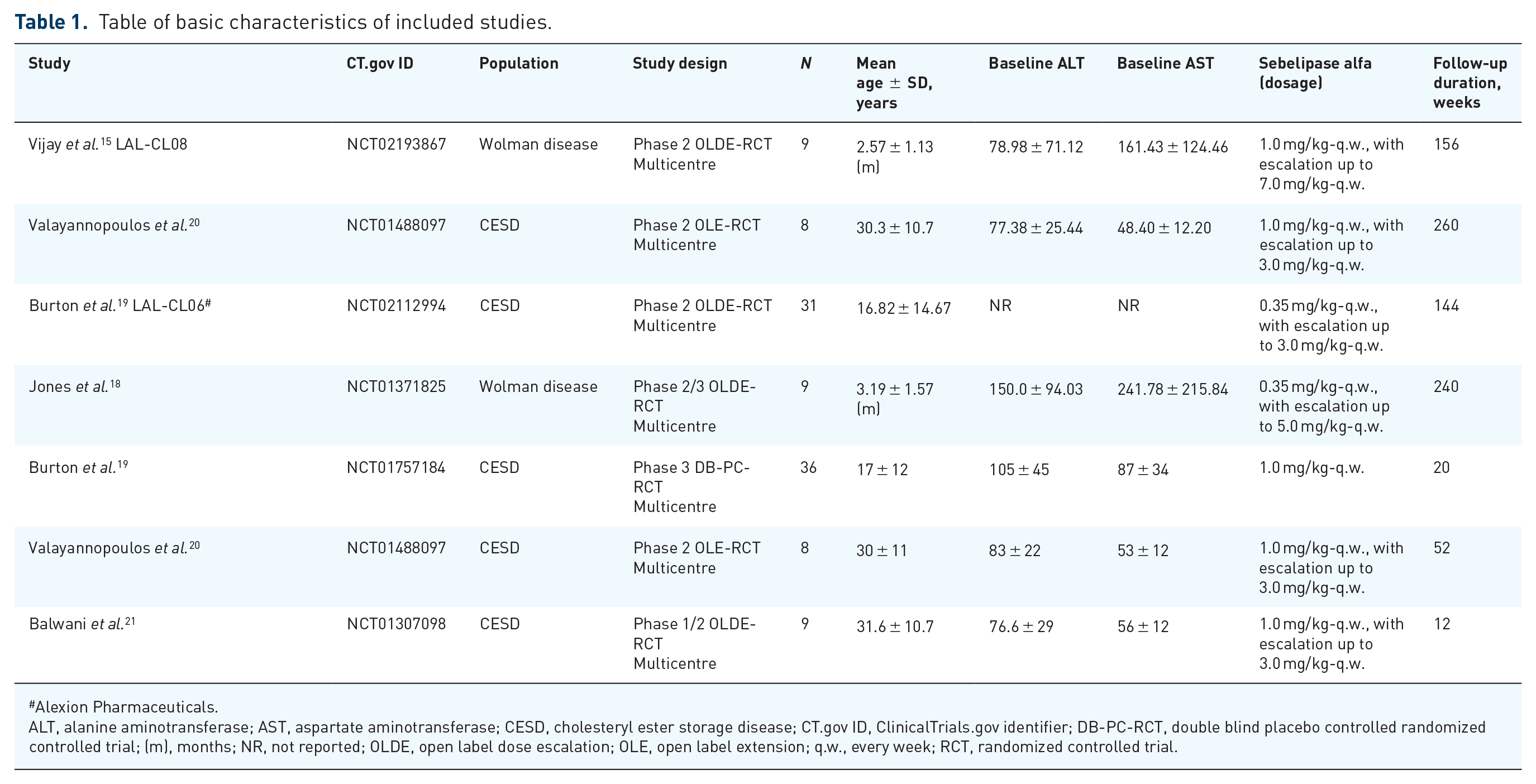
Alexion Pharmaceuticals.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CESD, cholesteryl ester storage disease; CT.gov ID, ClinicalTrials.gov identifier; DB-PC-RCT, double blind placebo controlled randomized controlled trial; (m), months; NR, not reported; OLDE, open label dose escalation; OLE, open label extension; q.w., every week; RCT, randomized controlled trial.
Risk of bias assessment
We used the Cochrane collaboration tool to identify the risk of bias, as shown in Figures 2 and 3. The risk of bias of all included open label studies was found to be high for almost all measures due to the low sample size and study design. However, the study by Burton et al. 19 with a double blind, placebo-control design showed a low risk of bias. A low risk of bias, in this work, was defined as the study fulfilling five to seven, a moderate risk of bias as three to four and a high risk of bias as the study fulfilling zero to two of the seven validity criteria.

Risk of bias graph.

Risk of bias summary.
Efficacy outcomes of sebelipase alfa
Different outcomes were measured to determine efficacy as change in serum transaminases, serum lipids and other parameters, such as GGT or liver volume. The pooled MDs (95% CI) for the efficacy outcomes are presented in Table 2. Statistically significant summary results point out a significant favourable change of measured outcomes after sebelipase alfa intervention.
Overall impact of sebelipase alfa intervention on included efficacy outcomes.

Alexion Pharmaceuticals.
Represents p < 0.05 showing statistically significant difference exist between baseline and after intervention.
Median (range) percentage change from baseline.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; GGT, gamma-glutamyltransferase; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; MD, mean difference; NR, not reported; TC, total cholesterol; TG, triglycerides.
Change in serum transaminases
ALT and AST levels show an elevation in patients with LAL-D, and their reduction is considered as a useful indicator for diagnosis and treatment response. 22 Keeping in view the upper limit of normal ALT as 67 U/l and AST as 50 U/l, all of the study patients had elevated serum transaminases. Sebelipase alfa as treatment showed a significantly higher rate of normalization for both the transaminases. In this analysis, six of the studies that reported ALT and AST as an outcome have shown a significant reduction after sebelipase alfa intervention. The pooled MDs (95% CIs) for ALT and AST were 45.94 (27.77–64.12, p < 0.01) and 31.92 (17.29–46.56, p < 0.01), respectively. For both ALT and AST, a significant minimum reduction in means was reported by Malinová et al., 16 as 24.35 (1.07–47.63, p = 0.04) and 13.92 (3.25–24.59, p = 0.01); however, a maximum reduction was seen in the study by Jones et al. 18 as 122.63 (60.63–184.63, p < 0.01) and 195.53 (54.41-336.65, p < 0.01) respectively.
Change in serum lipids
Disturbed lipid profile is also an important indication associated with deficiency of LAL enzyme activity. Total cholesterol, triglycerides and LDL-C show an abnormal elevation from the normal values, while a significant decline is seen in the levels of HDL-C. Sebelipase alfa treatment in all the studies has shown a favourable impact on lipid profile from baseline to the end of study. Statistically significant reduction in overall pooled MD (95% CI) was achieved in TC, TG and LDL-C as 78.00 (29.17–126.83) (p < 0.01), 51.01 (15.83–86.19) (p < 0.01) and 46.50 (18.73–74.27) (p < 0.01); however, a significant improvement was also found in HDL-C as –7.77 (–14.03 to –1.52, p = 0.01). Among the included studies, Vijay et al. 15 have reported the highest achieved MD (95% CI) for TC as 187.16 (–21.68 to 396.00) (p = 0.08) while for TG and LDL-C, Valayannopoulos et al. 20 have reported highest reduction as 102.00 (44.73–159.27) (p < 0.01) and 115.00 (64.39–165.61) (p < 0.01) respectively. Improvement in HDL-C after treatment was maximum seen in the study by Burton et al., 19 showing MD (95% CI) of –19.60 (–27.74 to –11.46) (p < 0.01). From the results of clinical trial LAL-CL06 (2019), we also found a significant percentage change in these parameters. 17
Change in other efficacy measures
The other efficacy outcomes measured were GGT and liver volume. Three of the included studies have reported GGT measurements with a marked decrease with sebelipase alfa intervention but the summary results were not statistically significant (p = 0.35).16,18,20 Jones et al. 18 have reported a higher reduction among other studies as 168.08 (–50.79 to 386.95) (p = 0.13). Liver volume also acts as an important predictor of diagnosis and treatment response in any fatty liver disease. In our analysis for liver volume change, the overall pooled reduction was not statistically significant; however, all three individual studies have reported a significant decline with the sebelipase intervention.15,16,18 Here too, Jones et al. 18 reported a higher reduction of 1.82 (0.68–2.96) (p < 0.01) in liver volume.
Safety of sebelipase alfa treatment
Overall, sebelipase alfa was well tolerated and reactions seen were non-serious, mild or moderate in severity. None of the patients in any trial discontinued treatment because of these events.
Among all the reports, Vijay et al. 15 reported the highest related or possibly related treatment emergent adverse events and infusion associated reactions (IARs) as risk ratio (RR)=0.89 (95% CI: 0.52–1.00) of each type. Maximum treatment-related or possibly related serious adverse events were reported by Jones et al. 18 [RR = 0.78 (95% CI: 0.40–0.97)]. Burton et al., 19 having the largest sample size of 36 patients among the included studies, have reported minimum experienced adverse drug reactions (ADRs) of any type in their patients. All of the treatment related events experienced by patients were addressed by dose modification to continue the ERT. Overall summarized proportionate pooled results of different safety outcomes are shown in Table 3 and Figure 4.
Overall distribution of safety outcomes reported from the included studies.

Alexion Pharmaceuticals.
IAR, infusion associated reaction.

Forest plots of safety outcomes. (a) Any adverse events experienced during the study. (b) Treatment-emergent adverse event experienced during the study. (c) Serious adverse event experienced during the study. (d) Infusion-associated reaction experienced during the study. (E) Dose modifications needed due to treatment-emergent adverse event during the study.
Discussion
LAL-D as an under-recognized liver disease is associated with a high rate of morbidity and mortality due to its nature of rapid progression. 23 Until the disease is diagnosed, patients are seen with frequent serious liver complications and other associated cardiovascular outcomes. Earlier management practices with statins and other lipid-lowering medications are not found efficient enough to restrict continued progression of liver damage. 13 Meanwhile, recently launched ERT with recombinant human sebelipase alfa is believed to address this progressive disorder.
In this systematic review, we report the efficacy and safety of all the clinical trials of sebelipase alfa, the only US Food and Drug Administration approved treatment for LAL-D, considering the dosing schedules and durations.
The enzyme LAL is important in mediating the normal lipid metabolism by hydrolysing the cholesterol esters and triglycerides and any reduction in its activity promotes increased cholesterol ester storage in lysosomes and alters the lipid haemostasis. 2 To maintain normal lipid metabolism, ERT with recombinant human LAL is the only available option to replace the deficient enzyme. To the best of the knowledge available, this work is the first analysis carried out to present cumulative clinical efficacy and safety of sebelipase alfa therapy.
As an enzyme replacement, sebelipase alfa was clinically found effective as reflected in the outcomes; overall improvement in serum transaminases, lipids and other related markers, with an acceptable safety profile. In the long run, these improvements are indirectly helpful in improving the physical and functional well-being of patients and ultimately reducing the overall morbidity and mortality. In the pooled results from all of the included studies, we found a statistically significant effect on both serum transaminases and lipids as (p < 0.01) and a statistically non-significant reduction for GGT and liver volume (p = 0.35 and p = 0.08; Table 2). GGT and liver volume were not reported by all the studies as their outcome, which might impact the actual effectiveness of the treatment reported through these outcomes.
In the very first human study reported by Balwani et al., 21 nine CESD patients divided into three dose cohorts received four once-weekly infusions of sebelipase with a dose of 0.35, 1, or 3 mg/kg 2 respectively. Both of the transaminases were normalized within 28 days of treatment with a significant pooled decrease of 41% for ALT and 32% for AST without any finite differences between the three cohorts. However, after treatment discontinuation, the level reversed within three weeks and subsequently treatment was restarted. After reinitiating the treatment to every other week dosing, again a significant overall decrease of 52% and 36% for ALT and AST at week 12 was achieved. The same patients were further enrolled into two extensions of the same study, whose reports were presented at week 52 and 260.20,16 The patients sustained the reduction in parameters as the observed MD (95% CI) from baseline to trial end was found to be 24.35 (1.07–47.63) (p = 0.04) for ALT and 13.92 (3.25–24.59) (p = 0.01) for AST. In this long-term trial, a significant improvement in lipid profile and other liver markers such as in GGT and liver volume had been documented.
These data were further supported with the ARISE study by Burton et al. 19 in their double blinded placebo controlled trial in CESD patients. They reported a MD of 58.00 (41.18–74.82) (p < 0.01) for ALT and 42.00 (29.11–54.89) (p < 0.01) for AST from baseline to treatment end at week 20 after 1.0 mg/kg-q.w. intervention. This study also found a significant reduction in TG 25.50 (5.94–45.06) (p = 0.01) and LDL-C 28.40 (6.51–50.29) (p = 0.01); however, an improvement in HDL-C as –19.60 (–27.74 to –11.46) (p < 0.01) was seen. In addition to the above discussed efficacy with sebelipase alfa, it is important to note that the patients were on adjuvant lipid lowering agents in both studies. The measured outcomes need to be seen in light of the confounders. And, these results need to be evaluated further in future research by exploring the efficacy of sebelipase alfa with and without adjuvant therapies.
Similarly, two of the included studies15,18 have reported the safety and efficacy data of sebelipase alfa in the infants having WD for longer treatment duration of 240 and 156 weeks. Both studies were complementary to earlier reports that have shown a significant reduction in the liver enzymes in enrolled CESD adult patients. The first study had reported a significant mean decrease of 122.63 (60.63–184.63) for ALT and 195.53 (54.41–336.65) for AST while the later study reported mean decrease of 30.38 (–25.59–86.35) and 101.09 (15.49–186.69), respectively. With improvement in serum lipids, both of the studies have also reported a significant reduction in liver volume as 1.82 (0.68–2.96) (p < 0.01) and 1.04 (0.14–1.94) (p = 0.02) independently.
In this review, we also included results of an unpublished trial of 31 CESD patients registered at ClinicalTrials.gov (NCT number NCT02112994). 17 The trial results have shown a significant overall percentage change in TG, LDL-C and HDL-C as 28%, 29% and 35%, respectively, after sebelipase intervention. In addition to the presented data, a number of studies are in progress to evaluate the clinical relevance of this ERT in the real clinical settings. The coming data will further extend the literature and better understanding of clinical application of this therapy.
Among the two age based clinical groups, WD starts during the sixth or seventh month of life in infants with almost nil LAL activity. The diagnosis of the disease is very challenging and shows diverse manifestations such as delayed growth, abnormal absorption, inflammation of liver and spleen, severe liver dysfunction, severe anaemia and multi-organ failure. Consequently, most of the infants die in their first year of life. However, CESD as a late-onset phenotype is seen with childhood, adolescence or adulthood and is diagnosed incidentally. The disease is characterized by dyslipidaemia, hepatic steatosis, hepatomegaly and raised transaminases. Both of these conditions are seen to disturb normal fat metabolism to cause multisystem abnormalities with fatal outcomes. 3 Therefore, it is necessary to have a standard, safe and effective treatment to prevent this rare category of patients.
On comparing the impact of therapy between WD and CESD for longer treatment durations, it is found that the WD patients are more responsive than CESD, as seen in outcomes presented in Table 2. Among Jones et al. 18 and Malinová et al., 16 who reported results at week 240 and 260 for WD and CESD respectively, the former study has presented higher mean differences in outcomes than the latter one. The results were further supported by Vijay et al., 15 who have also found higher mean differences in WD patients. However, it is of note that the dose escalation was from 0.35 mg/kg-q.w. to 5.0 mg/kg-q.w. (Jones et al. 18 ) and up to 7.0 mg/kg-q.w. (Vijay et al. 15 ) in WD while in CESD it was from 1.0 mg/kg-q.w. to 3.0 mg/kg-q.w. (Malinova et al. 16 ). Therefore, it is important to evaluate whether the differences in the treatment efficacy are due to differences in dosage or they exist because of phenotypic variation among the two.
In general, sebelipase alfa treatment evaluated up to 5 years was well tolerated with a consistent improvement in outcomes. 16 Mostly the adverse events (AEs) related to the infusions were infrequent and mild-to-moderate in severity. Mainly the patients who received ERT developed AEs as gastrointestinal disorders (abdominal pain, nausea, diarrhoea), pyrexia, headache, body pain, upper respiratory tract infection and nasopharyngitis (Table 3). However, after experiencing some issues with treatment, none of the patients in any study have died or discontinued the treatment. All the events were successfully managed by modifications in dosage of infusions or their rates. Symptomatic treatments were given to control other related complications to assure patient safety.
To summarize, our findings suggest that the treatment replaces the missing LAL enzyme activity in patients with LAL-D and simultaneously indicates a great need for long-term studies in both controlled and real life manner as the initial reports are subject to many drawbacks associated with study designs and reporting of primary results. Long-term, well designed evaluations are needed to study different genotypic and phenotypic factors such as finding gene mutations, biomarkers of liver fibrosis/cirrhosis, cardiovascular assessments, survival rate, et cetera which can predict the actual therapeutic response with this therapy in LAL-D.
Limitations
Our study provides the first insight into cumulative results as a systematic review over the safety and efficacy of sebelipase alfa in LAL-D patients. Despite that, the study is subject to several limitations. First, we have pooled results of different study designs including open label trials and both of the clinical variants of LAL-D together. Due to heterogeneity among the studies and a high risk of bias, the statistical analysis was not feasible. Second, given the small sample size in individual studies and the small number of published studies available for investigation, the results in the current work may be underpowered to give a general recommendation regarding clinical outcomes and AEs with this treatment. Third, we have included a limited number of efficacy outcomes among several others, such as survival, total bilirubin, hepatic fat content, serum ferritin et cetera, associated with LAL-D, which also need to be evaluated. Last, we have estimated the MDs with CIs for different included outcomes after converting median (range) into mean (SD) with appropriate methods24,25 from original measurements presented by primary studies, which might give some variation in pooled results.
Conclusion
Sebelipase alfa provides an effective, safe and well tolerated treatment by improving the clinical outcomes and attenuating the disease progression in both of the variants of LAL-D. The ERT should be exclusively evaluated for its potential benefits and risks to consider it as the standard of care for this genetic disorder. Future research through large clinical trials and evidence from real world studies are needed to explore its benefits when no other standard treatment is available.
Future perspectives
In the current review, the crude data investigated were not uniform. Neither dosage nor the course of treatment was standardized. Large trials and well-designed observational studies are needed to better understand the clinical relevance of this therapy by defining its dosing schedule, dose-related effects and treatment duration. Furthermore, the ERT needs to be evaluated with and without adjuvant therapies such as dietary therapy and lipid lowering agents to get an actual understanding of treatment benefits and risks. Since the initial reports indicate that this enzyme LAL is found deficient in non-alcoholic fatty liver disease (NAFLD) patients, it shall be meaningful to assess NAFLD patients vis-à-vis LAL activity as a matter of routine investigation. If a relationship evolves, the ERT needs to be considered for NAFLD patients as well. Also, as the disorder is seen worldwide, the global approval process should be worked out with great consideration given to its costing as the cost of the treatment is the biggest hurdle to its adherence.
Footnotes
Author contributions
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics statement
Ethical approval and informed consent were not required for this review.
