Abstract
Introduction
Individuals with chronic cardiac conditions are at increased risk for developing pneumococcal diseases. In this study, we aim to develop an evidence-based consensus document on the use of pneumococcal vaccines in individuals with cardiac conditions.
Methods
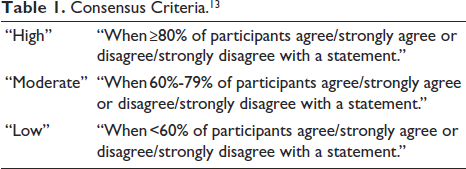
Modified Delphi consensus methodology was used to establish consensus. A committee of seven cardiologists from India formed the panel. Questions were drafted across three domains: (a) the risk of pneumococcal disease in patients with cardiac conditions, (b) evidence and optimal timing of pneumococcal vaccination, and (c) barriers to vaccination in cardiology settings. The consensus level was categorized into three groups: (a) low (<60%), (b) moderate (60%-79%), and (c) high (≥80%).
Results
Experts agreed that immunocompetent individuals with cardiac conditions, adults with a medical history of myocardial infarction/coronary artery disease, heart failure, individuals who fall under the coronary artery disease spectrum, older adults (>50 years of age) with chronic hypertension, and adults with chronic hypertension or a history of acute coronary syndrome/myocardial infarction, with multiple comorbidities, can benefit from pneumococcal vaccination.
Conclusion
This document will guide clinicians on the optimal timing for pneumococcal vaccination in patients with cardiac conditions and highlight specific patient subgroups that can benefit from it.
Introduction
Pneumococcal diseases (PDs) caused by Streptococcus pneumoniae significantly contribute to morbidity and mortality, especially among individuals with chronic underlying cardiac conditions.1–3 Clinically, PDs manifest across a broad spectrum, ranging from noninvasive infections (otitis media and sinusitis) to invasive forms (pneumonia with bacteremia and meningitis).1, 4–6 S. pneumoniae is a common cause of community-acquired bacterial pneumonia in adults.1, 3, 7 Cardiovascular (CV) events, such as myocardial infarction (MI), heart failure (HF), or arrhythmias, have been reported in patients with community-acquired pneumonia (CAP) due to S. pneumoniae. 8 Furthermore, patients hospitalized with invasive pneumococcal disease (IPD) are at an increased risk of major adverse CV events, including new/worsening HF, arrhythmia, and MI. 9 Individuals with preexisting cardiac conditions are at increased risk of developing these cardiac complications after pneumonia (all-cause CAP), which are associated with both short-term (e.g., within the first 24 h or 30 days) and long-term mortality risks (after hospital discharge extending over 10 years). 10 As a preventive measure, pneumococcal vaccination can be crucial in mitigating the burden of PD in patients with underlying cardiac conditions. Pneumococcal vaccination is associated with a reduced risk of all-cause mortality among adults with cardiovascular disease (CVD) or very high CV risk (N = 5 studies; 163,756 adult participants) versus no vaccination. 11 Herein, we aimed to review (a) the risks and consequences of CAP and IPD in patients with cardiac conditions; (b) the efficacy of pneumococcal vaccines (PVs) in patients with cardiac conditions; (c) recommendations for the administration of PVs in patients with cardiac conditions and risk factors; and (d) barriers to the administration of PVs in cardiology settings. We summarize expert opinions and practical consensus recommendations on vaccine sequencing, highlight patient subgroups with cardiac conditions that can benefit from PVs, and discuss the optimal timing of pneumococcal vaccination in cardiac conditions.
Methodology
A panel of seven cardiologists with significant experience in the field (Figure 1) participated in developing the consensus manuscript. A chair/moderator led and facilitated the consensus process. An evidence review was conducted using the PubMed database to identify relevant articles (published from January 2000 to June 2024). Keywords such as “risk,” “barriers,” “heart disease,” “chronic,” “cardiac,” “pneumococcal infections,” “pneumococcal disease,” “community-acquired pneumonia,” “pneumococcal pneumonia,” “invasive pneumococcal disease,” “pneumococcal vaccine,” “efficacy,” “effectiveness,” “safety,” “pediatric,” “adult,” “immunization,” “prevention,” and “guidelines” were used. Original research articles, review articles, systematic literature reviews, meta-analyses, surveys, guidelines, and consensus recommendations were included. In total, 26 clinically relevant statements and questions (statements: n = 19; other survey-type questions: n = 7) were drafted and finalized after discussions with the chair. An electronic survey link to these questions was sent to all the participants to gather their responses.
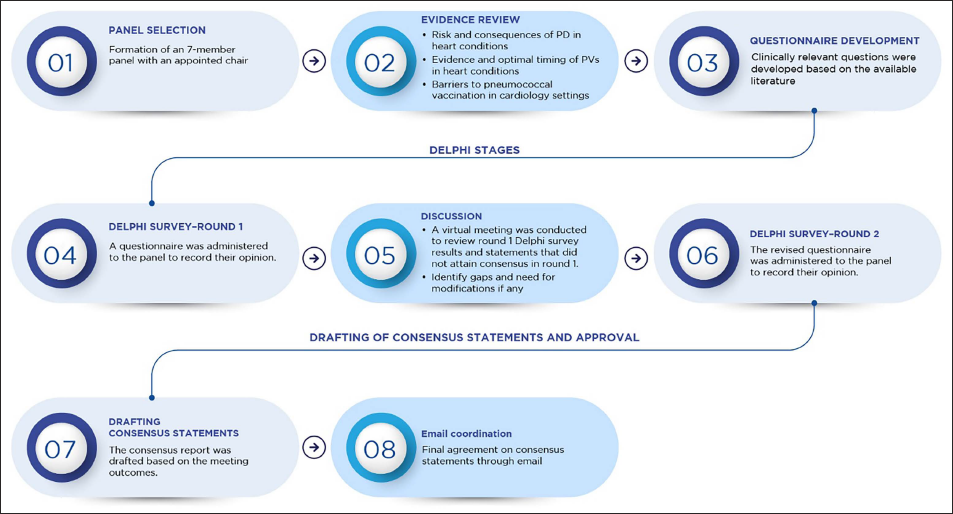
Expert consensus was achieved through the modified Delphi process. 12 The consensus level (Table 1) was categorized into three groups: (a) low (<60%), (b) moderate (60%-79%), and (c) high (≥80%). 13 The process involved two rounds of Delphi surveys and one round of discussion. A virtual meeting was conducted to review the Delphi round one survey results and to discuss the questions and statements receiving near or no consensus. The statements that did not attain consensus were modified and presented for voting in the second round. Experts arrived at the modifications based on the current scientific evidence, guideline recommendations, and real-world practice. In November 2024, another round of basic literature search was performed in PubMed to check for any updates. Consensus statements were formulated based on the opinions and agreements of the majority. The consensus report was shared with the expert panel for approval.
Consensus Criteria. 13
Results
In round one, a consensus was reached for 10 out of 19 statements (52.63%). Two statements that did not achieve consensus were excluded, whereas the remaining seven statements were modified and presented for voting in the second round. The seven statements were finalized after the Delphi survey round two. In this article, we first review the evidence for each study objective, followed by expert opinions and consensus recommendations.
Risk and Consequences of Community-acquired Pneumonia and Invasive Pneumococcal Disease in Patients with Cardiac Conditions
Community-acquired pneumonia (majorly caused by S. pneumoniae and influenza virus) poses a significant health risk to patients with preexisting cardiac conditions.1, 3, 8, 10, 14 Corrales-Medina et al. reported that individuals with CAP and preexisting HF, cardiac arrhythmias, coronary artery disease (CAD), or arterial hypertension were at an increased risk for developing cardiac complications. 14 Specifically, the odds ratios (ORs) for cardiac complications were 4.3 for HF, 1.8 for cardiac arrhythmias, 1.5 for CAD, and 1.5 for arterial hypertension. 14 However, the study did not specify the specific causative organism causing pneumonia (S. pneumoniae or influenza virus). 14 In the “PARADIGM-HF” (“Prospective Comparison of Angiotensin Receptor Neprilysin Inhibitor [ARNI] With Angiotensin-Converting Enzyme Inhibitor to Determine Impact on Global Mortality and Morbidity in HF”) and “PARAGON-HF” (“Prospective Comparison of ARNI with Angiotensin Receptor Blocker Global Outcomes in HF with Preserved Ejection Fraction”) trials, the incidence of pneumonia (all-cause) was 6.3% and 10.6%, respectively, translating to incidence rates of 29 and 39 per 1,000 patient-years. 15 The incidence of pneumonia was notably high in patients with HF, especially HF with preserved ejection fraction, at approximately three times higher than the expected rate, and a first episode of pneumonia was associated with a 4-fold mortality risk. 15 Torres et al. reported that individuals with chronic heart disease (CHD), including congestive HF, CVD, and valvular diseases, have up to a 3.3-fold risk of CAP and a 9.9-fold risk of IPD compared with those without CHD. 3 Mor et al. reported that patients with chronic HF with cardiomyopathy (OR: 2.05) and heart valve disease (OR: 2.19) were at an increased risk of hospitalization due to pneumonia. 16 The risk of pneumonia-related hospitalization was lower in patients with HF associated with previous acute MI (OR: 1.69) or atrial fibrillation (OR: 1.72). 16 Heart failure in combination with a history of heart valve disease or atrial fibrillation has also been associated with an increased mortality risk due to pneumonia. 17 These findings highlight that pneumococcal and influenza vaccinations are crucial preventative measures for adults with cardiac conditions to prevent pneumonia and associated complications, regardless of the specific causative organism.
Expert Opinions
In our study, the experts opined that patients with HF, CAD, and MI are at risk of PD based on their clinical practice. Acute coronary syndrome (ACS) and chronic hypertension (CH) may confer some degree of increased susceptibility to PD. Experts mentioned that, based on their clinical practice, 30%-70% of patients with underlying cardiac conditions show worsening of primary illness or cardiac complications after developing CAP, and 10%-50% after developing IPD. Furthermore, 10%-30% of patients with CHD who develop pneumococcal infections require hospitalization during the course of illness. Early identification and prompt management of PD are important for reducing mortality in this at-risk patient subgroup.
Evidence and Optimal Timing of Pneumococcal Vaccinations in Patients with Cardiac Conditions
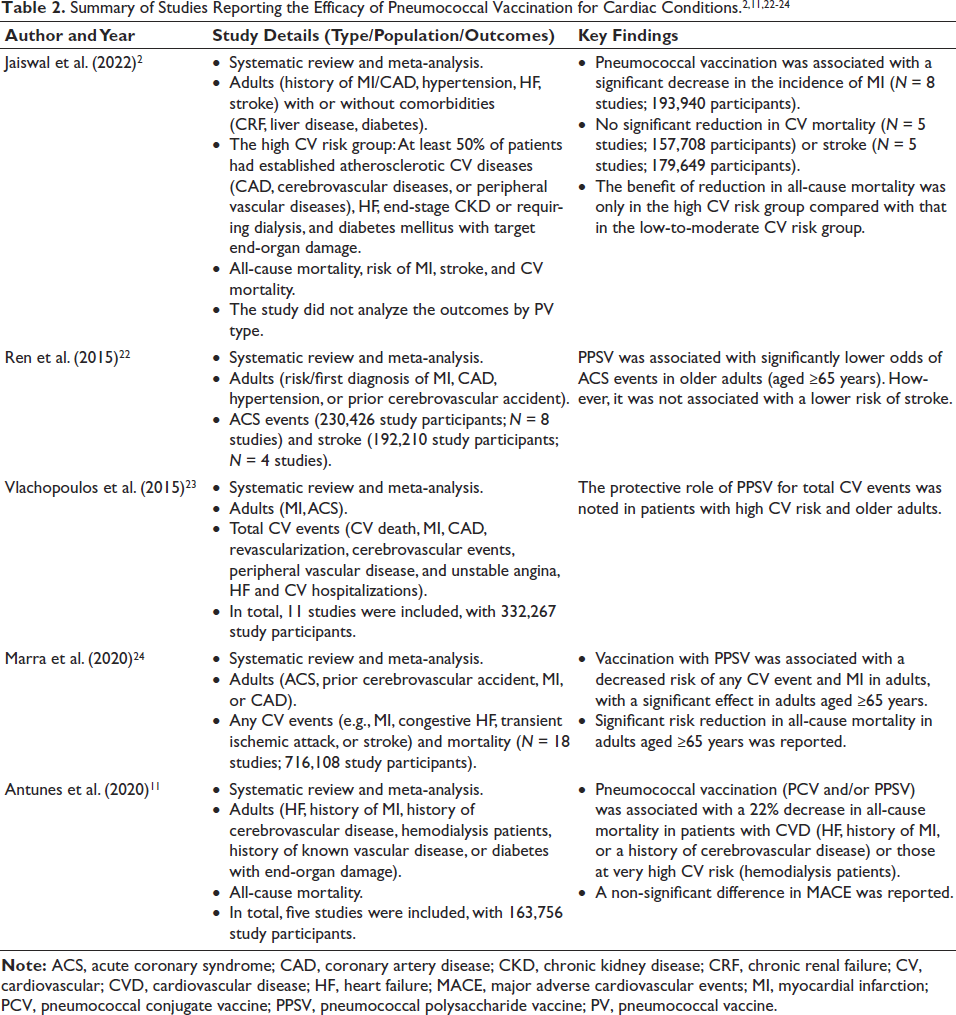
Two recent systematic reviews reported that pneumococcal conjugate vaccine 13 (PCV13) and pneumococcal polysaccharide vaccine 23 (PPSV23) protected against vaccine type (VT)-IPD and VT-pneumococcal pneumonia in adults.18, 19 Pneumococcal vaccination may provide economic benefits by lowering the hospitalization risk, as it plays a role in preventing PD.2, 20, 21 Pneumococcal vaccination offers significant protective effects for adults, especially those with underlying CV conditions (such as MI, ACS, HF, and CAD), and patients with high CV risk (Table 2).2, 11, 22–24 Ignatova et al. found that vaccinating patients (with PCV13) with chronic obstructive pulmonary disease (COPD), alongside chronic HF and ischemic heart disease, reduced healthcare costs by 74%-84%. 25 A meta-analysis by Vlachopoulos et al. reported that the protective effect of PPSV23 was particularly significant in individuals with elevated CV risk profiles. 23 This finding underscores the importance of pneumococcal vaccination in mitigating CV events in high-risk populations, as the analysis demonstrated a statistically significant reduction in both MI and cerebrovascular events among the elderly. 23 The benefit was most notable within the first year after vaccination, with a 28% reduction in CV events, though it waned beyond 1 year. Additionally, the protective effect was amplified in patients with CV and pulmonary comorbidities, further supporting its role in reducing risk in vulnerable populations. 23 Similarly, a study by Antunes et al. supports this finding, showing a 22% reduction in all-cause mortality in patients with CVD or those classified as having very high CV risk after pneumococcal vaccination (PCV13 and/or PPSV23). 11 Conversely, the absence of pneumococcal vaccination has been linked to a higher relative risk of developing HF and CAD, with older adults (between the ages of 80 and 89 years) facing an increased risk of hospitalization due to pneumococcal pneumonia in the absence of vaccination. 26 The use of PPSV was associated with a significantly lower risk of ACS events in older adults aged ≥65 years but not a significantly lower risk of stroke. 22 Local injection site and systemic reactions, such as fever and fatigue, were reported. 22 Jaiswal et al. reported that pneumococcal vaccination is associated with a decreased risk of all-cause mortality and MI in adult patients. 2 However, the study did not analyze the outcomes by PV type. 2 Tong et al. highlighted that sequential pneumococcal vaccination with PCV13 and PPSV23 was associated with lower CVD risk (ACS, congestive HF, or stroke), CVD-related deaths, and all-cause pneumonia versus single vaccination with either PCV13 or PPSV23 in older adults (≥65 years). 27
Children with cyanotic congenital heart disease and HF are at an increased risk of infections. Impaired nutrition and pulmonary congestion predispose one to pneumonia and other lower respiratory tract infections.28, 29 Pneumococcal conjugate vaccine can be considered in immunocompetent children with CHD (particularly cyanotic congenital heart disease and HF), who are at an increased risk of PD.30–32Table 3 provides a breakdown of pneumococcal vaccination schedules according to various global versus national guidelines for patients with cardiac conditions (CHD, pulmonary arterial hypertension, HF, and CAD).33–39
Expert Opinions/Consensus Recommendations
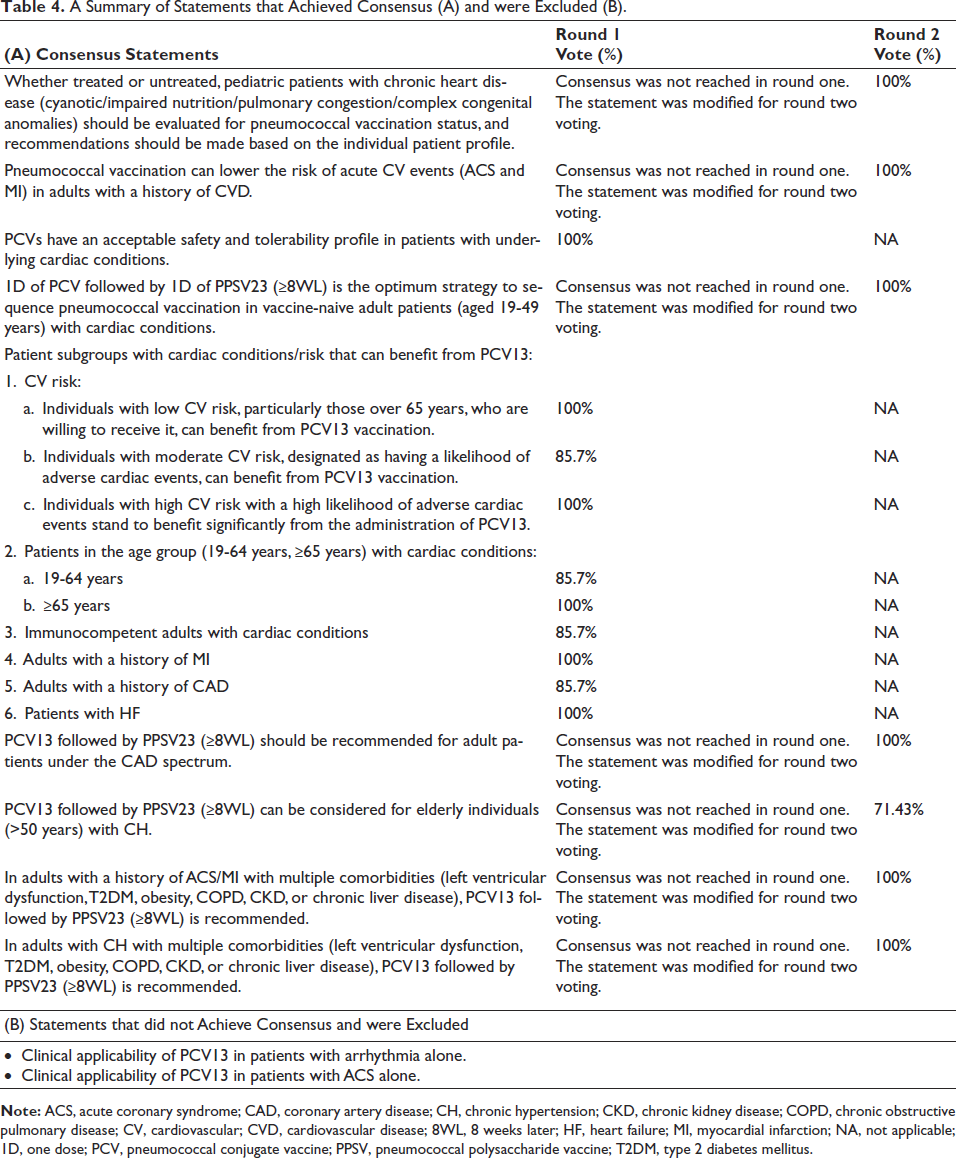
Table 4 lists the statements that achieved consensus (A) and the statements that were excluded (B). In our study, the experts agreed that, whether treated or untreated, pediatric patients with CHD (cyanotic congenital heart disease/impaired nutrition/pulmonary congestion/complex congenital anomalies) should be evaluated for pneumococcal vaccination status, and recommendations should be made based on the individual patient profile (Table 4A; high consensus). Experts came to a consensus that pneumococcal vaccination can lower the risk of acute CV events (ACS and MI) in adults with a history of CVD (high consensus). Experts recommended administering pneumococcal conjugate vaccine (PCV) followed by PPSV23 (≥8 weeks later) in vaccine-naïve adult patients (aged 19-49 years) with cardiac conditions, after the assessment of medical history, immune status, and risk factors (high consensus).
There was a moderate consensus on the administration of PCV13, followed by PPSV23 (≥8 weeks later), in elderly individuals (aged >50 years) with CH.
Experts agreed that administering PCV13 followed by PPSV23 after ≥8 weeks can be considered in adult patients under the CAD spectrum (high consensus).
The presence of multiple risk factors, such as smoking, type 2 diabetes mellitus (T2DM), and obesity, increases the susceptibility to pneumococcal infections. There was a low consensus on the clinical applicability of PCV13 in patients with ACS and arrhythmia alone. Hence, those two statements were excluded (Table 4B). The experts opined that patients with ACS and CH are at an increased risk of PD when complicated by additional multiple risk factors. Considering this, the panel also recommended administering PCV13 followed by PPSV23 after ≥8 weeks in adults (19 years and above) with CH/history of ACS/MI who had multiple comorbidities (left ventricular dysfunction, T2DM, obesity, COPD, chronic kidney disease [CKD], and/or chronic liver disease) (high consensus).
Immunocompetent individuals (aged >19 years) with cardiac conditions, adults (aged >19 years) with a medical history of MI/CAD, elderly individuals with cardiac conditions (≥65 years), individuals with moderate and high CV risk, and patients with HF (aged >19 years) could benefit from PCV13 vaccination (high consensus).
A Summary of Statements that Achieved Consensus (A) and were Excluded (B).
Barriers to Pneumococcal Vaccination in Cardiology Settings and the Recommended
Approach for Administering Pneumococcal Vaccines for Cardiac Conditions
Studies have highlighted that concerns about vaccine efficacy, lack of awareness about PD, and its complications, and poor understanding of vaccination schemes contribute to the lower uptake of pneumococcal vaccination.40–43 For patients with cardiac conditions, strategies such as patient education and direct physician recommendations for pneumococcal vaccination during clinical visits can play a major role in reducing vaccine hesitancy. 44
Expert Opinions
In our study, lack of awareness of PD and risk factors, lack of awareness of vaccine availability, and vaccine hesitancy (due to fear of adverse events) were identified as the most common perceived barriers to pneumococcal vaccination among patients with cardiac conditions. The most perceived barriers at the healthcare professional level were a lack of awareness regarding the burden of PD in cardiac conditions and uncertainty regarding clinical data on vaccine efficacy and recommendations (timing and schedule) for the use of pneumococcal vaccination in cardiac conditions. Experts agreed that counseling about the risk of PD early in the patient journey and the addition of flu and pneumococcal vaccination to the protocol for the management of cardiac conditions can decrease vaccine hesitancy and promote vaccination, especially among high-risk individuals.
Discussion
Pneumococcal diseases pose a significant public health concern in India, especially among older adults (>50 years of age) and those with preexisting cardiac conditions and/or chronic comorbidities (e.g., diabetes, chronic lung disease, obesity, and severe CKD).9, 42, 45, 46 Studies have reported that S. pneumoniae is one of the primary causes of IPD and CAP in India.47, 48 Furthermore, S. pneumoniae is the most frequently identified pathogen in studies of CAP etiology among adults, accounting for about 32.8%-55% of cases.49–52 Two studies from India reported a case fatality rate for IPD to be from 17.8% to 30%.48, 53
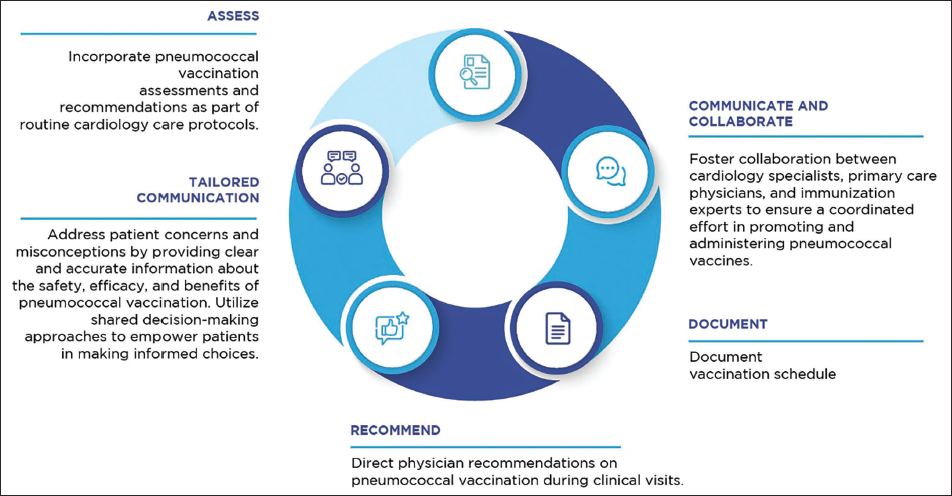
In 2017, PCV was introduced into the pediatric national immunization schedule in India. 54 Pneumococcal conjugate vaccine 13 offers broader protection, covering three additional prevalent serotypes (3, 6A, and 19A), than PCV10.55–57 Currently, both are licensed and available in the private sector in India. Solanki et al. confirmed PCV13’s immunogenicity and safety, with significant increases in opsonophagocytic assay geometric mean titers across all 13 vaccine serotypes, underscoring its suitability for addressing the PD burden among Indian adults aged 50-65 years. 58 Considering this, the experts reached a consensus that PCV13, followed by PPSV23, is recommended for vaccine-naive adult patients with various cardiac conditions in Indian settings. Experts further identified specific subgroups of patients who benefit significantly from pneumococcal vaccination, such as adults with a history of MI or CAD, patients with HF, older adults with CH, and those with a history of ACS/MI with multiple comorbidities. However, barriers to pneumococcal vaccination uptake persist in cardiology settings in India. Limited awareness of the burden of PD in individuals with cardiac conditions and vaccine hesitancy, driven by concerns over potential adverse events, exacerbates the gap (Figure 2). Addressing these barriers requires a multifaceted approach, including educational initiatives, collaboration, and integration of vaccination protocols into routine cardiac care pathways (Figure 3). Cardiologists, general practitioners, and primary care physicians should take an active role in improving vaccination awareness among this vulnerable patient population.
Barriers to Pneumococcal Vaccination in Cardiology Settings.

The Recommended Approach for Administering Pneumococcal Vaccines (PVs) for Cardiac Conditions.
Strengths and Limitations
Strengths: Our modified Delphi study provides a thorough synthesis of expert opinions. The panelists were selected based on their clinical expertise and in-depth knowledge of the field to ensure a fair and unbiased selection process. The questionnaire was designed after a thorough review of evidence to increase the study’s rigor. Furthermore, anonymity during the Delphi survey encouraged honest and unbiased input from panel members.
Limitation: The study did not include any patients’ opinions or views.
Conclusion
This expert consensus provides practical recommendations for clinicians, emphasizing the importance of pneumococcal vaccination for at-risk subgroups, such as individuals with HF, CAD, and a history of ACS/MI with comorbidities (notably diabetes and severe CKD). Addressing the barriers to vaccination necessitates coordinated efforts to raise awareness, reduce vaccine hesitancy, and implement customized vaccination protocols within cardiology practices. Although existing data emphasize the benefits of vaccination, more robust evidence is needed to refine strategies for at-risk subgroups with complex cardiac conditions. Future research should explore the long-term CV outcomes of pneumococcal vaccination and assess its cost-effectiveness in the adult population with/without multiple comorbidities in Indian settings.
Footnotes
Acknowledgment
The authors would like to thank BioQuest Solutions Pvt. Ltd. for its editorial support.
Authors Contribution
All authors contributed to data curation, formal analysis, methodology, validation, visualization, writing—original draft, and writing—review & editing. Additionally, conceptualization, investigation, resources, software, and supervision were carried out by Santosh Taur and Namrata Kulkarni. Funding acquisition was managed by Santosh Taur, while project administration was handled by Namrata Kulkarni. All authors have reviewed and approved the final version of the manuscript for publication and agree to take responsibility for all aspects of the work, ensuring that any concerns regarding its accuracy or integrity are thoroughly investigated and appropriately addressed.
Informed Consent
Informed consent was taken before answering the survey from all experts.
Data Availability
The datasets used during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.