Abstract
Background
Percutaneous closure of elliptical atrial septal defects remains challenging due to the circular configuration of conventional atrial septal defect occluders, making optimal device sizing difficult.
Case Presentation
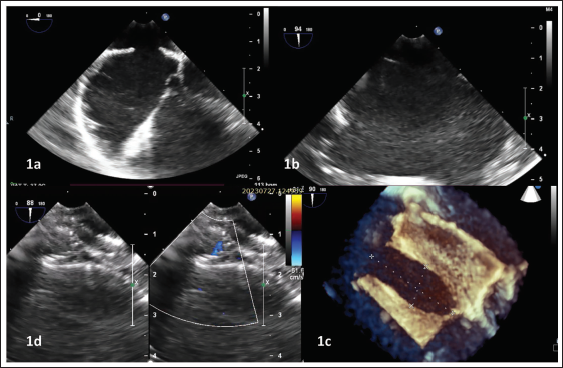
A 14-year-old child, weighing 40 kg, with a large atrial septal defect, was evaluated at our center for closure. Transesophageal echocardiography demonstrated a large, highly elliptical atrial septal defect. It measured 27 mm in the superoinferior plane in the bicaval view and 13 mm in the anteroposterior plane. A 24-mm atrial septal occluder was selected based on mathematical derivation of the circle with area equivalent to the elliptical atrial septal defect and was successfully deployed via the right upper pulmonary vein approach on the first attempt.
Conclusions
A circle is an ellipse with identical major and minor axes. This case illustrates the successful application of this mathematical concept in transesophageal echocardiography for atrial septal defect device closure, preventing unnecessary device oversizing.
Keywords
Introduction
Percutaneous closure of elliptical atrial septal defects (ASDs) still poses a challenge in optimal device size selection, as conventional ASD occluders are circular in configuration. Since a circle is an ellipse with identical major and minor axes, we explored whether this concept could optimize device sizing in elliptical ASDs. We demonstrate the successful application of this geometric concept in transesophageal echocardiography (TEE) for ASD device closure, thus avoiding unnecessary device oversizing.
Case 1
A 14-year-old child, weighing 40 kg, was referred to us with a large ASD for closure. Clinical examination indicated a hemodynamically significant pre-tricuspid left-to-right shunt without evidence of pulmonary hypertension. The 12-lead electrocardiogram revealed a crochetage pattern in the inferior leads and an incomplete right bundle branch block pattern, consistent with right ventricular volume overload.
Transesophageal echocardiography (TEE) demonstrated a large, highly elliptical ASD. It measured 27 mm in the superoinferior plane in the bicaval view and 13 mm in the anteroposterior plane (Figure 1). The ratio of the shortest to longest diameter was thus 0.48. The shortest posteroinferior rim measured 6 mm, and all other rims were adequate. Three-dimensional TEE demonstrated that all rims were sufficiently thick and not floppy.
Clockwise from Top Left. Transesophageal Echocardiography Stills of the 14-Year-Old Child at the Mid-esophageal Level in 0° (Panel 1a) and 90° (Panel 1b), Demonstrating the Secundum Atrial Septal Defect (ASD) Measuring 13 mm and 27 mm in the Respective Orthogonal Planes. Panel 1c is the 3D Still Demonstrating the Elliptical ASD Configuration. Panel 1d Shows the Well-positioned 24-mm ASD Device In Situ.
Geometry of an Elliptical Atrial Septal Defect
It is important to appreciate the differences between an ellipse and a circle for appropriate occluder selection in an elliptical ASD. Figure 2 is a cartoon depicting an elliptical ASD. The segment OA (measured as a) is the semi-major axis of the yellow, horizontally depicted ellipse, and segment OB (measured as b) is its semi-minor axis. Two distinct fixed points can be identified on the major axis of an ellipse, called focus points (or foci) and marked as F and F’, the sum of whose distances from each point on the ellipse is always constant and equal to the major axis of the ellipse (2a). 1 The mid-point of the foci is the center of the ellipse (point O). Each of the segments OF and OF’ (represented as “c”) constitutes the focal length of the ellipse. The eccentricity of an ellipse is the ratio of the focal length to the semi-major axis length. The eccentricity of an ellipse varies from 0 to 1. A circle is an ellipse where the eccentricity is zero.
Cartoon Depicting an Elliptical Atrial Septal Defect (ASD) (Yellow). O is the Center of the Ellipse. “a” Refers to Segment OA, the Semi-major Axis, and “b” Refers to Segment OB, the Semi-minor Axis. “c” Refers to Segment OF (or OF’), the Focal Length of the Ellipse. F and F’ are the Foci. Segment RR’ is the Latus Rectum; “p” Refers to the Semi-latus Rectum. “r” is the Length of Segment OR and Constitutes the Radius of the Blue Circle. The Red Circle has the Major Axis of the Ellipse as its Diameter, and the Green Circle has the Minor Axis.
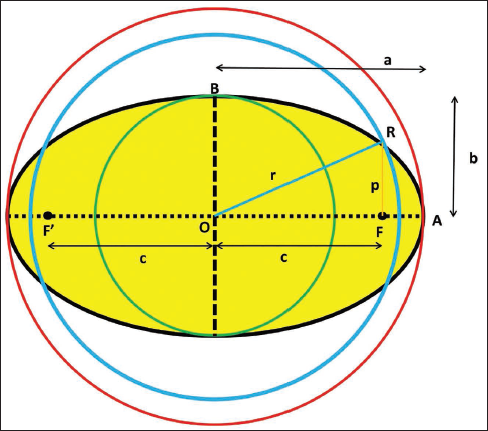
The latus rectum of an ellipse is a straight line passing through its foci, perpendicular to its major axis (segment RR’). Since an ellipse has two foci, it has two latus rectums. The segment FR, represented as “p” in the figure, is the semi-latus rectum. It is evident that a circular occluder of diameter 2b (represented by the green circle) is not sufficient to close the elliptical defect. A device with a diameter 2a (red circle) will completely occlude the defect but be extremely bulky. The conceptual circle that best fits the ellipse (blue circle) will have an area and perimeter comparable to the ellipse and is the one that intercepts the ellipse at its semi-latus rectum. The radius of this conceptual circle (r) can be mathematically derived as the hypotenuse of the right-angled triangle subtended by the focal length (c) and semi-latus rectum (p). The formulae involved are as follows1–3:
Focal length (c) = √(a² – b²)
Semi-latus rectum (p) = b2/a
Eccentricity of the ellipse = c/a = √(a² – b²)/a
Radius of the circle with equivalent area as the ellipse (r) = √(c² + p²). This provides the size of the ideal occluder for an elliptical ASD (2r).
The dimensions of the elliptical ASD in our patient were carefully analyzed. The relevant dimensions in the patient were as follows: semi-major axis (a) 13.5 mm, semi-minor axis (b) 6.5 mm, focal length (c) 11.83 mm, semi-latus rectum (p) 3.13 mm, defect eccentricity 0.88. The radius of the circle with equivalent area as the elliptical ASD was obtained to be 12.2 mm (diameter 24.4 mm). A 24 mm Amplatzer™ Septal Occluder (Abbott, St. Paul, MN) was chosen and successfully deployed via the right upper pulmonary vein approach on the first attempt. The device position was confirmed to be satisfactory on TEE, with no residual flow. The patient remains well at 9 months’ follow-up, with no residual flow or complications.
Case 2
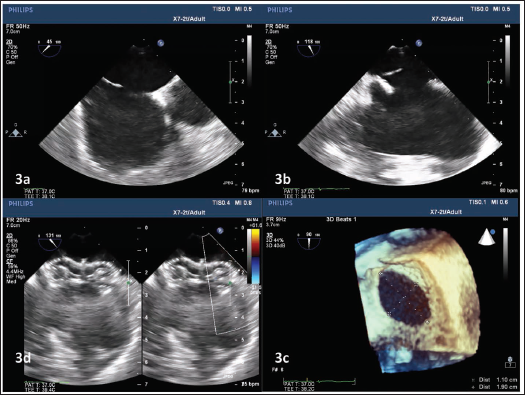
A 31-year-old lady was diagnosed with an elliptical ASD that measured 19 mm in its longest dimension and 11 mm in its shortest dimension (Figure 3). The relevant dimensions of the elliptical ASD were obtained to be as follows: semi-major axis (a) 9.5 mm, semi-minor axis (b) 5.5 mm, focal length (c) 7.75 mm, semi-latus rectum (p) 3.18 mm, defect eccentricity 0.81, and radius of the circle with equivalent area of the elliptical ASD (r) 8.38 mm. The optimal occluder diameter was derived as 16.8 mm. TEE analysis revealed relatively floppy posteroinferior rims, necessitating a 1–2 mm oversizing of the occluder. An 18 mm Cera (Lifetech Scientific, Shenzhen, China) occluder was successfully used to close the defect at first attempt, entirely by TEE guidance, avoiding the use of fluoroscopy.
Clockwise from Top Left. Transesophageal Echocardiography Stills of the 31-Year-Old Lady at the Mid-esophageal Level in 45° (Panel 3a) and 118° (Panel 3b) Views, Demonstrating the Secundum Atrial Septal Defect (ASD). Panel 3c is the 3D Still Demonstrating the Elliptical ASD Configuration Measuring 19 mm × 11 mm. Panel 3D Shows the Well-positioned 18-mm ASD Device In Situ.
Discussion
In conventional ASD closure, occluder selection typically involves oversizing the largest defect dimension by 2–4 mm. 4 In the absence of customized devices, oval and elliptical ASDs are also closed using the same circular ASD devices. Oversizing is known to be associated with device-related complications, including rim erosions. 5 Oval-shaped ASDs are reported to constitute approximately 42% of patients considered for device closure. 6 While an ellipse is not a circle, a circle is a special type of ellipse where the semi-major and semi-minor axes are identical. 2 The two foci points combine to become one in a circle, and the eccentricity becomes zero. Eccentricity refers to the ovalness of an ellipse, with a value closer to one indicating a higher degree of ovalness. The formula used in the index case becomes increasingly relevant for defects with higher grades of eccentricity. We consider a defect with an eccentricity of >0.8 as significantly oval. In our experience, ASDs with eccentricity >0.8 are significantly oval and may require alternative sizing strategies. We have noted that elliptical ASDs with sturdy rims and eccentricity >0.8 are optimally closed with an occluder smaller than the largest diameter of the defect.
Zanchetta previously described the application of this concept for choice of Amplatzer ASD devices using intracardiac echocardiography (ICE). 3 While ICE offers the advantages of avoiding general anesthesia and the need for a second operator, TEE is more cost-effective in the developing world. Our case demonstrates the relevance of meticulous imaging and reiterates the dwindling role of balloon sizing in ASD closure.7, 8
In addition to the shape of the defect, floppiness of the rims and malalignment of the interatrial septum are other factors that determine the size of the optimal device in secundum ASD, including elliptical ones, as illustrated in the second case.
It may be noted that other authors have reported significant complications while trying to close similar elliptical defects using the conventional approach, including cardiac perforation, pleural, and pericardial effusion. 9 The need for meticulous imaging in complex elliptical ASDs cannot be overemphasized.
Conclusions
Elliptical secundum ASDs can often be successfully closed with a device following thorough transesophageal echocardiographic evaluation. Optimal ASD occluder selection depends on factors such as defect eccentricity, the equivalent circular diameter, rim characteristics, and septal alignment. In many cases, the chosen occluder is equal to or smaller than the largest defect dimension.
Footnotes
Abbreviations
ASD: Atrial septal defect; ICE: Intracardiac echocardiography; TEE: Transesophageal echocardiography.
Author Contributions
Concept and data collection: AG.
Drafting article: GM and AG.
Editing: DR, AV, and KMK.
All authors accepted the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its amendments. The preparation of this manuscript did not require formal ethical approval from the institute ethics committee, as this involved data from two patients only and was not a clinical trial. Informed written consent was taken from the patients concerned. No patient identity particulars have been disclosed.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
