Abstract
A rare mechanical consequence that can occur after an acute myocardial infarction, particularly in the current thrombolytic era, is ventricular septal rupture. This condition exhibits a bimodal pattern of occurrence, with a higher frequency observed within the first 24 h and again between 3 and 5 days postinfarction. The risk of developing mechanical complications, such as ventricular septal rupture, is increased by delayed reperfusion of the artery affected by the infarction, which leads to tissue necrosis and death. Female gender, advanced age, raised Killip class, elevated cardiac biomarkers, delayed reperfusion, and ineffective thrombolysis are factors related to post-myocardial infarction ventricular septal rupture. While ventricular septal rupture is predominantly associated with ST elevation myocardial infarction, it has been infrequently documented in cases of acute non-ST segment elevation myocardial infarction (NSTEMI). The clinical presentation and progression of ventricular septal rupture are intricately related to the underlying pathophysiological mechanisms and the speed at which hemodynamic collapse and cardiogenic shock develop. Despite timely intervention, whether surgical or percutaneous, the prognosis remains grim, with a significant hospital mortality rate influenced by factors such as age, hemodynamic instability, and other pre-existing health conditions. The presentation, course, and results of five patients with ventricular septal rupture after myocardial infarction treated at our hospital during the previous two years are addressed in this case series.
Introduction
With an incidence of 0.25%-0.31%, postinfarction ventricular septal rupture (VSR) is an uncommon and sometimes severe mechanical consequence of an acute myocardial infarction (AMI).1, 2 Its occurrence has significantly decreased in the current thrombolytic era as a result of early reperfusion therapy with thrombolytics and percutaneous revascularization. 3 Delayed reperfusion of the infarct-related artery causes expansion of the infarcted zone, with myocardial thinning and necrosis leading to the formation of VSR. 4 In these cases, VSR occurs soon or subacutely after the initial infarction. With its bimodal distribution, VSR can appear hours or three to five days after the index MI. 5 The following are risk factors for VSR: Killip classes II-IV, female sex, older age group, prior cerebrovascular accidents, anterior wall myocardial infarction, ST-segment elevation myocardial infarction, and complete blockage of coronary arteries without established collateral circulation.2, 6, 7 The clinical presentation and pathophysiology of VSR depend on the size and location of the transmural infarct. 8 The patient’s hemodynamic condition typically dictates when surgery should be performed; early surgery is preferable for hemodynamically unstable patients, whereas stable patients are managed conservatively initially. 9 Despite the prompt treatment, either surgical, percutaneous closure, or conservative, the death rate remains high. 10 We describe the onset, progression, and outcome of five ventricular septal rupture cases associated with myocardial infarction occurring in our hospital over the past two years.
Case 1
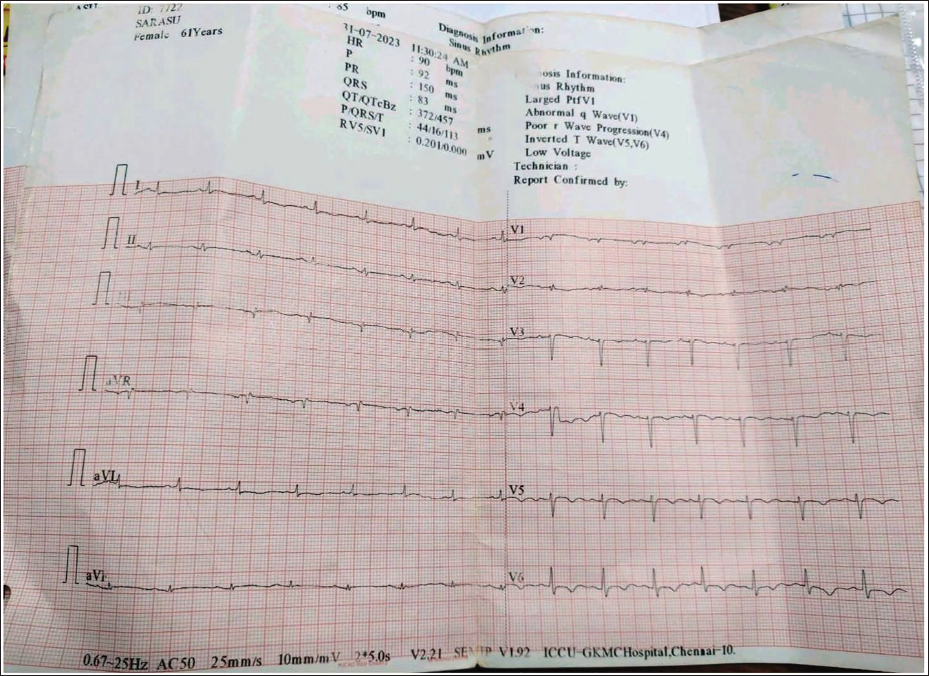
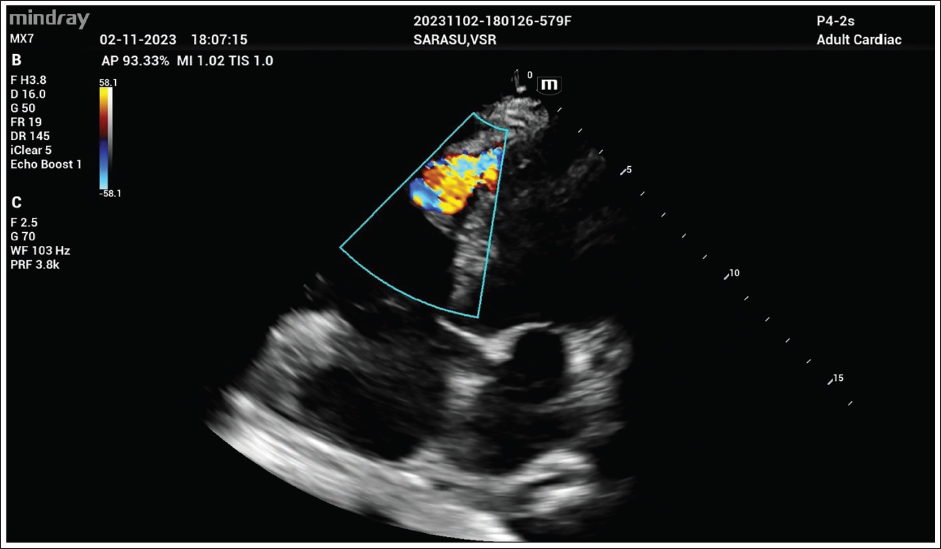
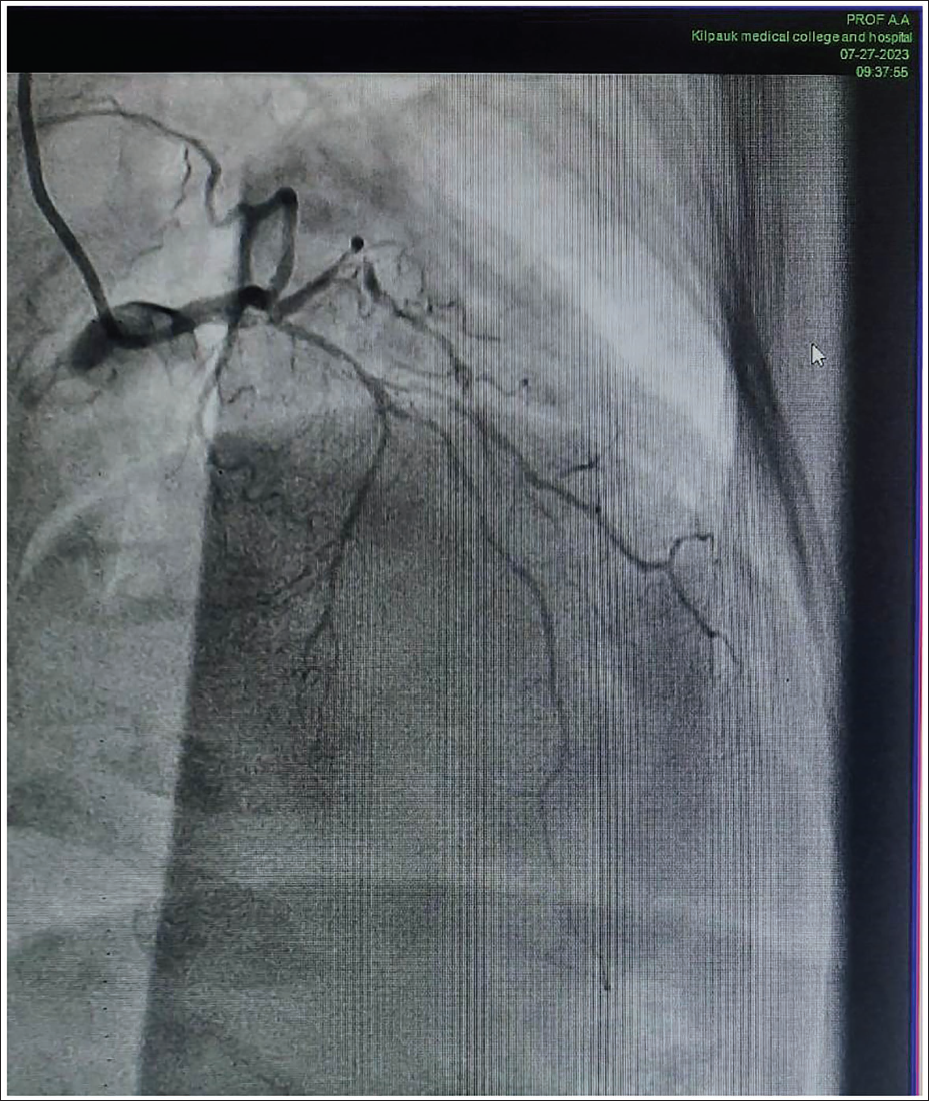
A 60-year-old woman reported having normal chest pain for the previous 12 h. She has a long history of diabetes mellitus and has been diagnosed with diabetic renal disease for the past three years. On admission, her vitals were stable; an electrocardiogram revealed inverted T waves in the anterior and lateral leads with delayed R wave transition (Figure 1), and on auscultation, a harsh systolic murmur was heard over the precordium. There were also right ventricular dysfunction, a dropout at the apex of the ventricular septum (4 mm) on echocardiography, a diffuse hypokinetic left ventricle with an ejection fraction of 36%, and turbulent flow from the left to the right ventricle (RV) on Doppler imaging (Figure 2). The tricuspid annular plane systolic excursion (TRPG) was 40 mmHg. The Troponin T test was positive, and coronary angiography revealed diffusely diseased left anterior descending artery (LAD) with significant branch vessel disease of the diagonal and obtuse marginal (Figure 3). Despite the recommendation for patch closure of ventricular septal rupture and coronary artery bypass grafting, she declined surgery. As of now, she is still alive and doing well with medications.
ECG Showing T Waves Inversion in the Anterior and Lateral Leads with Delayed R Wave Transition.
Transthoracic Echocardiography Showing VSR of 4 mm at Apical Location with Turbulent Flow from the Left to the Right Ventricle on Doppler Imaging.
CAG- Diffusely Diseased LAD with Significant Branch Vessel Disease of the Diagonal and Obtuse Marginal.
Case 2
A 65-year-old female presented with central chest discomfort associated with profuse sweating on and off for the last 9 h. She had no prior history of heart disease and was on treatment for diabetes and hypertension. At the time of admission, her vitals were stable, and an electrocardiogram (ECG) had significant J point elevation in anterolateral chest leads (Figure 4). Echocardiography showed hypokinesia of the anterior, anterolateral, anteroseptal segments of the mid and basal left ventricle, the distal third of IVS, and the apex of the left ventricle, with an ejection fraction of 35% and normal RV function (Figure 5). After receiving 1.5 million units of intravenous streptokinase, the patient’s ECG revealed that the thrombolysis attempt had failed. On day 3 of hospitalization, her clinical condition deteriorated, and on auscultation, she had a harsh murmur over the precordium with a palpable thrill. Repeat transthoracic echocardiography revealed an apical ventricular septal rupture (8 mm) in size with high-velocity turbulent flow to the RV, an ejection fraction of 30%, and a tricuspid annular plane systolic excursion (TAPSE) of 12 mm (Figure 5). The patient’s condition rapidly deteriorated, and she expired within 24 h of developing cardiogenic shock (CS).
Admission ECG Showing Acute Anterolateral MI.
2D Echocardiography Showing an Apical VSR of 8 mm with High-velocity Left to Right Shunt.
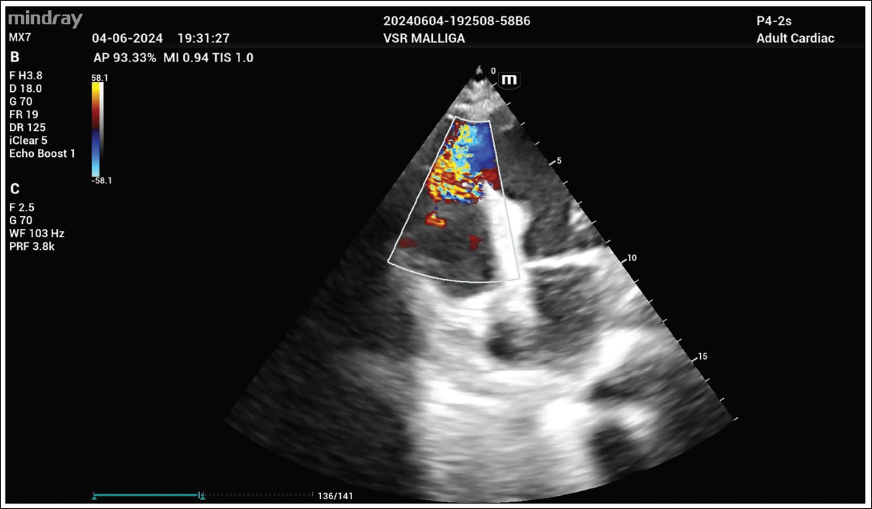
Case 3
A 75-year-old hypertensive lady with no other known comorbidities presented with central chest discomfort and dyspnea for the last 20 h. The ECG showed ST-segment elevation in the anterolateral chest leads, and she experienced persistent chest pain (Figure 6). She developed sudden hypotension and tachycardia, and a systolic murmur on the left side of the sternum was found during examination. Transthoracic echocardiography revealed a dilated left ventricle, significant tricuspid regurgitation, a 10-mm apical ventricular septal rupture with a bidirectional flow, and a hypokinetic area in the left anterior descending artery with an ejection fraction of 38% (Figure 7). In view of ventricular septal rupture, thrombolysis was deferred, and she was treated conservatively. However, the patient deteriorated rapidly due to CS, progressed to multiorgan failure, and ultimately succumbed.
ECG Showing ST-segment Elevation with T Inversion in the Anterolateral Chest Leads.
Transthoracic Echocardiography Showing, a 10 mm Apical Ventricular Septal Rupture with a Bidirectional Flow.
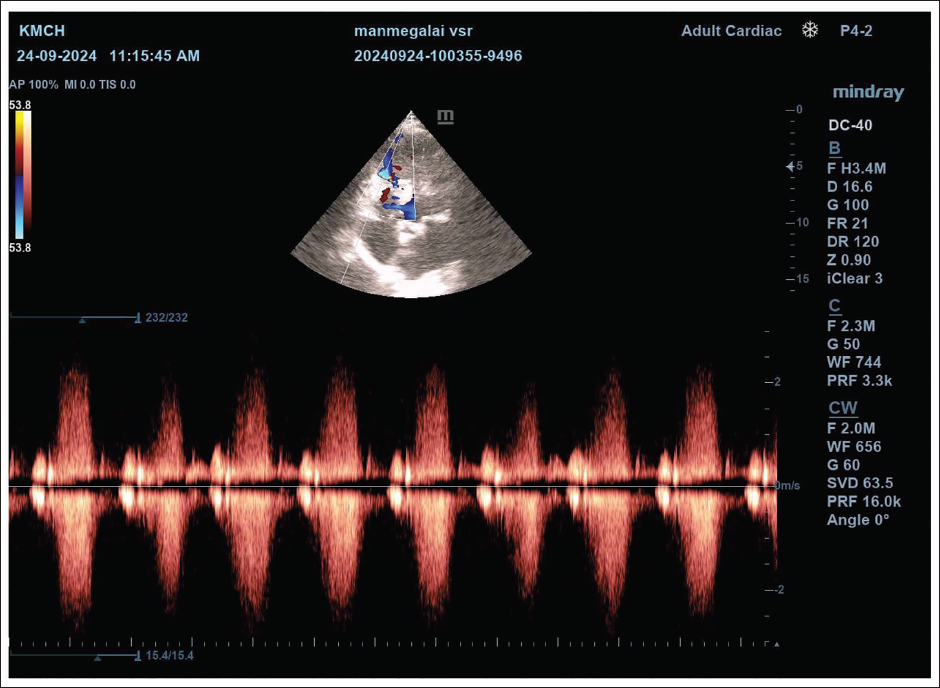
Case 4
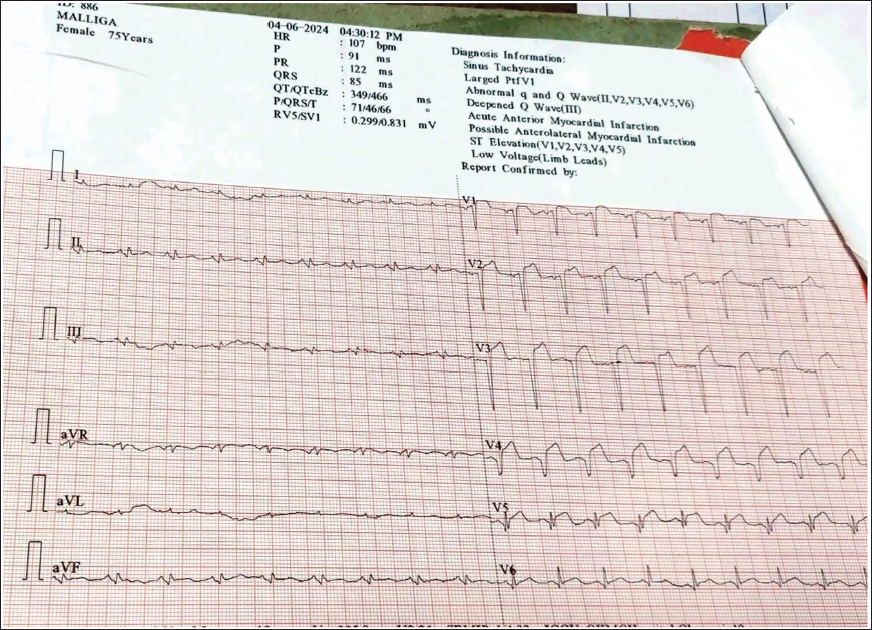
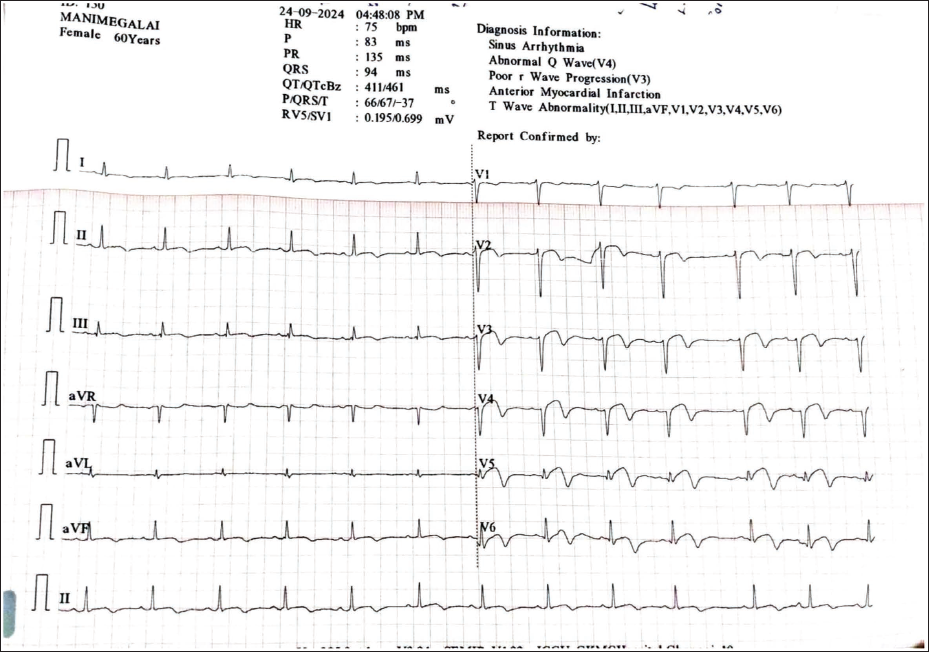
A 60-year-old female with a 20-year history of diabetes reported having chest pain for the previous 14 h. On admission, her vitals were stable, and the ECG showed ST-segment elevation with Q waves in V2-V5 (Figure 8). On auscultation, a harsh systolic murmur over the precordium was present. Two-dimensional (2D) echocardiography revealed an ejection fraction of 40%, a hypokinetic LAD territory, and the left ventricular apex, as well as a 6-mm-sized apical VSR (Figure 9). The patient received conservative treatment and ongoing monitoring; she is currently alive and receiving routine check-ups.
Admission ECG Showing ST-segment Elevation with Q Waves in V2-V5 Leads.
2D Echocardiography Showing a 6-mm-sized Apical VSR with a Left to Right Ventricle Turbulent Flow.
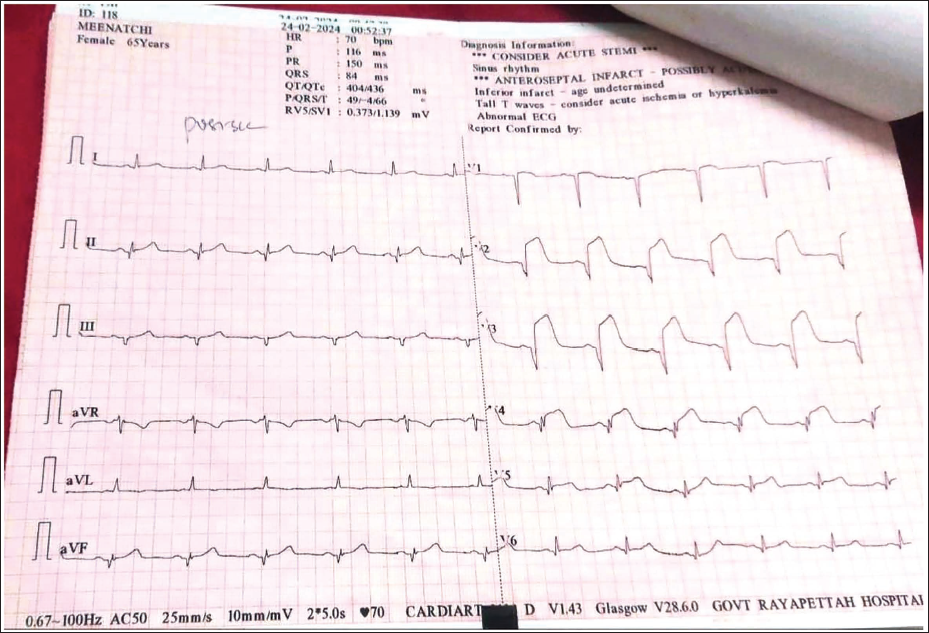
Case 5
A 56-year-old female patient with diabetes arrived with a history of usual chest pain that had persisted for the preceding 8 h, along with episodes of perspiration. Her ECG showed substantial ST-segment elevation in the precordial leads and ST-segment depression in the inferior lead. Her heart rate was 110 bpm, and her blood pressure was 130/90 mmHg at admission (Figure 10). The anterior and anteroseptal regions of the left ventricle were hypokinetic, with an ejection fraction of 48%, according to transthoracic echocardiography (Figure 11). An injection of 1.5 million units of streptokinase was administered in an attempt to reperfuse the heart. The patient’s congestive heart failure symptoms included hypotension, and a serial echocardiography showed decreasing heart functions, along with a 12-mm ventricular septal rupture and blood shunting into the RV. Ventricular septal rupture closure using an atrial septal defect (ASD) occluder device was carried out as the patient’s condition began to rapidly deteriorate. The Ceraflex ASD occluder device consists of a polyester membrane and a Nitinol wire frame. A waist-sized device measuring 14 mm in diameter and 4 mm in length was used, with a left disc diameter of 28 mm and a right disc diameter of 24 mm. A 9F delivery sheath was employed to deploy the ASD occluder device over the VSR (Figures 12 and 13). However, the patient died after the procedure after developing multiorgan failure as a result of refractory CS.
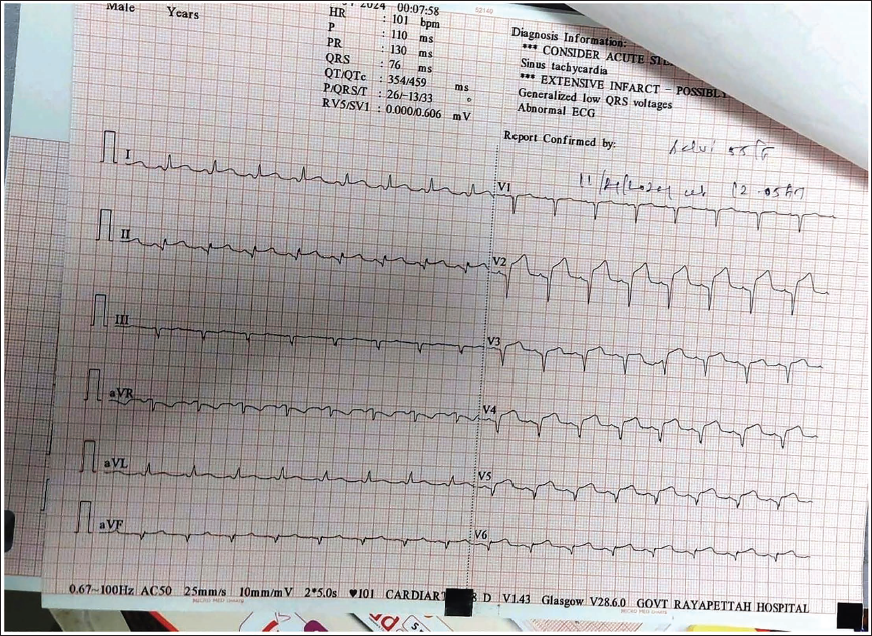
ECG Showing ST-segment Elevation in the Precordial Leads with Reciprocal Changes in the Inferior Lead.
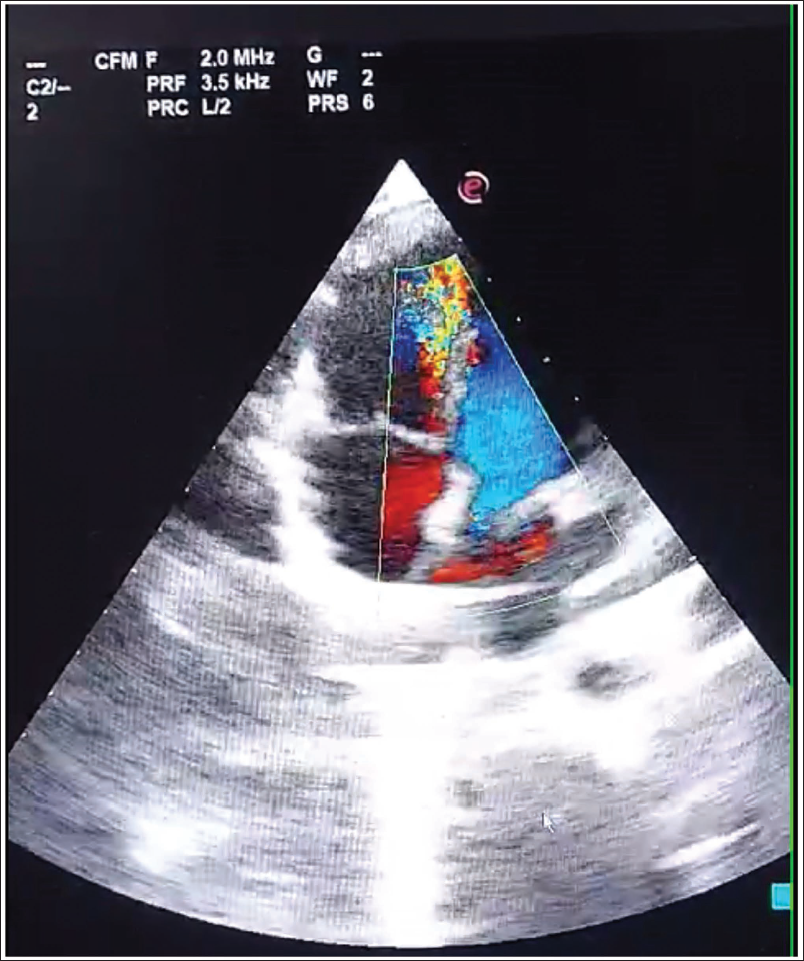
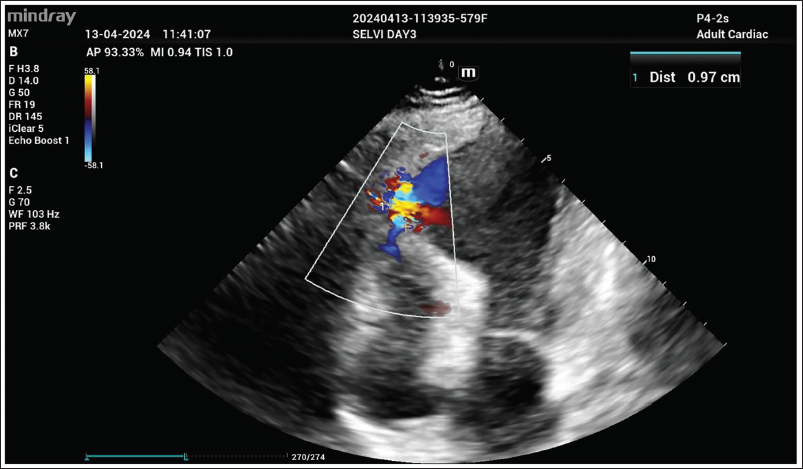
2D Transthoracic Echocardiography Showing a 12-mm Apical Ventricular Septal Rupture and Blood Shunting into the RV.
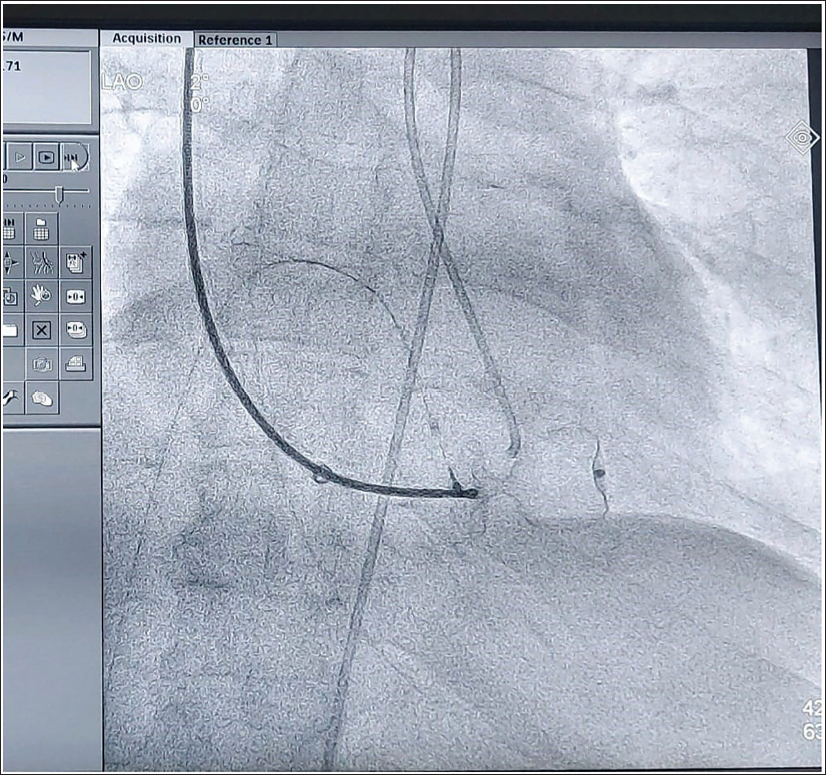
Images Showing the LV Angiogram and A-V Loop Formation to Deploy the ASD Device.
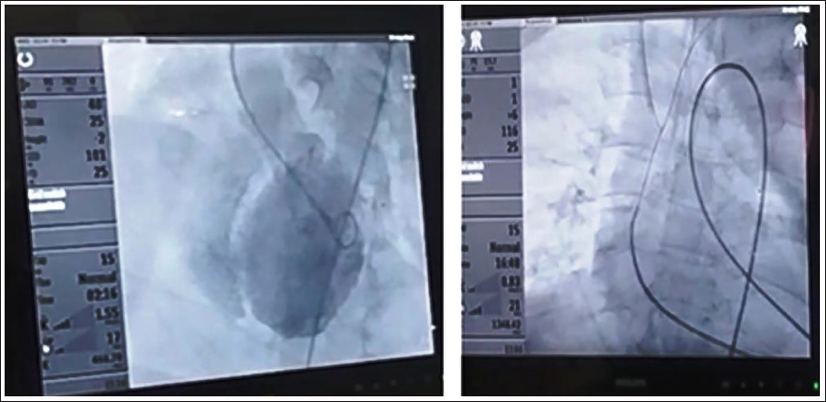
Image Showing the ASD Occluder Device Deployed over the VSR.
Discussion
Ventricular septal rupture can be classified based on its anatomical location into apical, anterior, or posterior types, as well as by its morphology into simple/complex forms. In cases of complex defects, one may observe multiple rupture sites along with a serpiginous tract.2, 7, 11 Because the septal perforator branches of the left anterior descending and posterior descending coronary arteries supply blood to the ventricular septum, a reduction in blood flow from either artery is more likely to cause a VSR.2, 12 Hemorrhagic transformation related to thrombolysis and intramyocardial dissection in the infarcted region, rather than necrosis, have been associated with the onset of VSR following thrombolytic therapy. 12 The surrounding myocardium’s shear stress and hypercontractile forces in the infarct zone worsen the septal tear, causing shunting and an abrupt rise in ventricular pressure and volume overload. 11 Due to the shunting of blood from the left to the right caused by VSR, the RV experiences a volume and pressure overload, which is followed by a left-sided volume overload and an increase in pulmonary venous return. The relative vascular resistances of the pulmonary and systemic circulations dictate the extent of shunting. The precordium experiences a systolic murmur due to this left-to-right shunt, which often peaks near the left sternal boundary and is frequently accompanied by a felt thrill. 11
In our observation, all the above-reported cases are females above 55 years of age and had an apical VSR. Among them, four patients had a long-standing history of diabetes, and two had chronic hypertension. Among these five patients, four presented with ST segment elevation myocardial infarction (STEMI) (anterior wall myocardial infarction [AWMI]), and one with non-ST segment elevation myocardial infarction (NSTEMI). One of these patients developed VSR about 72 h after presentation, while the other four patients had VSR within 24 h. All these patients had a harsh systolic murmur over the precordium, and 2D transthoracic echocardiography showed the VSR defect with shunting of blood.
The most common mechanical consequence after an AMI is ventricular septal rupture. 13 According to research by Elbadawi et al., 75% of mechanical problems following myocardial infarction were caused by ventricular septal rupture, with free wall rupture accounting for 6% and mitral regurgitation from ruptured papillary muscle for 19%. The development of left, right, or both types of heart failure, as well as the occurrence of CS, are the primary factors affecting outcomes in postinfarction ventricular septal defect. 14 About 0.21% of patients with ST-segment elevation myocardial infarction and 0.04% of cases with non-ST-segment elevation myocardial infarction result in post-myocardial infarction VSR. 14 Apical VSR is more common and usually associated with myocardial infarction of the anterior wall. 1 Heart auscultation can be used to diagnose postinfarction VSR, but it can be difficult to differentiate it from acute mitral regurgitation. To confirm the diagnosis, a transthoracic echocardiography is typically performed at the patient’s bedside. 9 The preferred first imaging method for individuals suspected of having VSR and for identifying mechanical problems associated with AMI is transthoracic echocardiography. In addition to helping to rule out alternative causes of hemodynamic instability, this imaging modality aids in assessing the magnitude and hemodynamic impact of VSR. Pulmonary artery catheterization may show an increase in oxygen saturation in the RV, which can be used to determine the Qp/Qs ratio. The diagnosis of VSR can be confirmed by left ventriculography in cases of unexplained hemodynamic instability.
Two of these patients arrived during the window period and received 1.5 million units of injectable streptokinase for thrombolysis; nonetheless, the procedure was unsuccessful. Thrombolysis was deferred for two patients; one patient arrived with NSTEMI, while two presented late with VSR upon admission. One among them developed VSR about 72 h after MI, whereas the remaining four had VSR within a day of the presentation. Out of the five patients, three have passed away, while two remain alive to this day.
Ventricular septal rupture demonstrates a bimodal distribution, characterized by a significant rise in incidence during the first 24 h, followed by a subsequent peak occurring 3 to 5 days post-acute MI. 5 Older age, female sex, persistent ST-segment elevation, elevated heart rate, hypotension, a higher Killip classification, the presence of multiple comorbidities, the severity of coronary artery disease, and hemodynamic instability are some of the factors that have been associated with poor outcomes in post-myocardial infarction VSR by previous research.15, 16
Surgical intervention was postponed for four of these patients; of these, two are still alive, whereas the other two succumbed after experiencing VSR. One patient underwent ASD device closure with an ASD occluder device via the transcatheter route, and this patient expired after the procedure.
In more than 50% of cases, septal rupture leads to an acute and progressive decline in hemodynamic stability, culminating in CS. For these individuals, conservative medical treatment is consistently associated with a fatal outcome.5, 11 Even with timely revascularization and surgical intervention, the mortality risk associated with post-myocardial infarction VSR remains significant, ranging from 20% to 87%. 17 Histopathology of the ventricular septum torn by MI shows intramyocardial bleeding with fibrotic unfavorable remodeling, contraction band necrosis, and extensive infiltration of inflammatory cells. The severity of inflammatory infiltration and adverse remodeling due to delayed perfusion increases the chance of ventricular septal rupture. 18
The optimal timing for VSR repair remains inadequately defined, despite its critical importance. It is generally accepted that surgical timing should be tailored to the patient’s condition: early intervention is recommended for those with unstable hemodynamics, while a delayed approach may be suitable for patients in a stable condition. 19 According to earlier research, delaying surgery is associated with higher 30-day and long-term survival rates, and there is a negative relationship between 30-day mortality and the time between diagnosis and surgical repair. The delicate necrotic myocardium presents serious technical difficulties for surgery in the immediate aftermath. 20 Percutaneous transcatheter alternatives may be investigated for patients who are not candidates for surgical surgery. 21 According to Schlotter et al., patients who had percutaneous closure of a postinfarction ventricular septal rupture within 2 weeks of the rupture had a 32% hospitalization mortality rate. 22 Therefore, patients with VSR have a generally good long-term prognosis, provided they survive the acute period. Therefore, it is essential to choose the best course of action and schedule the procedure based on the clinical situation of the patient. 23
Conclusion
The occurrence of VSR has significantly diminished in the present thrombolytic era; however, it remains essential to remain alert for this mechanical complication, as it can be life-threatening and is associated with poor outcomes. Patients who present with acute coronary syndrome should be closely watched for the early identification of VSR, particularly elderly females with known comorbidities like diabetes mellitus. Because of the high mortality and poor results, there is ongoing debate on the best time to perform surgery in VSR. Whenever hemodynamics remain steady, expectant management can be used. Even though VSR gives patients a slim but real chance of survival, it has a high mortality rate, so defect closure should be tried in cases of hemodynamic deterioration. Every patient who presents with AMI should have their VSR closely monitored. Prior to the initiation of thrombolytic therapy, a cardiac auscultation and a 2D echocardiogram should be performed. Furthermore, these patients require careful follow-up for any signs of post-myocardial infarction VSR, and appropriate management must be implemented, as this condition is associated with a high mortality rate.
Footnotes
Acknowledgments
I am deeply grateful to my professors for their valuable guidance and support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from all patients for participation and publication of data.
