Abstract
Ventricular septal rupture is a rare mechanical complication of acute myocardial infarction that continues to have high mortality outcomes in the 21st century. Mechanical complications rarely co-occur after myocardial infarction, posing significant management challenges. We present an extremely rare case of a true ventricular double rupture in a patient with inferior wall myocardial infarction and its management implications, wherein the second rupture may be missed.
Keywords
Introduction
Although rare, ventricular septal rupture remains a serious mechanical complication of acute myocardial infarction with high mortality even in the 21st century. 1 Surgical techniques have evolved from infarctectomy to modern methods such as infarct exclusion, double, triple, and sandwich patches. 2 However, mortality rates still depend largely on the clinical severity and timing of presentation, presence of right ventricular dysfunction, need for intra-aortic balloon pump (IABP), and the location of the defect. 3 Operative mortality in the repair of ventricular septal rupture is known to decrease with increasing time from the index myocardial infarction. 4
The advent of percutaneous device closure of ventricular septal ruptures offers options to close the defects in select cases of ventricular septal ruptures even in the early days of clinical presentation. However, device closure is also fraught with its risks, and the survival outcomes are not significantly different. 5 There is still no consensus on the optimal timing of intervention, and hemodynamic status is the primary guide to decision-making. Multiple mechanical complications can rarely co-exist in a patient following myocardial infarction and present various challenges in management. The clinical deterioration associated with the index mechanical complication often guides therapeutic decision-making. However, macroscopic tears of the ventricular myocardium may exceed expected boundaries, especially in relatively thin basal segments with adverse implications. Ventricular septal rupture in inferior wall myocardial infarction tends to have a worse prognosis due to complex morphology, involvement of thin basal segments, and associated right ventricular dysfunction. A double rupture compounds the trouble in inferior wall myocardial infarction.
Case History
A 75-year-old gentleman underwent coronary artery bypass surgery for triple vessel disease. Six months later, he presented to us with acute inferior wall ST segment elevation myocardial infarction. He underwent primary angioplasty to the proximal right coronary artery with a 3.5 × 38 mm drug-eluting stent with a total ischemic time of 8 hours. He remained well for 24 hours. However, he developed acute decompensated heart failure with cardiogenic shock on day 2 and was mechanically ventilated and put on inotropes. Cardiac troponin T was 17,300 ng ml–1 on day 2, and the N-terminal pro-brain natriuretic peptide recording was 21,400 pg ml–1. Transthoracic echocardiography revealed a 16 mm ventricular septal rupture in the mid-muscular septum extending posteriorly with an interventricular gradient of 45 mmHg. An urgent surgical consultation was taken for emergency surgical repair. The mortality risk was deemed to be 49% by EuroSCORE II, and the family refused surgery. An urgent heart team meeting was held, and it was decided for stabilization with IABP and an attempt at percutaneous closure of the ventricular septal rupture. The patient was rushed to the catheterization lab for device closure of the ventricular septal rupture and put on IABP support.
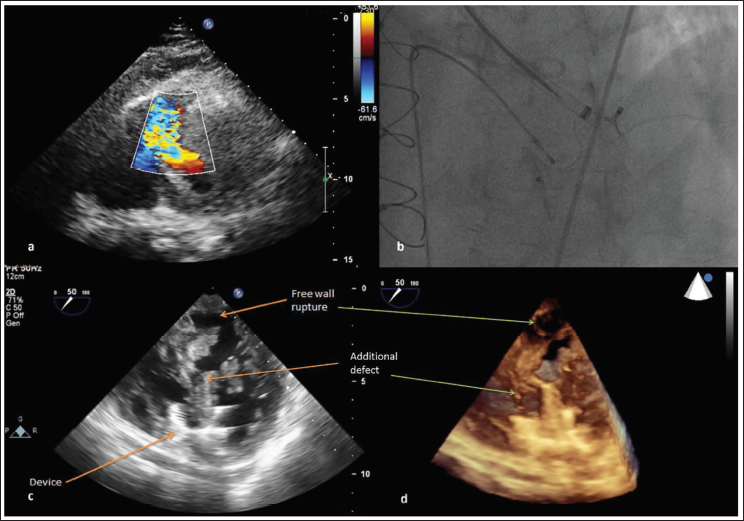
Transesophageal echocardiography suggested a serpiginous ventricular septal rupture from the mid-muscular septum extending to the apical septum (Figure 1a). Under transesophageal echo and fluoroscopy guidance, a 24 mm Amplatzer muscular ventricular septal defect device was deployed from the retrograde approach over a 12F TorqVue sheath advanced from the femoral artery (Figure 1b). Subsequent imaging revealed that the posterobasal aspect of the defect was not covered by the device, and there was residual left-to-right shunt. The defect extended to the basal posterior left ventricular free wall (Figure 1c and 1d) and was limited by the pericardium, though there was no pericardial effusion (Video 1). The residual defect could not be crossed despite multiple attempts from the retrograde and antegrade approaches. Even though the entry point of the defect on the left ventricular end appeared proximal to the deployed device, a hydrophilic guidewire could not be negotiated across the defect, and the procedure was abandoned. The patient succumbed shortly thereafter due to refractory hypotension.
(a) The Transthoracic Echocardiographic Color Doppler Image from the Modified Apical Four-chamber Projection Showing the Ventricular Septal Defect Extending from the Mid-Muscular Septum to the Apical Septum. (b) The Fluoroscopic Still in Left Anterior Oblique 60 Degrees Cranial Angulation 20 Degrees Projection Showing the Deployment of the Muscular Ventricular Septal Defect Device. The Transesophageal Echocardiography in 50 Degrees at the Mid-esophageal Level (2D [c] and 3D [d]) Shows the Ventricular Septal Rupture Closed by the Device and an Additional Defect Superiorly Extending to the Posterobasal Left Ventricular Free Wall (Ventricular Double Rupture).
Types of Ventricular Double Rupture.
Discussion
Free wall rupture with ventricular septal rupture (ventricular double rupture) is rare and is mostly reported after anterior wall myocardial infarction.6, 7 The perforation site is most commonly located adjacent to the apical septum. 6 Ventricular double rupture tends to occur in the basal posterior wall following inferior wall myocardial infarction and can be missed when associated with a basal ventricular septal rupture.
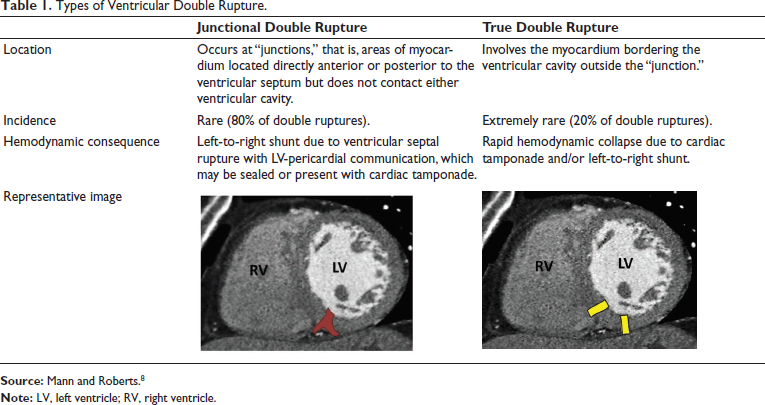
Ventricular double ruptures are classified into “junctional” and “true” ruptures (Table 1). 8 The former is the more common of the two and refers to defects located directly on the anterior or posterior ends of the interventricular septum, resulting in interventricular communication and a left-ventricle-to-pericardial communication. “True double ruptures,” like in the index case, involve extensive rupture of the interventricular septum outside the junction area and have associated free wall ruptures.
True ventricular double rupture carries a high risk and is unsuitable for percutaneous device closure. Surgical mortality is also exceedingly high in ventricular double rupture, with posterobasal ruptures faring worse. There are only rare reports of survival with surgery in posterobasal ventricular double rupture. 9 The key to mitigating the risk lies in the prevention of mechanical complications by reducing the time to myocardial revascularization in acute myocardial infarction and continued monitoring in the first 72 hours after primary angioplasty. Early identification is paramount. The use of mechanical circulatory support using IABP and/or extracorporeal membrane oxygenation followed by surgical repair is the treatment of choice. Mechanical circulatory support allows stabilization of hemodynamics, recovery or prevention of end-organ injury, and a strategy of bridge-to-decision.
Conclusions
Careful echocardiographic screening of the irregular edges of ventricular septal ruptures can help identify ventricular double rupture, a rare lethal complication of myocardial infarction. 3D transesophageal echocardiography is a useful tool in diagnosing double ventricular rupture. While its diagnosis is often challenging, in the absence of associated pericardial effusion or pseudoaneurysm, this should constitute a definite contraindication for percutaneous closure of the defect. Prompt mechanical circulatory support followed by high-risk surgery serves as the only method to salvage these patients.
Footnotes
Authors’ Contributions
Rohit Sunil Walse and Arun Gopalakrishnan prepared the manuscript.
Bijulal Sasidharan contributed to the data acquisition and edited the manuscript.
All authors approved the final version.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later revisions.
Informed Consent
Written informed consent was obtained from the close relative of the patient for being included in the case report. No patient identity particulars have been disclosed.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
