Abstract
Dual-antiplatelet therapy (DAPT) is a cornerstone treatment for patients diagnosed with acute coronary syndrome (ACS) and coronary artery disease (CAD). Although DAPT effectively reduces the occurrence of thrombotic events, it also substantially increases the likelihood of gastrointestinal (GI) bleeding. Proton pump inhibitors (PPIs) are essential for mitigating the GI bleeding risk, yet concerns persist regarding their prolonged use and potential adverse effects. Apart from rabeprazole, PPIs share the hepatic metabolism pathway with clopidogrel, resulting in unfavorable cardiovascular outcomes. The pairing of rabeprazole with clopidogrel emerges as a prudent recommendation, displaying superior efficacy in mitigating major adverse cardiovascular events (MACE) when juxtaposed with other PPIs. This narrative review underscores the complexity of managing PPIs in cardiac patients and emphasizes the importance of tailored approaches based on emerging evidence while providing valuable insights to clinicians navigating this challenging therapeutic landscape.
Background
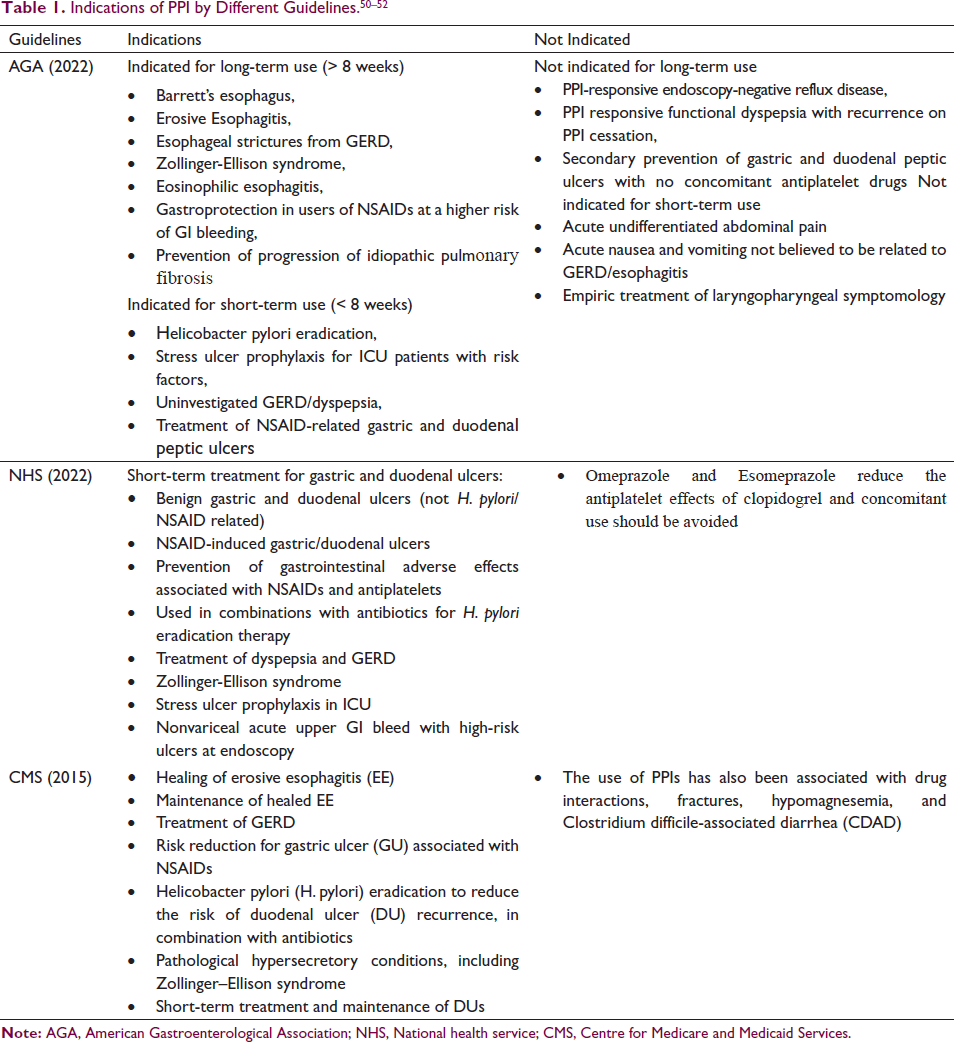
Dual-antiplatelet therapy (DAPT) is recommended as a long-term measure for patients with acute coronary syndrome (ACS) and coronary artery disease (CAD) who undergo either coronary artery bypass grafting (CABG) or percutaneous coronary intervention (PCI), to prevent thrombotic complications and stent thrombosis. DAPT typically comprises low-dose aspirin along with inhibitors of the P2Y12 receptor such as prasugrel, ticagrelor, or clopidogrel which together have a synergistic anti-thrombotic effect. 1 However, these antiplatelet drugs significantly increase the risk of gastrointestinal (GI) bleeding.2, 3 To prevent GI bleeding, proton pump inhibitors (PPIs) are advised in addition to DAPT. 4 In addition, they are commonly utilized for the entire gamut of gastric-acid-related conditions, such as dyspepsia, peptic ulcer disease, and gastroesophageal reflux disease (GERD) (Table 1). For such conditions, they are not only sold as prescription drugs but also as over-the-counter drugs.5, 6 Initially, they were investigated and authorized for the short-term treatment of conditions associated with gastric acid, limited to a 4-week period, with a maximum of only 2 treatment cycles per year. 7 However, in practice, there has been a significant rise in their prolonged use, surpassing the duration range approved by the US Food and Drug Administration (FDA). Over the years, accumulating data from several studies has highlighted the risk of multiple serious adverse outcomes due to the extended use of PPIs. 8
PPIs have performed better than earlier drugs, including anticholinergics, histamine-2-receptor antagonists (H2RAs), and synthetic prostaglandin analogs, in terms of decreasing acid production while also having a noteworthy safety profile and decent tolerability. Unlike other medications, they can successfully control gastric pH >4 for 15 to 21 hours per day. 6 The reported decline in the effectiveness of clopidogrel, a crucial component of DAPT, in patients who were also given PPI, has raised serious concerns. Numerous observational studies conducted over the past 10 years have shown that the H+-K+ ATPase inhibitor use is linked to an elevated risk of cardiovascular disease (CVD) events requiring re-hospitalization and higher mortality among patients with a prior history of CVD. This is believed to be due to the drug-drug interactions (DDI) between the proton pump inhibitors and clopidogrel caused by competition for the same pathway (cytochrome P450).9–14 Although all studies have not demonstrated an elevated risk of major adverse cardiovascular events (MACE) associated with PPI use, prolonged administration (≥ 6 months) tends to show an elevated risk in most cases. 8 In a review of 40 studies, Li et al. found that a combination of the proton pump inhibitors and DAPT showed no association with an increased risk of adverse clinical outcomes when analyzing data from randomized controlled trials (RCTs). However, when considering data from observational studies, an elevated risk of adverse clinical outcomes was observed in patients, although the heterogeneity among these studies was significant. 15 Similar findings were reported in a meta-analysis conducted by Khan et al. 16 involving 39 studies (comprising 4 RCTs and 35 observational studies) on subjects with CAD. Shang et al. in a meta-analysis involving 43,943 patients from 19 RCTs, also reported that a combination of PPIs with antithrombotic strategies was not found to have a negative impact on the occurrence of MACE, stent thrombosis, myocardial infarction, cardiovascular death or all-cause death. Moreover, this outcome was observed in patients who received lansoprazole, rabeprazole, pantoprazole, or omeprazole, both non-Asian and Asian patients, and patients taking a combination of clopidogrel and aspirin. The level of evidence, assessed using the GRADE system, was moderate for all reported outcomes. 4 In contrast, Pang et al. carried out a systematic review and meta-analysis that included 15 RCTs and 50,366 patients, and the results demonstrated that patients who received clopidogrel alone (non-PPI group) had a lower likelihood of fatal strokes, target vessel revascularization (TVR), stent thrombosis, myocardial infarction recurrence, and MACE than those who received the combination. However, both groups showed similar rates of bleeding events, all-cause death, and cardiovascular death. 17 Healthcare professionals are frequently faced with the issue of weighing and analyzing the risk-to-benefit ratio for each patient and developing a personalized method based on clinical evidence because of the inconsistent results that have been reported by studies on patient characteristics, management approach, and clinical presentation. It is crucial to note that in 2009, the FDA also issued a black box warning specifically cautioning against the concomitant use of clopidogrel with omeprazole. 18 This underscores the importance of careful consideration when prescribing medications, especially in situations where potential interactions may impact patient outcomes. In this review we look at the differences between the various proton pump inhibitors in terms of their likely interactions with DAPTs, the recommendations by various guidelines, attempts at some practical approaches by clinicians, and the opinions of some experts on the issue. The review process began with a comprehensive search of relevant literature from the past 15 years. Databases such as PubMed, Google Scholar, and Science Direct along with other academic databases were systematically explored to identify publications related to the role of PPIs in cardiac patients receiving DAPT.
Similarities and Differences of Individual PPIs in Cardiac Patients
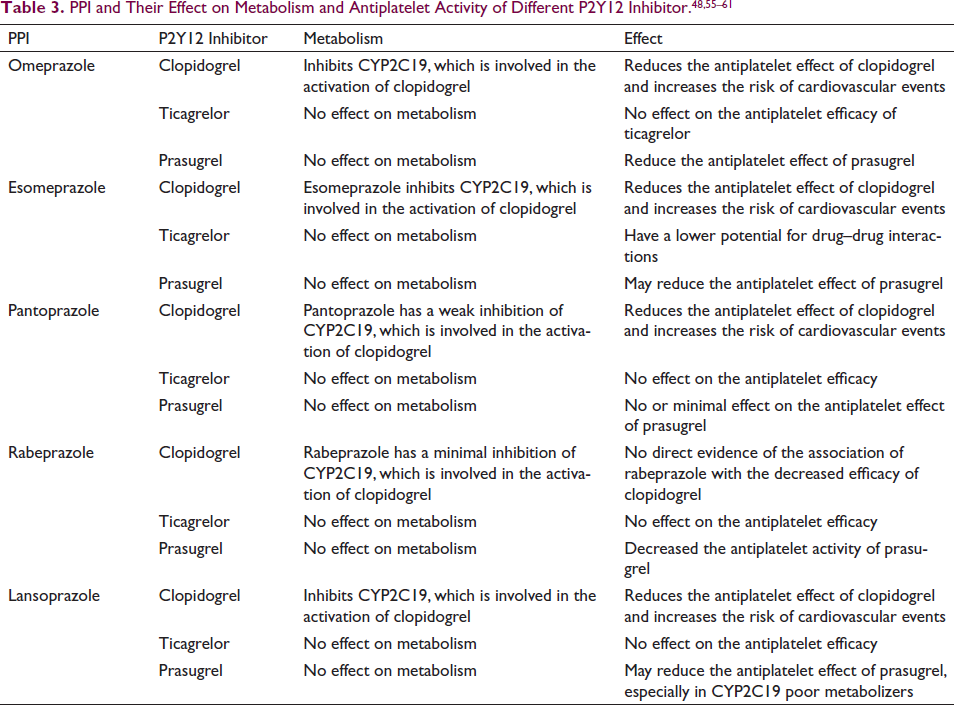
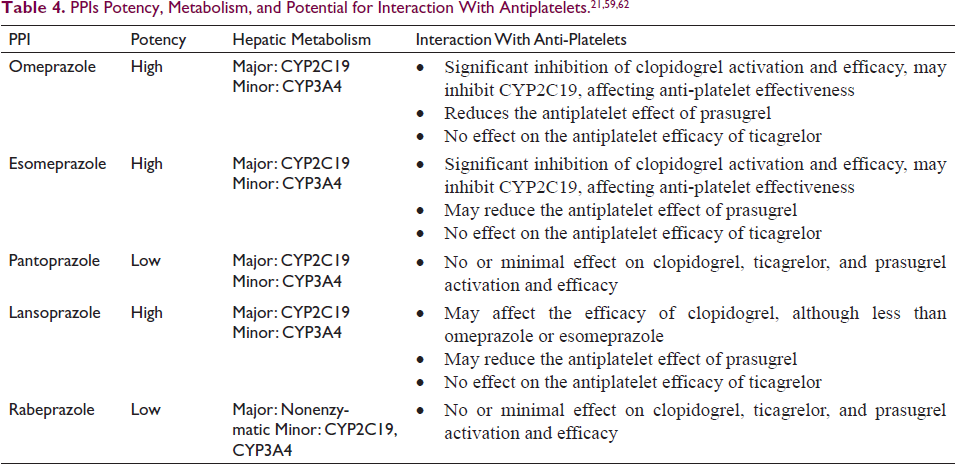
All the H+-K+ ATPase inhibitors, except rabeprazole, are extensively metabolized in the liver, mainly via the hepatic CYP2C19 and CYP3A4 enzymes. 19 Clopidogrel is also transformed to its active metabolite by the CYP2C19 enzyme. Hence, there is competitive behavior between clopidogrel and PPIs for the CYP2C19 enzyme. 20 However, differences in the extent of metabolism by the enzyme CYP2C19 result in variation in their pharmacodynamics and pharmacokinetic characteristics and the extent to which each competes with clopidogrel for CYP2C19. 21 The impact of each PPI on CYP metabolism varies, leading to potential differences in clopidogrel metabolism and subsequent cardiovascular outcomes. 22
Omeprazole is almost completely metabolized in the liver by CYP3A4 and CYP2C19; however, the role of CYP2C19 is predominant. 19 Esomeprazole undergoes relatively lower metabolism by CYP2C19 as opposed to omeprazole. CYP2C19 is responsible for around 70% clearance of esomeprazole and approximately 90% of the clearance of omeprazole. 21 Esomeprazole is available in two salt form, esomeprazole magnesium and esomeprazole strontium. Magnesium salt can improve the stability and absorption of the drug. The stability of esomeprazole magnesium depends on the pH of the environment. It degrades quickly in acidic conditions but remains stable under alkaline conditions. In a pH 6.8 buffer, the half-life of the magnesium salt is approximately 19 hours at 25°C and around 8 hours at 37°C. 23 Esomeprazole strontium, an incrementally modified version of esomeprazole magnesium, uses strontium salts instead of magnesium salts. Esomeprazole strontium and esomeprazole magnesium are considered bioequivalent, as defined by the FDA. This means there’s no significant difference in the rate and extent of availability of the active ingredient at the site of drug action when administered at the same molar dose under similar conditions. Bioequivalence was demonstrated with 49.3 mg esomeprazole strontium achieving a Cmax at approximately 1.7 hours compared to 1.6 hours for esomeprazole magnesium 40 mg. 24 Pantoprazole and lansoprazole are also metabolized by CYP3A4 and CYP2C19. 25 Pantoprazole is primarily metabolized by CYP2C19, which accounts for more than 80% of its metabolism, with the remaining portion metabolized by CYP3A4. 26 In contrast to omeprazole and esomeprazole, pantoprazole exhibits a metabolic profile where the influence of CYP2C19 is significant. However, lansoprazole has a weaker affinity substrate to CYP2C19 compared to omeprazole. Thus, omeprazole and esomeprazole have the highest propensity for drug interaction. 6 Rabeprazole undergoes non-enzymatic reduction as its primary metabolic pathway, and the contribution of CYP2C19 and CYP3A4 to this pathway is minor. 19 Rabeprazole is a weak competitive CYP2C19 inhibitor and has negligible impact on the metabolizing process of clopidogrel when viewed alongside the remainder of the 4 PPIs. 20
Dexlansoprazole modified release (MR) is an R-enantiomer of lansoprazole and represents a new generation of PPIs known for its effectiveness in managing symptoms and lesions associated with erosive esophagitis caused by GERD. It employs a unique dual delayed-release (DDR) formulation, which extends the duration of acid suppression by prolonging plasma concentration. This dual release occurs in the duodenum and the small intestine, leading to two peak concentrations at different times, typically within two and five hours post-administration. Dexlansoprazole exhibits a slower elimination rate than S-lansoprazole and remains in the bloodstream for a longer duration. Metabolism primarily occurs in the liver, involving oxidation and reduction followed by conjugation with sulfates, glucuronate, and glutathione to produce inactive metabolites. Oxidation metabolites are generated through the cytochrome P450 (CYP) enzymatic system, predominantly via hydroxylation by CYP2C19 and oxidation to sulphone by CYP3A4. 27 A new benzimidazole compound, ilaprazole, also known as IY81149, was recently synthesized by Il-Yang Pharmacy Co. in Seoul, Korea. Ilaprazole demonstrates significant and dose-dependent suppression of gastric acid secretion, along with a prolonged plasma half-life and comparable safety profile to omeprazole. 28 According to a study conducted by Li et al., there is no apparent correlation between ilaprazole metabolism and CYP2C19 genotypes. 29 Variations in CYP3A5 or CYP2C19 genotypes do not seem to impact ilaprazole metabolism significantly. Ilaprazole is primarily metabolized by CYP3A, with only partial involvement of CYP2C19. Therefore, the influence of CYP450 polymorphism on ilaprazole pharmacokinetics is less pronounced compared to previous PPIs. 28
A systematic review conducted by Kwok et al. involved 23 studies to find any distinct intra-class variations between the PPIs for the hypothesized clopidogrel interaction. However, 19 of these were cohort studies, and there were only 2 RCTs. Each PPI was found to have statistically significant adverse effects, as demonstrated by two or more studies. 30 However, the aggregated mean estimates of the elevated risk observed for every proton pump inhibitor were limited due to moderate to significant variations between the investigations. Moreover, a meta-analysis of seven observational studies solely focused on PPI therapy (without concurrent clopidogrel use) also revealed an elevated odds ratio for adverse cardiovascular use. Consequently, the researchers concluded that the increased cardiovascular risk associated with PPIs, even in clopidogrel absence; suggests a strong likelihood of confounding and bias in the data. They emphasized that the clinical significance or relevance of the presumed interaction between clopidogrel and PPIs remains uncertain and wants further investigation. 30 In a later study, the COGENT RCT, compared omeprazole 20 mg and clopidogrel 75 mg combination with clopidogrel 75 mg alone. Every study participant received enteric-coated aspirin. In patients who underwent PCI, omeprazole was found to significantly decrease the occurrence of combined GI events, while not leading to an increase in combined cardiovascular events. In patients presenting with ACS, Omeprazole was found to decrease the risk of primary GI events without causing a significant increase in cardiovascular events. 31 However, Niu et al. conducted a meta-analysis comprising 21 studies, and their findings indicated that pantoprazole, esomeprazole, lansoprazole, and omeprazole increased the risk of MACE except rabeprazole. 20 Yasu et al. conducted a comparison between patients receiving DAPT with clopidogrel and aspirin after drug-eluting stent (DES) implantation and those who were only on DAPT to determine the effectiveness and safety of rabeprazole. Patients who had more severe coronary artery lesions in the rabeprazole group required more DES. However, co-administration of rabeprazole was not associated with an increased risk of stent thrombosis. The use of rabeprazole did not increase the incidence of MACE. 32 Similarly, Hokimoto et al. measured the platelet aggregation and assessed platelet reactivity 24 hours after concomitant clopidogrel and rabeprazole administration vs. clopidogrel alone. Over 18 months of follow-up, they assessed the likelihood of cardiovascular events as well. Cardiovascular events did not differ between participants using rabeprazole and the ones who did not take rabeprazole. Additionally, there were no variations in the degree of platelet reactivity between the rabeprazole and non-rabeprazole groups. 33 It can be seen that various studies and meta-analyses over the years have reported conflicting outcomes regarding the risks of DDI between clopidogrel and each PPI, although no study has reported any negative interaction between DAPT and rabeprazole.
The clopidogrel-PPI interaction also varies with varying CYP2C19 genotypes. Individuals having the allele CYP2C19*2 and/or CYP2C19*3 are weak metabolizers of clopidogrel. Such individuals have significantly reduced enzyme activity and cannot activate clopidogrel via CYP2C19, leading to a reduced antiplatelet effect of the drug. Moreover, these individuals require higher doses of clopidogrel and more frequent dosing for PPI. 34 Rabeprazole is not metabolized by the CYP2C19 enzyme and has minimal impact on its activity, thus it is devoid of the genetic polymorphism associated with CYP2C19*2 and*3 alleles. This characteristic makes Rabeprazole a safer option for individuals with the CYP2C19 poor metabolizer genotype as it does not interfere with the activation and efficacy of clopidogrel and it does not require dose adjustments or impact the antiplatelet effects of the drug in such individuals.35, 36
Guidelines About the Co-prescription of DAPT and PPI
According to the 2010 Expert Consensus jointly formed by the American College of Cardiology Foundation (ACCF), American College of Gastroenterology (ACG), and American Heart Association (AHA) (Table 2), proton pump inhibitors should be administered exclusively to patients at a higher risk of bleeding.37, 38 The 2016 guideline from the ACC/AHA also emphasized this approach, highlighting their utilization only in high-risk patients. The guidelines suggest that individuals who have previous instances of GI bleeding managed using DAPT (Class I) need to take PPIs. The use of PPIs (Class IIa) is appropriate in individuals who have a higher risk of GI bleeding, such as those who are elderly or who are taking warfarin, nonsteroidal anti-inflammatory medications, or steroids concurrently. Patients with a low risk of GI bleeding should not regularly take PPIs (Class III: No Advantages). 39
As per the European Society of Cardiology (ESC) 2015, regulations for managing acute coronary syndromes in individuals displaying without persistent ST-segment elevation, PPI with DAPT was only advised in patients with a greater-than-average likelihood of GI bleeding (i.e., history of gastrointestinal ulcer/hemorrhage, age ≥65 years, chronic alcohol use, Helicobacter pylori infection, GERD, dyspepsia, chronic NSAID/ corticosteroid use, anticoagulant therapy). 40 In contrast, to the 2017 ESC guidelines on DAPT in CAD, it is recommended to routinely use for all patients on DAPT. It is noted that esomeprazole and omeprazole show the highest likelihood for clinically significant interactions, while rabeprazole and pantoprazole exhibit the lowest propensity for such interactions. 41
As per the 2020 International Consensus Recommendations for the Management of Patients with Nonvariceal Upper GI Bleeding, patients with a history of ulcer bleeding who are on cardiovascular prophylaxis with either single- or dual-antiplatelet treatment should receive PPI medication. 42 The Italian Association of Hospital Gastroenterologists (AIGO) and the National Association of Hospital Cardiologists (ANMCO) also advise the use of PPI in patients taking single antiplatelet agents if there are existing GI risk factors. These risk factors include patients who have history of Peptic Ulcer Disease (PUD) and receiving antiplatelet therapy, concomitant use of vitamin K-antagonists (VKAs), concomitant use of another antiplatelet agent, non-steroidal anti-inflammatory drugs (NSAIDs), direct-acting oral anticoagulants (DOACs), or steroids should be further taken into consideration for PPI use due to the increased risk of bleeding. Other risk factors include GERD, dyspeptic symptoms, and age over 65 years. 1
Expert Opinions and Practical Approach
Among the most frequent causes of early termination of antiplatelet treatment is gastrointestinal bleeding. Discontinuation of DAPT owing to failure to comply or bleeding greatly increases the likelihood of detrimental results after PCI. GI bleeding is closely related to composite ischemia in ACS patients, cardiac mortality, and 30-day all-cause mortality. 43 Hence, a practical approach to balance the need for compliance with DAPT and minimizing the purported risk of DDI between DAPT and PPI is necessary. However, there is real-world data on the rate of PPI co-prescription with DAPT. Only 35% of individuals at greater risk of bleeding from the GI tract took the prescribed PPI and DAPT treatment, according to a Danish registry. 44 Only 62% of the patients in Shen et al.’s cohort who received DAPT at a tertiary cardiac center in Montreal, Canada, additionally received PPI medication. 45
The Royal Wolverhampton Hospitals in the UK undertook a quality improvement project to highlight the present compliance rates of co-prescription of PPI with DAPT and introduce effective interventions to ensure a 100% co-prescription. Low-cost measures were implemented, including classes for trainee doctors, cardiac nurses, and pharmacists. Additionally, posters were displayed at appropriate places, which served as visual prompts for the treating clinicians. They also devised a standard procedure that the pharmacy staff cross-checked with the treating physicians to determine whether a patient who has been prescribed DAPT should also be on PPI. Within 14 weeks of initiating the project, the co-prescription of PPI with DAPT increased from 52% to 100%. 43
Improvements in coronary stent technology have gradually reduced the time frame of DAPT. Therefore, present regulations for stable CAD suggest a minimum 6-month DAPT for patients receiving drug-eluting stents (DES) and a minimum 1-month DAPT for patients receiving bare-metal stents. The suggested duration of DAPT for patients with ACS receiving PCI is at least 12 months. DAPT may be prolonged to >12 months (30-36 months) in patients who have minimal bleeding risk, or it may be limited to 6 months in patients with an elevated bleeding risk (or to 3 months in extremely elevated bleeding-risk patients). 41 The shortened duration of DAPT also reduces the duration of concomitant PPI intake. Some clinicians have also tried to use H2-blockers instead of PPIs in patients on DAPT. However, it was seen that PPIs provide better gastric acid suppression for a more prolonged period than H2-blockers, although H2-blockers are efficient in averting GI complications for individuals taking antiplatelet medications for vascular protection. Due to this, ulcer healing rates are improved, and gastroprotection is increased. PPIs provide better defense against GI side effects than H2-blockers in people who are prescribed DAPT. 46 In such cases where PPIs are necessary to prevent GI complications and provide gastroprotection in patients on DAPT, rabeprazole could be a preferred option.
Several expert recommendations have been published to guide clinicians on strategies they can follow to prescribe PPIs in combination with DAPT. Some experts have suggested that ticagrelor or prasugrel might be used instead of clopidogrel in some patients to prevent DDI (Table 3). Ticagrelor is a more effective antiplatelet agent than clopidogrel and is not dependent on metabolic activation to show its effects. Prasugrel has a more straightforward hepatic activation process, requiring fewer steps, and is more potent than clopidogrel. However, the increased potency of the antiplatelet effect of these agents also leads to a higher risk of bleeding. 1 Moreover, despite fewer steps, prasugrel still requires CYP2C19 to be metabolized into its active form, and there is inadequate evidence about the interaction between prasugrel and PPIs. A study reported that the use of proton pump inhibitors did not have a significant impact on the comparative effectiveness or bleeding risk of prasugrel compared to clopidogrel. 47 Patients with ACS were prescribed a combination of H+-K+ ATPase inhibitors along with clopidogrel or ticagrelor, a study revealed that there were no significant differences observed in the risk of severe bleeding, re-infarction, all-cause death, and GI bleeding between the two groups.48, 49
If PPIs are prescribed, according to Ariel and Cook, they should only be used for highly particular indications and for a brief period (4 weeks, followed by 1 to 2 weeks of gradually reducing the dose) for which there might not be an effective substitute therapy—cases such as GERD, gastric and duodenal ulcers, and Helicobacter pylori. They are commonly prescribed in patients with cardiac disorders for gastroprotective purposes, especially in conjunction with DAPT or other medications used in the treatment of cardiovascular diseases. As discussed earlier, the duration of DAPT has been adjusted based on advancements in coronary stents, often leading to concomitant PPI use to reduce GI complications. Nevertheless, it is imperative to regularly review and document the indications for long-term PPI use in cardiac patients, considering the potential for associated adverse events. In cases where de-prescribing is considered, this becomes particularly relevant for cardiac patients who may already be on multiple medications for their cardiovascular condition. A judicious approach involves selecting a PPI with minimal inhibitory effects on CYP2C19, such as rabeprazole, to optimize gastro protection while minimizing potential drug interactions (Table 4).
When a combination of antiplatelet therapy and gastric mucosal protection is needed, the use of rabeprazole with clopidogrel is suggested, as this strategy circumvents the clopidogrel-PPI interaction related to an elevated possibility of MACE observed with some PPIs. By opting for rabeprazole, healthcare providers can offer effective gastro protection without compromising the antiplatelet effects of clopidogrel or other CYP2C19-metabolized medications, thus promoting better patient outcomes. When deciding whether to de-prescribe PPIs needs to be driven primarily in the absence of clear evidence, the unique pharmacological characteristics of rabeprazole may offer an additional advantage in cardiac patients, particularly those receiving concurrent antiplatelet therapy.
Conclusion
The management of PPIs in cardiac patients undergoing DAPT represents a nuanced balance between gastrointestinal protection and potential drug interactions. While concerns persist regarding the potential for adverse interactions between PPIs and clopidogrel, particularly with regard to cardiovascular outcomes, emerging evidence suggests that individual proton pump inhibitors exhibit varying degrees of metabolic interaction. Notably, rabeprazole emerges as a promising option due to its unique metabolic profile, minimizing interference with clopidogrel antiplatelet efficacy. Guidelines offer varying perspectives on PPI use in cardiac patients on DAPT, ranging from selective application to routine prescription. Although most International guidelines recommend co-prescription of PPIs to only those who are at higher risk of GI bleeding. Practical strategies, such as considering alternative antiplatelet agents and reviewing long-term PPI indications, are essential for optimizing patient care while minimizing potential risks.
Footnotes
Acknowledgements
The authors thank Shalaka Marfatia of pharmEDGE and her team for their assistance in conducting the research, developing the model, and drafting the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The article does not involve animal and human studies, so no ethical clearance was obtained.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
This article does not involve human studies, so patient written informed consent was not obtained.