Abstract
The population with chronic kidney disease (CKD) is unique in the sense that they are more prone to heart failure and ischemic heart disease. This is mainly due to the presence of hypertension, anemia, and associated fluid overload, which is common to them. Managing heart failure in this cohort of patients is challenging as treatment of heart failure leads to worsening of kidney parameters.
Keywords
Chronic kidney disease (CKD) is increasingly prevalent in patients with heart failure (HF), and HF is one of the leading causes of hospitalization, morbidity, and mortality in patients with an impaired renal function. There is a significant co-occurrence of HF and CKD—it is reported that almost half of patients with HF have a degree of renal impairment, and HF is prevalent in 17%–50% of patients with CKD, depending on the stage of the CKD and age of the patients.1, 2 In addition, the prevalence and mortality of HF increase with worsening renal failure. 3 Renal function is an independent predictor for inpatient mortality of patients with acute HF, length of hospital stay, and re-admission rate. 3
With improving survival in both patients with HF and those with CKD, it is likely that the numbers of patients presenting with both these conditions will continue to rise.
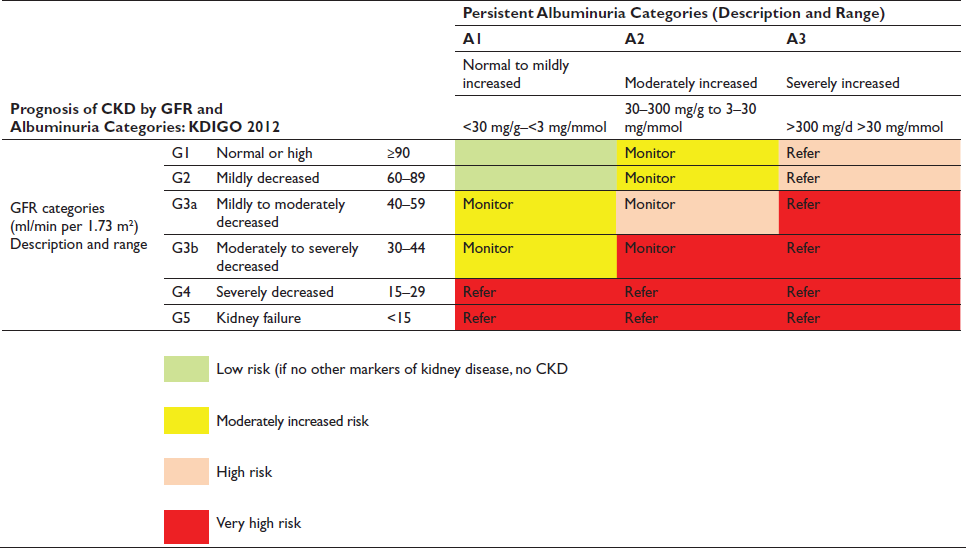
Case 1: A 56-year-old man is admitted to the hospital with worsening dyspnea and 8 kg weight gain and is diagnosed with acute decompensated heart failure (ADHF). He has a history of hypertension, type 2 diabetes, coronary artery disease, chronic kidney disease (CKD), glomerular filtration rate (GFR) category 3b and albuminuria category 2 (G3bA2), and ischemic cardiomyopathy with an ejection fraction of 30%. He is adherent to prescribed medications, including losartan, furosemide (40 mg twice daily), atorvastatin, and insulin. His blood pressure (BP) is 162/92 mm Hg and heart rate is 104 beats per minute. Physical examination reveals an S3 gallop, bilateral crackles, and pitting edema (3+). Admission laboratory data include serum sodium level of 132 mEq/L, serum potassium level of 5.2 mEq/ L, serum urea nitrogen level of 63 mg/dL, and serum creatinine (Scr) level of 2.1 mg/dL (baseline, 1.4 mg/dL).
Question 1: Which of the following is the next best step in management? Prescribe furosemide intravenous (IV) infusion Prescribe metolazone Prescribe dapagliflozin Prescribe isolated ultrafiltration Discontinue losartan
Volume overload is defined as excess total body sodium and water with expansion of extracellular fluid volume. Volume overload characterizes several common disorders, including congestive heart failure (CHF), cirrhosis or end-stage liver disease (ESLD), CKD (including kidney failure), and nephrotic syndrome. Diuretics are the cornerstone of therapy for volume overload. There are several classes of diuretics, whose mechanisms of action, pharmacokinetics, and clinical use constitute basic principles of nephrology. The patient in case 1 presents with ADHF associated with resistance to oral furosemide. The best next step in management is changing furosemide from oral to IV dosing, thereby bypassing slowed absorption from gastrointestinal (GI) edema. Additionally, most contemporary guidelines suggest using 2–2.5 times the home dose (here, 80–100 mg IV). Alternatively, furosemide could be changed to an equivalent dose of bumetanide or torsemide; some small studies have shown better intermediate outcomes with torsemide for symptomatic decongestion, but large outcome studies conflict as to whether loop diuretic choice affects long-term outcomes, including mortality. Metolazone could be added to block distal tubule sodium reabsorption in conjunction with, but not in place of, IV furosemide. Dapagliflozin improves outcomes in CHF but would not replace an IV loop diuretic for prompt decongestion. Isolated ultrafiltration removes fluid as effectively as furosemide but is associated with more frequent adverse events. Stopping losartan may be necessary if acute kidney injury (AKI) or hyperkalemia worsen, but these conditions frequently improve with diuresis alone. So, the correct answer to question 1 is (a).
Case 1, continued: Furosemide 40 mg IV followed by an infusion of 5 mg/h is administered, and urine output increases to 2.4 l/d. After 2 days, the patient’s dyspnea and peripheral edema have improved, and BP has decreased to 146/84 mm Hg. Scr has also decreased from 2.1 to 1.7 mg/dL. Preparing the patient for discharge, the inpatient team converts medications from IV to oral dosing and schedules primary care follow-up within 1 week.
Question 2: Which of the following is the most appropriate diuretic regimen for discharge?
Furosemide 120 mg twice daily and metolazone 10 mg/d Furosemide 80 mg twice daily Furosemide 160 mg/d Bumetanide 4 mg twice daily Bumetanide 4 mg/d
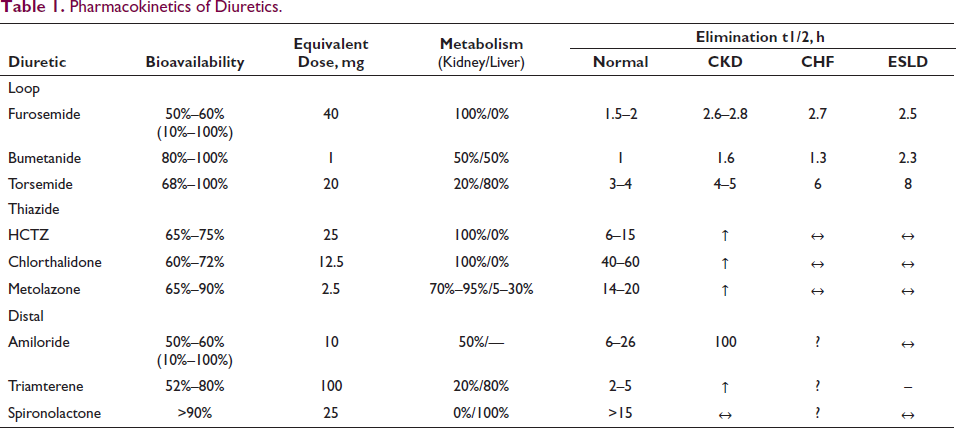
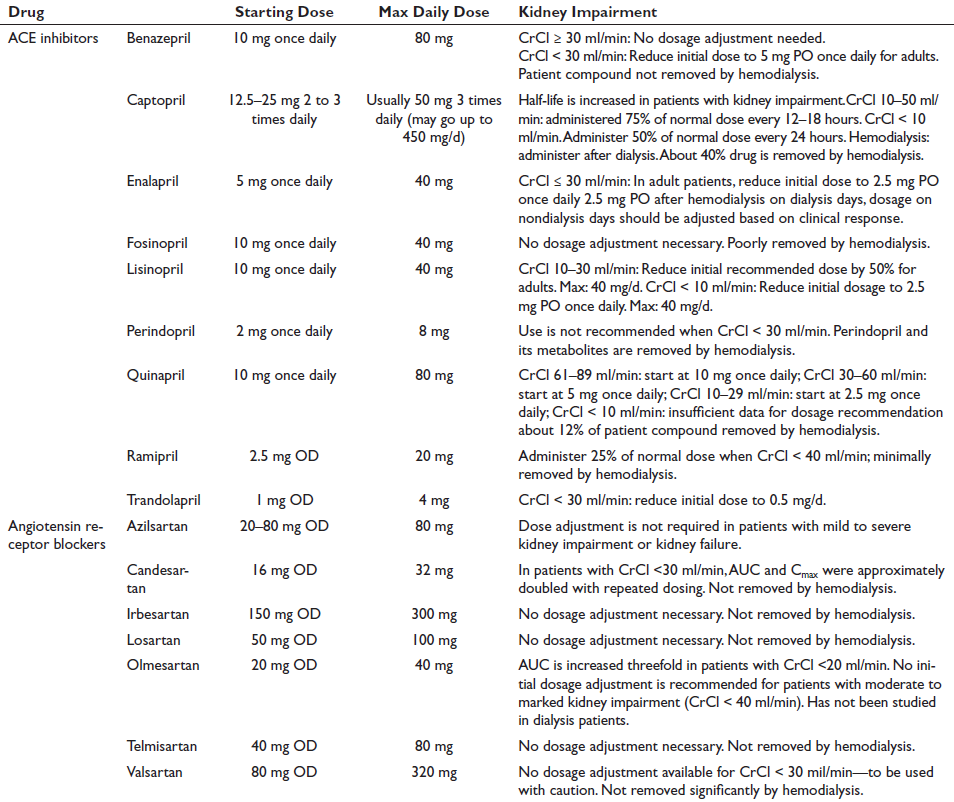
Pharmacokinetics describe how a drug is absorbed, distributed, metabolized, and eliminated by the body. For example, furosemide is absorbed similarly in the stomach and duodenum, although gastric absorption is slow and includes first-pass metabolism. This effect may account for the lower (50%–60%) bioavailability of furosemide compared with other loop diuretics such as bumetanide and torsemide (>80%). Such differences in bioavailability have implications for IV-to-oral dosing conversions: generally, the furosemide dose should be doubled when making this change, whereas bumetanide and torsemide doses should stay the same, although these rules are only approximations. Ultimately, the effective dose is determined by the urine output response. GI tract edema decreases the velocity but not the total amount of furosemide absorbed. However, this slowed absorption may prevent orally administered furosemide from reaching its threshold plasma concentration (Figure 1(A)). Finally, the dose-response curve for loop diuretic agents is sigmoidal and logarithmic, so exponential dose increases may be needed for patients who do not show a response to an algorithmic or empirical dose (Figure 1(B)). Conversely, doses that yield plasma concentrations greater than the ceiling may increase toxicity without increasing the response. Once absorbed, most diuretics bind to serum albumin for distribution, followed by metabolism and elimination by the liver and kidneys. Hypoalbuminemia and impaired renal blood flow (as in AKI and CKD) decrease diuretic delivery, so albumin and loop diuretic coinfusion has been proposed to improve drug response, but this practice has not been beneficial in critically ill adult patients. Furosemide is metabolized completely by the kidneys (35% furosemide glucuronide and 65% parent molecule excretion), whereas torsemide is metabolized mainly by the liver. Triamterene is a prodrug that requires hepatic activation to hydroxytriamterene, so it is relatively ineffective in ESLD. Routes of diuretic metabolism and elimination may be important considerations for patients with kidney or liver disease (Table 1). Peak serum concentrations of loop diuretics occur within 0.5–2 hours, even though drug effects may last for 6–8 hours, especially with impaired kidney, heart, or liver function. The short elimination half-life (t1/2) of most loop diuretics, except possibly torsemide, means they are typically dosed at least twice daily. Despite this short t1/2, more frequent administration than twice daily is usually unnecessary because sodium intake is zero during sleep. Conversely, if loop diuretics are prescribed only once daily, the kidneys are diuretic free for many hours, allowing considerable rebound sodium reabsorption in most cases. On the contrary, thiazide and distal diuretics have much longer t1/2 values and may be given daily. A new extended-release preparation of torsemide has been approved by the US Food and Drug Administration (FDA) for the treatment of edema from heart and kidney failure. In case 1, the patient was initially prescribed a furosemide loading dose of 80 mg IV to rapidly achieve a therapeutic serum level, followed by a maintenance dose of 5 mg/h (120 mg/d) IV to maintain a steady state; this management strategy has effectively achieved decongestion. This dose is equivalent to 240 mg/d orally, which should be given as 120 mg twice daily to avoid postdiuretic sodium reabsorption. However, because this patient’s signs and symptoms of ADHF and AKI resolved and 2.4 L/d urine output is no longer necessary, a lower oral dose of furosemide, such as 80 mg twice daily, may be appropriate. Furosemide 120 mg twice daily with metolazone 10 mg/d would further increase diuresis and could cause hypovolemia and/or hypokalemia. Similarly, bumetanide 4 mg orally twice daily, which is equivalent to furosemide 160 mg IV twice daily or 320 mg orally twice daily, would also risk hypovolemia. Unless sodium intake can be severely restricted, neither furosemide nor bumetanide should be dosed once daily. The correct answer to question 2 is (b). 4
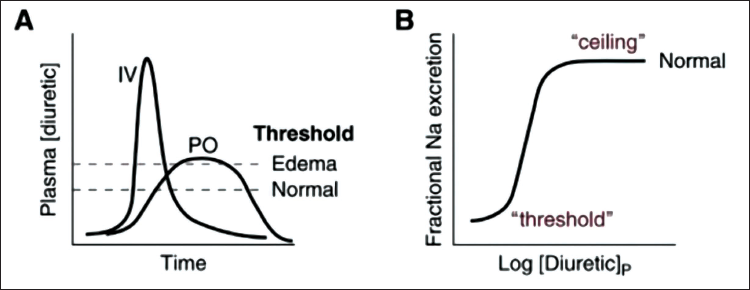
Pharmacok inetics of Diuretics.
Case 1, continued: He is adherent to prescribed medications, including losartan, furosemide (80 mg twice daily), atorvastatin, and insulin. His BP is 162/92 mm Hg and heart rate is 104 beats per minute. What should be the next step for better management ?

Treatment with beta blockers reduces the risk of death and the combined risk of death and hospitalization in patients with HFrEF.5–7 In addition, this treatment can improve LVEF, lessen the symptoms of HF, and improve clinical status.5–10 Clinical trials have shown that beta blockers should be prescribed to all patients when HFrEF is diagnosed, including in-hospital, unless contraindicated or not tolerated.5–10 These benefits of beta blockers were observed in patients with or without CAD, and in patients with or without diabetes, in older patients, as well as in women and across racial and ethnic groups but not in patients with AF.5–7,9–11 Even if symptoms do not improve, long-term treatment should be maintained to reduce the risk of major cardiovascular events. Beta blockers should be initiated at low doses, and every effort should be made to achieve the target doses of the beta blockers shown to be effective in major clinical trials, as tolerated.1–8
Three beta blockers have been shown to be effective in reducing the risk of death in patients with HFrEF: bisoprolol, sustained-release metoprolol (succinate), and carvedilol.5–7 The favorable findings with these three agents, however, should not be considered a beta-blocker class effect in HFrEF. Other beta blockers are not included in this recommendation for use.12–14 Even when asymptomatic, or when symptoms are mild or improve with other therapies, beta-blocker therapy is important and should not be delayed until symptoms return or disease progression is documented. 15
The Metoprolol CR/XL Randomized Intervention Trial in Chronic HF (MERIT-HF) randomized patients with symptomatic HFrEF to metoprolol or placebo and included many patients with eGFR < 45 ml/min per 1.73 m2. The hazard ratio for total mortality was 0.41 in favor of metoprolol for the CKD subgroup and demonstrated higher risk reduction than did the reference group with eGFR > 60 ml/min per 1.73 m2. 16
A similar analysis was performed in the Cardiac Insufficiency Bisoprolol Study II (CIBIS-II), which included patients having a serum creatinine level up to 300 mmol/l (3.4 mg/dl) and demonstrated sustained benefit of bisoprolol with worsening kidney function. 17
A small study of carvedilol in patients with dialysis-dependent CKD and HFrEF also conferred a mortality benefit. 18 Therefore, it seems reasonable to use beta blockers for managing HFrEF in patients with CKD, except for beta blockers that have significant renal excretion and have the potential for overexposure, such as atenolol, nadolol, or sotalol. 19 Atenolol can be used as part of the management approach for hypertension and coronary disease if given 3 times per week in ESKD during hemodialysis. 20 Consideration should be given to the potential for dialyzability of certain beta blockers, as a 1.4-fold increased mortality risk was observed in the group treated with highly dialyzable beta blockers such as metoprolol. 21
Case 1, continued: The patient is adherent to prescribed medications including losartan, furosemide (80 mg twice daily), atorvastatin, insulin, and beta blockers. His BP is 162/92 mm Hg and heart rate is 104 beats per minute.
Which is better to continue, ACEi, ARB, or ARNi?
Renin-angiotensin System Inhibition with ACEi or ARB or ARNi
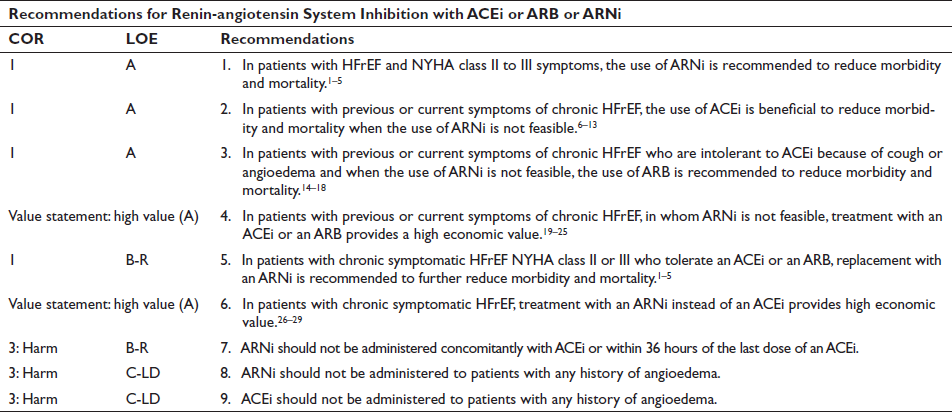
Inhibition of the renin-angiotensin system is recommended to reduce morbidity and mortality for patients with HFrEF, and ARNi, ACEi, or ARB is recommended as a first-line therapy.22–39 If patients have chronic symptomatic HFrEF with NYHA class II or III symptoms and they tolerate an ACEi or ARB, they should be switched to an ARNi because of improvement in morbidity and mortality.22–26 An ARNi is recommended as a de novo treatment in hospitalized patients with acute HF before discharge, given there is improvement in health status, reduction in the prognostic biomarker NT-proBNP, and improvement in LV remodeling parameters compared with ACEi/ARB. Although data are limited, the use of an ARNi may be efficacious as a de novo treatment in patients with symptomatic chronic HFrEF to simplify management. ARB may be used as an alternative to ACEi in the setting of intolerable cough or as an alternative to ACEi and ARNi in patients with a history of angioedema. If patients are switched from an ACEi to an ARNi or vice versa, there should be at least 36 hours’ gap between ACEi and ARNi doses.
In the Prospective Comparison of ARNi with ACEi to Determine Impact on Global Mortality and Morbidity in Heart Failure (PARADIGM-HF) trial, an RCT that compared the first approved ARNi, sacubitril-valsartan, with enalapril in symptomatic patients with HFrEF tolerating an adequate dose of either ACEi or ARB, sacubitril-valsartan significantly reduced the composite endpoint of cardiovascular death or HF hospitalization by 20% relative to enalapril. 22
Case 1, continued: The patient is prescribed sacubitril-valsartan in place of losartan, furosemide (80 mg twice daily), atorvastatin, and insulin. His BP is 162/92 mm Hg and heart rate is 104 beats per minute. This patient has a creatinine of 1.7 mg% (eGFR 47 ml/min).
How to monitor renal functions when a patient is on ARNi/ACEi/ARB?
Renin-angiotensin-aldosterone System Inhibitor Therapy in Patients with Heart Failure with Reduced Ejection Fraction and CKD
Randomized controlled trials have shown improvement of survival in patients with HF with the use of ACEi (or ARB) and mineralocorticoid receptor antagonists. Most of these studies included patients with mild-to-moderate (i.e., stages 1–3) CKD. 40 Patients with severe CKD have been excluded from most ACEi studies.
The use of ACEi and mineralocorticoid receptor antagonists, particularly in patients with CKD, is often associated with hyperkalemia and rising creatinine. Rising creatinine of up to 40% has been observed in ACEi trials, without significant effects on long-term outcomes.41, 42 A rise of up to 30% can be viewed as resulting from hemodynamic changes owing to RAASis. 43 In fact, such a change may be beneficial and is named permissive AKI as opposed to true AKI due to other reasons in patients with HFrEF, which requires careful history taking and physical examination. RAASis should be stopped when kidney blood flow autoregulation is necessary in a patient, for example, with diarrhea causing volume depletion.
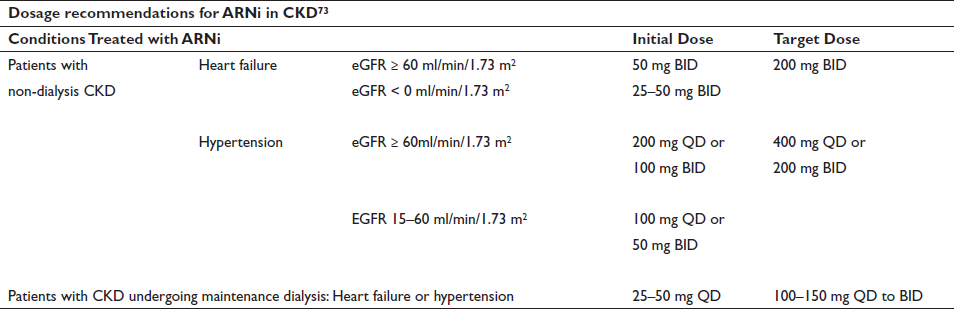
A large randomized controlled trial of dual angiotensin receptor and neprilysin inhibitor therapy excluded patients with an eGFR of 30 ml/min per 1.73 m2 (see Figure 1, Table 1) but included patients with only mild CKD. 44 A randomized controlled trial of angiotensin receptor neprilysin inhibitors, including patients with eGFR as low as 20 ml/min per 1.73 m2, demonstrated safety and efficacy similar to irbesartan. 45 More recently, angiotensin receptor and neprilysin inhibitor therapy has been shown to slow the progression of CKD in patients with HF with preserved ejection fraction (HFpEF) more effectively than valsartan. 46 The recommended starting dosage for patients with an eGFR of 60 ml/min per 1.73 m2 is 24 mg sacubitril and 26 mg valsartan, administered twice per day, at least 36 hours after stopping ACEi or ARB; the dose is then increased, with careful monitoring of creatinine, potassium, and BP.
Patients are referred back and forth, between nephrologists and cardiologists, with RAASi-induced changes in creatinine and potassium, resulting in multiple hospital attendances and, often, discontinuation of RAASi. An analysis of 194,456 patient records showed up to 30% increase in the frequency of hyperkalemia in patients with CKD stages 4 and 5 if measured more than 4 times a year and discontinuation of ACEi/ARB in 24% of patients. 47 Nephrologists traditionally have managed hyperkalemia with judicious use of diuretics and correction of acidosis, but oral potassium binders such as patiromer or sodium zirconium cyclosilicate may be useful. 48 Randomized controlled studies of new potassium binders for maximization of RAASi therapy in patients with advanced CKD and heart failure are necessary. A close collaboration between nephrologists and cardiologists is necessary for successful initiation and continuation of RAASi in patients with CKD and HF.
Case 1, continued: This patient’s creatinine was repeated 2 weeks after starting ARNi. His creatinine jumped to 2.4 mg% (eGFR 31 ml/min). Should ARNi be continued or not?
As 30% rise in creatinine is expected after starting ARNi. We continued ARNi with frequent monitoring of renal functions.
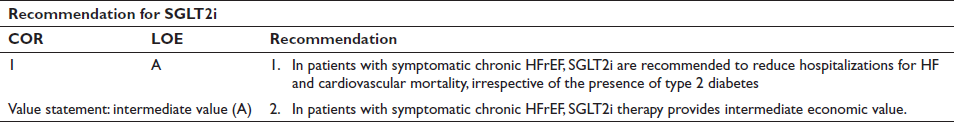
Several RCTs in patients with type 2 diabetes and either established CVD or high risk for CVD have shown that SGLT2i prevent HF hospitalizations compared with placebo.49–51
The Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure (DAPA-HF) trial and EMPagliflozin outcomE tRial in patients with chrOnic heaRt failure with Reduced ejection fraction (EMPEROR-Reduced) showed the benefit of SGLT2i (dapagliflozin and empagliflozin, respectively) versus placebo on outcomes (median follow-up, 16–18 months).49, 52 Patients who enrolled had symptomatic chronic HFrEF (LVEF ≤ 40%, NYHA class II to IV, and elevated natriuretic peptides) and were already on GDMT. Important exclusions were eGFR < 20 ml/min (EMPEROR-Reduced) < 30 ml/min (DAPA-HF), type 1 diabetes, or lower SBP.
In the DAPA-HF and EMPEROR-Reduced trials, SGLT2i compared with placebo reduced the composite of cardiovascular death or HF hospitalization by approximately 25%.50, 52–54 The benefit in reduction of HF hospitalization was greater (30%) in both trials. 9 Risk of cardiovascular death was significantly lowered (18%) with dapagliflozin, as was risk of all-cause mortality (17%).
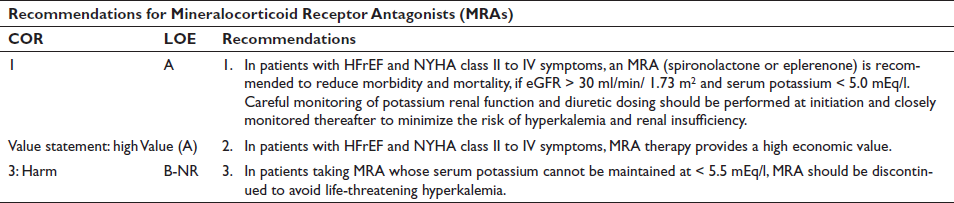
MRA (also known as aldosterone antagonists or anti-mineralocorticoids) show consistent improvements in all-cause mortality, HF hospitalizations, and SCD across a wide range of patients with HFrEF.55–57
Clinical trials taken on MRA together—Randomized Aldactone Evaluation Study (RALES), which randomized highly symptomatic patients with LVEF ≤ 35%; Eplerenone Post–Acute Myocardial Infarction Heart Failure Efficacy and Survival Study (EPHESUS), which randomized patients post-MI with LVEF ≤ 40%; and Eplerenone in Mild Patient Hospitalization and Survival Study in Heart Failure (EMPHASIS-HF), which randomized patients with mild symptoms and LVEF ≤ 30%—suggest a benefit of MRA across the spectrum of HFrEF, inclusive of a wide range of etiologies and disease severities. Initiation in the ambulatory or hospital setting is appropriate. 58 The starting dose of spironolactone and eplerenone is 25 mg orally daily, increased to 50 mg daily orally after a month; for eGFR 31–49 m;/min/1.73 m2, dosing should be reduced by half. Regular checks of serum potassium levels and renal function should be performed according to clinical status, approximately 1 week, then 4 weeks, then every 6 months after initiating or intensifying MRA, with more frequent testing for clinical instability.
Spironolactone and eplerenone are partially excreted through the kidneys, raising concerns about safety when eGFR ≤ 30 ml/min/1.73 m2.59, 60 Spironolactone and eplerenone decrease renal potassium excretion, raising the risk of hyperkalemia, particularly when MRA is initiated at serum potassium ≥ 5.0 mEq/L and continued at serum potassium ≥ 5.5 mEq/L. Evidence around the use of any MRA in patients with HF and CKD stages 4–5 is lacking (ECR).
Vasodilators
Two RCTs, Vasodilator Heart Failure Trial (V-HeFT I) and African American Heart Failure Trial (A-HeFT), established the benefit of the combination of hydralazine-isosorbide dinitrate in self-identified African Americans.61, 62
In a large-scale trial that compared the vasodilator combination with placebo, the use of hydralazine and isosorbide dinitrate reduced mortality in patients with HF treated with digoxin and diuretics but not an ACEi or beta blocker. 4 However, in two other trials that compared the vasodilator combination with an ACEi, the ACEi produced more favorable effects on survival. 63
There are insufficient data to guide the use of hydralazine-isosorbide dinitrate with ARNi. In patients with HFrEF who cannot receive first-line agents such as ARNi, ACEi, or ARB, referral to a HF specialist can provide guidance for further management because the use of hydralazine and isosorbide dinitrate in these patients is uncertain.
Ivabradine
The ivabradine and outcomes in chronic heart failure (SHIFT) study demonstrated improved hospitalization and deaths owing to HF with ivabradine treatment in patients with HFrEF with a heart rate > 70 beats per minute despite beta-blocker therapy, which included patients with creatinine 2.5 mg/dl. The study included 1,589 patients with CKD stage 3, and benefits in patients with CKD were similar to those in patients without CKD. 64 Ivabradine is metabolized by the CYP3A4 enzyme in the liver and gut, and elimination by kidneys is minimal. The dosage is 2.5–7.5 mg twice per day and does not require dose adjustment with creatinine clearance of 15 ml/min.

Management of Iron Deficiency and Anemia in Patients with Heart Failure with Reduced Ejection Fraction and CKD
Nephrologists have been using intravenous iron for the past three decades to treat anemia in patients with CKD, both before and during dialysis. In the United Kingdom, a recent study in patients receiving dialysis has shown benefits of high-dose intravenous iron in reducing mortality and morbidity, together with HF hospitalizations by 44%. 65 A previous trial of benefits of intravenous iron in patients with HFrEF included patients with early stages of CKD. 66 A collaboration between cardiologists and nephrologists may assist the management of iron deficiency in patients with HFrEF and CKD.
RRT
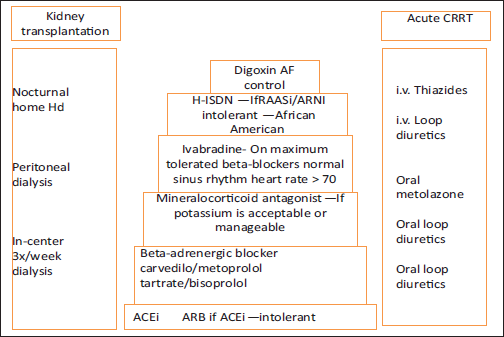
CKD is a progressive disease with declining kidney function over time, causing frequent episodes of fluid overload and hyperkalemia, further complicated by episodes of AKI. With an eGFR of 20 ml/min per 1.73 m2, decisions need to be made, in discussion with the patient, about indications, appropriateness, and modality of KRT. Careful consideration of factors such as patient expectations, comorbidities, frailty, and quality of life is necessary to consider when starting KRT. Patients with HF on dialysis have very poor prognosis, with a five-year survival rate of 12.5%. 67 There may be symptomatic relief and fewer hospitalizations with peritoneal dialysis in carefully selected patients. 68 In a study of 118 patients with HF and CKD, peritoneal dialysis was associated with improvement in quality of life and New York Heart Association class. 69 A meta-analysis of 23 studies demonstrated benefits of peritoneal dialysis in patients with HF and CKD, with improved hospitalization rates and heart function. 70 In patients with refractory HF, overnight ultrafiltration with icodextrin solution improved quality of life, heart function, and New York Heart Association class. 71 Hemodialysis may be tricky in patients with low BP, but more frequent dialysis and longer nocturnal dialysis may be useful for fluid removal. However, creation of arteriovenous fistula or graft for hemodialysis may cause dilation of left atrium and right ventricle and associated HF. 72 For patients with acute HF and low BP, slow ultrafiltration with continuous KRT may be useful.
The Way Forward
To achieve appropriate therapy and the best possible outcome, combined cardiology–nephrology clinics are necessary, which manage kidney and heart care, together with intravenous iron and advice on dialysis therapy. 49 Patients appreciate the multiple benefits from a single visit and provide excellent feedback. The multidisciplinary “one-stop shop” clinic is very patient centered and helps with decision making regarding RAASi, device therapy, and intravenous iron administration but requires proper resources, including the presence of a nephrologist, cardiologist, and nurse and appropriate space (Figure 2). With improved knowledge among cardiologists and nephrologists and multidisciplinary approach, evidence-based therapy can be implemented in patients with early-to-moderate CKD. However, we recognize that more studies are necessary to strengthen the evidence base in patients with advanced CKD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethical clearance and patient consent were obtained for the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
