Abstract
Beta blockers (BBs) are widely acknowledged as a standard care for numerous cardiovascular conditions. They have a long-established history in treating angina, regulating heart rate in atrial fibrillation and ventricular tachycardia, and managing heart failure with reduced ejection fraction (HFREF). The effectiveness of BBs in treating acute coronary syndrome was established during a time when reperfusion therapies were not readily available. However, with the recent advancements in coronary revascularization techniques, the role of BB therapy in managing patients after a myocardial infarction and in those with stable angina has become a subject of debate. Likewise, there are uncertainties about the efficacy of BBs in individuals with HFREF who also suffer from atrial fibrillation, as well as in patients with heart failure with preserved ejection fraction (HFPEF). Furthermore, there are numerous concerns about using BBs as the initial therapy, as was customary in hypertension management in the past, and safety concerns regarding their use in patients undergoing non-cardiac surgery. This comprehensive review aims to thoroughly examine all existing recent evidence concerning BB therapy in various cardiovascular scenarios and to provide a valuable guidance to the clinicians in the field of cardiology.
Keywords
Introduction
Ever since Sir James Black introduced the first beta blocker (BB) in 1958, for which he was awarded the Nobel Prize in 1988, these drugs have become a vital class of medications and have played a crucial role in the standard treatment of many cardiovascular conditions. They offer various advantages by acting in multiple ways with their anti-catecholamine effects: (a) Effects on inotropy and chronotropy, (b) antiarrhythmic effects, and (c) anti-ischemic effects. The major side effects are bradycardia, hypotension, bronchospasm, fatigue, and depression. BBs are an excellent class of drugs in the armamentarium of various cardiac diseases such as coronary artery disease (CAD), congestive heart failure, and hypertension. Given the tremendous advances in the field of cardiology, the launch of new drugs, and the latest interventional and therapeutic procedures, it became necessary to review the role of BB therapy in contemporary medicine.
Coronary Artery Disease
Rationale for the Use of BBs in CAD
The use of BBs in CAD is grounded in their ability to inhibit sympathetic overdrive, effectively reducing the heart rate. This, in turn, leads to a decrease in myocardial demand for oxygen consumption, which is pivotal in the management and treatment of CAD. Anti-ischemic effects, prevention of reinfarction, and reduction in arrhythmia-induced sudden death also promote the use of BBs in CAD, particularly acute coronary syndrome.
The earlier guidelines that showed potential benefits of BBs in the MI setting were based on RCTs done before the reperfusion era. But the robust growth of intervention and healthcare management in recent years has led to a discussion on the role of BBs in MI and post-MI situations, particularly in the long term. Timely coronary reperfusion and potent antiplatelet agents and statins reduce the infarct size, thereby lowering sympathetic drive and decreasing myocardial oxygen demand. So, the risk of heart failure and negative inotropy leading to cardiogenic shock should be carefully weighed against the potential benefits of BBs.
Acute Coronary Syndrome
Intravenous BBs, particularly metoprolol (medications without intrinsic sympathetic activity), have shown a more pronounced protective effect in the setting of ongoing acute coronary occlusion (STEMI) and primary percutaneous coronary intervention (PPCI) for patients without significant acute heart failure and SBP above 120 mmHg (Class IIa). This is not validated in NSTEMI. In ACS patients with atrial fibrillation in the absence of HF or hypotension, they are recommended for rate control (Class 1). In patients with ACS and ventricular tachycardia/VF, intravenous BBs are still recommended1, 2.
Various trials have been done and are still ongoing on various BBs in acute myocardial infarction. The initial trial, ISIS-I (1986), 3 was done in the pre-reperfusion era, when antiplatelets and statins were not in proper use; it showed a significant reduction in death by atenolol. But the COMMIT trial 4 (2005) failed to show a benefit with metoprolol. CAPRICORN 5 (2001) was a study done in the reperfusion era that showed survival benefit with carvedilol in patients with MI and LVEF < 40%. A meta-analysis done on RCTs in 2013 concluded that BB therapy in ACS patients decreases hospital mortality, reinfarction, and arrhythmias. Data from the Melbourne Interventional Group (MIG) 6 (2020) have also shown a reduction in mortality in patients undergoing PCI for ACS with EF < 50% (but not in EF > 50%). The KAMIR-NIH 7 registry suggested that BB therapy may offer clinical advantages not only to patients with a reduced LVEF (≤40%) but also to those with a mid-range LVEF (40%–49%) in acute MI patients, but not in patients with preserved LVEF.
But, most importantly, the treatment duration and dosage of drugs were not directly investigated in many RCTs. In the present era of prompt revascularization for patients experiencing myocardial infarction (MI), the recommendation for prolonged (>1 year) BB therapy in the absence of left ventricular systolic dysfunction has been questioned. A 2020 observational study confirmed benefit for up to 3 years post-MI but not longer. 8 The Swedish registry (2023) 9 suggested that BB treatment beyond 1 year of MI for patients without heart failure or LVSD (EF < 50%) was not associated with improved CV outcomes. Regarding BB dosage, an analysis of The OBTAIN 10 registry (2021) has suggested that patients treated with >12.5% to 25% of the target dose (used in earlier trials) beyond 1 year after MI may experience enhanced survival compared to other BB dosages. These findings indicate that it may be appropriate to consider LVEF as a guiding factor for long-term use of BB therapy. The duration of treatment is advisable for 1 year and can be extended up to 3 years. In patients with high-risk features such as heart failure or left ventricular dysfunction, BBs can be continued indefinitely.
Chronic Coronary Disease
In stable CAD/CCD (chronic coronary disease), the role of BBs is still debatable as there is a paucity of RCTs in this subset of patients. Patients with CCD encompass individuals with or without a history of MI and/or left ventricular systolic dysfunction. These factors will decide whether to prescribe BB therapy for patients with CCD or not. The main reasons for considering BBs may involve angina, poorly managed hypertension, or arrhythmias.
The investigators of the REACH 11 study conducted in 2012 reported that BBs do not provide any significant benefit in reducing cardiovascular events in stable CAD patients who have not experienced a prior myocardial infarction (MI). Similarly, the CHARISMA 12 trial (2014), which involved patients without a previous MI or heart failure, did not demonstrate any reduction in cardiovascular events. Likewise, The CATH PCI 13 registry analysis (2016), focusing on individuals with stable angina who did not have a history of MI, left ventricular systolic dysfunction, or systolic heart failure (HF) and who were undergoing percutaneous coronary intervention (PCI), did not reveal any significant improvement in cardiovascular morbidity or mortality rates at either the 30-day mark or the 3-year follow-up. But the latest study done by Godey et al 14 (2023) showed a small but significant reduction in the occurrence of cardiovascular events in patients with stable CAD without MI or HF at 5 years. This study was more powered by the latest definition of stable CAD and supported by imaging modalities.
The current American guidelines gave a Class IIb recommendation for BBs in stable CAD who are already on BBs for previous MI without a history of or LVEF ≤ 50%, angina, arrhythmias, or uncontrolled hypertension. A Class III recommendation has been issued for long-term BB therapy in the absence of MI in the past year, LVEF ≤ 50%, or another primary indication for BB therapy. 15 The European and Canadian guidelines also gave no recommendation for BBs in stable CAD. Ongoing large-scale RCTs in patients without LVSD, such as REBOOT–CNIC, BETAMI, and REDUCE-SWEDEHEART, can provide greater insights into this specific group of patients, whereas AβYSS and SMART-DECISION can shed light on the duration of BBs in uncomplicated ACS without LVSD.
In conclusion, BBs are indicated: (a) Intravenous (IV) or oral in ACS for relief from angina, (b) IV in AF and VT during ACS/PPCI, (c) long term in patients with ACS and LV dysfunction (EF < 50%) and chronic heart failure, (d) All patients of MI up to 1 year and may be extended to 3 years, and (e) Not recommended in chronic CAD without MI or LV dysfunction/heart failure. Further trials are needed to evaluate their usage in the long term in stable CAD patients without HF, prior MI, or LV dysfunction.
Choice of Agent
The choice of agent was driven by RCTs, which supported the clinical efficacy of drugs such as bisoprolol, carvedilol, and metoprolol succinate among patients with LV systolic dysfunction in reducing cardiovascular death and MACE. Start at low doses, and titrate gradually every 2 weeks to avoid adverse effects.
Heart Failure
Rationale of BB Use in HF
β receptor blockade antagonizes the cardiotoxic effects of catecholamines and is helpful in the upregulation of myocardial β receptors, suppression of RAS and endothelin systems, improvement of coronary flow, and decrease in HR, BP, and atrial and ventricular arrhythmias. These mechanisms help in reducing hospitalizations, preventing LV remodeling, promoting reverse remodeling, and increasing survival in heart failure with reduced ejection fraction (HFREF).
Decompensated Heart Failure/Acute Heart failure/Severe Heart Failure
In patients with persistent NYHA Class IV HF symptoms or stage D HF, the use of BBs should be approached with extreme caution and reserved for exceptional cases as these individuals are particularly vulnerable to decompensation due to the negative inotropic effect of BBs.
HFREF
Though the pathophysiological mechanism of BB use was known earlier, there was a delay in acceptance due to its transient negative inotropic effects and worsening of HF. MERIT-HF 16 (1999), CIBIS II 17 (1999), CAPRICORN (2001), and COMET 18 (2003) have all established the survival benefit and reduction in all-cause mortality in patients with HF and LV systolic dysfunction. To date, no study or trail has yielded contradictory findings in this perspective. Hence, BBs have a Class I recommendation and stand first among the fantastic four drugs in chronic HF with sinus rhythm 19 (fantastic four drugs—BBs, mineralocorticoid antagonists, ACEI/ARB/ARNI, and SGLT 2i).
HF with Atrial Fibrillation
BBs have been shown to improve symptoms but not survival in AF and HF as the evidence is not clearly established. While Koteche et al 20 have shown no mortality benefit or decrease in hospitalizations, the AF–CHF trial 21 sub-study showed mortality benefit but no decrease in hospitalization. Further RCTs are warranted to address this gap in patients of HF and AF.
Heart Failure with Mildly Reduced EF (HFMREF)
Although research consistently indicates that BBs are effective in reducing morbidity and mortality among patients with reduced ejection fraction (EF), the data is less conclusive for those with heart failure and only a mildly reduced ejection fraction (HFmrEF). A recent study conducted by Arnold et al 22 has revealed that BBs were linked to a reduced risk of HF hospitalization and mortality in patients with heart failure with HFmrEF. However, there was an increased risk of heart failure hospitalization in patients with heart failure and HFpEF, particularly when their ejection fraction exceeded 60%. The recent focused update of ESC on heart failure gave IIb recommendation for BBs in HFMREF. 23
Heart Failure with Preserved Ejection Fraction
While HFPEF and HFREF share similar symptoms, they represent different pathological entities. The management strategies effective in HFREF patients have not demonstrated significant benefits in those with HFPEF, including the use of BBs. Many trials have shown inconsistent results, while a few have proven to be beneficial. 24
HF in Chronic Kidney Disease
BBs are often used in patients with chronic kidney disease not only for controlling hypertension but also for providing cardio protection, given the increased cardiovascular risk in these patients. A meta-analysis done by Badve et al 25 have shown that treatment with BBs improved all-cause mortality in patients with CKD and chronic systolic heart failure. A similar study conducted by Zhou H et al 26 over a span of 10 years, which examined patients with HF and CKD who were initiated on dialysis, found that metoprolol (highly dialyzable) but not carvedilol (poorly dialyzable) was associated with reduced mortality in patients with an EF greater than 40%, as opposed to those with EF less than 40%. This benefit was seen at 1 year follow-up but not at 6 months, and there was no difference in hospitalizations.
Drugs of Choice
Carvedilol is the agent of choice in patients with hypertension, diabetes, and peripheral arterial disease due to its vasodilatory properties and less alteration of glycemia. Metoprolol is often preferred in patients with HF with atrial fibrillation and thyrotoxicosis due to its excellent control of heart rate. BBs such as bisoprolol and nebivolol, which exhibit higher β1 receptor selectivity, are generally better tolerated by patients with asthma and COPD as they minimize the chances of broncho-constrictive side effects. However, nebivolol has not shown statistically significant reduction in all-cause mortality in HF patients in SENIORS trial. 27
In conclusion, BBs are definitely indicated in HFREF in sinus rhythm. Further data is needed in HFPEF and HF with AF.
Hypertension
Numerous trials and research studies have demonstrated that BBs are no longer the first-line preferred treatment for essential hypertension (HTN). BBs are considered less effective than calcium channel blockers (CCBs) and renin-angiotensin system (RAS) antagonists in achieving the target blood pressure and preventing stroke. 28 The current American guidelines, specifically the Joint National Committee VIII, position BBs as a third-line option for the management of hypertension. Likewise, the NICE (National Institute for Health and Care Excellence) guidelines state that BBs are not the preferred initial treatment for hypertension. They are primarily recommended for cases of resistant hypertension. But the ESC/ESH guidelines have suggested BBs as the first-line choice in a special category of patients with cardiovascular comorbidities, such as reduced ejection fraction, CHF, myocardial infarction, angina, and arrhythmias, due to their anti-ischemic and anti-arrhythmic properties.
It’s important to note that most of these studies were conducted using atenolol. In the subset of younger and middle-aged individuals with essential hypertension, especially when there is a requirement to manage high sympathetic activity and blood pressure response to physical activity, BBs can be considered as an initial choice of medication. Therefore, further randomized controlled trials (RCTs) should explore the use of BBs with vasodilating properties, such as nebivolol and carvedilol, which can reduce central blood pressure and potentially offer cerebrovascular protection.
Arrhythmias
Rationale
BBs reduce the risk of proarrhythmia by diminishing the influence of the sympathetic nervous system on the heart, resulting in negative effects on inotropy, chronotropy, bathmotropy, and dromotropy. They also prevent sympathetically mediated triggers and functional reentrant substrates and slow down the rates of the SA and AV nodes.
AF
BBs slow AV node conduction and are preferred for controlling ventricular rates, especially during exercise, in patients with a history of MI, HF, or post-cardiothoracic surgery. BBs exhibit weaker antiarrhythmic properties compared to Class I (Na channel blockers) and Class III agents (K channel blockers) and are generally not considered suitable for atrial rhythm control. In acute atrial fibrillation (AF) cases, intravenous administration of esmolol, propranolol, and metoprolol has been found effective, while for chronic AF, oral BBs such as atenolol, bisoprolol, metoprolol, nadolol, propranolol, and sotalol (a K channel blocker) are effective for ventricular rate control. However, it’s worth noting that among different BBs, metoprolol is more effective than carvedilol for rate control.
However, this recommendation lacks strong evidence from large randomized trials, and their effectiveness in preventing atrial fibrillation in patients in sinus rhythm remains unproven. Small randomized studies comparing BBs to digoxin or non-dihydropyridine calcium-channel blockers showed unfavorable effects such as reduced functional capacity and higher NT-proBNP levels. 29 The RATE-AF trial 30 comparing digoxin to bisoprolol raised safety concerns, with more adverse events in the BB group, including heart failure hospitalizations and office visits for atrial fibrillation. LIFE Hypertension 31 and ASCOT Hypertension 32 studies suggested that BBs, by reducing sinus rate, may increase AF risk. BBs, by lowering heart rate, can lead to intracardiac congestion and elevated natriuretic peptide levels. This effect is compounded by the Frank–Starling mechanism and reflected peripheral pressure waves, increasing the risk of atrial fibrillation and heart failure with preserved ejection fraction.
Given the limited evidence from outcome studies and the availability of alternative treatments, there is a need for large RCTs to address concerns about the over-prescription of BBs for atrial fibrillation. Significantly, their adverse effects are more prominent in individuals who have a normal ejection fraction and lower heart rates, a situation frequently observed in those with paroxysmal atrial fibrillation who receive high doses that substantially reduce sinus rates.
MI
Specific anti-arrhythmic effects of BBs post-MI may encompass prolonging the ventricular effective refractory period, suppressing triggered activity, automaticity, and electrophysiological heterogeneity, while also slowing the heart rate. Additionally, they may mitigate the influence of sympathetic nerve activity on reentrant circuits near infarct borders, potentially reducing the risk of conduction block. In the MADIT-II trial, the use of high-dose BBs in patients with implantable cardioverter-defibrillators (ICDs) resulted in a notable reduction in the occurrence of recurrent VT/VF episodes, necessitating ICD interventions when compared to those who did not receive BBs. 33
HF
By downregulation of β1 (~60%) and upregulation of β2 (~40%) and β3-AR receptors, the antiarrhythmic action of BBs in HF might be mediated by attenuating both the triggers and the functional or structural arrhythmia substrates.
LQTS
Elevated sympathetic activity serves as a primary arrhythmia trigger in LQT1 (due to reduced IKs current) and can be effectively prevented by BBs. Notably, among various BBs, nadolol demonstrated the highest efficacy, particularly in the more severely affected LQT2 patient group. Recent findings have shown that BBs also reduce the risk in LQT3 patients despite prior studies. 34
CPVT
CPVT causes arrhythmia through diastolic calcium leak. First-line treatment includes exercise restriction and BBs (without intrinsic sympathomimetic activity). Guidelines recommend β-blockers for all CPVT patients with ventricular arrhythmias and suggest considering them for asymptomatic mutation carriers (RyR2). 34
Perioperative Risk
Research on the role of perioperative BBs has generated mixed findings. Much of the support for using perioperative BBs relied on the DECREASE 35 [(DECREASE-I (1999) IV (2009)] trials, which were discredited in 2011 due to issues related to data fabrication. The POISE 36 trial in 2008 demonstrated that while metoprolol reduced the risk of perioperative MI in patients undergoing non-cardiac surgery, this advantage was outweighed by an increased incidence of mortality and strokes. The indiscriminate use of high-dose metoprolol succinate (100 mg initiated 2–4 hours before surgery and continued at 200 mg afterward) in this study might have led to a higher occurrence of strokes and deaths attributed to hypotension. In the DIPOM 37 study done in diabetic patients undergoing surgery, metoprolol showed no statistically significant difference between the metoprolol CR/XL and placebo groups with regard to the incidence of serious adverse events. The MaVS trial 38 did not support a clinically useful effect for metoprolol in reducing the cardiac event rate in patients undergoing abdominal aortic surgery and infra-inguinal or extra-anatomic revascularization. In the context of cardiac surgery, where it would be reasonable to expect a positive effect on patient prognosis from BBs, evidence did not show a significant decrease in adverse events, including the acute coronary events.
The ACC/AHA guidelines (2014 update) recommend that patients who have been on BBs chronically should continue taking them when undergoing non-cardiac surgery (Class I). Additionally, it may be considered reasonable to initiate perioperative BBs for patients with intermediate- or high-risk myocardial ischemia or those who exhibit three or more risk factors according to the Revised Cardiac Risk Index (Class IIb). These risk factors include conditions such as heart failure, coronary artery disease, renal insufficiency, diabetes mellitus, or a history of cerebrovascular accidents. However, it’s crucial that the initiation of BB therapy is done with sufficient lead time before surgery to assess its safety and tolerability. A notable caution is to avoid initiating BBs on the day of surgery, as it is discouraged in view of increased incidence of stroke, mortality, bradycardia, and hypotension (Class III). 39 The ESC and European Society of Anesthesiology favored atenolol and bisoprolol when initiating a BB prior to surgery.
CVD and COPD
Individuals with chronic obstructive pulmonary disease (COPD) encounter a 2–5 times elevated risk of CVD attributable to factors such as smoking and systemic inflammation. These factors contribute to processes such as atherosclerosis, endothelial dysfunction, plaque development, and eventual plaque rupture and thrombosis, advancing the progression of CVD. Despite the 2016 European Society of Cardiology guidelines recommending BBs for patients with both COPD and CVD, there remains a tendency to under-prescribe BBs in COPD patients. A recent meta-analysis showed that the utilization of BBs has been linked to a decrease in both in-hospital and overall mortality in individuals who have both COPD and CVD. The use of cardioselective BBs is also connected to a lower likelihood of exacerbations in COPD. The positive effects of BBs may, to some extent, be linked to their capacity to decrease the heart rate. When cardioselective BBs are used in conjunction with bronchodilators, they do not diminish respiratory indices or impair the effectiveness of bronchodilators. 40
Others
In hyperthyroidism, the sympathetic nervous system is upregulated, which produces symptoms such as sinus tachycardia, palpitations, anxiety, and tremor. BBs have been used for decades to alleviate these symptoms until other treatments can normalize thyroid hormone levels. Propranolol is typically the drug of choice, due to its non-selective blockade and additional inhibition of peripheral conversion of thyroxine (T4) to the more active triiodothyronine (T3). Lipophilic BBs, specifically propranolol and metoprolol, have the ability to penetrate the blood–brain barrier and are strongly recommended for the preventive treatment of migraine attacks (Class A indication). 41 Topical BBs are used as second-line or first-line (in selected patients) therapy in glaucoma patients. Recently, as beta-adrenergic receptors influence the growth processes of both cancer and immune cells, blocking these receptors has been linked to a reduction in metastasis in various epithelial and solid organ tumors. 42 But further research is warranted in this area.
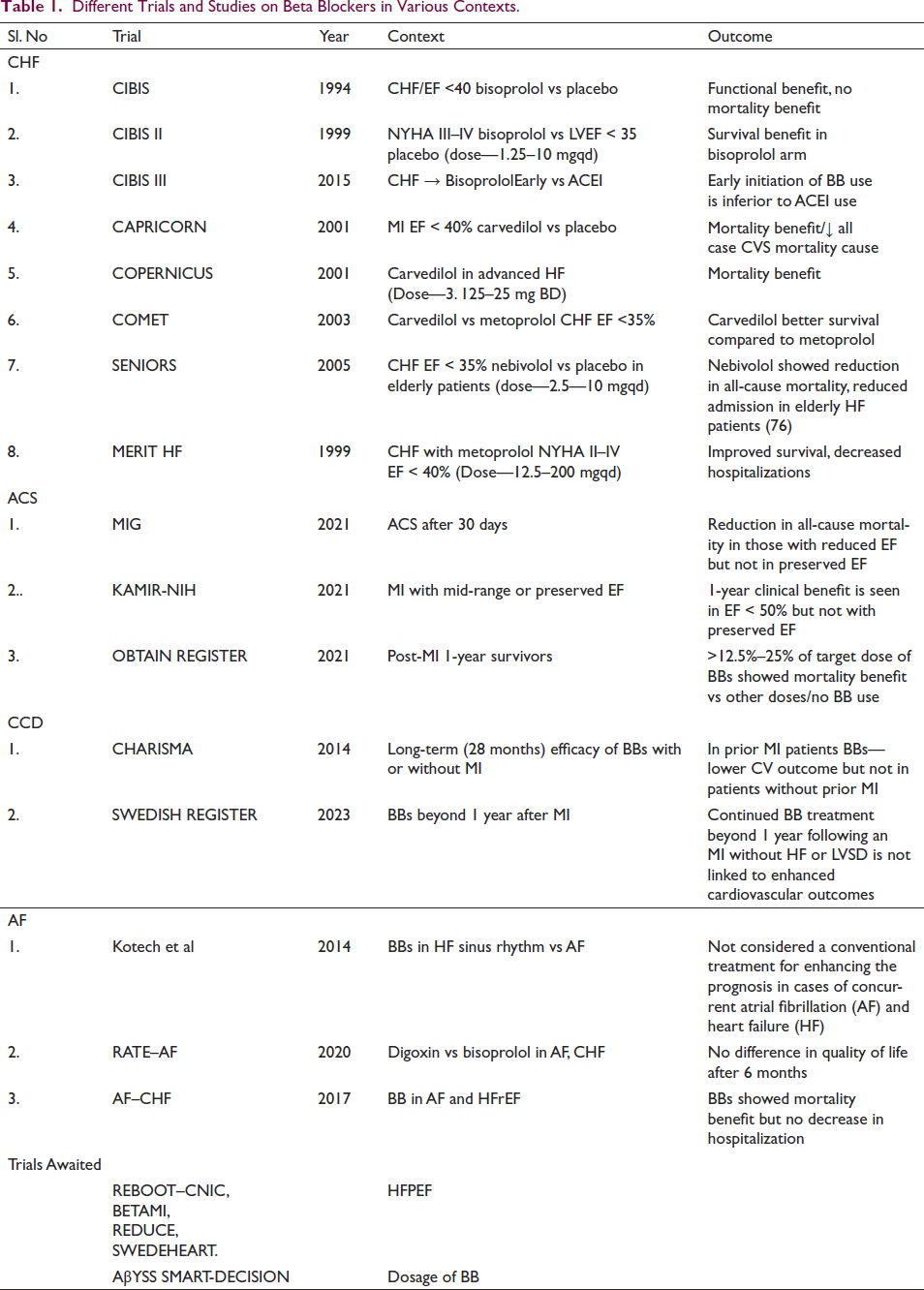
Various trials and studies pertaining to BBs are mentioned in Table 1.
Different Trials and Studies on Beta Blockers in Various Contexts.
Adverse Effects and Contraindications
Bradycardia
Beta blockers can cause sinus bradycardia and AV node conduction block, potentially leading to heart block, though these effects are milder with beta blockers having ISA.
COPD
Using BBs in patients can increase airway resistance. However, caution is required in those with moderate to severe COPD, as indicated by reduced FEV1 (<50% of predicted) and significant reversibility(≥20%) in airway obstruction with salbutamol.
Withdrawal
Withdrawal of BBs is a critical concern, as it can worsen ischemic symptoms. This withdrawal effect is linked to heightened sympathetic activity. Beta receptor upregulation amplifies their responsiveness to circulating catecholamines, especially with short-acting drugs like propranolol. Gradual withdrawal, reducing the dose by half daily and then taking on alternate days, is preferred over abrupt discontinuation.
Peripheral Vascular Disease
In spite of worries regarding the deterioration of peripheral vascular disease (PVD), a meta-analysis of documented studies encompassing individuals with mild to moderate peripheral artery disease showed no exacerbation of symptoms with the use of BBs. 43 Thus, the concern may be overstated.
Glucose Metabolism
Epinephrine, through beta-adrenergic receptors, boosts glucose production and safeguards against hypoglycemia. It also triggers early warning symptoms of low blood sugar. Nonselective BBs can hinder recovery from insulin-induced hypoglycemia, but beta-1 selective drugs and those with intrinsic sympathomimetic activity (ISA) may have milder effects on glucose metabolism. Moreover, carvedilol seems to improve glucose utilization and reduce insulin levels in individuals with type 2 diabetes.
Depression, Fatigue, and Sexual Dysfunction
These are commonly mentioned as side effects of BBs. It was previously believed that lipophilic drugs, such as propranolol and metoprolol, had a higher occurrence of central nervous system effects, including fatigue and depression. However, a systematic review of randomized trials found that BB therapy did not increase the risk of depression and only resulted in small increases in fatigue and sexual dysfunction. 44
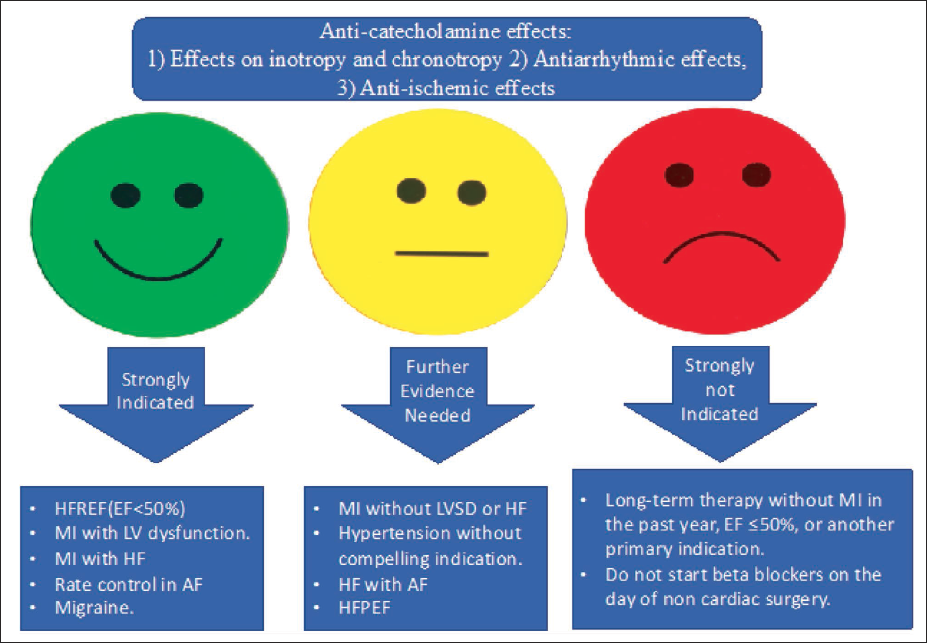
An overall summary of the BB mechanism of action, their usefulness in various conditions, and the areas where additional evidence is needed is depicted in Figure 1.
Summary of Beta Blockers’ Mechanism of Action, Their Usefulness in Various Conditions, and the Areas Where Additional Evidence Is Needed.
Conclusion
While BBs are commonly employed in everyday clinical practice, this analysis shows that new evidence and guidelines underscore that their clinical impact is highly influenced by the specific clinical scenarios. BBs are still a boon in heart failure with reduced ejection fraction in sinus rhythm. They are still recommended in acute coronary syndrome with ongoing ischemia and up to 1 year post-primary event and long term in patients with reduced EF (<50%) or heart failure. In patients with chronic coronary disease, the present evidence shows that the survival benefit in patients without MI or LV dysfunction is debatable. The results of ongoing RCTs will provide valuable insights and essential evidence regarding the role of long-term BB therapy. The current recommendations of BBs in various cardiovascular diseases are summarized in Table 2.
Current Recommendations on Available RCTs’ ACC/AHA and ESC Guidelines for Beta Blockers in Various Cardiovascular Diseases.

aESC; bAHA/ACC, cAHS/AAN.
Abbreviation List
BB—Beta blockers
HF—Heart failure
CAD—Coronary artery disease
PVD—Peripheral vascular disease
RCT—Randomized controlled trials
SBP—Systolic blood pressure
MI—Myocardial infarction
STEMI—St elevation myocardial infarction
PCI—Percutaneous coronary intervention
PPCI—Primary percutaneous coronary intervention
NSTEMI –Non-elevation myocardial infarction
ACS—Acute coronary syndrome
AF—Atrial fibrillation
VF—Ventricular fibrillation
AWMI—Anterior wall myocardial infarction
LVEF—Left ventricular ejection fraction
CV—Cardiovascular
CVD—Cardiovascular disease
CCD—Chronic coronary disease
MACE—Major adverse cardiovascular events
RAS—renin-angiotensin system
HR—Heart rate
BP—Blood pressure
HFREF—Heart failure with reduced ejection fraction
HFPEF—Heart Failure with preserved ejection fraction
ACEI—Angiotensin-converting enzyme inhibitors
ARB—Angiotensin receptor or blocker
ARNI—Angiotensin receptor/neprilysin inhibitor
SGLT—Sodium-glucose cotransporter
CKD—Chronic kidney disease
COPD—Chronic obstruction pulmonary disease
CCB—Calcium channel blockers
RAS—Renin-angiotensin system
NICE—National Institute for Health and Care Excellence
ESC—European Society of Cardiology
ACC—American College of Cardiology
AHA—American Heart Association
AHS—American Headache Society
AAN—American Academy of Neurology
Ethical Approval
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Informed Consent
Consent was not needed/not applicable, as this is a review article compiled from various research articles and guidelines and not from patients directly.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
