Abstract
Objectives
Postoperative residual shunts in the form of ventricular septal defects (VSD) and patent ductus arteriosus (PDA), if hemodynamically significant, can lead to symptoms of left ventricular volume overload and delayed recovery. The standard management is redo surgery, which can lead to significant morbidity and mortality. Device closure of such defects offers an alternative management strategy in selected cases. This study aims to examine the utility of device closure of residual left-to-right shunts in the pediatric age group in the immediate postoperative period.
Method
A retrospective analysis of hospital records was done from March 2018 to December 2022 to analyze the device closure in postoperative residual shunts. The success rate, complications, hospital length of stay, and midterm follow-up were analyzed.
Results
A total of four postoperative patients underwent VSD device closure and one patient underwent PDA device closure. The median age at transcatheter procedure was 6 years (range 3–10 years). The median time interval between the surgical procedure and the device closure procedure was 4 days (range 2–15 days). The indication for device closure was left ventricular volume overload in three patients and two patients had persistent pleural effusion. Qp:Qs was more than 1.5:1 in all five patients. The amplatzer duct occluder I (ADO-I) device was used in four patients and the ADO-II in one patient. There was no residual shunt post-deployment, with no major complication. The mean follow-up period was 2 years and all the patients had no residual shunt.
Conclusion
This case series, with its limitation of only five cases, has shown that device closure of hemodynamically significant residual postoperative left-to-right shunt lesions is an effective alternative strategy to redo surgery in older children. Further study is required with a larger sample size, and a longer follow-up is needed to evaluate its long-term safety and efficacy.
ADO – Amplatzer duct occlude
A-V Loop – Arterio-venous loop
ECG – Electrocardiograph
LAO – Left anterior oblique
LPA – Left Pulmonary artery
LV – Left ventricle
PDA – Patent ductus arteriosus
PA – pulmonary artery
PVRI – Pulmonary vascular resistance index
Qp:Qs – Pulmonary; systemic blood flow ratio
RVOT – Right ventricular outflow tract
TAP – Trans annular patch
TOF – Tetralogy of Fallot
Introduction
Postoperative residual defects after corrective pediatric cardiac surgery are not unknown. These can be in the form of residual ventricular septal defects (VSD), patent ductus arteriosus (PDA), right ventricular outflow tract (RVOT) obstruction, regurgitant lesions, and so on. Out of them, residual left-to-right shunt lesions in the form of VSD and PDA are quite common and, if hemodynamically significant, can lead to left ventricular volume overload and failure, pulmonary hypertension, persistent pleural effusion, and features of right ventricular failure. Residual VSD in the postoperative period is seen to occur in 5%–25% of patients in cases of only VSD closure and around 10% in cases of Tetralogy of Fallot (TOF).1, 2 Residual PDA after surgical ligation is seen in 0.6%–5% of cases in one study. 3 The standard treatment for hemodynamically significant residual VSD and PDA is redo surgery. However, due to the need for cardiopulmonary bypass, there is an increased risk of myocardial edema and scarring. 4 Transcatheter closure of these defects is an alternative approach that has significantly less morbidity and mortality as compared to a redo surgery. However, this is not as widely accepted as the native VSD and PDA device closure. Especially in the case of residual VSD, where the location is around the pericardial or dacron patch, the morphology of the defect is not as straightforward as in the case of native VSD. Thus, making transcatheter closure a challenging endeavor. This is even more so in the immediate postoperative period, especially in the pediatric age group. We present our experience with transcatheter closure of hemodynamically significant residual VSD and PDA in the immediate postoperative period in the pediatric age group.
Methods
This is a single-center retrospective observational study conducted at a newly established pediatric cardiac centre. Hospital records from April 2018 to December 2022 were analyzed. A transthoracic echocardiogram was performed on all patients in the postoperative period according to a fixed protocol. Once a residual VSD or PDA was found on transthoracic echocardiography, the following were the indications for cardiac catheterization:
Clinical evidence of left ventricular volume overload. Pulmonary hypertension due to the residual shunt. Persistent pleural effusion not responding to medical management.
A pre-procedure echocardiographic analysis was done to determine the number, location, approximate size of the VSD, and proximity of the defect to the tricuspid and aortic valves. For a PDA, its narrowest diameter (usually at the pulmonary artery (PA) end) was measured to determine the appropriate size of the device to be used. Also, other residual lesions that warranted redo surgery were ruled out.
Device Closure Procedure
The procedure was done under general anesthesia. Both femoral artery and vein access were taken. Systemic heparinization (100 units/kg) was done. Right heart catheterization and oximetry at room air were done to look for the hemodynamic significance of the residual defect. In cases of hemodynamically significant shunts, a left ventricular angiogram was done in the left anterior oblique (LAO) 60/cranial30 and in LAO 30/cranial20 view for delineating the size and location of the VSD. For the PDA, a descending aortogram was done in lateral view to determine the morphology and size of the PDA. The device size was taken as being 2 mm bigger than the narrowest diameter of the defect.
Two approaches were used for VSD device closure, that is, anterograde and retrograde. An anterograde approach was used in three patients in whom amplatzer duct occluder I (ADO-I) was deployed. A retrograde approach was used in one patient where ADO-II had to be used.
Anterograde approach: The defect was crossed retrogradely with a Judkins right catheter parked in the ascending aorta using an exchange length (260 cm) 0.032-inch J tip teremo wire. The crossed teremo wire was parked in either one of the branch pulmonary arteries or the inferior venacava, and an arterio-venous (A-V) loop was created by snaring it through the venous side. Through this A-V loop, a long sheath (appropriate for the device size) was introduced from the venous side and parked in the ascending aorta. The device was deployed under fluoroscopic and transthoracic echocardiography guidance anterogradely and by coming back from the ascending aorta by the peeling method. Retrograde approach: The defect was crossed as described in the anterograde approach but an A-V loop was not created. A guiding catheter was introduced via the teremo wire through the defect and parked in the right ventricle. The device was then deployed retrogradely by pulling the guiding catheter from the RV into the ascending aorta.
Post-deployment, the device location and presence of any residuals were seen on the LV angiogram in the case of antegrade deployment. A transthoracic echocardiogram was done to look for residual shunt, the development of new aortic or tricuspid regurgitation, and LV function.
PDA device closure was done anterogradely through the femoral vein. Arterial access was taken for the purpose of oximetry. A floppy-end hard wire was passed from the venous end (femoral vein) via the right ventricle to the descending aorta across the PDA. Over the hard wire, a long sheath of suitable size was passed and parked in the descending aorta. Then, after the removal of the wire, an appropriately sized device was delivered via a delivery cable through the long sheath. The ADO-I device was deployed via a peeling-off method. A post-deployment transthoracic echocardiogram was done to look for device position, residual shunt, flow in the LPA and descending aorta, and LV function.
Outcome analysis: The following outcomes were analyzed:
Procedure-related complications. Hospital length of stay. Presence of any residual shunt. Relief of volume overload symptoms.
Follow-up
The patients were evaluated by echocardiography and ECG. Follow-up was done at 1 month, 3 months, 6 months, and yearly after that.
Ethical Consideration
The study was conducted on ethical guidelines for biomedical research on human subjects given by the Central Ethical Committee on Human Research, New Delhi, in addition to principles enunciated in the “Declaration of Helsinki.” Proper informed consent was taken by the patients/guardians of the patients.
Statistical Analysis
Descriptive statistics for categorical variables were reported as frequency and percentage. Continuous variables were reported as medians and ranges.
Results
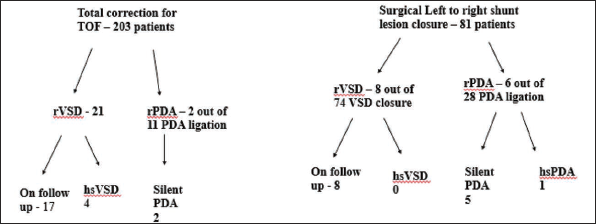
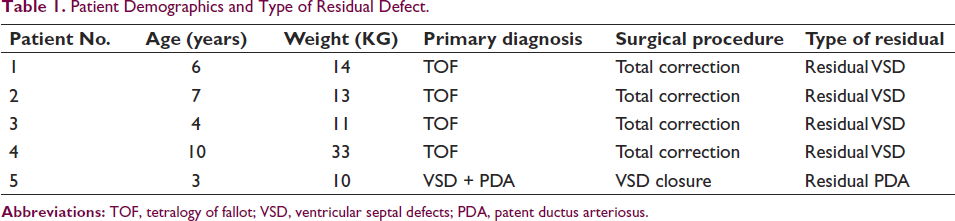
A total of 203 patients underwent complete repair for TOF; 74 patients underwent surgical VSD closure; and seven patients underwent isolated surgical PDA ligation. Out of the total 277 patients who underwent patch closure of VSD, 29 (10.4%) had a residual VSD. Thirty-nine patients underwent PDA ligation, of whom eight (20.5%) had a residual PDA, as shown in Figure 1. Five patients underwent closure of hemodynamically significant residual left-to-right shunts of which four were residual VSD and one was residual PDA. The patient demographics and diagnosis are shown in Table 1. Table 2 depicts the hemodynamic data and devices used.
Patient Demographics and Type of Residual Defect.
Showing Device Closure Data.
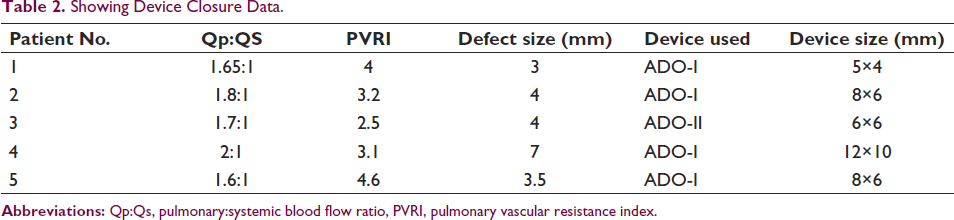
The median age at the transcatheter procedure was 6 years (range 3–10 years), with the median weight being 13 kg (range 10–33 kg). The median time interval between the surgical procedure and the device closure procedure was 4 days (range 2–15 days). The oximetry showed a median Qp:Qs of 1.7:1, and the median PVRI was 3.2 wuxm 2 (range 2.5–4.6 wuxm 2 ). The median fluoroscopy time was 17.2 min (range 11.6–32.3 min). The median defect size was 4 mm (range 3–7 mm).
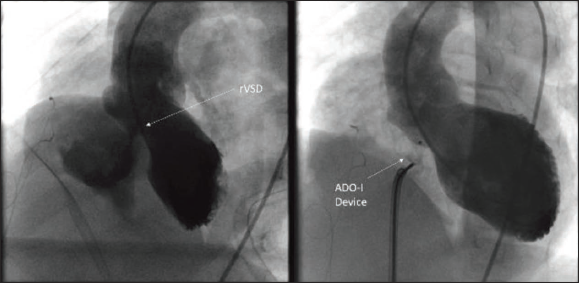
Patient 1 was a 6-year-old boy diagnosed with TOF and underwent complete repair with a transannular patch (TAP). The patient was extubated uneventfully but developed bilateral persistent pleural effusion, which persisted for 15 days postoperatively. On echocardiography, the patient had a residual VSD, and since the child was not responding to medical management, it was decided to do cardiac catheterization. Oximetry showed Qp:Qs of 1.65:1. The LV angiogram showed a residual defect of size 3 mm in the lower end of the VSD patch, as shown in Figure 2. A 5×4 ADO-I device was deployed in an anterograde fashion using a 6F amplatzer sheath. Post-deployment, there was no residual aortic regurgitation or tricuspid regurgitation. Post-procedure, the hospital stay was uneventful with a resolution of pleural effusion, and the patient was discharged on postoperative day 20.
Patient 2 was a 7-year-old patient with TOF who underwent complete repair with TAP. He developed features of left ventricular volume overload with tachypnoea and basal crepts on postoperative day 2, and echocardiography was suggestive of residual VSD. Oximetry revealed a hemodynamically significant shunt. The LV angiogram showed a residual VSD of 4 mm which was closed using an 8 × 6 ADO-I device, as shown in Figure 3. The device was deployed in an anterograde fashion using a 6F amplatzer sheath. Post-deployment, there was no residual, and the patient was extubated on postoperative day 3 and subsequently discharged on postoperative day 11.
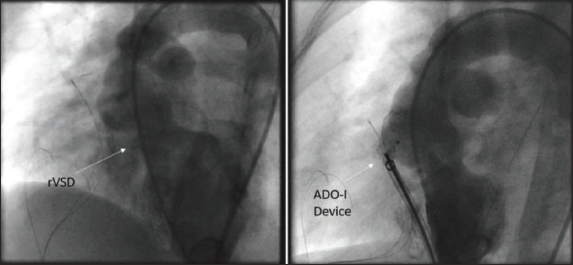
Patient 3 was a 4-year-old boy diagnosed with TOF who underwent complete repair with TAP. The indication for device closure was LV volume overload symptoms due to residual VSD on postoperative day 4. The LV angiogram showed a defect of 4 mm, and the ADO-I 8×6 device was planned for closure. A-V loop was formed, and a 6F amplatzer sheath was planned to cross the defect. But on attempting to cross the VSD anterogradely, the child developed transient bradycardia due to the tenting of the heart. So, it was planned to cross the defect retrogradely using a 5F guide catheter, and the defect was closed using a 6 × 6 ADO-II device as shown in Figure 4. Post-procedure, there was no residual, and the patient improved symptomatically and was discharged on postoperative day 7.
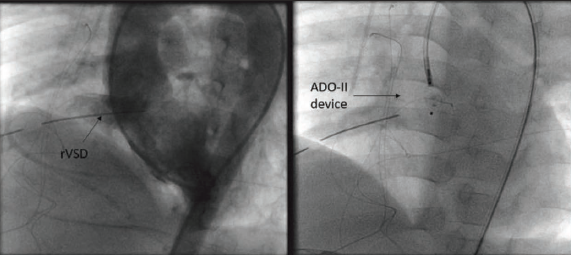
Patient 4 was a 10-year-old case of TOF who had a turbulent hospital course. The patient presented in a sick state with very low saturation (30%) and was taken for emergency RVOT stenting due to a poor general condition. After deploying the stent, it embolized into the right PA, and the patient had to be taken for emergency stent retrieval and complete repair. The patient was extubated successfully and was hemodynamically stable. The patient developed persistent pleural effusion, and echocardiography showed a significant residual VSD. The patient was taken for device closure on postoperative day 11. An LV angiogram showed a large defect of size 7 mm, and a 12 × 10 ADO-I device was deployed anterogradely using a 7F amplatzer sheath as shown in Figure 5. Despite this, the pleural effusion continued and a trial of somatostatin followed by pleurodesis was done. The child improved gradually and was discharged on postoperative day 36.
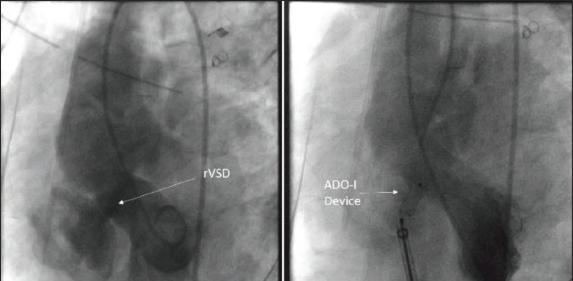
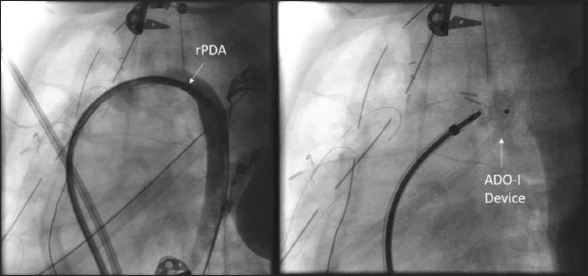
Patient 5 was a 3-year-old case of large perimembranous VSD with a PDA. The patient underwent surgical VSD closure with PDA ligation. On postoperative day 3, the patient developed symptoms of LV volume overload, and an echo showed a residual PDA, so the patient was taken for PDA device closure. A descending aortogram showed a residual PDA with the PA end size being 3.5 mm, as shown in Figure 6. The PDA was closed using an 8 × 6 ADO-I device anterogradely using a 6F amplatzer sheath. There was no residual shunt post-procedure and the patient showed symptomatic improvement. The patient was discharged on postoperative day 6.
Outcome
All five patients improved symptomatically and were discharged in satisfactory condition. A procedure-related complication was seen in patient 3, who developed transient bradycardia while trying to maneuver the long sheath across the VSD. Patient 3 also developed a temporary loss of femoral artery pulse, which was managed by a heparin infusion. The median total hospital length of stay was 11 days (range 6–36 days). The median hospital length of stay after the device closure procedure was 5 days (range 3–25 days). There was no residual shunt seen in pre-discharge echocardiography, showing a 100% success rate of device closure.
Follow-up
The median follow-up period was 2 years, with a range of 1–3.4 years. At the last follow-up, all five patients had no residual shunt and normalized LV dimensions. There was trace AR in three patients and mild TR in two patients. The rest of the 25 residual VSD and five silent PDA patients are on follow-up and are asymptomatic.
Discussion
In this study, we found that the transcatheter closure of postoperative residual VSD and PDA is a safe procedure with minimal procedure-related complications, even though the procedures were done in the immediate postoperative period. The median time interval between the surgery and device closure was 4 days (range 2–15 days) and the median age at the transcatheter procedure was 6 years (range 3–10 years). In other studies, the time interval and age of the procedure were fairly similar as compared to our study. In a study done by Kouakou et al. 5 the median time interval between surgery and device closure was 10.4 years (range 0.3–33.0 years), and the median age was 17.7 years (range 7 months–48 years). Similarly, in another study, the mean time interval between the original operation and the first transcatheter VSD procedure was 8 ± 2.3 years. The median age at the procedure was 32.5 years (16 months–79 years), which was higher as compared to our study. 6
Postoperative VSDs can be due to an additional VSD that was not detected during preoperative assessment, a residual through the patch used to close the VSD, or a fenestration created in cases of PA hypertension. Residual VSD in the perimembranous region can be detected at the upper end of the patch near the aortic valve or at the lower end below the septal leaflet of the tricuspid valve. In previous studies, it was shown that almost one-third of patients had color Doppler evidence of a residual VSD during intraoperative assessment. Two-thirds of these close spontaneously before discharge. 7 Most residual VSDs that are less than 2 mm in size close spontaneously with time and can be followed up. 8 In our series, there was no residual VSD seen on intraoperative transoesophageal echocardiographic analysis, suggesting that they could have been due to postoperative patch dehiscence. The incidence of postoperative patch dehiscence is less than 5%, but not much data is available. The most common location of patch dehiscence is the posteroinferior and posterosuperior quadrants, near the location of the conductive tissue; thus, the surgeon is not able to take continuous sutures in these areas, making them prone to dehiscence. 9 Similarly, residual PDA is not uncommon, as it may be missed in the preoperative assessment or may be present if the ligation is not tight enough. Closure of residual VSD and PDA is indicated if it is associated with symptoms of volume overload or if there is associated infective endocarditis. The ideal management of residual lesions, including residual VSD and PDA, is surgical closure. But it is not free from complications, which include complete heart block, infection, neurological complications, and even mortality. 10 Surgical closure is also not technically easy as it involves a redo sternotomy, the need for a second cardiopulmonary bypass run, increased bleeding, difficulty in finding the residual, and the presence of myocardial edema, which makes taking sutures difficult. Thus, the percutaneous closure provides an alternative for these residual defects.
Device closure of residual VSD has its own challenges. Unlike the native muscular VSD and, in some cases, perimembranous VSD, where device closure has become a standard of care, 11 there is no consensus guideline regarding the device closure of residual VSD. In postoperative residual VSDs, the location and the reasons for their development must be considered to identify ideal candidates for this type of closure. It is sometimes difficult to determine the precise location of the residual VSD in postoperative cases. Therefore, pre-procedure imaging and surgical notes must be taken into consideration before planning the device closure. Echocardiography must give us an idea of the number of defects, their size, location, and proximity to the tricuspid and aortic valves. As the residual is in close proximity to the patch, the angiographic morphology of the VSD is not like the typical perimembranous or muscular VSD and multiple angiograms may be needed in certain cases. An LV angiogram in LAO 60/cranial 30 and LAO 30/cranial 20 views should be taken to look for the number of residual defects, size, location, and proximity to the aortic valve. Device closure is not advisable if there are multiple defects or the defect is very close to the aortic valve (less than 4 mm). All four patients with VSD devices in this study had a single residual defect through the lower end of the patch, away from the aortic valve. The indication for device closure was LV volume overload in two patients and persistent pleural effusion in two. The hemodynamic assessment revealed Qp:Qs of more than 1.5 in all of the VSD patients, which is an indication of the closure of the defect. 11
The device used for VSD closure was the single disc ADO-I in three patients and the double disc ADO-II in one patient. The ADO-I device is deployed anterogradely, and the ADO-II can be deployed both anterogradely and retrogradely. Anterograde deployment is preferred as a larger, longer sheath can be safely introduced through the venous system. In patient 3, anterograde deployment could not be achieved as the patient developed bradycardia as the long sheath was introduced through the defect. Procedure-related complications are not uncommon during VSD device closure and include device embolization, complete heart block, aortic or tricuspid regurgitation, hemolysis requiring blood transfusion, and the need for surgical intervention. The rate of major complications ranges from 0% to 8.6%, as seen in various studies.12, 13 The most feared of these is the complete atrioventricular block, seen in 0%–5.7% cases of native VSD device closure cases. In postoperative residual VSD cases, the presence of patches and scar tissue may provide some protection against the mechanical/compressive effect of the device, reducing the incidence of this complication. 1 There were only two procedure-related complications in our study, that is, transient bradycardia and transient loss of femoral arterial pulse, both seen in the same patient. The published reports for transcatheter perimembranous VSD device closure have shown a success rate of 80%–99%. 12 In our study, the success rate was 100%, with no patient having any residual VSD. The postoperative course was also uneventful, with early discharge for all four patients in satisfactory condition. The median follow-up period was 2 years. All four patients were asymptomatic with normal LV dimensions and normal sinus rhythm.
Similarly, the presence of a residual PDA after surgical ligation can be seen, and it is due to suboptimal occlusion or recanalization. 14 Most of these are silent PDAs and close spontaneously in 3–6 months, but 7% of patients will need a second procedure to close the duct. 15 In this study, there were eight cases (20.5%) of residual PDA of which only one was hemodynamically significant enough to warrant closure in the immediate postoperative period. The rest of the seven cases were silent PDAs, and it was decided to keep these patients on follow-up. Device closure of residual PDA is a good alternative to redo surgical ligation as it has less morbidity and mortality. 16 One issue with a residual PDA is that it can be less distensible than a native PDA due to reactive fibrosis. This can make device closure technically more challenging, and a retrograde or snare technique is often used instead of a standard anterograde approach. 17 In this study, the PDA was closed anterogradely without any technical difficulty, and there was no procedure-related complication.
Study Limitations
This was a retrospective analysis based on a small number of cases from a single center.
Conclusion
Transcatheter device closure of residual postoperative VSD and PDA is an effective alternative to redo surgery in older children. Our analysis has a small number of cases to draw any firm conclusion from, but it has shown that it is feasible even in the immediate postoperative period, with good success and closure rates and very low morbidity. Further study is required with a larger sample size, and a longer follow-up is needed to evaluate its long-term safety and efficacy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
