Abstract
Coronary artery calcification impacts outcomes after percutaneous coronary interventions owing to stent under expansion causing increased risk of stent thrombosis and restenosis. Therefore, adequate calcium modification before stent placement is the key to get desired outcomes in the patients with coronary artery calcification. Many devices are available for calcium modification, which includes orbital or rotational atherectomy, cutting balloon, scoring balloon, and intravascular lithotripsy. All these techniques have inherent risks of complications, such as coronary dissection, perforation, and slow or no reflow. We report a case of intravascular lithotripsy in calcified proximal left circumflex artery (LCX) lesion in a 63-year-old female leading to the left anterior descending artery (LAD) with no flow and ischemic ventricular tachycardia requiring DC cardioversion. Intravascular ultrasound from LAD revealed subintimal hematoma from ostium to mid LAD with intimal flap continuation into ostio-proximal LCX. The intimal flap in LAD has not showed any entry or exit tears which might increase the possibility of intramural hematoma shift to adjacent areas and branches leading to no reflow. Hence, cutting balloon fenestration of the intimal flap, followed by left main coronary artery bifurcation stenting was done to get a good result with flowing distal branches.
Keywords
Introduction
Coronary artery calcification impacts outcomes after percutaneous coronary intervention (PCI) because of stent under expansion causing increased risk of stent thrombosis and restenosis. Ongoing evolution of calcium modification devices, operator skills, and coronary imaging guidance for operators during coronary procedures to achieve the best possible final minimum stent area, changed the outcomes of these patients.1, 2 Intravascular lithotripsy (IVL), the latest addition to this armamentarium revolutionized calcium modification and also made procedure complex and shortened learning curve. 2 Intracoronary imaging information contributes significantly to decision-making at every step of calcium modification and also helps in identifying complications such as dissections and intramural hematoma (IMH) to devise further treatment strategies. 3 Early data on IVL safety and efficacy showed acceptable procedural outcomes with minimal complications, such as dissections in 5% with no incidence of slow flow or no flow.4, 5 Here, we report a case of calcified lesion in the proximal left circumflex artery (LCX) treated with IVL resulting in slow flow in the left anterior descending artery (LAD) owing to IMH extending retrogradely from the proximal LCX into the ostio-proximal LAD. To the best of our knowledge, this is the first such case reported in literature.
Case
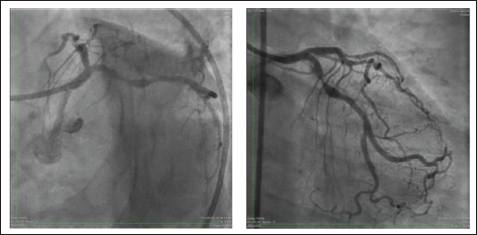
A 63-year-old diabetic and hypertensive female presented with unstable angina. On evaluation, the electrocardiogram (ECG) showed ST depressions in lateral leads, and the echocardiogram revealed the normal left ventricular function and no regional wall motion abnormality. Serial biomarkers were negative. The exercise stress test revealed positive for inducible ischemia. Hence, coronary angiogram (CAG) was performed, which showed double vessel disease with critical calcific proximal LCX stenosis as shown in Figure 1A and B.
(A) LAO Caudal View Proximal LCX 80%-90% Lesion. (B) AP Caudal View Mid LAD 70%-80% Lesion, Proximal LCX 80%-90% Lesions and Mild Lesions in Both Ostium’s of LAD and LCX.
As the lesion showed severe calcium on fluoroscopy, intravascular ultrasound (IVUS)-guided PCI with stent to proximal LCX was planned. IVUS could not be navigated through the proximal LCX lesion, so we predilated with the 1.5-mm balloon and the advanced IVUS catheter (OPTICROSS HD, Boston Scientific, India). IVUS showed circumferential calcium (Figure 2A) at the lesion and also dog boning observed during predilatation with the 3.5 × 12 mm2 NC balloon (Apollo Balloon, BrosMed Medical Corporation Ltd, China) at 22 atmospheres (ATM) (Figure 2B). Therefore, calcium modification with the 3.5-mm IVL balloon (Shockwave Medical, Translumina Therapeutics LLP, India) was done, and the balloon waist disappeared (Figure 3A) after 5 cycles of pulses. Soon after successful IVL calcium modification in the LCX, the patient started complaining of angina, and the ECG showed VT, requiring cardioversion with a 200-J synchronized DC shock. The angiogram revealed a critical occlusion of LAD with Thrombolysis in Myocardial Infarction 1 (TIMI 1) flow (Figure 3B).
(A) IVUS Images Showing More than 270o of Calcium. (B) 3.5/12 NC Balloon with Dog Boning Even at 22 ATM.
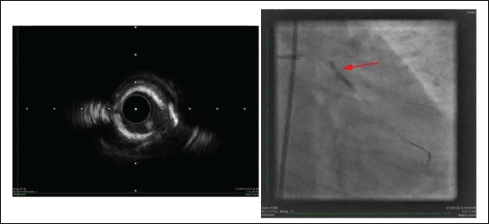
(A) Well Expanded IVL Balloon After 5 Pulses. (B) Reduced LAD Flow After IVL in LCX.
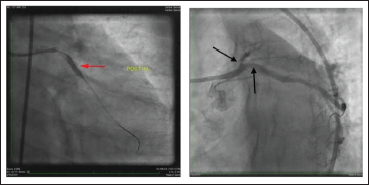
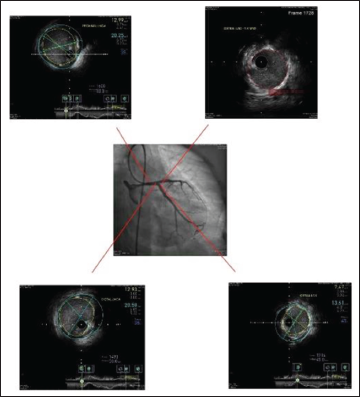
LAD was wired swiftly with a Sion blue wire (Asahi Intecc Co., Ltd, Japan) with microcatheter support. IVUS interrogation from LAD revealed subintimal hematoma extending from ostium to mid LAD and also flap continuation into ostio-proximal LCX (Figure 4). As the patient was unstable, we decided to go ahead with left main bifurcation stenting in view of IMH extending into both ostia of LAD and LCX. So, ostial LCX is quickly stented using the 3.25 × 33 mm2 drug eluting stent (DES) (XIENCE Xpedition TM, Abbot Vascular, USA) with minimal protrusion into the left main coronary artery (LMCA), followed by crushing with the 3.5 × 12 mm2 NC balloon (Shockwave Medical, Translumina Therapeutics LLP, India) and the 3 × 10 mm2 cutting balloon (Wolverine, Boston Scientific, India). Dilatation was done in the proximal LAD to create fenestrations in the intimal flap to prevent IMH migration distally and into the branches of the proximal LAD. Subsequent IVUS from LAD showed clear fenestrations in the flap with reduced intensity of speckling, which is suggestive of the increased flow (Figure 4). Further, LMCA to LAD stenting was done with 3 × 33 mm2 stent (XIENCE AlpineTM, Abbot Vascular, USA), followed by POT, LCX Re-cross, kissing balloon and REPOT. The final result showed good stent expansion and TIMI 3 flow into the distal LAD filling all septal arteries and diagonals (Figure 5).
IVUS from LAD before cutting balloon 4A, 4B, 4C, 4D, IVUS from LAD after cutting balloon 4E, 4F, 4G, 4H. (A) Distal reference without IMH. (B) IMH with increased speckling intensity from 2-6 o clock, branches opening into true lumen at 9 & 11 O clock. (C) Alternated echogenicity in IMH. (D) Ostial LAD IMH at 3-6o clock in ostial LAD extending into LCX ostium. (E) Distal end of IMH with intact flap. (F) Fenestration in flap at 2 o clock with similar intensity of speckling. (G) Intact flap in proximal LAD, (H) Ostial LAD fenestration seen at 2o clock.
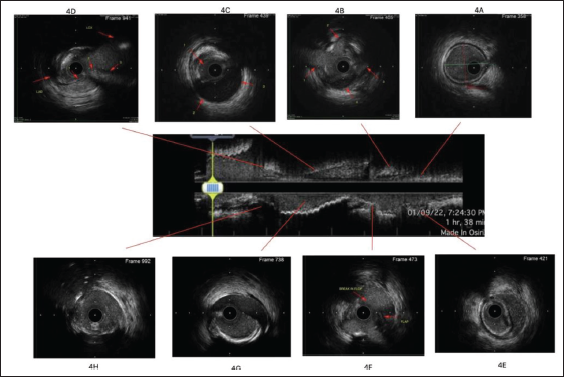
Angiogram and Final IVUS.
Discussion
IVL, a balloon with the shaft containing emitters to convert the electrical energy into the acoustic pressure wave, delivers pressure waves equivalent to 50 ATM on to plaques, leading to the comminution of calcium in it. 5 Generally, it is more controlled calcium modification, but it has its own share of complications, such as dissection, coronary perforation, or IMH. 6 Often these complications are limited to the vessel treated, causing downstream ischemia. Nevertheless, as in this case, dissection with IMH can extend into the adjacent branch in a retrograde manner and can cause ischemia-driven complications in upstream neighborhood. Being a pulsatile acoustic pressure wave, IVL can propagate IMH, which is further facilitated by inter–pulse balloon deflation, allowing flow into subintimal dissection. IMH usually propagates distally in the direction of blood flow; however, if intramural tissue resistance offered to split is increased by the atherosclerotic process, it might propagate to low resistance proximal tissue planes, as happened in this case. Hence, it might be prudent to park a safety wire in significant side branches so that timely bailout becomes feasible. Undoubtedly, coronary imaging helps in unraveling the unprecedented complications like this during PCI. Intravascular imaging, preferably IVUS, as there is no need to inject into coronaries for imaging, is a better tool to define the extent of IMH and to locate the possible entry tear. 7 As in this case where no entry tear was seen in LAD and the dissection flap is traced back into the LCX with corroborative evidence of prior IVL in the proximal LCX, entry tear is in the LCX as seen in fluoroscopy but not confirmed with IVUS as the LCX stented quickly to stop further hemodynamic deterioration. Variable degree of speckling intensity can be noticed at different locations of IMH because of variations in the speed of flow, red cell aggregation, and fibrin content.8, 9 IMH can also contain echolucent areas (Figure 4c) that presumably represent the accumulation of saline or radiographic contrast. Stent placement in IMH, in the absence of fenestrations in the flap, can squeeze it to adjacent segments of the vessel or into the side branches around, resulting in a no-flow situation. Hence, fenestration in the flap of the IMH would facilitate the evacuation of intramural blood collection, preventing propagation when the stent is deployed. Fenestration in the flap can be achieved by cutting balloon, scoring balloon, or by perforation with stiffer wires.10–12 IVUS findings, such as a gap in the continuity of the flap, drop in the intensity of speckling in IMH, and speckling intensity equivalence with true lumen help in identifying successful fenestration.
Conclusion
Ischemia of the upstream neighboring territory can occur while doing IVL in the proximal LAD or LCX owing to the retrograde propagation of IMH. IVUS helps in unraveling the anatomy in such an unprecedented complication and guides further interventional strategy to achieve the better end of the procedure flow into the distal vessel. Further, having a safety wire in the neighboring vessel at the start of IVL procedure would help in minimizing the rescue response time.
Footnotes
Acknowledgement
I would like to acknowledge Dr. Chandana from the clinical research department for drafting the manuscript and submitting to the journal.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Inform Consent
Informed consent was taken before undergoing the procedure.
